UNITED STATES SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the fiscal year ended December 31 , 2025
or
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the transition period from _____ to _____
Commission file number : 001-35803
(Exact name of registrant as specified in its charter)
(State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | |||||||
(Address of principal executive offices) (Zip Code)
Telephone: +353 1 696 0000
(Registrant's telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act: None
Securities registered pursuant to Section 12(g) of the Act: Ordinary shares, par value $0.01 per share
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large Accelerated Filer | ☐ | Accelerated Filer | ☐ | ☒ | Smaller Reporting Company | Emerging Growth Company | ||||||||||||||||||||||||||||||||||||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management's assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No ☒
Indicate by check mark whether the registrant has filed all documents and reports required to be filed by Section 12, 13 or 15(d) of the Securities Exchange Act of 1934 subsequent to the distribution of securities under a plan confirmed by a court. ☒ Yes ☐ No
The number of shares of the registrant's ordinary shares outstanding as of April 8, 2026 was 39,581,987 .
DOCUMENTS INCORPORATED BY REFERENCE
Certain portions of the registrant's definitive proxy statement for its annual meeting of shareholders, to be filed with the Securities and Exchange Commission within 120 days after December 31, 2025, are incorporated by reference into Part III of this Annual Report on Form 10-K.
KEENOVA THERAPEUTICS PLC | ||||||||
| TABLE OF CONTENTS TO FORM 10-K | ||||||||
2
Presentation of Information
Unless the context requires otherwise, references to “Keenova Therapeutics plc,” “Keenova,” “we,” “us,” “our” and “the Company” refer to Keenova Therapeutics plc (formerly Mallinckrodt plc), an Irish public limited company, and its consolidated subsidiaries. References to “dollars” or “$” refer to United States (“U.S.”) dollars.
Trademarks and Trade Names
Keenova owns or has rights to use trademarks and trade names that it uses in conjunction with the operation of its business. One of the more important trademarks that it owns or has rights to use that appears in this Annual Report on Form 10-K (this “Annual Report”) is “Keenova,” which is a registered trademark or the subject of pending trademark applications in the United States and other jurisdictions. Solely for convenience, the Company only uses the ™ or ® symbols the first time any trademark or trade name is mentioned. Such references are not intended to indicate in any way that the Company will not assert, to the fullest extent permitted under applicable law, its rights to its trademarks and trade names. Each trademark or trade name of any other company appearing in this Annual Report is, to the Company's knowledge, owned by such other company.
Forward-Looking Statements
The Company has made forward-looking statements in this Annual Report that are based on management's beliefs and assumptions and on information currently available to management. Forward-looking statements include, but are not limited to, information concerning the Company's possible or assumed future results of operations, business strategies, financing plans, competitive position, potential growth opportunities, potential operating performance improvements, the effects of competition and the effects of future legislation or regulations. Forward-looking statements include all statements that are not historical facts and can be identified by the use of forward-looking terminology such as the words “believe,” “expect,” “plan,” “intend,” “project,” “anticipate,” “approximately,” “estimate,” “predict,” “potential,” “continue,” “may,” “will,” “could,” “should” or the negative of these terms or similar expressions.
Forward-looking statements involve risks, uncertainties and assumptions. The Company’s actual results and financial condition may differ materially from those indicated in the forward-looking statements. Therefore, you should not place undue reliance on any of these forward-looking statements.
The risk factors included in Item 1A. Risk Factors of this Annual Report could cause the Company's actual results and financial condition to differ materially from those indicated in the forward-looking statements. There may be other risks and uncertainties that the Company is unable to predict at this time or that the Company currently does not expect to have a material adverse effect on its business.
These forward-looking statements are made as of the filing date of this Annual Report. The Company expressly disclaims any obligation to update these forward-looking statements other than as required by law.
3
Summary of Selected Risk Factors
Our business is subject to numerous material and other risks and uncertainties that you should be aware of. These risks and uncertainties are described more fully in “Item 1A. Risk Factors” in this Annual Report, and include, but are not limited to, the following:
Risks Related to the Business Combination with Endo and Separation of Par Health
•We may not realize the anticipated benefits and synergies from our Business Combination (as defined below) with Endo.
•The estimated fair values of the net assets acquired by us in connection with the acquisition of Endo are preliminary and subject to change if new information becomes available.
•We could incur additional payment obligations pursuant to the U.S. Government Economic Settlement upon the achievement of certain EBITDA outperformance targets.
•We may not achieve growth opportunities, profit improvements, cost savings and other benefits, and may incur unanticipated costs associated with the Separation (as defined below), and our results of operations, financial condition and valuation could be adversely affected as a result.
•As a result of the Separation, we may lose the benefits of services provided by Par Health or certain of its subsidiaries and we may incur incremental costs as a result.
•We are a smaller and less diversified company than before the Separation.
•The Separation may result in litigation and/or regulatory inquiries and investigations, which could harm our business, financial condition and operating results and could divert management attention.
Risks Related to Our Business
•Pharmaceutical companies like us have been under increasing scrutiny and non-compliance with relevant policies, laws, regulations or government guidance may result in adverse actions.
•We have various contractual and court-ordered compliance obligations that, if violated, could result in penalties.
•We face significant competition and may not be able to compete effectively.
•We experience pricing pressure on certain of our products, which could reduce our future revenue and profitability.
•Sales of our products are affected by the reimbursement practices of governmental health administration authorities, private health coverage insurers and other third-party payers.
•Our reporting and payment obligations under governmental purchasing and rebate programs are complex. Any determination of failure to comply could have a material adverse effect on our business.
•Cost-containment efforts of our customers and other parties could materially adversely affect our business.
•Extensive laws and regulations govern the industry in which we operate, and any failure to comply may materially adversely affect us.
•Our approved and investigational products may cause undesirable side effects that limit their commercial profile or result in other negative consequences.
•We have limited resources and may not seek to, or be successful in our efforts to, identify or discover additional products or product candidates at the rate we expect.
•We may be unable to successfully develop, commercialize or launch new products or expand commercial opportunities for existing products.
•We may not achieve the anticipated benefits of price increases for our products.
•Our customer or product concentration may materially adversely affect our business.
•We may be unable to protect our intellectual property rights or we may be subject to claims that we infringe on the intellectual property rights of others.
•Clinical trials demonstrating the efficacy of Acthar® Gel are limited, which could cause physicians not to prescribe Acthar Gel, or payers not to reimburse the drug.
•Clinical studies are expensive and time-consuming, and their outcome is highly uncertain. If any such studies are delayed or yield unfavorable results, regulatory approval may be delayed or become unobtainable or, even if approved, physicians might not prescribe our products.
4
•We may incur litigation liability, including but not limited to product liability losses.
•If our business development activities or other transactions are unsuccessful, it may adversely affect us.
•We may be unable to attract and retain qualified personnel in key fields.
•Our business depends on the continued effectiveness of our information technology infrastructure, and external attacks or any failures could harm our operations.
•Our business could suffer if we, or our suppliers, encounter manufacturing or supply problems.
•Our operations expose us to risks and challenges associated with conducting business internationally.
•New or increased tariffs and evolving trade relations between the United States and other countries, as well as changes in U.S. international trade and taxation policy, could adversely affect us.
•Our actual financial results are not comparable to our historical financial statements.
Risks Related to Our Indebtedness and Settlement Obligation
•Our substantial indebtedness and settlement obligation could adversely affect our financial condition and future access to capital and may prevent us from fulfilling our obligations.
•The terms of the agreements that govern our indebtedness and settlement obligation restrict our current and future operations.
•Our variable-rate indebtedness exposes us to interest rate risk.
•Despite current and anticipated indebtedness levels, we may incur additional debt in the future.
•Future financings may not be available on favorable terms and may be dilutive, and the use of proceeds therefrom may be subject to restrictions from our existing indebtedness.
Risks Related to Tax Matters
•Our status as a foreign corporation for United States federal tax purposes could be affected by a change in law.
•Future changes to U.S. and foreign tax laws, including the Pillar Two global minimum tax, may increase our effective tax rate and cash tax obligations.
•The United States could treat Keenova Therapeutics plc as a United States taxpayer or otherwise subject it to certain adverse tax consequences under Internal Revenue Code Section 7874.
•The Internal Revenue Service may interpret Internal Revenue Code Section 382 limitation and cancellation of debt income attribution rules differently.
•A loss of a major tax dispute, if one were to arise, or a challenge to our operating structure or intercompany pricing policies could result in a higher tax rate on our worldwide earnings, which could result in a material adverse effect on our financial condition, results of operations and cash flows.
Risks Related to Our Jurisdiction of Incorporation
•Irish law differs from United States law and may afford less protection to holders of our securities.
•Irish law imposes restrictions on certain aspects of capital management.
Risks Related to Our Ordinary Shares
•Our ordinary shares are not listed on any national securities exchange and our plans to list on an exchange and to conduct a concurrent underwritten public offering are subject to a variety of factors, several of which are outside our control.
•Our ability to pay dividends and fund share repurchases is limited, and Irish law requires that we meet certain financial requirements before we pay dividends or fund repurchase our ordinary shares.
5
PART I
| Item 1. | Business. | ||||
Overview
Keenova Therapeutics plc, formerly Mallinckrodt plc, and its consolidated subsidiaries is a leading U.S.-focused branded therapeutics company that strives to help patients with rare or unaddressed conditions live happier and healthier lives.
We operate our business in one operating and reportable segment with a clear and focused strategy centered on our branded therapeutics. Keenova’s rare disease capabilities underpin our diversified brands portfolio, which is focused across a wide range of therapeutics areas of significant unmet need. These include rheumatology, ophthalmology, nephrology, neurology, pulmonology, orthopedics, urology, and neonatal respiratory critical care. For further information on our products, refer to “Our Businesses and Products” within this Item 1. Business.
On July 31, 2025, we completed our business combination with Endo Inc. (which has since been converted to Endo LP, “Endo”) (the “Business Combination”), which resulted in increased scale and enhanced capabilities to develop, manufacture and commercialize branded therapeutics, generic pharmaceuticals, and sterile injectables. Our operating results for the year ended December 31, 2025 reflect the operating results of Endo following the closing of the Business Combination on July 31, 2025. On November 10, 2025, we completed the separation of our generic pharmaceuticals and sterile injectables businesses into an independent, private company named Par Health, Inc. (“Par Health”) (the “Separation”). As a result, we are a smaller and less diversified company than before the Separation and our operations are now centered on our branded therapeutics portfolio. Further, following the Separation, our financial statements and accompanying notes have been recast to reflect Par Health’s assets, liabilities, results of operations and cash flows as discontinued operations for all periods presented. Refer to Note 5 and Note 6 of the Notes to Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for further information on the Business Combination and the Separation, respectively.
Our principal executive offices are located at College Business & Technology Park, Cruiserath, Blanchardstown, Dublin 15, Ireland, with additional office facilities in New Jersey, Pennsylvania, Missouri, the District of Columbia (“D.C.”) and Japan, among others. We have a strong U.S. manufacturing footprint, with facilities in Louisiana, New Jersey, New York, Pennsylvania and Wisconsin. We also have seven regional service centers in the U.S.
In the fourth quarter of fiscal year 2025, we approved a change in our fiscal year end from a 52-53-week year ending on the last Friday of December to a calendar year ending on December 31, 2025.
Our Business and Products
We develop, manufacture and commercialize a portfolio of branded therapeutics for the treatment of rare or unaddressed diseases in the specialty areas of rheumatology, ophthalmology, nephrology, neurology, pulmonology, orthopedics, urology, and neonatal respiratory critical care. Our marketed products are positioned to serve patients with significant unmet medical needs.
Our strategy is to:
•increase patient access to our existing products and support their appropriate utilization through dedicated teams;
•advance label expansion opportunities for our marketed products by targeting new indications that represent substantial unmet medical need, underpinned by our rare disease capabilities; and
•selectively acquire or license products that are strategically aligned with our product portfolio and could leverage our commercial infrastructure and research and development (“R&D”) capabilities across our key therapeutic areas.
We promote our products directly to physicians in office settings, hospitals, and ambulatory surgical centers across our key therapeutic areas with our own direct sales force of over 300 sales representatives as of December 31, 2025. Our products are distributed through independent wholesale drug distributors, specialty pharmaceutical distributors, retail pharmacy chains, and hospital procurement departments, among others. We also contract directly with payer organizations through our team of reimbursement managers to assist with relevant reimbursement processes.
The following is a description of select products in our product portfolio:
•Acthar® Gel (repository corticotropin injection) (“Acthar Gel”) is a complex mixture of peptides approved by the U.S. Food and Drug Administration (“FDA”) for use in 19 indications across five specialty areas. Acthar Gel currently generates substantially all of its net sales from 11 of the on-label indications, including adjunctive therapy for short-term administration for an acute episode or exacerbation in rheumatoid arthritis (“RA”), including juvenile RA; treatment of severe acute and chronic allergic and inflammatory processes involving the eye and its adnexa including keratitis and uveitis; treatment during an exacerbation or as maintenance therapy in selected cases of systemic lupus erythematosus; treatment of acute exacerbations of multiple sclerosis (“MS”) in adults; including a diuresis or a remission of proteinuria in nephrotic syndrome without uremia of the idiopathic type or that is due to
6
lupus; treatment during an exacerbation or as maintenance therapy in selected cases of systemic dermatomyositis (polymyositis); treatment of symptomatic sarcoidosis; and monotherapy for the treatment of infantile spasms in infants and children under two years of age. Acthar Gel has one listed patent in the FDA publication, “Approved Drug Products with Therapeutic Equivalence Evaluations” (“Orange Book”) that expires in 2041.
There is significant clinical evidence supporting the effectiveness of Acthar Gel. This includes data generated from company-sponsored controlled clinical trials, as well as previously completed and largely independent clinical case series and smaller investigator initiated studies that have broadened the product's evidence base and strengthened its clinical profile. We launched Acthar Gel Single-Dose Pre-filled SelfJect™ Injector (“SelfJect”) in August 2024, following FDA approval on March 1, 2024. SelfJect offers an easier and more patient-friendly application for single unit dosage indications. SelfJect continues to receive positive physician and patient feedback, reflecting momentum with both new and returning healthcare providers, and providing patients with an important new option to manage challenging chronic and acute inflammatory and autoimmune conditions, underscoring our investment to modernize the brand for patients and enhance patient affordability.
•Xiaflex® (collagenase clostridium histolyticum) (“Xiaflex”) is an FDA‑approved biologic injectable enzyme therapy used in the treatment of Dupuytren’s contracture and Peyronie’s disease. Xiaflex is approved for the treatment of adult patients with Dupuytren’s contracture who present with a palpable cord, offering a nonsurgical option to reduce contracture and restore finger extension. Xiaflex is also indicated for adult men with Peyronie’s disease who have a palpable penile plaque and a curvature deformity of at least 30 degrees at treatment initiation. In addition to these FDA-approved indications, we are also advancing certain Xiaflex pipeline development programs for potential future indications, including plantar fibromatosis and hammer toe, advancing our strategy of developing non-surgical musculoskeletal care interventions.
•In addition to our two flagship products, Acthar Gel and Xiaflex, our product portfolio also includes various other branded therapeutics, including INOmax®, Supprelin® and Terlivaz®, among others.
•We believe that the details described in the section captioned “Intellectual Property” below underpin the durability of our two flagship products and their future growth.
Our efforts to further develop our key products, including new indications, are described in the section captioned “Research and Development” below.
Business Strategy Developments and Transactions
Our Board of Directors (the “Board”) may determine, from time to time, to implement changes in our business strategy, underpinned by our rare disease capabilities, which may affect our operations and our future direction and which may differ materially from those in the past. Significant transactions, including the Separation and the Business Combination and the related financing, each discussed further below, have substantially reshaped our organization. Our business strategy focuses on developing, manufacturing, and commercializing branded therapeutics that address key areas of significant unmet need, including rheumatology, ophthalmology, nephrology, neurology, pulmonology, orthopedics, urology, and neonatal respiratory critical care. Consistent with this strategy, and at the direction of our Board of Directors, we continue to explore a variety of transactions intended to maximize shareholder value, including potential acquisitions, divestitures, financings and other strategic transactions. In connection with this process, we intend to exit from our remaining opioid business and are currently pursuing the divestiture of our Percocet® business.
Separation of Par Health
On November 10, 2025 (the “Redemption Date”), we completed the Separation of our generic pharmaceuticals and sterile injectables businesses into an independent, private company named Par Health. The Separation was implemented by way of a redemption of all of our issued and outstanding preferred shares, upon which the preferred shares were automatically cancelled (the “Redemption”). In connection with the Redemption and pursuant to Irish law, we allocated the right to receive one hundred percent (100%) of the outstanding shares of Par Health common stock as of the Redemption Date, to holders of record of our preferred shares as of October 27, 2025, who complied with certain certification procedures and certified as to being a qualified institutional buyer as defined in Rule 144A under the Securities Act of 1933, as amended (the “Securities Act”), an accredited investor as defined in Rule 501(a) under the Securities Act or Company director or officer or a Par Health director or officer as of the Redemption Date and also an accredited investor. Holders of record of our preferred shares who complied with the certification procedures and certified that they do not qualify as any of the foregoing categories of investors received a per share amount in cash that the Board of Directors determined was equal in value to the Par Health common stock allocated to qualified shareholders for each of our preferred shares.
We and Par Health entered into several agreements that govern our relationship following the Separation, including a separation agreement, a transition services agreement, a tax matters agreement, an employee matters agreement, a manufacturing and supply agreement and an amended and restated multi-tenant lease agreement.
Also in connection with the Separation, we and the Opioid Disbursement Trust II (“Trust”) entered into a termination agreement (“CVR Termination Agreement”) to cancel the contingent value rights (“CVRs”) issued under the agreement (“CVR Agreement”)
7
entered into in connection with our emergence from the 2023 Bankruptcy Proceedings (as defined below) and terminate the CVR Agreement in exchange for a payment by us of $35.0 million to the Trust. Pursuant to the CVR Termination Agreement, on November 10, 2025, the CVRs were cancelled and the CVR Agreement was terminated. The CVR Termination Agreement also included customary representations and warranties and a waiver of certain claims. Refer to Note 6 to the Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for further discussion on the CVR Termination Agreement.
As a result of the Separation, neither we nor any of our subsidiaries continue to be borrowers or guarantors of the indebtedness entered into in connection with the Par Health Credit Agreement (as defined below). All borrowers and guarantors in respect of such indebtedness are subsidiaries of Par Health.
Business Combination with Endo
On July 31, 2025, we completed the Business Combination with Endo, whereby we acquired all of the issued and outstanding shares of common stock of Endo in exchange for a combination of cash and our ordinary shares in accordance with the Transaction Agreement (as amended on April 23, 2025) (“Transaction Agreement”) entered into on March 13, 2025, among us, Endo, and our wholly owned subsidiary, Salvare Merger Sub LLC (“Merger Sub”). Outstanding shares of common stock of Endo were cancelled and converted into the right to receive 0.2575 of our ordinary shares (“Per Share Stock Consideration”) and approximately $1.31 in cash (“Per Share Cash Consideration”) without interest and subject to applicable withholding. The aggregate amount of cash paid to Endo stockholders was $100.0 million and the aggregate amount of our ordinary shares issued to former Endo stockholders was 19,650,663 shares.
Refer to Note 5 to the Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for further information regarding the Business Combination.
Other Developments and Transactions
On November 29, 2024, we completed the sale of our Therakos business to affiliates of CVC Capital Partners IX for total cash consideration of $887.6 million, which amount was net of preliminary purchase price adjustments, including an adjustment based on estimated net working capital at close, and we recorded a gain on sale of $754.4 million. We used the net proceeds of such sale to mandatorily prepay and redeem a portion of our senior secured debt, resulting in makewhole payments and a net loss on extinguishment of debt. We paid $6.2 million for the final working capital settlement during the year ended December 31, 2025.
Research and Development
Our R&D strategy is primarily focused on identifying areas of high unmet medical and clinical need that may be addressed by our existing products, advancing the development of those products to address such needs, and evaluating the viability of new product candidates that we may acquire or license through business development activities.
Our current R&D infrastructure is focused on supporting late-stage product development, maximizing new product launches, and accelerating additional lifecycle management opportunities, inclusive of new product enhancements, and line extensions that provide value to patients, physicians and payers.
Evidence generation is a key strategic imperative within R&D for all our products and therapeutic areas of interest, as it supports approved uses, label enhancements, and new indications. Our integrated evidence generation strategy is realized through investments in both clinical and health economic activities. We seek to support research that advances the understanding and treatment of various diseases states and further the development of our currently marketed products, notably Acthar Gel, and Xiaflex.
We are currently developing Xiaflex for additional indications including plantar fibromatosis (“PFI”) and hammer toe.
The registrational Phase 3 study for the treatment of PFI is on-going, and results are expected in the third quarter of 2026.
A Phase 1/2 study for the treatment of hammer toe was completed in February 2026. Topline data demonstrated a favorable safety profile and met secondary and exploratory efficacy endpoints enabling progression of the program into a registrational Phase 3 study that, subject to the outcome of discussion with the FDA, is planned to be initiated in the fourth quarter of 2026.
We are also considering further development for plantar fasciitis (“PFA”). To date, the development program for PFA consists of completed Phase 1 and Phase 2 clinical studies. Results from these studies provided clinically meaningful information to design a registrational Phase 3 trial for patients with moderate to severe plantar fasciitis, which we currently plan to initiate subject to the results of our PFI study and other considerations.
We may develop our Xiaflex product for potential additional indications in the future.
Competition
Our products face competition from alternative forms of treatment that a prescriber may utilize. To successfully compete for business from managed care and pharmacy benefits management organizations, we must often demonstrate that our products offer not only superior health outcomes but also cost and service advantages, as compared with other forms of care. For example, while there is
8
no therapeutically substitutable generic alternative for Acthar Gel, it faces significant competition from earlier-line treatment alternatives including high-dose steroids, and is generally prescribed when earlier-line treatments have failed to provide positive outcomes or are not well tolerated by the patient.
We continue to differentiate Acthar Gel through pre-clinical studies and through product enhancements including the continued growth of SelfJect, which received FDA approval on March 1, 2024 and launched in the U.S. in August of 2024, and is designed to create an easier and more patient-friendly application for single-unit dosage indications. SelfJect requires less preparation with fewer materials and steps for the administration of Acthar Gel compared to the multi-dose vial and syringe. The latex-free device also has additional safety elements, including a hidden needle intended to help protect patients against needle sticks.
While there is no therapeutically substitutable non-surgical treatment alternative for Xiaflex, it faces significant competition from surgical intervention and other procedural approaches used to treat Dupuytren’s contracture and Peyronie’s disease. Xiaflex is differentiated as a non‑surgical treatment option, and is generally utilized in patients for whom a non‑surgical approach may be preferred due to considerations such as invasiveness, recovery time, site‑of‑care, or patient and physician treatment preferences.
We are also exposed to direct competition in the U.S. market for INOmax. However, we believe INOmax's highly differentiated service offering and the next generation delivery system will help to differentiate the product and mitigate the impact of competition longer-term.
The highly competitive environment in which we operate requires us to continually seek out new products to treat diseases and conditions in areas of high unmet medical need, to create technological innovations, and to market our products effectively. Most new products that we introduce must compete with other products already on the market, as well as other products that are subsequently developed by competitors. We may be granted market exclusivity either through the FDA, the U.S. Patent Office, or similar agencies internationally. Regulatory exclusivity is granted by the FDA for new innovations, such as new clinical data, a new chemical entity, or orphan drugs, and patents are issued for inventions, such as composition of matter or method of use. While patents offer a longer period of exclusivity, there are more bases to challenge patent-conferred exclusivity than with regulatory exclusivity. Generally, once market exclusivity expires, competition will likely intensify as generic forms of the product are launched. Products that do not benefit from regulatory or patent exclusivity must rely on other competitive advantages, such as confidentiality agreements or product formulation trade secrets for difficult to replicate products.
Branded products face pricing pressure when generic products are launched, as generic products do not carry the sales, marketing, R&D and product support costs borne by the innovators. These lower cost generics generally erode brand profitability over time. The generic form of a drug may also enjoy a preferred position relative to the branded version under third-party reimbursement programs, or be routinely dispensed in substitution for the branded form by pharmacies. If competitors introduce new products, delivery systems or processes with therapeutic or cost advantages, our products can be subject to progressive price reductions, decreased sales volume or both. To successfully compete for business with managed care and pharmacy benefits management organizations, we must often demonstrate that our branded products offer not only superior health outcomes but also cost advantages, as compared with other forms of care. Certain of our products are targeted for niche patient populations with unmet medical needs, for example Acthar Gel, that may not be prescribed unless a clear benefit in efficacy or safety is demonstrated or until alternatives have failed to provide positive patient outcomes or are not well tolerated by the patient.
Intellectual Property
We own or license a number of patents in the U.S. and other countries covering certain products and have also developed brand names and trademarks for those and other products. Generally, our business relies upon patent and trade-secret protection to protect our products, related inventions, and product innovations that are important to our business.
In a broad sense, patents provide the innovator companies with the right to exclude others from practicing an invention related to a product. Protection for individual products extends for varying periods in accordance with the expiration dates of patents in the various countries. The protection afforded, which may also vary from country to country, depends upon the type of patent, its scope of coverage, and the availability of meaningful legal remedies in the country. In the United States, certain patents relating to products that are the subject of an approved new drug application (“NDA”) are listed in the Orange Book. The Orange Book does not include a listing of patents related to biological products approved pursuant to a biologics license application (“BLA”). We may have other relevant regulatory protection or patents that may extend beyond the expiration dates provided below.
Intellectual property that relates to our major marketed products include:
•Acthar Gel. We have one Orange Book listed patent that relates to Acthar Gel. This patent is set to expire in 2041. We also have pending patent applications relating to the product. Acthar Gel is subject to complex and proprietary manufacturing processes, and is protected by trade secrets and proprietary know-how.
•Xiaflex. We own or have licensed rights to patents and patent applications related to Xiaflex worldwide, including U.S. drug product and methods of manufacture patents and patent applications that expire between 2028 and 2038. We also have pending patent applications related to the development of a potential PFI and PFA indication for Xiaflex and intend
9
to seek patent protection on a potential hammer toe indication. Xiaflex is also protected by trade secrets and proprietary know-how, with restricted access to the bacterial strain used to produce the enzymes.
•INOmax. We have a portfolio of patents and patent applications for INOmax and related technologies worldwide. The portfolio includes numerous U.S. issued patents, expiring between 2029 and 2041, and numerous pending U.S. patent applications.
In the U.S. branded pharmaceutical industry, an innovator product’s market exclusivity is generally determined by two forms of intellectual property: (i) patent rights held by the innovator company and listed in the Orange Book and (ii) any regulatory forms of exclusivity to which the innovator is entitled. In addition, commercial durability may also partially depend upon product-related trade secrets, confidentiality agreements, know-how, and trademark and copyright laws. These additional items may not prevent competitors from independently developing similar technology or a bioequivalent or biosimilar product.
Many developed countries provide certain non-patent incentives for the development of pharmaceuticals. Regulatory exclusivity is independent of any patent rights and can be particularly important when a drug lacks broad patent protection. However, many regulatory forms of exclusivity do not prevent a competitor from gaining regulatory approval prior to the expiration of regulatory exclusivity on the basis of the competitor's own safety and efficacy data on its drug, even when that drug is identical to that marketed by the innovator.
In addition to our patent estate and trade secret protections, we have rights to a number of trademarks and service marks, and pending trademark and service mark applications, in the U.S. and elsewhere in the world to further protect the proprietary position of our products.
We estimate the likely market exclusivity period for each of our products on a case-by-case basis. It is not possible to predict with certainty the length of market exclusivity for any of our products because of the complex interaction between patent and regulatory forms of exclusivity, the relative success or lack thereof by potential competitors' experience in product development and inherent uncertainties concerning patent litigation. There can be no assurance that a particular product will enjoy market exclusivity for the full period of time that we currently estimate or that the exclusivity will be limited to the estimate.
We consider the overall protection of our patents, trademarks and license rights to be of material value and act to protect these rights from infringement. For a discussion of the challenges we face in obtaining or maintaining patent and/or trade secret protection, see the risk factor captioned “We may be unable to protect our intellectual property rights, intellectual property rights may be limited or we may be subject to claims that we infringe on the intellectual property rights of others” included within Item 1A. Risk Factors of this Annual Report.
Regulatory Matters
Quality Assurance and Current Good Manufacturing Practice Requirements
The FDA enforces regulations to ensure that the methods used in, and the facilities and controls used for, the manufacture, processing, packaging and holding of drugs, biologics and medical devices conform to current good manufacturing practice (“cGMP”) or, for medical devices, the Quality System Regulation (“QSR”)/Quality Management System Regulation (“QMSR”).
The cGMP regulations that the FDA enforces are comprehensive and cover all aspects of manufacturing operations, from receipt of raw materials to finished product distribution, and are designed to ensure that the finished products meet all the required identity, strength, quality and purity characteristics. Compliance with cGMP includes adhering to requirements relating to organization and training of personnel, buildings and facilities, equipment, control of components and drug product containers and closures, production and process controls, quality control and quality assurance, packaging and labeling controls, holding and distribution, laboratory controls and records and reports. The QMSR is also comprehensive and covers all aspects of device manufacture, from product design to distribution and servicing, to post-market surveillance, insofar as they bear upon the safe and effective use of the device and whether the device otherwise meets the requirements of the U.S. Federal Food, Drug and Cosmetic Act (“FFDCA”). Combination drug medical device products may be subject to the applicable cGMP requirements as well as the QMSR requirements related to management responsibility, design and development, purchasing controls and complaint handling, among others. The QMSR, which replaces the QSR, is substantially similar to those set forth in the existing QSR although the QMSR has heightened emphasis on risk-based, lifecycle quality management and integration with business operations and leadership priorities. The FDA began to enforce the QMSR requirements on February 2, 2026.
Failure to comply with applicable cGMP/QMSR requirements or the conditions of the product’s approval or clearance may lead the FDA to take enforcement actions, such as issuing a warning letter, or to seek sanctions, including fines, civil penalties, injunctions, suspension of manufacturing operations, imposition of operating restrictions, withdrawal of FDA approval, seizure or recall of products and criminal prosecution. Although we periodically monitor FDA compliance of the third parties on which we rely for manufacturing certain of our products, we cannot be certain that our present or future third-party manufacturers will consistently comply with cGMP, QMSR, or other applicable FDA regulatory requirements. Other foreign regulatory authorities have their own good manufacturing practice and quality system requirements for drugs and medical devices, implemented through local laws and
10
regulations and enforced under each authority’s statutory framework. Efforts to ensure compliance require a continuous commitment of time, money and effort in all operational areas.
United States
In general, drug, medical device, and biological product manufacturers operate in a highly regulated environment. In the U.S., we must comply with laws, regulations and standards promulgated by a variety of regulatory agencies, including the FDA, the Department of Health and Human Services (“HHS”), the Drug Enforcement Administration (“DEA”), the Environmental Protection Agency (“EPA”), the Customs Service, and state boards of pharmacy.
In the U.S., the FDA regulates drug, medical device, and biological products (collectively referred to as “pharmaceutical products,” “drug products,” or “products”) under the FFDCA. Biological products are also regulated under the Public Health Service Act (“PHSA”).
The FFDCA provides several distinct pathways for the approval of new drugs. Under current law, drug applications are required to demonstrate safety and efficacy of a new drug. Prior to 1962, FDA only required that drugs were shown to be safe. Drugs approved only for safety between 1938 and 1962, were later evaluated for effectiveness by FDA under a process entitled the Drug Efficacy Study Implementation (“DESI”). Today, a drug may be approved under one of three sections of the FFDCA. An NDA under Section 505(b)(1) of the FFDCA is a comprehensive application to support approval of a product candidate that includes, among other things, data and information to demonstrate that the proposed drug is safe and effective for its proposed uses, that production methods are adequate to ensure the identity, strength, quality, and purity of the drug, and that proposed labeling is appropriate and contains all necessary information. A 505(b)(1) NDA generally contains results of the full set of pre-clinical studies and clinical trials conducted by or on behalf of the applicant to characterize and evaluate the product candidate. Section 505(b)(2) of the FFDCA provides an alternate regulatory pathway to obtain FDA approval; it permits the filing of an NDA where at least some of the information required for approval comes from studies not conducted by or for the applicant and for which the applicant has not obtained a right of reference. The applicant may rely to some extent upon the FDA’s findings of safety and effectiveness for an approved product that acts as the reference drug, and submit its own product-specific data – which may include data from pre-clinical studies or clinical trials conducted by or on behalf of the applicant – to address differences between the product candidate and the reference drug. Drug manufacturers may also submit an abbreviated new drug application (“ANDA”) under section 505(j) of the FFDCA to market a generic version of an approved branded drug product. The ANDA must show that the generic version is “therapeutically equivalent,” or expected to have the same clinical effect and safety profile as the branded drug product when administered to patients under the conditions specified in the labeling.
The FDA uses several different pathways for non-exempt medical devices to obtain market authorization. Specifically, to market and sell a new medical device in the U.S., the manufacturer generally must follow one of three paths, which are described under Medical Devices below.
Certain of our products are or could become regulated and marketed as biologic products pursuant to a BLA. Our BLA-licensed product was licensed based on a determination by the FDA of safety, purity and potency as required under the PHSA. Under the Biologics Price Competition and Innovation Act of 2009 (the “BPCIA”), manufacturers can pursue approval of a biological product using an abbreviated licensure pathway (submitted pursuant to section 351(k) of the PHSA). Under the PHSA, as amended by the BPCIA, following the expiration of a 12-year reference exclusivity period granted to certain referenced FDA-licensed biologic products, the FDA may license under section 351(k) of the PHSA a biological product that it determines is biosimilar to, or interchangeable with, a reference product licensed under section 351(a) of the PHSA. FDA deems a proposed product as biosimilar to an FDA-licensed biological reference product if it is “highly similar” to the reference product and there are no clinically meaningful differences in terms of safety, purity and potency. A proposed biosimilar product must have the same route of administration, dosage form, strength and mechanism of action (if known) as the reference product, and must be proposed only for uses for which the reference product is approved. Although licensure of biosimilar or interchangeable products is generally expected to require less than the full complement of product-specific preclinical and clinical data required for innovator products, the FDA has considerable discretion over the kind and amount of scientific evidence required to demonstrate biosimilarity and interchangeability.
The FDA and other federal regulatory agencies also closely regulate the marketing and promotion of pharmaceutical and medical device products sold in the U.S. through, among other things, standards and regulations for direct-to-consumer advertising, communications regarding unapproved uses, industry-sponsored scientific and educational activities and promotional activities, including those involving the Internet. A pharmaceutical or medical device product cannot be commercially promoted before it receives marketing authorizations through approval or clearance. After approval or clearance, product promotion can include only those claims relating to safety and effectiveness that are consistent with the labeling approved by the FDA. Healthcare providers are permitted to prescribe drugs or medical devices for “off-label” uses - that is, uses not approved by the FDA and therefore not described in the drug or medical device’s labeling - because the FDA does not regulate the practice of medicine. However, FDA regulations impose stringent restrictions on manufacturers’ communications regarding off-label uses. In general, a manufacturer may not promote a drug or medical device for off-label use, but may engage in non-promotional, balanced communication regarding off-label use under specified conditions. Failure to comply with applicable FDA requirements and restrictions in this area may subject a company to adverse publicity and enforcement action by the FDA, the U.S. Department of Justice (“DOJ”) or the Office of the
11
Inspector General (“OIG”) within the HHS, as well as state authorities. Enforcement action could subject a company to a range of penalties that could have a significant commercial impact, including civil and criminal penalties and agreements that materially restrict the manner in which a company promotes or distributes drug or medical device products.
In addition, the manufacture, marketing, and selling of certain drug products that are controlled substances may be limited by quota grants and other requirements or restrictions enforced by the DEA. Refer to “Drug Enforcement Administration” within this Item 1. Business - Our Business and Products for further information.
The path leading to FDA approval of a marketing application for a new pharmaceutical product begins when the product is merely a chemical formulation in the laboratory. In general, the process involves the following steps:
•Completion of formulation and laboratory testing that fully characterizes the drug product from a pre-clinical perspective and provides preliminary evidence that the drug product is safe to test in human beings, conducted in accordance with good laboratory practices (“GLP”) and applicable requirements for the humane use of laboratory animals or other applicable regulations;
•Filing an investigational new drug (“IND”) application with the FDA, which must become effective before the conduct of clinical trials (testing in human beings under adequate and well-controlled conditions);
•Approval by an independent institutional review board (“IRB”) or ethics committee representing each clinical trial site before each trial may be initiated;
•Designing and conducting adequate and well-controlled human clinical trials to show the safety and efficacy of the product candidate for the proposed indication in accordance with the applicable IND and other clinical trial-related regulations, sometimes collectively referred to as good clinical practice (“GCP”);
•Submitting the marketing application for FDA review, which provides a complete characterization of the product;
•Determination by the FDA within 60 days of its receipt of a marketing application to accept and file the application for review;
•Satisfactory completion of potential FDA pre-approval inspections of the designated facility or facilities where the product is produced to assess compliance with cGMP requirements;
•Potential FDA audit of the non-clinical and/or clinical trial sites that generated the data in support of the marketing application;
•Payment of applicable user fees;
•If applicable, satisfactory completion of an FDA Advisory Committee meeting in which the FDA requests views from outside experts in evaluating the application;
•FDA approval of the application, including prescribing information, labeling and packaging of the drug product; and
•Implementation of a risk evaluation and mitigation strategy (“REMS”) program, if applicable, and conduct of any required Phase 4 studies and compliance with post-approval requirements, including ongoing monitoring and reporting of adverse events related to the product.
•Clinical Trials. Clinical trials are typically conducted in four sequential phases, although they may overlap. The four phases are as follows:
•Phase 1. Phase 1 includes the initial introduction of an investigational product candidate into humans. Phase 1 trials generally are conducted in healthy volunteers but in some cases are conducted in patients with the target disease or condition. These trials are designed to evaluate the safety, metabolism, pharmacokinetic properties and pharmacologic actions of the investigational product candidate in humans, the side effects associated with increasing doses, and if possible, to gain early evidence on effectiveness. During Phase 1 trials, sufficient information about the investigational product candidate’s pharmacokinetic properties and pharmacological effects may be obtained to permit the design of Phase 2 trials. The total number of participants included in Phase 1 trials varies, but is generally in the range of 20 to 80.
•Phase 2. Phase 2 includes the controlled clinical trials conducted in patients with the target disease or condition, to determine dosage tolerance and optimal dosage, identify possible adverse side effects and safety risks associated with the product candidate and obtain initial evidence of the effectiveness of the investigational product candidate for a particular indication. Phase 2 trials are typically well-controlled, closely monitored and conducted in a limited subject population, usually involving no more than several hundred participants.
12
•Phase 3. Phase 3 trials are controlled clinical trials conducted in an expanded subject population at geographically dispersed clinical trial sites. They are performed after preliminary evidence suggesting effectiveness of the investigational product candidate has been obtained, and are intended to further evaluate dosage, clinical effectiveness and safety, to establish the overall benefit-risk relationship of the product candidate, and to provide an adequate basis for drug approval. Phase 3 trials usually involve several hundred to several thousand participants. In most cases, the FDA requires two adequate and well controlled Phase 3 trials to demonstrate the efficacy and safety of the drug; however, the FDA may find a single Phase 2 or Phase 3 trial with other confirmatory evidence to be sufficient in rare instances, particularly in an area of significant unmet medical need and if the trial design provides a well-controlled and reliable assessment of clinical benefit.
•Phase 4. In some cases, the FDA may condition approval of an NDA, BLA, or supplement to such an application on the sponsor’s agreement to conduct additional clinical trials after approval. In other cases, a sponsor may voluntarily conduct additional clinical trials after approval to gain more information about the product. Such post-approval trials are typically referred to as Phase 4 clinical trials.
Clinical trials may not be completed successfully within a specified period of time, if at all. The decision to terminate development of an investigational product candidate may be made by a health authority (such as the FDA), an IRB/ethics committee, or by a company for various reasons. At any time, the FDA may order the temporary or permanent discontinuation of a clinical trial, which is referred to as a clinical hold, or impose other sanctions, if the agency believes the clinical trial is not being conducted in accordance with FDA requirements or presents an unacceptable risk to the clinical trial patients. The suspension or termination of development can occur during any phase of clinical trials if it is determined that the participants or subjects are being exposed to an unacceptable health risk. In addition, there are requirements for the registration of ongoing clinical trials of product candidates on public registries and the disclosure of certain clinical trial results and other trial information after completion.
Assuming successful completion of all required testing in accordance with all applicable regulatory requirements, detailed investigational product candidate information is submitted to the FDA in the form of a marketing application to request market approval or clearance for the product in specified indications.
Marketing Applications. In order to obtain approval to market a drug in the U.S., a marketing application (i.e., an NDA or BLA) must be submitted to the FDA that provides data establishing the safety and effectiveness of the product candidate for the proposed indication. The application includes all relevant data available from pertinent preclinical studies and clinical trials, including negative or ambiguous results as well as positive findings, together with detailed information relating to the product’s chemistry, manufacturing, controls and proposed labeling, among other things. Data can come from company-sponsored clinical trials intended to test the safety and effectiveness of a product or from a number of alternative sources, including studies initiated by investigators. To support marketing approval, the data submitted must be sufficient in quality and quantity to establish the safety and effectiveness of the investigational product candidate to the satisfaction of the FDA.
Under the Prescription Drug User Fee Act, the FDA has the authority to collect fees from drug manufacturers who submit pharmaceutical marketing applications for review and approval, although there may be some instances in which the user fee is waived. These user fees help the FDA fund the drug approval process. For the federal fiscal year 2026, the user fee rate has been set at approximately $4.7 million for a marketing application requiring clinical data (which would be most NDAs and BLAs), and approximately $2.3 million for an application not requiring clinical data (such as an ANDA and some 351(k) biosimilar applications). No user fees are assessed on marketing applications for products designated as orphan drugs, unless the product also includes a non-orphan-designated indication. Similarly, under the Medical Device User Fee Act, the FDA has the authority to collect fees from manufacturers who submit applications for clearance or approval. For fiscal year 2026, the user fee rate for 510(k) notifications is approximately $26,000, for premarket approval applications (“PMAs”) is approximately $579,000, and for De Novo classification requests is approximately $174,000. We expense these fees as they are incurred.
Once an NDA, BLA or supplement has been compiled and submitted, FDA performs an initial review before it accepts the application for filing. FDA may refuse to file an application and/or request additional information before acceptance. Once accepted for filing, FDA begins an in-depth review of the application. The FDA has agreed to certain performance goals in the review of applications. The FDA does not always meet these goal dates, and in certain circumstances, the goal date may be extended. The FDA reviews the application to determine, among other things, whether the proposed product is safe and effective for its intended use, and whether the product is being manufactured in accordance with cGMP. The FDA may also refer an application to an advisory committee, typically a panel of clinicians, for review, evaluation and a non-binding recommendation as to whether the application should be approved. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions. A similar process exists for the review of medical device marketing applications though the rigor and timelines vary depending upon the risk level of the medical device and the specific pathway.
Before approving an application, the FDA often will inspect the facilities at which the product is manufactured for cGMP compliance, and may inspect one or more clinical sites to assure compliance with GCP. After it evaluates the application and the results of inspections, the FDA issues either an approval letter or a complete response letter. A complete response letter generally outlines the deficiencies in the submission and may require substantial additional testing or information in order for the FDA to
13
reconsider the application. If those deficiencies are addressed to the FDA’s satisfaction in a resubmission of the marketing application, the FDA will issue an approval letter. Notwithstanding the submission of any requested additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval.
An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition of approval, the FDA may require a REMS to help ensure that the benefits of the drug outweigh the potential risks. A REMS can include a medication guide, communication plan for healthcare professionals, and elements to assure safe use (“ETASU”). ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patient registries. The requirement for a REMS can materially affect the potential market and profitability of the drug. Moreover, product approval may require substantial post-approval testing and surveillance to monitor the drug’s safety or efficacy. Once granted, product approvals may be withdrawn if compliance with regulatory requirements is not maintained or problems are identified following initial marketing.
Changes to some of the conditions established in an approved application, including changes in indications, labeling, or manufacturing processes or facilities, may require submission and prior FDA approval of a supplemental application (or, in some cases, a new application) before the change can be implemented. A supplemental application for a new indication typically requires clinical data similar to that in the original application, and the FDA uses the same procedures and actions in reviewing supplements as it does in reviewing original marketing applications. Likewise, certain changes to a medical device approved under a PMA will require that a supplemental PMA be submitted or, in the case of a 510(k), an entirely new 510(k) submission.
Expedited Programs. The FDA maintains certain expedited programs to facilitate the development and review processes for certain qualifying pharmaceutical product candidates, including fast track designation, breakthrough therapy designation, priority review, accelerated approval, regenerative medicine advanced therapy designation, and medical devices under a Breakthrough Device Designation. A pharmaceutical product candidate may be granted fast track designation if it is intended for the treatment of a serious or life-threatening condition and demonstrates the potential to address unmet medical needs for such condition. With fast track designation, the sponsor may be eligible for more frequent opportunities to obtain the FDA’s feedback, and the FDA may initiate review of sections of an application before the application is complete. This rolling review is available if the applicant provides and the FDA approves a schedule for the remaining information. Even if a product receives fast track designation, the designation can be rescinded and provides no assurance that a product will be reviewed or approved more expeditiously than would otherwise have been the case, or that the product will be approved at all.
The FDA may designate a product candidate as a breakthrough therapy if it finds that the product candidate is intended, alone or in combination with one or more other product candidates or approved products, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the product candidate may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints. For product candidates designated as breakthrough therapies, more frequent interaction and communication between the FDA and the sponsor can help to identify the most efficient path for clinical development. Product candidates designated as breakthrough therapies by the FDA may also be eligible for priority review. Even if a product receives Breakthrough Therapy Designation, the designation can be rescinded and provides no assurance that a product will be reviewed or approved more expeditiously than would otherwise have been the case, or that the product will be approved at all.
Accelerated approval under FDA regulations allows a product designed to treat a serious or life-threatening disease or condition that provides a meaningful therapeutic advantage over available therapies to be approved on the basis of either an intermediate clinical endpoint or a surrogate endpoint that is reasonably likely to predict clinical benefit. Approvals of this kind typically include requirements for confirmatory clinical trials to be conducted with due diligence to validate the surrogate endpoint or otherwise confirm clinical benefit and for all promotional materials to be submitted to the FDA for review prior to dissemination.
The FDA may also grant priority review designation to a product candidate, which sets the target date for FDA action on the application at six months from FDA filing, or eight months from the sponsor’s submission. Priority review may be granted where a product is intended to treat a serious or life-threatening disease or condition and, if approved, has the potential to provide a safe and effective therapy where no satisfactory alternative therapy exists or a significant improvement in safety or efficacy compared to available therapy. If criteria are not met for priority review, the standard FDA review period is ten months from FDA filing or 12 months from sponsor submission. Priority review designation does not change the scientific/medical standard for approval or the quality of evidence necessary to support approval.
Orphan Drug Designation. Under the Orphan Drug Act, the FDA may grant orphan designation to a drug product intended to treat a “rare disease or condition,” which is generally a disease or condition that affects fewer than 200,000 individuals in the U.S., or more than 200,000 individuals in the U.S., but for which there is no reasonable expectation that the cost of developing and making a drug product available in the U.S. for this type of disease or condition will be recovered from sales of the product. If orphan product designation is sought, it must be requested before submitting a marketing application for the drug for the proposed rare disease or condition. If the FDA grants orphan drug designation, the common name of the therapeutic agent and its designated orphan use are disclosed publicly by the FDA. Orphan product designation does not, by itself, convey any advantage in or shorten the duration of the regulatory review and approval process.
14
If a product that has orphan designation subsequently receives the first FDA approval for the disease or condition for which it has such designation, the product is entitled to orphan product exclusivity, which the FDA has interpreted to preclude approving for seven years any other sponsor’s application to market the same drug for the same use for which the drug has been granted orphan drug designation, except in limited circumstances, such as a showing that the subsequent product is clinically superior to the product with orphan exclusivity. Orphan exclusivity operates independently from other regulatory exclusivities and other protection against generic competition, including patents that we hold for our products, and applies even if the competing product is supported by its own data. A sponsor of a product application that has received an orphan drug designation generally is exempt from the $4.7 million application fee and may qualify for tax incentives for clinical research undertaken to support the application.
Orphan drug exclusivity does not block approval of competing products intended for the orphan-protected indication but containing a different active moiety, or containing the same moiety but intended for a different use. Orphan product exclusivity that could block a competitor to one of our products also could block the approval of one of our products for seven years if a competitor obtains approval of a product containing the same moiety for the same orphan disease or condition.
Marketing Exclusivity. Upon NDA approval of a new chemical entity, which is a drug substance that contains no active moiety that has previously been approved by the FDA in any other NDA, that drug receives five years of marketing exclusivity during which the FDA cannot accept for review any ANDA or 505(b)(2) NDA for which the new chemical entity is a reference product. (An application that contains a challenge to a patent associated with the reference product may be submitted at four years after reference product approval.) There are provisions that operate to preclude approval of the application for an additional period of time after submission. Certain changes to an approved drug, such as the approval of a new indication, may qualify for a three-year period of exclusivity during which the FDA cannot approve an ANDA or 505(b)(2) NDA for a similar drug that includes the change.
Biologic products approved under a BLA also may benefit from a period of exclusivity that protects a reference product from competition from biosimilars. FDA may not accept a biosimilar application for review until four years after the date of first licensure of the reference product, and the biosimilar cannot be licensed until 12 years after the reference product was first licensed.
Pediatric Exclusivity. Under certain circumstances, if the sponsor submits pediatric data that fairly respond to a written request from FDA for such data, the exclusivity periods applicable to drugs and biologics and the patent-related protections applicable to drugs may be extended by six months. This extension may be granted even if the data does not support a pediatric indication.
Patent Term Restoration. A portion of the patent term lost during product development and FDA review of an application is restored if approval of the application is the first permitted commercial marketing of a drug containing the active ingredient. The patent term restoration period is generally one-half the time between the effective date of the IND or the date of patent grant (whichever is later) and the date of submission of the application, plus the time between the date of submission of the application and the date of FDA approval of the product. The maximum period of restoration is five years, and the patent cannot be extended to more than 14 years from the date of FDA approval of the product. Only one patent claiming an approved product or a method of using or manufacturing it is eligible for restoration and the patent holder must apply to the U.S. Patent and Trademark Office (“USPTO”) for restoration within 60 days of FDA approval. The USPTO, in consultation with the FDA, reviews and approves the application for patent term restoration.
Post-Approval Regulation. After regulatory approval of a drug is obtained, a sponsor is required to comply with a number of post-approval requirements. For example, as a condition of approval of an application, the FDA may require post-marketing testing, including Phase 4 clinical trials, and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization. In addition, as a holder of an approved NDA, a sponsor is required to report adverse reactions and production problems to the FDA, provide updated safety and efficacy information, submit annual reports and comply with advertising and promotional labeling requirements. Manufacturing must continue to conform to cGMP after approval, and the FDA periodically inspects manufacturing facilities to assess compliance with cGMP, as discussed in Quality Assurance and Current Good Manufacturing Practice Requirements above.
The distribution of prescription pharmaceutical products is subject to the Prescription Drug Marketing Act, which regulates the distribution of samples at the federal level and sets minimum standards for the registration and regulation of drug distributors by the states. Moreover, the Drug Supply Chain Security Act imposes requirements on manufacturers and their trading partners related to identifying and tracing prescription drug products distributed in the U.S. to ensure accountability in distribution and to identify, trace and remove counterfeit and other illegitimate or harmful drugs from the market.
Newly discovered or developed safety or effectiveness data may require changes to a product’s approved labeling, including the addition of new warnings, contraindications, or limitations of use, and also may require the implementation of other risk management measures, including the development and implementation of a REMS. Product approvals may be withdrawn if compliance with regulatory requirements is not maintained or if problems concerning safety or efficacy occur following approval. Also, new government requirements, including those resulting from new legislation, may be established, or the FDA’s policies may change, which could impact our approved products or delay or prevent regulatory approval of our products under development.
Abbreviated New Drug Application (ANDA) Process. The path leading to FDA approval of generic drug product under an ANDA is different from that of an NDA, a biologics license application or even a biosimilar. Generally, a generic drug product is one that is
15
the same as an approved “brand” or “innovator” drug product (the reference listed drug or “RLD”) in active ingredient, dosage form, strength, and route of administration, bioequivalent to the RLD, and labeled the same as the RLD. Generic drug applications are termed “abbreviated” because they are not required to include data to establish safety and effectiveness, instead relying on demonstrating the sameness to the RLD, which FDA has already found to be safe and effective.
Medical Devices. There are three primary pathways to receive authorization to distribute a new device in the U.S. The first pathway is pre-market notification or the 510(k) process. Under this pathway, the manufacturer must demonstrate to the FDA that the new device is substantially equivalent to a legally marketed predicate device, generally by showing it has the same intended use and substantially similar technological characteristics or that any technological differences do not raise new questions of safety or effectiveness. The FDA will make a determination as to whether the new device is substantially equivalent to the predicate device before commercial distribution occurs. Changes that do not significantly affect the safety or efficacy of a legally marketed device may generally be made without additional 510(k) premarket notifications.
The second primary pathway is a premarket approval application (“PMA”). This pathway is generally more complex, time-consuming and expensive than the 510(k) process. Under the PMA pathway, the manufacturer must demonstrate that the device is safe and effective for its intended use. This generally requires data from clinical trials, which must be performed in accordance with the applicable Investigational Device Exemption regulations. PMA review may also include manufacturing and clinical inspections, advisory panel input, and post approval requirements.
Third, novel low to moderate risk devices that lack a predicate are by default subject to the PMA pathway. However, the manufacturer may instead request a risk-based classification through the De Novo process. If granted, the device receives marketing authorization and establishes a new device type that may serve as a future predicate. Regardless of pathway, after devices receive marketing authorization, they are subject to FDA and state requirements, including establishment registration, device listing, manufacturer/distributor licensing, quality system obligations, adverse event reporting, and reporting of certain corrections and removals, along with periodic inspections.
Patent Period. A sponsor of an NDA is required to identify in its application any patent that claims the drug or a use of the drug subject to the application. Upon NDA approval, the FDA lists these patents in the Orange Book. Any person that files a Section 505(b)(2) NDA, the type of NDA that relies upon the data in the application for which the patents are listed, or an ANDA to secure approval of a generic version of a previous drug, must make a certification in respect to listed patents. The FDA may not approve such an application for the drug until expiration of the listed patents unless the generic applicant certifies that the listed patents are invalid, unenforceable or not infringed by the proposed generic drug and gives notice to the holder of the NDA for the RLD of the bases upon which the patents are challenged, and the holder of the RLD does not sue the later applicant for patent infringement within 45 days of receipt of notice. If an infringement suit is filed, the FDA may not approve the later application until the earliest of: (a) 30 months after receipt of the notice by the holder of the NDA for the RLD; (b) entry of an appellate court judgment holding the patent invalid, unenforceable or not infringed; (c) such time as the court may order; or (d) the expiration of the patent.
One of the key motivators for challenging patents is the 180-day market exclusivity period (“generic exclusivity”) granted to the developer of a generic version of a product that is the first to file an ANDA containing a Paragraph IV certification and that prevails in litigation with the manufacturer of the branded product over the applicable patent(s) or is not sued or enters into a settlement agreement with the manufacturer of the branded product. For a variety of reasons, there are situations in which a company may not be able to take advantage of an award of generic exclusivity. The determination of when generic exclusivity begins and ends is very complicated as it depends on several different factors.
Risk Evaluation and Mitigation Strategies. The FDA has the authority to require a pharmaceutical manufacturer to develop and implement a REMS for certain products if the agency finds that a REMS is necessary to ensure that the benefits of a drug outweigh the risks. Failure to comply with REMS requirements could result in enforcement action such as product seizure, injunction or civil money penalties. Certain of our products are subject to REMS, namely, the Opioid Analgesic REMS, Aveed® REMS, and XIAFLEX REMS, and others may be subject to REMS programs in the future.
Drug Enforcement Administration. The DEA is the U.S. federal agency responsible for domestic enforcement of the federal Controlled Substances Act of 1970 (“CSA”). A drug product approved by FDA may be subject to scheduling as a controlled substance under the CSA. Compounds that have a potential for dependence and abuse are scheduled as controlled substances under the CSA and similar state and foreign laws based on the drug’s potential for abuse. Controlled substances that are pharmaceutical products are subject to a high degree of regulation under the CSA, which establishes, among other things, certain registration, manufacturing quotas, security, recordkeeping, reporting, import, export, inspection and other requirements administered by the DEA.
The DEA classifies controlled substances into five schedules. Schedule I substances by definition have a high potential for abuse, have no currently “accepted medical use” in the U.S., lack accepted safety for use under medical supervision, and may not be prescribed, marketed or sold in the U.S. Pharmaceutical products approved for use in the U.S. may be classified as Schedule II, III, IV or V, with Schedule II substances considered to present the highest potential for abuse or dependence and Schedule V substances the lowest relative risk of abuse. Percocet, and the active pharmaceutical ingredient (“API”) of Percocet, oxycodone, (in addition to acetaminophen), are regulated by the DEA as a Schedule II controlled substance. In addition, our branded testosterone drug products, Aveed, Testim®, and Testopel®, are regulated by the DEA as Schedule III controlled substances.
16
The DEA limits the quantity of certain Schedule I and II controlled substances that may be manufactured and procured in the U.S. in any given calendar year through a quota system and, as a result, DEA quotas to manufacture and procure oxycodone in the U.S. are needed. Given that the DEA has discretion to grant or deny quota requests, the quota the DEA grants may be insufficient to meet our business needs. Furthermore, our scheduled drug products are subject to DEA and state regulations relating to the importation, manufacturing, storage, distribution and physician prescription procedures, including limitations on prescription refills. In addition, the third parties who perform certain activities related to our products that contain controlled substances, including for Percocet, are required to maintain necessary DEA registrations and state licenses and comply with federal and state controlled substance requirements. The DEA periodically inspects facilities for compliance with its rules and regulations. For all controlled substances, there are potential criminal and civil penalties that apply for the failure to meet applicable legal requirements, and, in general, healthcare professionals may be required to have a federal and/or state license in order to handle, prescribe or dispense controlled substances.
Government Benefit Programs. Statutory and regulatory requirements for Medicaid, Medicare, Tricare and other government healthcare programs govern provider reimbursement levels, as well as require that each pharmaceutical manufacturer that participates in the Medicaid Drug Rebate Program pay rebates to individual states based on their Medicaid program-reimbursed products utilization. The federal and state governments may continue to enact measures in the future aimed at containing or reducing payment levels for, or increasing rebates on, prescription pharmaceuticals paid for in whole or in part with government funds. We cannot predict the nature of such measures, which could have material adverse consequences for the pharmaceutical industry as a whole and, consequently, also for us. However, we believe we have provided for our best estimate of potential rebates based on current information available.
The Centers for Medicare & Medicaid Services (“CMS”), the agency that administers the Medicare and Medicaid programs, may implement or revise reimbursement or coverage restrictions under those programs, and a state may do likewise under the Medicaid program. Any reduction in reimbursement or restriction of coverage under Medicare, Medicaid or other government programs may result in a similar reduction in payments or restriction of coverage by private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability or commercialize our products.
From time to time, legislative changes are made to government healthcare programs that impact our business. For example, the Medicare Prescription Drug Improvement and Modernization Act of 2003 created a prescription drug coverage program for people with Medicare through a system of government-regulated private market drug benefit plans. This law provides a prescription drug benefit to seniors and individuals with disabilities in the Medicare program (“Medicare Part D”). The U.S. Congress continues to examine various Medicare policy proposals that may result in pressure on the prices of prescription drugs in the Medicare program.
In addition, the Affordable Care Act provided for major changes to the U.S. healthcare system, which impacted the delivery and payment for healthcare services in the U.S. Our business has been impacted by, among other things, changes to the rebates under the Medicaid Fee-For-Service Program and new rebates on Medicaid Managed Care utilization and the imposition of an annual fee on branded prescription pharmaceutical manufacturers. Medicaid provisions reduced net sales by $68.6 million, $51.6 million, $15.8 million, and $110.6 million for the years ended December 31, 2025, December 27, 2024, the period November 15, 2023 through December 29, 2023 (Successor), and the period December 31, 2022 through November 14, 2023 (Predecessor), respectively. Our business was also impacted by the annual fee on branded prescription pharmaceutical manufacturers, which is reflected within selling, general and administrative expenses (“SG&A”). During the years ended December 31, 2025 and December 27, 2024 (Successor), the period November 15, 2023 through December 29, 2023 (Successor), and the period December 31, 2022 through November 14, 2023 (Predecessor), we recorded an expense of $6.0 million, $2.3 million, $0.4 million, and $3.5 million, respectively.
The Affordable Care Act also established a Medicare Part D coverage gap discount program, under which manufacturers must agree to offer point-of-sale discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during the coverage gap period, as a condition for the manufacturer’s outpatient drugs to be covered under Medicare Part D.
On August 16, 2022, President Biden signed into law the Inflation Reduction Act of 2022 (“Inflation Reduction Act”) which, among other things, established Medicare Part B and Part D inflation rebate schemes, under which, generally speaking, manufacturers will owe rebates if the average sales price of a Part B drug or the average manufacturer price of a Part D drug increases faster than the pace of inflation. Failure to timely pay an inflation rebate is subject to a civil monetary penalty. The Inflation Reduction Act also created a drug price negotiation program under which the prices for Medicare units of certain high Medicare spend drugs and biologics without generic or biosimilar competition will be capped by reference to, among other things, a specified non-federal average manufacturer price, starting in 2026. Failure to comply with requirements under the drug price negotiation program is subject to an excise tax and/or a civil monetary penalty. The Inflation Reduction Act further made changes to the Medicare Part D benefit, including sunsetting the coverage gap discount program and replacing it with a new manufacturer discount program in 2025, pursuant to which manufacturers of applicable brand drugs are required to provide mandatory discounts on Part D drugs dispensed to Medicare beneficiaries in both the initial coverage phase and the catastrophic phase of the benefit. Failure to offer discounts under this program could be subject to civil monetary penalties. Under the Inflation Reduction Act, certain drug products may be eligible for a small biotech exemption based on specified thresholds around Medicare program spending. Products with this designation are exempt from the drug price negotiation program in 2026, 2027, and 2028. Similar phase-ins are available regarding manufacturer discount liability
17
for the Medicare Part D benefit redesign over a number of years. The U.S. Congress continues to examine various policy proposals that may result in pressure on the prices of prescription drugs in the government health benefit programs. The Inflation Reduction Act or other legislative changes could impact the market conditions for our product candidates.
On July 4, 2025, the “One Big Beautiful Bill Act,” or OBBBA, was signed into law. Changes under the OBBBA to ACA marketplace enrollment are projected to decrease the number of individuals with marketplace coverage. The OBBBA is also projected to decrease federal health care spending by approximately $1 trillion by reducing Medicaid spending and enrollment and making changes to federal Medicare spending. It is unclear if these changes will impact demand for our products.
As discussed above, we anticipate that the U.S. Congress, state legislatures, and federal and state regulators may adopt or accelerate adoption of new healthcare policies and reforms intended to regulate drug pricing or the way in which such prices are made available on the market. This includes efforts by individual states in the United States to pass legislation and implement regulations designed to control pharmaceutical and biological product pricing, such as by passing laws that regulate how manufacturers make the 340B Drug Pricing Program (“340B program”) ceiling price (“340B ceiling price”) available on the market, and/or establishing Prescription Drug Affordability Boards (or similar entities) that may review high-cost drugs, set upper payment limits, and implement marketing cost disclosure and transparency measures.
Pharmaceutical Pricing and Reimbursement. Certain of our affiliates that are manufacturers participate in the Medicaid Drug Rebate Program and other governmental programs. Each manufacturer that participates in the Medicaid Drug Rebate Program is required to pay a rebate to each state Medicaid program for its covered outpatient drugs that are dispensed to Medicaid beneficiaries and paid for by a state Medicaid program, as a condition of having federal funds available for that manufacturer’s drugs under Medicaid and Medicare Part B. Those rebates are based on pricing data the manufacturer reports on a monthly and quarterly basis to CMS, the federal agency that administers the Medicare and Medicaid programs. The data include the average manufacturer price and, in the case of innovator products, the best price for each drug, which best price, in general, represents the lowest price available from the manufacturer to any wholesaler, retailer, provider, health maintenance organization, nonprofit entity, or governmental entity in the U.S. in any pricing structure, calculated to include all sales and associated rebates, discounts, and other price concessions. Where the average manufacturer price of a drug increases faster than the pace of inflation, the drug may be subject to an additional rebate paid by its manufacturer in the amount that the average manufacturer price has exceeded the pace of inflation. The Medicaid rebate is no longer subject to a cap effective January 1, 2024, which caused our rebate per unit to increase significantly for certain products in 2024, notably for Acthar Gel, and further increases continue to be applied. In December 2020, CMS issued a final rule that modified prior Medicaid Drug Rebate Program regulations to permit reporting multiple best price figures with regard to value‑based purchasing arrangements (beginning in 2022); and provide definitions for “line extension,” “new formulation,” and related terms, with the practical effect of expanding the scope of drugs considered to be line extensions that are subject to an alternative rebate formula (beginning in 2022). While the regulatory modifications that purported to affect the applicability of the best price and average manufacturer price exclusions of manufacturer-sponsored patient benefit programs, in the context of PBM “accumulator” programs, were invalidated by a court and rescinded, such programs may continue to negatively affect us in other ways. A manufacturer’s failure to comply with these price reporting and rebate payment options could negatively impact its financial results. CMS periodically issues updates to its regulations under the Medicaid Drug Rebate Program that could impact our price reporting obligations in various ways. For example, in September 2024, CMS further modified the regulations governing the Medicaid Drug Rebate Program, which could increase manufacturer costs and the complexity of compliance, impact rebate liabilities, and be time-consuming to implement.
The U.S. Congress also could enact additional changes that affect overall rebate liability and the information manufacturers report to the government as part of price reporting calculations, which could impact the market conditions for our products. We further expect continued scrutiny on government price reporting and pricing more generally from the U.S. Congress, federal or state agencies, and other bodies, and are seeing an increase in state interest in price reporting, transparency, and other policies to address drug pricing concerns. For additional information about the risk associated with these programs, please see “Our reporting and payment obligations under the Medicare and Medicaid rebate programs, and other governmental purchasing and rebate programs, are complex. Any determination of failure to comply with these obligations or those relating to healthcare fraud and abuse laws could have a material adverse effect on our business” included within Item 1A. Risk Factors of this Annual Report.
Federal law requires that each manufacturer that participates in the Medicaid Drug Rebate Program also participate in the 340B program in order for federal funds to be available for the manufacturer’s drugs under Medicaid and Medicare Part B. The 340B program, which is administered by the Health Resources and Services Administration (“HRSA”), requires participating manufacturers to agree to offer statutorily defined covered entities no more than the 340B ceiling price for the manufacturer’s covered outpatient drugs. These 340B program-covered entities include a variety of community health clinics and other entities that receive health services grants from the Public Health Service, as well as hospitals that serve a disproportionate share of low-income patients, certain free-standing cancer hospitals, critical access hospitals, rural referral centers and sole community hospitals. The Affordable Care Act exempts “orphan drugs” from the ceiling price requirements for certain hospital covered entities. The 340B ceiling price is calculated using a statutory formula, which is based on the average manufacturer price and rebate amount for the covered outpatient drug as calculated under the Medicaid Drug Rebate Program, and, in general, products subject to Medicaid price reporting and rebate liability are also subject to the 340B ceiling price calculation and discount requirement. Where a drug is subject to an additional rebate, as noted previously, or a low best price, the 340B ceiling price may calculate as low as, but not lower than, $0.01 per unit. Changes to the
18
Medicaid Drug Rebate amount also could affect a manufacturer’s 340B ceiling price calculations and negatively impact results of operations.
HRSA issued a final regulation regarding the calculation of the 340B ceiling price and the imposition of civil monetary penalties on manufacturers that are found to have knowingly and intentionally overcharged covered entities, which became effective on January 1, 2019. It is unclear how the government will apply its enforcement authority under the regulation. Manufacturers also are required to report 340B ceiling prices to HRSA on a quarterly basis, and HRSA then publishes them to covered entities. Moreover, under a final regulation effective January 13, 2021, HRSA established an administrative dispute resolution (“ADR”), process for claims by covered entities that a manufacturer has engaged in overcharging, and by manufacturers that a covered entity violated the prohibitions against diversion or duplicate discounts. Such claims are to be resolved through an ADR panel of government officials rendering a decision that could be appealed in federal court. HRSA issued a final rule that, effective June 2024, modified aspects of the ADR process, which could impact the procedures that are used to determine whether a manufacturer owes additional 340B program discounts. An ADR proceeding could subject a manufacturer to onerous procedural requirements and result in additional liability.
Manufacturers are required to report the average sales price for certain Medicare Part B-covered products under the Medicare program. Manufacturers calculate the average sales price based on a statutorily defined formula as well as regulations and interpretations of the statute by CMS. CMS may use these submissions to determine payment rates for drugs under Medicare Part B. Since 2023, manufacturers must pay refunds to Medicare for single source drugs or biologics, or biosimilar biological products, reimbursed under Medicare Part B and packaged in single-dose containers or single-use packages, for units of discarded drug reimbursed by Medicare Part B in excess of ten percent of total allowed charges under Medicare Part B for that drug. Manufacturers that fail to pay refunds could be subject to civil monetary penalties of 125 percent of the refund amount. In addition, as noted previously, a manufacturer may be liable for Part B inflation rebates for utilization in quarters starting with the first quarter of 2023. Manufacturers may be liable for civil monetary penalties for violations of this program.
Statutory or regulatory changes or CMS guidance could affect the average sales price calculations for approved products and the resulting Medicare payment rate, and could negatively impact results of operations. Also, the Medicare Part B drug payment methodology is subject to change based on legislation enacted by the U.S. Congress.
In order to be eligible to have our products paid for with federal funds under the Medicaid and Medicare Part B programs and purchased by the Department of Veterans Affairs (“VA”), Department of Defense (“DoD”), Public Health Service, and Coast Guard (collectively, the “Big Four agencies”) and certain federal grantees, we are required to participate in the VA Federal Supply Schedule (“FSS”) pricing program, established under Section 603 of the Veterans Health Care Act of 1992. Under this program, we are obligated to make our “covered” drugs (i.e., innovator drugs and biologics) available for procurement on an FSS contract and charge a price to the Big Four agencies that is no higher than the Federal Ceiling Price (“FCP”), which is a price calculated pursuant to a statutory formula. The FSS program also allows us (but does not require us) to list certain non-covered drugs on an FSS contract at negotiated pricing, not capped at the FCP. The FCP is derived from a calculated price point called the “non-federal average manufacturer price” (“non-FAMP”), which we are required to calculate and report to the VA on a quarterly and annual basis. Pursuant to applicable law, knowing provision of false information in connection with a non-FAMP filing can subject a manufacturer to significant civil monetary penalties for each item of false information. The FSS contract also contains extensive disclosure and certification requirements. In addition, Section 703 of the National Defense Authorization Act for FY 2008 requires us to pay quarterly rebates to DoD on utilization of covered drugs that are dispensed through DoD’s Tricare network pharmacies to Tricare beneficiaries. The rebates are calculated as the difference between the annual non-FAMP and FCP for the calendar year that the product was dispensed. If we overcharge the government in connection with the FSS contract or Tricare Retail Pharmacy Rebate Program, whether due to a misstated FCP or otherwise, we will be required to refund the difference to the government. Failure to make necessary disclosures and/or to identify contract overcharges can result in allegations against us under the False Claims Act (“FCA”) and other laws and regulations. Unexpected refunds to the government, and any response to government investigation or enforcement action, would be expensive and time-consuming, and could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
Healthcare Fraud and Abuse Laws
We are subject to various federal, state, and local laws and regulations targeting fraud and abuse in the healthcare industry in the countries where we operate. For example, in the U.S., there are federal and state anti-kickback, false claims and other related laws that apply to healthcare products and services that are ultimately paid for by government health care programs. These laws include the following:
•The U.S. federal Anti-Kickback Statute prohibits, among other things, any person or entity from knowingly and willfully offering, paying, soliciting or receiving anything of value to induce (or in return for) the referral of business, including the purchase, recommendation or prescription of a particular drug, medical device or biologic reimbursable under Medicare, Medicaid or other federally financed healthcare programs. The statute has been interpreted to apply to arrangements between pharmaceutical companies on one hand and patients, prescribers, purchasers and formulary managers on the other. Although there are a number of statutory exemptions and regulatory safe harbors protecting certain common manufacturer business arrangements and activities from prosecution and administrative sanctions, the
19
exemptions and safe harbors are drawn narrowly and are subject to regulatory revision or changes in interpretation by the DOJ and OIG within the HHS. Practices or arrangements that involve remuneration may be subject to scrutiny if they do not qualify for an exemption or safe harbor. Additionally, there are no safe harbors for common practices, such as educational and research grants, charitable donations, product support and patient assistance. Violations of the federal Anti-Kickback Statute may be established without proving specific intent to violate the statute.
•The federal civil FCA prohibits, among other things, any person or entity from knowingly presenting, or causing to be presented, a false or fraudulent claim for payment of federal funds, or knowingly making, or causing to be made, a false record or statement material to a false or fraudulent claim to the federal government, or avoiding, decreasing, or concealing an obligation to pay money to the federal government. A claim resulting from a violation of the federal Anti-Kickback Statute constitutes a false or fraudulent claim. The FCA also permits a private individual acting as a “whistleblower” to bring actions on behalf of the federal government alleging violations of the statute and to share in any monetary recovery.
•The healthcare fraud provisions under the Health Insurance Portability and Accountability Act of 1996 (“HIPAA”), which extend to non-government health benefit programs and which impose criminal liability for, among other things, knowingly and willfully executing, or attempting to execute, a scheme to defraud any health care benefit program, including private third party payors, or falsifying or covering up a material fact or making any materially false or fraudulent statement in connection with the delivery of or payment for health care benefits, items or services. The government does not have to establish actual knowledge of the statute or specific intent in order to prove a violation.
Violations of these laws can lead to administrative, civil, and criminal penalties, fines (including mandatory penalties on a per claim or statement basis for violations of the FCA), damages, imprisonment and exclusion from participation in federal healthcare programs. These laws apply to hospitals, physicians and other potential purchasers of our products and are applicable to us as both a manufacturer and a supplier of products reimbursed by federal healthcare programs. In addition, some states in the U.S. have enacted compliance and reporting requirements aimed at drug manufacturers. Many states also have statutes or regulations similar to the federal anti-kickback law and the FCA and which apply to items and services reimbursed under Medicaid and other state programs, or, in several states, apply regardless of the payor. Other states restrict whether and when pharmaceutical companies may provide meals to health care professionals or engage in other marketing-related activities, and certain states and cities require identification or licensing of sales representatives.
We are also subject to the Foreign Corrupt Practices Act of 1977 (“FCPA”) and similar worldwide anti-bribery laws in non-U.S. jurisdictions, such as the United Kingdom (“U.K.”) Bribery Act of 2010, which generally prohibit companies and their intermediaries from making improper payments to non-U.S. officials for the purpose of obtaining or retaining business. Because of the predominance of government-sponsored healthcare systems around the world, most of our customer relationships outside of the U.S. are with governmental entities and are therefore subject to such anti-bribery laws. Our policies mandate compliance with these anti-bribery laws; however, we operate in many parts of the world that have experienced governmental corruption to some degree and, in certain circumstances, strict compliance with anti-bribery laws may conflict with local customs and practices. Despite our training and compliance programs, our internal control policies and procedures may not protect us from reckless or criminal acts committed by our employees or agents.
Sunshine Act and Transparency Laws
The U.S. Physician Payment Sunshine Act (“Sunshine Act”) requires tracking of payments and transfers of value to physicians, certain advanced practice practitioners, and teaching hospitals and ownership interests held by physicians and their families, and reporting to the federal government and public disclosure of these data. Certain states also require pharmaceutical companies to report expenses relating to the marketing and promotion of pharmaceutical products and to report transfers of value made to healthcare providers in the applicable state. Many of the non-U.S. jurisdictions in which we operate also have equivalent laws requiring us to report transfers of value to healthcare professionals.
Data Protection and Privacy
We are also subject to laws and regulations governing the privacy and security of health-related and other personal data we collect and maintain (e.g., European Union’s (“E.U.”) General Data Protection Regulation (“GDPR”) and the U.K. GDPR), Section 5 of the Federal Trade Commission Act (“FTC Act”), HIPAA, the California Consumer Privacy Act (“CCPA”), as amended by the California Privacy Rights Act (“CPRA”), and other state comprehensive privacy laws.
In Europe, the GDPR governs the collection, use, disclosure, or other processing of personal data of individuals within the European Economic Area and the transfer of personal data out of the European Economic Area and/or the U.K. to other countries, including the U.S. Among other things, the GDPR imposes requirements regarding the security of personal data and notification of data breaches to the competent national data processing authorities, and requires having lawful bases for processing personal data. The GDPR imposes substantial fines for breaches and violations (up to the greater of €20 million or 4% of our annual global turnover for the most serious breaches) and confers the right for data subjects to lodge complaints with supervisory authorities, seek judicial remedies and obtain compensation for damages resulting from violations of the GDPR.
20
The Federal Trade Commission (“FTC”) sets expectations for failing to take appropriate steps to keep consumers’ personal information secure, or failing to provide a level of security commensurate to promises made to individuals about the security of their personal information (such as in a privacy notice), which may constitute unfair or deceptive acts or practices in violation of Section 5(a) of the FTC Act. The FTC expects a company’s data security measures to be reasonable and appropriate in light of the sensitivity and volume of consumer information it holds, the size and complexity of its business, and the cost of available tools to improve security and reduce vulnerabilities. Individually identifiable health information is considered sensitive data that merits stronger safeguards. With respect to privacy, the FTC also sets expectations that companies honor the privacy promises made to individuals about how the company handles consumers’ personal information; any failure to honor promises, such as the statements made in a privacy policy or on a website, may also constitute unfair or deceptive acts or practices in violation of the FTC Act. While we do not intend to engage in unfair or deceptive acts or practices, the FTC has the power to enforce promises as it interprets them, and events that we cannot fully control, such as data breaches, may result in FTC enforcement. Enforcement by the FTC under the FTC Act can result in civil penalties or enforcement actions.
HIPAA imposes privacy and security obligations on covered entity health care providers, health plans, and health care clearinghouses, as well as their “business associates” - certain persons or covered entities that create, receive, maintain, or transmit protected health information in connection with providing a specified service or performing a function on behalf of a covered entity. While we are not directly subject to HIPAA as a covered entity or business associate, we could potentially be subject to criminal penalties if we, our affiliates, or our agents knowingly receive individually identifiable health information maintained by a HIPAA-covered entity in a manner that is not authorized or permitted by HIPAA.
In California, the CCPA and the CPRA establish certain requirements for data use and sharing transparency and grants certain rights for California consumers. Failure to comply with the CCPA and the CPRA may result in, among other things, significant civil penalties and injunctive relief, or statutory or actual damages. In addition, California residents have the right to bring a private right of action in connection with certain types of incidents. These claims may result in significant liability and damages. Other states, including, for example, Virginia, Colorado, Utah, Indiana, Iowa, Tennessee, Montana, Texas, Delaware, New Jersey, Oregon, Nebraska, New Hampshire and Connecticut, have enacted similar privacy laws that impose new obligations or limitations in areas affecting our business, and we continue to assess the impact of this state legislation, on our business as additional information and guidance becomes available. These laws and regulations are evolving and subject to interpretation, and may impose limitations on our activities or otherwise adversely affect our business.
Compliance with these laws and regulations may require significant additional cost expenditures or changes in products or our business that increase competition or reduce revenue. Noncompliance could result in the imposition of fines, penalties, or orders to stop noncompliant activities, or withdrawal of noncompliant products from a market.
Evolving Legal and Regulatory Landscape
The U.S. Supreme Court issued several decisions in 2024 that affect how courts analyze federal regulations, including the deference courts pay to federal agency decisions. The full impact of these decisions is not yet known, but they could lead to meaningful changes to the federal regulatory landscape, both generally and specifically with regard to the pharmaceutical industry. In addition, the presidential administration may implement significant changes in the federal regulation of our business and the pharmaceutical industry.
There has been increasing scrutiny of foreign-sourced drugs and foreign drug supply chains, resulting in legislative and executive actions, including imposition of tariffs and issuance of executive orders intended to incentivize moving drug manufacturing operations to the United States. There is significant uncertainty with respect to the current legal, regulatory, and policy environment, and we are unable to predict the impact of any future legislative, regulatory, third-party pay, or policy actions on us. There may be changes that we, and any third parties we might engage, are unable to adapt to, and we could face difficulties in maintaining profitability, or otherwise experience a material adverse impact on our business, financial condition and results of operations.
Compliance Programs
In order to systematically and comprehensively mitigate the risks of non-compliance with legal and regulatory requirements described within this Item 1. Business, we have developed what we believe to be robust compliance programs based on the April 2003 OIG Compliance Program Guidance for Pharmaceutical Manufacturers, 2023 OIG General Compliance Program Guidance, the U.S. DOJ Guidance on the Evaluation of Corporate Compliance Programs, the U.S. Federal Sentencing Guidelines, the Pharmaceutical Research and Manufacturers of America Code on Interactions with Healthcare Professionals, the U.K. Anti-Bribery guidance, FCPA guidance and other relevant guidance from government and national or regional industry codes of behavior. As further described below, we also operate under a corporate integrity agreement (“CIA”) and the Endo VOI (as defined below). We conduct ongoing compliance training of all employees, including for specific roles and functions, and maintain a 24-hour integrity and compliance reporting hotline with a strict policy of non-retaliation. Our compliance programs are implemented and facilitated by our Chief Compliance Officer, who reports to the Chief Executive Officer (“CEO”), is independent of other functions and acts under the oversight of the Governance and Compliance Committee of our Board of Directors.
21
As part of our compliance program, we have implemented internal cross-functional processes designed to facilitate review and approval of product-specific promotional materials, presentations and external communications to address the risk of misbranding, mislabeling or making false or misleading claims about our products through our promotional efforts. In addition, we monitor business activities (such as speaker programs and business arrangements with healthcare professionals (“HCPs”)) through our compliance monitoring program, which includes real-time observations of interactions with HCPs, review of records and data related to such interactions. We have also implemented a controlled substances compliance program, including suspicious order monitoring (“SOM”) and anti-diversion efforts and we regularly assist federal, state and local law enforcement and prosecutors in the U.S. As part of this program, we also work with some of our customers to help develop and implement what we believe are best practices for SOM and other anti-diversion activities.
Corporate Integrity Agreement
We entered into a CIA with the OIG within the HHS in March 2022. The CIA has a five-year term and requires, among other things, maintaining all elements of our compliance program, fulfillment of self-reporting, monitoring and training obligations, management certifications and resolutions from our Board of Directors. In addition, we were required to retain an independent review organization to conduct annual reviews of certain company systems and transactions related to our government pricing and patient assistance activities. We believe we are in compliance in all material respects with our CIA obligations.
Endo VOI
Certain of our subsidiaries have been, and continue to be, subject to a voluntary operating injunction (the “Endo VOI”), which prevents them from manufacturing high-dose opioid pills, advertising or marketing opioids to patients and doctors, offering compensation incentives based on opioid sales and engaging in opioid-related lobbying, among other restrictions.
Outside the United States
Outside the U.S., we must comply with laws, guidelines and standards promulgated by other regulatory authorities that regulate the development, testing, manufacturing, distribution, marketing and selling of medicinal products and medical devices, including, but not limited to, Health Canada, the European Medicines Agency, the European Commission and the Competent Authorities of E.U. member states of the E.U. such as the Health Products Regulatory Agency, the Therapeutic Goods Administration in Australia, the Ministry of Health and Welfare in Japan, the European Pharmacopoeia of the Council of Europe and the International Conference on Harmonization. Although international harmonization efforts continue, many laws, guidelines and standards differ by region or country. We currently market our products in various countries, including in Japan, Canada, Australia and a number of countries in Latin America and the Asia-Pacific regions. The approval requirements and processes vary by country, and the time required to obtain a marketing authorization may vary from that required for FDA approval. Certain drug products and variations in drug product lines also must meet country-specific and other local regulatory requirements. The following discussion highlights some of the differences in the approval process in other regions or countries outside the U.S.
Canada
In Canada, the Regulations Amending the Patented Medicines Regulations (Additional Factors and Information Reporting Requirements) (the “Amendments”) came into force on July 1, 2022. The Amendments made a number of changes to the reporting and regulation of Canadian drug prices by the Patented Medicine Prices Review Board (“PMPRB”). The PMPRB is an administrative board with a mandate to protect Canadians from excessive pricing of patented medicines. Pharmaceutical manufacturers that are patentees are required to report applicable patents and file sales information so the PMPRB can monitor for excessive pricing as long as the product is considered to be a patented medicine. If it is determined that the average price, over a given reporting year, for a patented medicine is too high based on pricing tests developed by the PMPRB, the manufacturer may be required to make payments to offset revenues determined to be excessive and/or to reduce the price of the medicine. The PMPRB’s authority to regulate the price of a drug product is linked to patent status, specifically, when there is a patent to an invention that is intended or capable of being used for medicine or for the preparation or production of medicine. In addition, in Canada, in order to be eligible to market a drug in Canada, a notice of compliance (“NOC”) must be obtained. If a NOC is issued for a new drug that meets the definition of an “innovative drug”, eight years of data protection, running from the date of first marketing of the new drug, will be available. If not, the protection does not apply.
European Union
E.U. Medical Devices. In the E.U., medical devices must currently comply with the General Safety and Performance Requirements laid down in Annex I to the E.U. Medical Devices Regulation (“MDR”). Compliance with these requirements is a prerequisite to be able to affix the Conformité Européenne (“CE”) mark on products, without which they cannot be marketed or sold in the E.U. To demonstrate compliance with the General Safety and Performance Requirements of the E.U. MDR and obtain the right to affix the CE mark, medical devices manufacturers must undergo a conformity assessment procedure, which varies according to the type of medical device and its classification. Apart from low risk medical devices (Class I with no measuring function, which are not sterile and are not reusable surgical instruments), in relation to which the manufacturer may issue an EC Declaration of Conformity based on a self-assessment of the conformity of its products with the General Safety and Performance Requirements, a conformity assessment procedure requires the intervention of a notified body, which is an organization designated by a Competent Authority of an
22
E.U. member state to conduct conformity assessments. Depending on the relevant conformity assessment procedure, the notified body would audit and examine the technical documentation and the quality system for the manufacture, design and final inspection of the medical devices. The notified body issues a CE Certificate of Conformity following successful completion of a conformity assessment procedure conducted in relation to the medical device and its manufacturer and their conformity with the General Safety and Performance Requirements. This Certificate and the related conformity assessment process entitles the manufacturer to affix the CE mark to its medical devices after having prepared and signed a related EC Declaration of Conformity. Notified bodies must be designated by the authority responsible for notified bodies in the relevant E.U. member states to conduct assessment procedures for medical devices in accordance with the E.U. MDR. The time required to obtain a CE Certificate of Conformity from a notified body in the E.U. is lengthy and may be unpredictable. On average, the time-to-certification under the MDR for all device categories ranges between 13 and 18 months. The E.U. has also adopted directives and other laws that govern the labeling, marketing, advertising, supply, distribution of medicinal products and medical devices. Such directives set regulatory standards throughout the E.U. and permit member states to supplement such standards with additional requirements.
Environmental Matters
Our operations, like those of other pharmaceutical companies, are subject to substantial federal, state and local environmental laws and regulations concerning, among other matters, the generation, handling, storage, transportation, treatment and disposal of, and exposure to, hazardous substances. We cannot provide assurance that we have been or will be in full compliance with environmental, health and safety laws and regulations at all times. Violation of these laws and regulations, which may change, can lead to substantial fines and penalties. Many of our operations require environmental permits and controls to prevent and limit pollution of the environment. As part of our corporate responsibility strategy, we are committed to operating our business in a responsible manner that seeks to minimize environmental impact, while promoting the safe, efficient and responsible use of global resources. Environmental laws are complex and generally have become more stringent over time. We believe that we have planned sufficiently for future capital and operating expenditures to comply with these laws.
Raw Materials
We contract with various third-party manufacturers and suppliers to provide us with raw materials used in our products. We mitigate raw material supply risks through inventory management or alternate sources of supply, where possible. However, certain raw materials are only available from a single source. If we are unable to obtain sufficient quantities of any of the raw materials required for our products, it could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
Sales, Marketing and Customers
Sales and Marketing
We market our products to physicians (including rheumatologists, ophthalmologists, hepatologists, nephrologists, neurologists, pulmonologists, neonatologists and orthopedic surgeons), other health care providers including pharmacists, pharmacy buyers, and specialty pharmacies. We distribute our products through independent channels, including wholesale drug distributors, specialty pharmaceutical distributors, retail pharmacy chains, hospital procurement departments, and governmental agencies, among others. In addition, we contract with group purchasing organizations (“GPO(s)”) and managed care organizations to improve access to our products.
For further information on our sales and marketing strategies, refer to “Item 1. Business - Our Business and Products” above.
Customers
Net sales to distributors that accounted for more than 10.0% of our total net sales in the years ended December 31, 2025 and December 27, 2024 (Successor), the period November 15, 2023 through December 29, 2023 (Successor), and the period December 31, 2022 through November 14, 2023 (Predecessor) were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| FFF Enterprises, Inc. | 46.0 | % | 42.2 | % | 40.2 | % | 38.1 | % | ||||||||||||||||||
| Cencora, Inc. | 12.1 | * | * | * | ||||||||||||||||||||||
* Net sales to this distributor were less than 10.0% of total net sales during the respective periods presented above.
No other customer accounted for 10.0% or more of our net sales in the above periods presented.
23
Manufacturing and Distribution
As of December 31, 2025, we had seven manufacturing sites, including five located in the U.S., as well as sites in Ireland and Japan, which handle production, assembly, quality assurance testing, packaging, and sterilization of our products. Approximately 66% of our manufacturing production (as measured by cost of production) was performed within the U.S. in fiscal 2025.
As of December 31, 2025, we maintained distribution centers in three countries specific to INOmax, including in the U.S. In addition, we utilize various third-party distribution centers in the U.S. and in limited instances, outside the U.S. Products generally are delivered to these distribution centers from our manufacturing facilities and then subsequently delivered to the customer. In some instances, product is delivered directly from our manufacturing facility to the customer. We contract with a wide range of transport providers to deliver our products by road, rail, sea and air.
We utilize contract manufacturing organizations to manufacture certain of our finished goods that are available for resale, including certain activities for Acthar Gel and Xiaflex.
Seasonality
We have historically experienced fluctuations in our business resulting from seasonality. For example, Acthar Gel has typically experienced lower net sales during the first calendar quarter compared to other calendar quarters, which we believe is partially attributable to effects of annual insurance deductibles and the lack of warm temperatures that may exacerbate certain medical conditions.
Human Capital
Our mission at Keenova, to help patients with rare or unaddressed conditions live happier and healthier lives, cannot be accomplished without the dedication, collaboration and engagement of our workforce. We work hard to identify, retain and attract a workforce that shares our mission so we can improve the lives of patients with rare and unaddressed conditions. We aim to create a culture that encourages collaboration and respect, enabling employees to perform at their best. We invest in human resources programs designed to develop capabilities to deliver on our critical business priorities. Following our recent Business Combination and Separation, we are currently focused on team integration and effective team leadership. We offer competitive compensation and benefit programs, investing in our employees’ growth and development and fostering a safe and healthy work environment. We empower employees to bring their whole, authentic selves to work. Further, we encourage and support our employees in being active members of their communities.
As of December 31, 2025, we employed approximately 1,600 people, of which approximately 25% worked in manufacturing and distribution sites located across the U.S., Ireland and Japan, approximately 35% were field based, working across multiple countries and engaging with healthcare professionals and facilities, and approximately 40% worked within our corporate services locations, including locations in Bridgewater, New Jersey; Malvern, Pennsylvania; Hazelwood, Missouri; D.C.; Tokyo, Japan; and Dublin, Ireland. Of our total workforce, 99% were full-time.
Employee Total Rewards
Keenova is committed to providing a comprehensive and competitive total rewards package to meet the needs of our employees.
Keenova’s compensation and benefits offerings include, among other things, salaries, short- and long-term incentives, retirement benefits, healthcare and insurance benefits, health savings and flexible spending accounts, student loan matching programs, caregiver resources, paid time off, family leave, employee assistance programs, employee recognition programs and an employee well-being program. We continuously consider market data when evaluating our compensation and benefits offerings.
Talent Development and Employee Engagement
At Keenova, we are committed to a culture of continuous learning. Our talent strategies are closely aligned with our business priorities, creating opportunities for employees to develop professional and technical skills and advance their careers. Through our talent review and individual development planning processes, we align employee aspirations with business needs, facilitating professional growth and effective succession planning.
We offer a flexible approach to learning. Through our learning platforms, employees can build skills across a wide range of topics, from business fundamentals and technical expertise to leadership development. We also offer resources to enable professional development and support formal education through tuition reimbursement, helping employees pursue accredited programs that advance their careers. As we evolve, we remain focused on expanding development opportunities and exploring innovative ways to nurture talent.
At Keenova, we highly value employee feedback. We are committed to creating a culture where employees feel empowered to speak freely and ask questions. We actively seek feedback from our employees to obtain valuable insights that we translate into actionable steps to enhance the employee experience, so that our employees feel engaged and supported, both personally and professionally.
24
Culture
Keenova is dedicated to creating a vibrant workplace where our employees can thrive by providing access to opportunities, resources and pathways for growth and advancement. By nurturing an environment where our employees’ voices are heard and valued, we unlock the full potential of our talent, drive collaboration and enhance our ability to serve patients and communities worldwide.
Creating a supportive workplace requires deliberate action. We do this through various initiatives, such as offering training and education programs to promote understanding and supportive behaviors across all levels of the organization. We also offer platforms for employees to connect, share experiences and drive initiatives that promote collaboration across functions and sites to help enhance our unique culture.
Social Impact and Corporate Charitable Giving Program
Keenova is committed to making a positive difference in the lives of patients and our global community. Our social impact strategy is centered on improving patient health, building stronger communities and empowering our employees to participate in these efforts. We provide grants and charitable donations to eligible nonprofits globally and have programs that encourage our employees’ philanthropic efforts.
We support nonprofit organizations that align with our mission to address unmet medical needs with innovative solutions. Our patient-centric charitable contributions prioritize programs that benefit public health and advance medical care within our therapeutic areas of focus. We also invest in community-based programs in focus areas, such as education, health and wellness, and sustainability. We support patient advocacy and STEM-education organizations in the therapeutic areas we focus on and communities in which we operate.
Information About Our Executive Officers
Set forth below are the names, ages as of April 15, 2026, and current positions of our executive officers.
| Name | Age | Title | ||||||
| Sigurdur O. Olafsson | 57 | President, Chief Executive Officer and Director | ||||||
| Christiana Stamoulis | 55 | President and Chief Financial Officer | ||||||
| Tracy Basso | 51 | Executive Vice President and Chief Human Resources Officer | ||||||
| Lisa French | 57 | Executive Vice President and Chief Commercial Officer | ||||||
| Dr. Marek Honczarenko | 56 | Executive Vice President and Chief Scientific Officer | ||||||
| Henriette Nielsen | 60 | Executive Vice President and Chief Transformation Officer | ||||||
Paul O'Neill | 56 | Executive Vice President and Chief Operations Officer | ||||||
| Cheryl Stouch | 54 | Executive Vice President and Chief Information Officer | ||||||
| Mark Tyndall | 50 | Executive Vice President, Chief Legal Officer and Corporate Secretary | ||||||
| Susan Williamson | 50 | Executive Vice President and Chief Compliance Officer | ||||||
Set forth below is a brief description of the position and business experience of each of our executive officers.
Sigurdur (Siggi) O. Olafsson serves as our President, Chief Executive Officer and a director, a role he assumed in June 2022, and is also a member of the Company’s executive committee. Before joining Keenova, Mr. Olafsson served as chief executive officer of Hikma Pharmaceuticals plc, a multinational pharmaceutical company publicly traded on the London Stock Exchange, from February 2018 to June 2022. Prior to Hikma, Mr. Olafsson served as president and chief executive officer of the Global Generic Medicines Group of Teva Pharmaceuticals, from 2014 to 2017. Before that, he was President of Actavis plc (formerly, Watson Pharmaceuticals, Inc.) from 2010 to 2014 and served in other leadership roles at Actavis ehf from 2003 to 2010. Mr. Olafsson previously held a number of positions of increased responsibility in Pfizer’s Global R&D organization in the U.K. and U.S., focused on branded drug development, and served as head of drug development for Omega Farma in Iceland. Mr. Olafsson previously served as a director on the boards of directors of Hikma from 2018 to 2022, Pfenex Inc. from 2017 to 2019 and as chairman of Oculis ehf from 2017 to 2018. Mr. Olafsson holds a Master of Science degree in pharmacy (Cand Pharm) from the University of Iceland, Reykjavik. Mr. Olafsson’s qualifications to serve on our Board include his more than 30 years of diverse pharmaceutical experience, in-depth knowledge of all aspects of our business, extensive and diverse industry and managerial expertise, and a proven record of leadership to serve as President, Chief Executive Officer, and director.
Christiana Stamoulis serves as our President and Chief Financial Officer, a role she assumed in September 2025, and is also a member of the Company’s executive committee. She brings 30 years of experience in the biotechnology industry, with expertise in corporate finance, capital markets, strategy, and the origination, structuring, and execution of strategic partnerships and M&A transactions. Before Keenova, Ms. Stamoulis served as executive vice president and chief financial officer of Incyte Corp., a biopharmaceutical company, from February 2019 to September 2025. Prior to joining Incyte, she served as president from February 2018 to January 2019 and chief financial officer from January 2015 to January 2019 of Unum Therapeutics, a biopharmaceutical company. Prior to joining Unum, Ms. Stamoulis was a senior vice president of corporate strategy and business development at Vertex Pharmaceuticals, Inc., a biopharmaceutical company. Prior to joining Vertex, Ms. Stamoulis spent nearly 15 years in the investment
25
banking and management consulting industries. She was a managing director in the investment banking division of Citigroup and, prior to that, she was a senior investment banker in the healthcare investment banking group of Goldman Sachs. Ms. Stamoulis started her career as a strategy consultant at The Boston Consulting Group. From November 2011 to April 2026, Ms. Stamoulis served on the board of directors of Hologic Inc., a medical technology company. She holds two Bachelor of Science degrees from the Massachusetts Institute of Technology (MIT) and a Master of Business Administration from the MIT Sloan School of Management.
Tracy Basso is our Executive Vice President and Chief Human Resources Officer, a role she assumed in July 2025, leading all aspects of human resources, enabling the development and growth of the company. Ms. Basso is also a member of the Company’s executive committee. She served as chief human resources officer of Endo, Inc. and its predecessor Endo International plc from July 2019 to July 2025. Prior to that, Ms. Basso was senior vice president, human resources, leading human resources support for Endo’s sterile injectable and generic businesses, as well as operations, quality, and enterprise-wide talent acquisition. She also worked for GlaxoSmithKline and started her career at Best Foods Baking Company. Ms. Basso holds a Master’s degree in Human Resources from Fordham University and a Bachelor of Science in Business Management from Iona College.
Lisa French is our Executive Vice President and Chief Commercial Officer, a role she assumed in October 2022. She has executive responsibility for all commercial and market-access activities for the Company’s branded therapeutics, as well as new product launch execution for assets in the Company’s near-term development portfolio. Ms. French is a member of the Company’s executive committee. Ms. French has more than 30 years of experience in U.S. go-to-market commercialization strategy development and operating experience across a multitude of therapeutic areas: chronic care, vaccine, hospital, and rare disease, and at various life-cycle stages. Before joining the Company, Ms. French served as U.S. business unit lead of the women’s health franchise at Organon & Co., a global healthcare company, where she led the commercial team from January 2021 through September 2022. Prior to that, she held various positions of increasing responsibility at Merck, a pharmaceutical company, where she ultimately led all aspects of a multi-billion dollar brand, executed commercial innovation initiatives and oversaw multiple sales teams, including as associate vice president, U.S. marketing lead, HPV franchise, from October 2019 to January 2021, and as associate vice president, U.S. strategy and commercial model innovation, from January 2016 to October 2019. Ms. French holds a Bachelor of Science degree in Biology from West Chester University and completed Harvard Business School’s Emerging Leaders and Leadership & Strategy executive programs.
Dr. Marek Honczarenko is our Executive Vice President and Chief Scientific Officer, a role he assumed in September 2025, and is also a member of the Company’s executive committee. He brings more than 25 years of experience in drug development, including achievements in all stages of the pharmaceutical lifecycle. Before Keenova, from 2023, he served as senior vice president and head of global innovative medicines development at Sun Pharmaceutical Industries. At Sun Pharma, he had full responsibility for driving strategy and execution of the specialty medicines pipeline with oversight of clinical development, clinical operations, biostatistics, experimental medicine, pharmacology, translational medicine, data management, quality assurance, and regulatory strategies across all therapeutic areas and geographies. From December 2020 to August 2023, he was senior vice president and head, development clinical sciences, GlaxoSmithKline, responsible for strategies and programs across all specialty therapeutic areas; vice president and head, global immunology development, AbbVie; head of immunoscience and fibrosis, Bristol Myers Squibb; head, translational immunology, Pfizer; medical director, rheumatology, inflammation and autoimmunity, MedImmune; and director, immunobiology and translational medicine, Biogen. From October 2019 to August 2025, Dr. Honczarenko served as an industry representative for the FDA Arthritis Advisory Committee. He received degrees of Doctor of Medicine and Doctor of Philosophy summa cum laude from Pomeranian Medical University, Szczecin, Poland, and completed his post-graduate training at the Perelman School of Medicine at the University of Pennsylvania. He then joined the faculty at Boston Children’s Hospital and Harvard Medical School, and served as a medical director at Harvard’s Centre for Human Cell Therapies.
Henriette Nielsen is our Executive Vice President and Chief Transformation Officer, a role she assumed in August 2022, and is also a member of the Company’s executive committee. Ms. Nielsen has an impressive track record of enhancing operations at pharmaceutical companies and significant experience across a range of corporate functions. Before joining the Company, Ms. Nielsen served at Hikma Pharmaceuticals plc, a multinational pharmaceutical company publicly traded on the London Stock Exchange as executive vice president, business operations, a role she held from June 2018 to July 2022. Before that, Ms. Nielsen served at Teva Pharmaceuticals, a global pharmaceutical company, as senior vice president and chief transformation officer, from January 2015 to June 2018. Prior to her role at Teva Pharmaceuticals, she was the founder of System Matters ApS, a healthcare and impact investing consultancy, from April 2011 to December 2014, and the general counsel and an executive vice president at Actavis Group, from January 2006 to March 2011. Ms. Nielsen began her career as a commercial lawyer in Denmark at Kromann Reumert. She presently serves as vice chair of Think Equal USA, a not-for-profit providing and advocating for early-age social emotional learning, and is an advisor to EIR, which promotes women’s sports globally. Ms. Nielsen was a candidate of law at the University of Copenhagen, received her Master of Laws at the University of Edinburgh, and completed a Leading Sustainable Corporation Programme at the University of Oxford.
Paul O'Neill is our Executive Vice President and Chief Operations Officer, a role he assumed in November 2025, after serving as our Executive Vice President, Quality & Operations, Specialty Brands, from February 2024. In this capacity, he leads internal and external manufacturing, supply chain distribution, device engineering, quality, technical services and product support, and is also a member of the Company’s executive committee. Mr. O’Neill joined the Company in March 2023, initially serving as Senior Vice President of Quality & Operations, Specialty Brands. Mr. O’Neill has more than 25 years of experience in manufacturing operations,
26
plant start-ups, technology transfer and supply chain management, and has held numerous leadership positions at biopharmaceutical companies, including Merck, Pfizer and Wyeth. Prior to joining the Company, Mr. O’Neill served as executive director, biologics operations, at Merck, and was responsible for overseeing the end-to-end supply strategy of Merck’s Keytruda and biologics (mABs) pipeline portfolio. Prior to that, Mr. O’Neill held leadership positions at Pfizer and Wyeth in plant operations, supply chain management, new product launches, site start-ups and network design. Mr. O’Neill holds a Master of Business Administration degree from the Alfred Lerner College of Business & Economics at the University of Delaware and a Bachelor of Science in Food Science and Technology from University College of Cork.
Cheryl Stouch is our Executive Vice President and Chief Information Officer, a role she assumed in July 2025, and is also a member of the Company’s executive committee. Ms. Stouch is responsible for the overall IT strategy and roadmap to support the Company’s strategy and vision, ensuring digital capabilities for all global operations and team members. She served as senior vice president, information technology and chief information officer of Endo, Inc. and its predecessor Endo International plc from June 2022 to July 2025. Ms. Stouch joined Endo in May 2020 as executive director of IT corporate functions & end user services. She championed the development and implementation of the company’s enterprise collaboration strategy and was a founding member of AWE (Alliance for Women at Endo). Prior to joining Endo, Ms. Stouch spent 13 years at Shire (now Takeda), where she held various positions of increasing responsibility within IT, partnering with leaders from across the business. Her most recent role at Shire was head of IT cross function & platforms, where she was accountable for eight global enterprise capabilities supporting 32,000 team members. Ms. Stouch began her career in the food industry and then in management consulting, working for IBM and focused on SAP enterprise solutions. Ms. Stouch is a member of the Healthcare Businesswomen’s Association (HBA). She holds a Bachelor of Business and a Bachelor of Science for Information Systems from Dakota State University.
Mark Tyndall is our Executive Vice President, Chief Legal Officer and Corporate Secretary, a role he assumed in August 2022. Mr. Tyndall has executive responsibility for all legal functions and serves as the primary liaison to the Board of Directors. He also has responsibility for the Company’s Government Affairs and Patient Advocacy functions, and is also a member of the Company’s executive committee. Previously, from February 2021 to August 2022, Mr. Tyndall served as the Company’s Senior Vice President and U.S. General Counsel, where he had responsibility for the U.S. and international commercial legal teams, corporate litigation and investigations, legal operations, and the corporate privacy function, and oversaw the Government Affairs team. Before that, Mr. Tyndall held the roles of Senior Vice President of Government Affairs & Chief Counsel of Litigation (from February 2019 to February 2021), and Vice President of Government Affairs, Policy and Patient Advocacy (from June 2014 to February 2019). Prior to joining the Company, Mr. Tyndall served as head of global policy and public affairs at Bayer Healthcare’s consumer health division, a role he served in from January 2013 to June 2014. Prior to joining Bayer, Mr. Tyndall practiced healthcare and political law in the Washington, D.C. office of Sidley Austin LLP, where he focused on healthcare regulatory issues, fraud and abuse matters, and legislative and policy issues. He is also a former professional staff member of the U.S. Senate Committee on Agriculture, Nutrition and Forestry. Mr. Tyndall holds a Juris Doctor from George Washington University Law School, a Master’s degree in Public Policy from the College of William and Mary, and a Bachelor of Arts degree in Economics from Christopher Newport University. He also completed the International Human Rights Law Summer Program at the University of Oxford, New College.
Susan Williamson is our Executive Vice President and Chief Compliance Officer, a role she assumed in July 2025, and is also a member of the Company’s executive committee. Ms. Williamson is responsible for the strategic direction and operations of the Company’s Global Corporate Compliance program. Ms. Williamson previously served as senior vice president and chief compliance officer of Endo, Inc. and its predecessor Endo International plc from April 2018 to July 2025. She joined Endo in 2012 and was a valued member of the corporate compliance leadership team, previously serving as vice president and U.S. and Canadian compliance officer where she was responsible for oversight of the company’s Corporate Compliance program, including management of its CIA, enterprise-wide corporate compliance training and monitoring, and aggregate spend reporting. Prior to Endo, Ms. Williamson worked at Pfizer in operational risk management supporting Pfizer’s CIA from 2009 to 2012. Prior to her employment with Pfizer, Ms. Williamson spent over five years at Wyeth Pharmaceuticals and held key roles in building, developing, and implementing a global corporate compliance program. Ms. Williamson began her career in public accounting at Ernst & Young in the assurance and advisory practice. Ms. Williamson is a Certified Public Accountant and holds a Bachelor of Science degree in Accounting from Saint Joseph’s University as well as a Master of Business Administration from the Erivan K. Haub School of Business at St. Joseph’s University.
Emergence from Voluntary Reorganization
2023 Bankruptcy Proceedings
On August 28, 2023, we voluntarily initiated Chapter 11 proceedings (“2023 Chapter 11 Cases”) under chapter 11 of title 11 (“Chapter 11”) of the U.S. Code in the U.S. Bankruptcy Court for the District of Delaware (“Bankruptcy Court”). On September 20, 2023, our then serving directors initiated examinership proceedings with respect to Mallinckrodt plc by presenting a petition to the High Court of Ireland pursuant to Section 510(1)(b) of the Companies Act 2014 seeking the appointment of an examiner to Mallinckrodt plc. On October 10, 2023, the Bankruptcy Court entered an order confirming a plan of reorganization (“2023 Plan”). Subsequent to the Bankruptcy Court’s order confirming the 2023 Plan, the High Court of Ireland made an order confirming a scheme of arrangement on November 10, 2023, which is based on and consistent in all respects with the 2023 Plan (“2023 Scheme of Arrangement”). The 2023 Plan and the 2023 Scheme of Arrangement became effective on November 14, 2023, (“2023 Effective Date”), and we emerged from the 2023 Chapter 11 Cases and the Irish examinership proceedings (together, the “2023 Bankruptcy
27
Proceedings”) on that date. Refer to Note 2 to the Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for further information on the 2023 Plan and emergence from the 2023 Bankruptcy Proceedings.
Adoption of Fresh-Start Accounting
Upon emergence from the 2023 Bankruptcy Proceedings on November 14, 2023, we adopted fresh-start accounting in accordance with the provisions of Financial Accounting Standards Board Accounting Standards Codification (“ASC”) Topic 852 - Reorganizations (“ASC 852”), and became a new entity for financial reporting purposes as of the 2023 Effective Date. References to “Successor” relate to the financial position as of December 31, 2025 and December 27, 2024 and results of operations of the reorganized Company subsequent to November 14, 2023, while references to “Predecessor” relate to the financial position as of December 30, 2022 and results of operations of the Company for the period December 31, 2022 through November 14, 2023. All emergence-related transactions related to the 2023 Effective Date were recorded as of November 14, 2023. Accordingly, the consolidated financial statements for the Successor are not comparable to the consolidated financial statements for the Predecessor periods. Refer to Note 3 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for further information.
Available Information
Financial results, news, and other information about Keenova can be accessed from our website at https://www.keenova.com/. This site includes important information on our locations, products and services, financial reports, news releases, and career opportunities. Our Annual Reports on Form 10-K, Quarterly Reports on Form 10-Q, Current Reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934 (“Exchange Act”) are available on our website, free of charge, as soon as reasonably practicable after they are electronically filed with or furnished to the U.S. Securities and Exchange Commission (“SEC”), and are available on the SEC's website at https://www.sec.gov. Information contained on, or that may be accessed through, our website is not incorporated by reference in this Annual Report and, accordingly, you should not consider that information part of this Annual Report.
We use our website as a channel of distribution for important company information, such as press releases, investor presentations and other financial information. We also use our website to expedite public access to time-critical information regarding our Company in advance of or in lieu of distributing a press release or a filing with the SEC disclosing the same information. Therefore, investors should look to the Investor Relations page of our website for important and time-critical information. Visitors to our website can also register to receive automatic e-mail and other notifications alerting them when new information is made available on the Investor Relations page of our website.
| Item 1A. | Risk Factors. | ||||
We operate in a rapidly changing environment that involves a number of risks, some of which are beyond our control. You should carefully consider the risks described below in addition to all other information provided to you in this Annual Report, any one of which could cause our actual results to vary materially from recent results or from our anticipated future results and could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows. The risks and uncertainties described below are those that we currently believe may materially affect our company. These and other risks could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
Risks Related to the Business Combination with Endo and Separation of Par Health
We may not realize the anticipated benefits and synergies from our Business Combination with Endo.
The success of the Business Combination depends, in part, on our ability to realize the anticipated benefits from successfully combining our and Endo’s businesses. We have devoted and continue to devote substantial management attention and resources to integrating our business practices and operations with Endo’s so that we can fully realize the anticipated benefits of the Business Combination. Nonetheless, difficulties may arise during the process of combining the operations of our business and Endo’s business that could result in the failure to achieve the synergies that we anticipate, the loss of key employees that may be difficult to replace in the competitive pharmaceutical industry, the disruption of each company’s ongoing businesses, complexities associated with managing inconsistencies in standards, controls, procedures and policies that adversely affect our ability to maintain relationships with customers, suppliers, distributors, collaborators, creditors or other business partners. As a result, the anticipated benefits of the Business Combination may not be realized fully within the expected timeframe or at all or may take longer to realize or cost more than expected, which could materially impact our business, cash flow, financial condition or results of operations as well as adversely impact the price of our ordinary shares.
We have also incurred, and will continue to incur, a number of costs associated with combining our business with Endo’s business. Additional unanticipated costs may be incurred in the integration of our business and Endo’s business. The elimination of duplicative costs, as well as the realization of other efficiencies related to the integration of the two companies, may not initially offset integration-related costs or achieve a net benefit in the near term, or at all. In addition, at times, the attention of certain members of
28
management, other key employees and resources may be focused on the integration of the businesses of the two companies and diverted from day-to-day business operations, which may disrupt our business.
We, along with our current or former officers or directors, could become subject to litigation in connection with the Business Combination, which could result in substantial costs.
Our future success will depend, in part, on our ability to manage our business by, among other things, integrating the assets, operations and personnel of our company and Endo in an efficient and timely manner; consolidating systems and management controls and successfully integrating relationships with customers, suppliers and other business partners. Failure to successfully manage our combined company may have an adverse effect on our business, cash flow, reputation, financial condition and results of operations.
The estimated fair values of the net assets acquired by us in connection with the acquisition of Endo are preliminary and subject to change if new information becomes available.
In accordance with applicable accounting standards for business combinations, we have made preliminary estimates of the fair value of Endo’s assets and liabilities based on currently available information, which remain subject to change as we finalize our purchase accounting during the measurement period following the acquisition date. For a period of up to 12 months from the acquisition date, adjustments to these preliminary estimates may result from additional information becoming available to us about facts and circumstances that existed as of the acquisition date. Such adjustments could have a material impact on our financial statements, including, but not limited to, the recognition of goodwill, intangible assets, deferred taxes and other assets and liabilities. In accordance with applicable accounting standards, any such measurement period adjustments may require that we make retroactive adjustments to the estimated fair value of Endo’s acquired net assets, which in turn may impact our operating results in the periods subsequent to the acquisition.
There can be no assurance that future adjustments will not have a material adverse effect on our financial condition, results of operations or cash flows.
We could incur additional payment obligations pursuant to the U.S. Government Economic Settlement upon the achievement of certain EBITDA outperformance targets.
The U.S. Government Economic Settlement provides for payment of contingent consideration of $25.0 million per year for each calendar year between 2024 and 2028 (capped at $100.0 million in the aggregate) if Endo LP’s annual EBITDA for the corresponding calendar year exceeds defined baselines (the “EBITDA Outperformance Targets”), as set forth in the U.S. Government Economic Settlement. In accordance with the provisions of the U.S. Government Economic Settlement, in the event Endo LP acquires or sells assets, such EBITDA Outperformance Targets shall be adjusted upward or downward dollar for dollar in an amount equal to the EBITDA contribution of such acquired or sold assets. The EBITDA Outperformance Targets for 2024 and 2025 were not met and we do not expect to meet the EBITDA Outperformance Targets in any of the fiscal years 2026 through 2028. No payments have been made or accrued for related to the achievement of certain EBITDA outperformance targets. Such contingent payments continue to apply after the closing of the Business Combination and the Separation.
We may not achieve growth opportunities, profit improvements, cost savings and other benefits, and may incur unanticipated costs associated with the Separation, and our results of operations, financial condition and valuation could be adversely affected as a result.
We believe that the Separation will provide significant benefits. However, there can be no assurance that we will successfully execute our strategy, or that the expected growth opportunities, profit improvements, cost savings and other benefits of the Separation will be realized.
The process of implementing the Separation has been and is expected to continue to be time-consuming and involve significant costs and expenses. The costs may be significantly higher than what we currently anticipate and may not yield a discernible benefit if the Separation is not executed efficiently, or the expected benefits of the Separation are not realized. Implementing the Separation has required and will continue to require significant amounts of management’s time and effort, which may divert management’s attention from operating and growing our business. We may also experience increased difficulties in attracting, retaining and motivating employees as a result of the Separation. Additionally, we continue to receive certain services relating to administrative and corporate services, among others, from Par Health for a period of time. See the risk factor captioned “As a result of the Separation, we may lose the benefits of services provided by Par Health or certain of its subsidiaries and we may incur incremental costs as a result.” for additional information.
Pursuant to the terms of the separation agreement with Par Health, Par Health agreed to indemnify us for certain liabilities relating to the generic pharmaceuticals (including APIs) and sterile injectables businesses assumed by Par Health including all related pending, threatened and unasserted legal matters. However, third parties could also seek to hold us responsible for any of the liabilities that Par Health agreed to assume, and there can be no assurance that the indemnity from Par Health will be sufficient to protect us against the full amount of such liabilities, or that Par Health will be able to fully satisfy its indemnification obligations in the future. Even if we ultimately succeed in recovering from Par Health any amounts for which we are held liable, we may be temporarily required to bear these losses.
29
To the extent that we incur additional costs, achieve lower profit improvements or have lower-than-expected cost savings, our results of operations, financial condition and valuation could be adversely affected.
As a result of the Separation, we may lose the benefits of services provided by Par Health or certain of its subsidiaries and we may incur incremental costs as a result.
Certain of our subsidiaries have received administrative and corporate services from Par Health or certain of its subsidiaries and, for a transition period, continue to receive administrative and corporate services, among others, pursuant to an agreement for Par Health or its subsidiaries, as applicable. The effective and appropriate performance of such services is critical for a successful transition but also for the success of our operations. We are working to replicate or replace the services we continue to need in the operation of our business that are provided currently by or through Par Health or its subsidiaries, but it is possible that we may not be able to replace these services in a timely manner, or on the same or similar terms and conditions that we received them from Par Health or its subsidiaries, which may put further constraints on our human resources, capital and other resources.
Additionally, a subsidiary of Keenova has entered into an agreement for a subsidiary of Par Health to provide certain manufacturing and other related services (in accordance with our specifications) related to Xiaflex and, subject to mutual agreement, may agree to manufacture and supply additional products, which will result in incremental costs to us.
We are a smaller and less diversified company than before the Separation.
As a result of the Separation, we are a less diversified company with a more concentrated product portfolio. As a result, we may be more vulnerable to changing market and regulatory conditions, which could have a material adverse effect on our business, financial condition and results of operations. In addition, the diversification of our revenues, costs and cash flows are reduced, such that our results of operations, cash flows, working capital, effective tax rate and financing requirements may be subject to increased volatility, and our ability to fund capital expenditures and investments, pay dividends and service debt may be diminished. This increased volatility could have a material adverse effect on our business, financial condition, results of operations and cash flows.
The Separation may result in litigation and/or regulatory inquiries and investigations, which could harm our business, financial condition and operating results and could divert management attention.
In the past, securities class action litigation and/or shareholder derivative litigation and inquiries or investigations by regulatory authorities have often followed certain significant business transactions, such as the sale of a company or announcement of any other strategic transaction, such as the Separation. Any Separation-related litigation or investigation against us, whether or not resolved in our favor, could result in substantial costs and divert management’s attention from other business concerns, which could adversely affect our business, financial condition, results of operations and cash flows.
Risks Related to Our Business
Pharmaceutical companies like us have been under increasing scrutiny from governments, legislative bodies and enforcement agencies related to sales, marketing and pricing practices, and changes to, or non-compliance with, relevant policies, laws, regulations or government guidance may result in actions that could adversely affect our business.
In the U.S., over the past several years, a significant number of pharmaceutical and biotechnology companies have been subject to inquiries and investigations by various federal and state regulatory, investigative, prosecutorial and administrative entities in connection with the promotion of products for unapproved uses and other sales, marketing and pricing practices, including the DOJ, the OIG within the HHS, the FDA, the FTC and various state Attorneys General offices. These investigations have alleged violations of various federal and state laws and regulations, including alleged violations of antitrust laws, the FFDCA, the FCA, the Prescription Drug Marketing Act, anti-kickback laws, data and patient privacy laws, export and import laws, consumer protection laws and other alleged violations in connection with the promotion of products for unapproved uses, pricing and Medicare and/or Medicaid reimbursement. These laws are described in greater detail in Item 1. Business included within this Annual Report. The DOJ and the SEC have also increased their focus on the enforcement of the FCPA, particularly as it relates to the conduct of pharmaceutical companies.
Many companies, including us, have faced government investigations or lawsuits by whistleblowers who bring a “qui tam” action under the FCA on behalf of themselves and the government for a variety of alleged improper promotional and marketing activities, including providing free product to customers expecting that the customers would bill the federal programs for the product and inflating prices reported to private price publication services, which are used to set drug reimbursement rates under government healthcare programs. In addition, the government and private whistleblowers have pursued FCA cases against pharmaceutical companies for causing false claims to be submitted as a result of the promotion and marketing of their products for unapproved uses or violations of the federal Anti-Kickback Statute, such as providing consulting fees, grants, free travel and other benefits to physicians to induce them to prescribe the company’s products and providing assistance to patients with their insurance or co-insurance obligations and providing donations to third-party charities that provide patients with such assistance. In October 2025, Endo received a Civil Investigative Demand (“CID”) from the DOJ under the FCA seeking documents and information from January 2020 through the present. The CID concerns allegations that (1) Endo violated the FCA by paying kickbacks to induce the purchase of Xiaflex, in violation of the Anti-Kickback Statute and (2) Endo inflated reimbursement rates for Xiaflex by excluding applicable price
30
concessions from average sales price reports submitted to the CMS. Endo is cooperating with the investigation and is in the process of responding to the CID. The Company cannot predict the eventual scope, duration or outcome of this matter at this time. We have in the past been, and may in the future become, the subject of an FCA or other government investigation or whistleblower suit and we may incur substantial legal costs (including settlement costs) and business disruption responding to any such investigation or suit, regardless of the outcome.
If we are deemed to have failed to comply with any applicable laws, regulations or government guidance, we could be subject to additional criminal and/or civil sanctions, including significant fines, damages, civil monetary penalties and exclusion from participation in government healthcare programs, including Medicare and Medicaid, actions against executives overseeing our business, consent decrees and corporate integrity agreements pursuant to which our activities would be subject to ongoing scrutiny and monitoring to ensure compliance with applicable laws and regulations and/or burdensome remediation measures. Any such fines, awards, other sanctions or required remediation could have an adverse effect on our competitive position, business, financial condition, results of operations and cash flows. See Item 3. Legal Proceedings included within this Annual Report for additional information regarding various legal proceedings and claims.
We have various contractual and court-ordered compliance obligations that, if violated could result in monetary, injunctive or other penalties.
We have various contractual and court-ordered compliance obligations, including pursuant to the CIA and the Endo VOI, each of which are discussed in greater detail in Item 1. Business included within this Annual Report. The CIA, which was entered into with the OIG within the HHS in March 2022, has a five-year term and requires, among other things, enhancements to our compliance program, fulfillment of self-reporting, monitoring and training obligations, management certifications and resolutions from our Board of Directors and the retention of an independent review organization to conduct annual reviews of certain systems and transactions related to our government pricing and patient assistance activities. The Endo VOI prevents the relevant subsidiaries of Endo from manufacturing high-dose opioid pills, advertising or marketing opioids to patients and doctors, offering compensation incentives based on opioid sales and engaging in opioid-related lobbying, among other restrictions.
In addition, on November 30, 2023, we reached an agreement with the SEC to resolve the SEC staff’s previously disclosed investigation into certain of our disclosures. As part of the agreement, we consented to the entry of an SEC order (“SEC Order”) that, among other things, required us to retain a compliance consultant to review our disclosure controls and procedures relating to collection and assessment of information concerning potential risks, contingencies, trends and uncertainties, and the implementation and sufficiency of our internal accounting controls related to generally accepted accounting principles in the U.S. (“GAAP”) ASC 450. Under the terms of the SEC Order, we have adopted and implemented the recommendations of the compliance consultant.
Compliance with our contractual and court-ordered obligations requires the expenditure of significant resources and management time. Further, the failure to comply with any of our obligations may result in adverse action by courts, one or more state Attorneys General, the SEC or other enforcement authorities; monetary, injunctive or other penalties; exclusion from participation in federal healthcare programs, including Medicare; increased legal fees and costs; negative publicity; and/or an increased risk of future lawsuits or other actions by third parties.
We face significant competition and may not be able to compete effectively.
The industries in which we operate are highly competitive. Competition takes many forms, such as price reductions on products that are comparable to our own, development of new or improved products, processes or technologies that make our products or proposed products less competitive or obsolete, acquisition or in-licensing of new products that may be more cost-effective than, or have performance superior to, our products, the introduction of generic versions when our proprietary products lose their patent protection or market exclusivity and technologies that are similar to our devices but may operate either more effectively or less expensively. This competition may limit the effectiveness of any price increases we initiate. Following any price increase by us, competitors may elect to maintain a lower price point that may result in a decline in our sales volume. Our failure to compete effectively could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
In addition, manufacturers of generic pharmaceuticals typically invest far less in R&D than research-based pharmaceutical companies, allowing generic versions to typically be significantly less expensive than the related branded products. The generic form of a drug may also enjoy a preferred position relative to the branded version under third-party reimbursement programs, or be routinely dispensed in substitution for the branded form by pharmacies. To successfully compete for the business, we must often demonstrate that our branded products offer medical benefits and cost advantages as compared with generic versions or other forms of care. If competitors introduce new products, delivery systems or processes with therapeutic or cost advantages, our products can be subject to progressive price reductions, decreased sales volume or both.
For further discussion on the competitive nature of our business, as well as the intellectual property rights and market exclusivity that play a key role in our business, refer to Item 1. Business included within this Annual Report. Our failure to compete effectively could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
31
We experience pricing pressure on certain of our products due to legal changes or changes in insurers' reimbursement practices, including as a result of increased public scrutiny of healthcare and pharmaceutical costs, which could reduce our future revenue and profitability.
Public and governmental scrutiny of the cost of healthcare generally, and pharmaceuticals in particular, especially in connection with price increases of certain products, have affected and are expected to continue to affect our ability to maintain or increase the prices of one or more of our products, which could negatively impact our future revenue and profitability. For instance, press reports and other commentary have criticized the increases in the price of Acthar Gel over time, including related to the period prior to our acquisition of the product. In addition, federal prosecutors and state attorneys general continue to investigate and file legal proceedings challenging pricing increases and practices, and the U.S. Congress continues to investigate pharmaceutical costs and pricing practices. We cannot predict whether any particular legislative or regulatory changes or changes in insurers' reimbursement practices may result from any such public scrutiny, what the nature of any such changes might be or what impact they may have on us. If legislative or regulatory action were taken or insurers changed their reimbursement practices in a manner that limits our ability to maintain or increase the prices of our products, our financial condition, results of operations and cash flows could be negatively affected.
Sales of our products are affected by, and we may be negatively impacted by any changes to, the reimbursement practices of governmental health administration authorities, private health coverage insurers and other third-party payers. In addition, reimbursement criteria, policies and practices outside the U.S. could reduce prices for our products or reduce our market opportunities.
Sales of our products depend, in part, on the extent to which the costs of our products are reimbursed by governmental health administration authorities, private health coverage insurers and other third-party payers. The ability of patients to obtain appropriate reimbursement for products and services from these third-party payers affects the selection of products they purchase and the prices they are willing to pay. In the U.S., there have been, and we expect there will continue to be, a number of state and federal proposals that limit the amount that third-party payers may pay to reimburse the cost of drugs, including with respect to Acthar Gel. We believe the increasing emphasis on managed care in the U.S. has and will continue to put pressure on the usage and reimbursement of Acthar Gel. Our ability to commercialize our products depends, in part, on the extent to which reimbursement for the costs of these products is available from government healthcare programs, such as Medicaid and Medicare, private health insurers and others. We cannot be certain that, over time, third-party reimbursements for our products will be adequate for us to maintain price levels sufficient for realization of an appropriate return on our investment. Furthermore, demand for new products may be limited unless we obtain reimbursement approval from governmental and private third-party payers prior to introduction. Reimbursement criteria, which vary by country, are becoming increasingly stringent and require management expertise and significant attention to obtain and maintain qualification for reimbursement.
With regard to private payers, reimbursement of highly-specialized products, such as Acthar Gel, is typically reviewed and approved or denied on a patient-by-patient, case-by-case basis, after careful review of details regarding a patient's health and treatment history that is provided to the insurance carriers through a prior authorization submission and appeal submission, if applicable. During this case-by-case review, the reviewer may refer to coverage guidelines issued by that carrier. These coverage guidelines are subject to ongoing review by insurance carriers. Because of the large number of insurance carriers, there is a large number of guideline updates issued each year.
We also anticipate that the U.S. Congress, state legislatures and federal and state regulators may adopt or accelerate adoption of new healthcare policies and reforms intended to regulate drug pricing or the way in which such prices are made available on the market. This includes efforts by individual states in the United States to pass legislation and implement regulations designed to control pharmaceutical and biological product pricing, such as by passing laws that regulate how manufacturers make the 340B ceiling price available on the market and/or by establishing Prescription Drug Affordability Boards (or similar entities) which may review high-cost drugs, set upper payment limits and implement marketing cost disclosure and transparency measures.
In addition, a number of markets outside the U.S. in which we operate may implement policies that limit price increases or reimbursement for our products.
We are unable to predict what additional legislation or regulation or changes in third-party coverage and reimbursement policies may be enacted or issued in the future or what effect such legislation, regulation and policy changes would have on our business.
Our reporting and payment obligations under the Medicare and Medicaid rebate programs, and other governmental purchasing and rebate programs, are complex. Any determination of failure to comply with these obligations or those relating to healthcare fraud and abuse laws could have a material adverse effect on our business.
The regulations regarding reporting and payment obligations with respect to Medicare and Medicaid reimbursement programs, and rebates and other governmental programs, are complex. Because our processes for these calculations and the judgments used in making these calculations involve subjective decisions and complex methodologies, these accruals may have a higher inherent risk for material changes in estimates. In addition, they are subject to review and challenge by the applicable governmental agencies, and it is possible that such reviews could result in material adjustments to amounts previously paid. See the risk factor captioned “Sales of our products are affected by, and we may be negatively impacted by any changes to, the reimbursement practices of governmental health
32
administration authorities, private health coverage insurers and other third-party payers. In addition, reimbursement criteria, policies or practices outside the U.S. could reduce prices for our products or reduce our market opportunities.” for additional information.
Pricing and rebate calculations vary among products and programs. The calculations are complex and are often subject to interpretation by us, governmental or regulatory agencies and the courts. If a manufacturer becomes aware that its Medicaid reporting for a prior period was incorrect, or has changed as a result of recalculation of the pricing data, the manufacturer is obligated to resubmit the corrected data. Such restatements and recalculations could increase our costs for complying with the laws and regulations governing the Medicaid Drug Rebate Program. Any corrections to our rebate calculations could result in an overage or underage in our rebate liability for past quarters, depending on the nature of the correction. Price recalculations also may affect the ceiling price at which we are required to offer its products to covered entities under the 340B program, and may require us to issue refunds to 340B program-covered entities, which can be costly and burdensome. Restatements may also impact a manufacturer’s liability with respect to the Part B and Part D inflation rebates, passed as part of the Inflation Reduction Act.
Civil monetary penalties can be applied if a manufacturer is found to have made a misrepresentation in the reporting of its average sales price for each misrepresentation and for each day in which the misrepresentation was applied, or if the manufacturer is found to have charged 340B program-covered entities more than the statutorily mandated ceiling price. In addition to retroactive rebates and the potential for 340B program refunds, if a manufacturer is found to have knowingly submitted false average manufacturer prices or best price information to the government, or to have misrepresented that information, the manufacturer may be liable for significant civil monetary penalties per item of false information. A manufacturer’s failure to submit monthly/quarterly average manufacturer price and best price data on a timely basis could result in a significant civil monetary penalty per day for each day the information is late beyond the due date. Such failures also could be grounds for CMS to terminate the manufacturer’s Medicaid drug rebate agreement, pursuant to which it participates in the Medicaid program, or, if the manufacturer fails to comply with 340B program requirements, the HRSA could decide to terminate its 340B program participation agreement. In the event that CMS terminates a manufacturer’s rebate agreement or HRSA terminates its 340B program participation agreement, no federal payments would be available under Medicaid or Medicare Part B for the manufacturer’s covered outpatient drugs. Finally, manufacturers that fail to offer discounts under the Medicare Part D manufacturer discount program or that fail to comply with Part B and Part D inflation rebate program requirements may be liable for additional civil monetary penalties.
CMS periodically issues updates to its regulations under the Medicaid Drug Rebate Program that could impact our price reporting obligations in various ways. Failure to comply with these price reporting requirements could negatively impact our financial results. Regulatory and legislative changes, and judicial rulings relating to the Medicaid Drug Rebate Program and related policies (including coverage expansion), have increased and will continue to increase our costs and the complexity of compliance, have been and will continue to be time-consuming to implement, and could have a material adverse effect on our results of operations, particularly if CMS or another agency challenges the approach we take in our implementation.
CMS and the OIG within HHS have pursued manufacturers that were alleged to have failed to report these data to the government in a timely manner. Governmental agencies may also make changes in program interpretations, requirements or conditions of participation, some of which may have implications for amounts previously estimated or paid. Manufacturers cannot guarantee that a submission will not be found by CMS to be incomplete or incorrect.
As discussed within Item 1. Business - Regulatory Matters, the Inflation Reduction Act established and altered a number of schemes and programs. Manufacturers may be subject to civil monetary penalties for certain violations of the negotiation and inflation rebate provisions and an excise tax during a noncompliance period under the negotiation program. Drug manufacturers may be subject to civil monetary penalties with respect to their compliance with the new Part D manufacturer discount program. Manufacturers thus could be subject to additional liability with respect to these programs as well.
As discussed within Item 1. Business - Regulatory Matters, we are required to participate in the FSS pricing program in order to maintain eligibility to have our products paid for with certain federal funds. Pursuant to applicable law, knowing provision of false information in connection with a non-FAMP in connection with the FSS program filing can subject a manufacturer to significant civil monetary penalties for each item of false information. The FSS contract also contains extensive disclosure and certification requirements. In addition, if we overcharge the government in connection with the FSS contract or Tricare Retail Pharmacy Rebate Program, whether due to a misstated FCP or otherwise, we will be required to refund the difference to the government. Failure to make necessary disclosures and/or to identify contract overcharges can result in allegations against us under the FCA and other laws and regulations. Unexpected refunds to the government, and any response to government investigation or enforcement action, would be expensive and time-consuming, and could have a material adverse effect on our business, financial condition, results of operations and growth prospects.
Any governmental body or agency that has commenced, or may commence, an investigation or other claim or action of or against us relating to the sales, marketing, pricing, quality or manufacturing of our products could seek to impose, based on a claim of violation of fraud and false claims laws or otherwise, civil and/or criminal sanctions, including fines, penalties and possible exclusion from federal healthcare programs including Medicare and Medicaid. Some of the applicable laws may impose liability even in the absence of specific intent to defraud. Furthermore, should there be ambiguity with regard to how to properly calculate and report payments, and even in the absence of any such ambiguity, a governmental authority may take a position contrary to a position we have
33
taken, and may impose civil and/or criminal sanctions. For example, in May 2019, CMS issued a final decision directing the Company to revert to the original base date average manufacturer price (“AMP”) used to calculate Medicaid drug rebates for Acthar Gel despite having granted Questcor Pharmaceuticals, Inc. (“Questcor”) written authorizations to reset the base date AMP in 2012. In addition, from time to time, state attorneys general have brought cases against us that allege generally that we and numerous other pharmaceutical companies reported false pricing information, including in connection with certain drugs that are reimbursable under Medicaid, resulting in overpayment by state Medicaid programs for those drugs, and generally seek monetary damages and attorneys' fees. Any such penalties or sanctions that we might become subject to in this or other actions could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
For any marketed drug products which are covered in the U.S. by federal or state healthcare programs, such as the Medicare and Medicaid programs, we have various obligations, including government price reporting and rebate requirements, which generally require medicines be offered at substantial rebates and/or discounts to the government and certain private purchasers including “covered entities” purchasing under the 340B Drug Discount Program. Some of these programs require submission of pricing data and calculation of discounts and rebates pursuant to complex statutory formulas, as well as entry into government procurement contracts governed by the Federal Acquisition Regulations, and the guidance governing such requirements is not always clear or precise. Compliance with such requirements can require significant investment in personnel, systems and resources, but failure to properly calculate our prices, or offer required discounts or rebates, could subject us to substantial penalties. One component of the rebate and discount calculations under the Medicaid and 340B programs, respectively, is the “additional rebate,” a complex calculation which is based, in part, on the extent that a branded drug's price increases over time more than the rate of inflation (based on the Consumer Price Index for All Urban Consumers). Because, effective January 1, 2024, the Medicaid rebate amount is no longer capped at 100% of a drug's “average manufacturer price,” this “additional rebate” calculation can result in an increase in Medicaid rebate liability beyond such price. In addition, this “additional rebate” calculation can result in a 340B ceiling price of one penny when such price calculates to less than $0.01. With respect to Acthar Gel, the “additional rebate” scheme, as applied to the historical pricing of Acthar Gel both before and after we acquired the medicine, has resulted in a 340B ceiling price of one penny, which has negatively impacted and is expected to continue to negatively impact our net sales of Acthar Gel. See the risk factor captioned “We have implemented changes to our Acthar Gel patient assistance program, which may receive additional review from governmental regulators and, if challenged, could have a material adverse effect on future net sales of Acthar Gel.” for more information.
Cost-containment efforts of our customers, purchasing groups, third-party payers and governmental organizations could materially adversely affect our business.
In an effort to reduce costs, many existing and potential customers for our products within the U.S. are members of GPOs and integrated delivery networks (“IDNs”). GPOs and IDNs negotiate pricing arrangements with healthcare product manufacturers and distributors and offer the negotiated prices to affiliated hospitals and other members. GPOs and IDNs typically award contracts on a category-by-category basis through a competitive bidding process. Bids are generally solicited from multiple manufacturers with the intention of driving down pricing. Due to the highly competitive nature of the GPO and IDN contracting processes, there is no assurance that we will be able to obtain or maintain contracts with major GPOs and IDNs across our product portfolio. Furthermore, the increasing leverage of organized buying groups may reduce market prices for our products, thereby reducing our profitability. While having a contract with a GPO or IDN for a given product can facilitate sales to members of that GPO or IDN, having a contract is no assurance that sales volume of those products will be maintained. GPOs and IDNs increasingly are awarding contracts to multiple suppliers for the same product category. Even when we are the sole contracted supplier of a GPO or IDN for a certain product, members of the GPO or IDN generally are free to purchase from other suppliers. Furthermore, GPO and IDN contracts typically are terminable without cause upon 60 to 90 days prior notice. Accordingly, our net sales and results of operations may be negatively affected by the loss of a contract with a GPO or IDN. In addition, although we have contracts with many major GPOs and IDNs, the members of such groups may choose to purchase from our competitors, which could result in a decline in our net sales. Distributors of our products have and may continue to form strategic alliances and negotiate terms of sale more aggressively in an effort to increase their profitability. Failure to negotiate distribution arrangements having advantageous pricing and other terms of sale could cause us to lose market share to our competitors or result in lower pricing on volume we retain, both of which could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows. Outside the U.S., we have experienced pricing pressure due to the concentration of purchasing power in centralized governmental healthcare authorities and increased efforts by such authorities to lower healthcare costs. We frequently are required to engage in competitive bidding for the sale of our products to governmental purchasing agents. Our failure to maintain volume and pricing with historical or anticipated levels could materially adversely affect our business, financial condition, results of operations and cash flows.
Extensive laws and regulations govern the industry in which we operate and any failure to comply with such laws and regulations, including any changes to those laws and regulations, may materially adversely affect us.
The testing, development, manufacture, quality control, safety, effectiveness, approval, labeling, storage, record-keeping, reporting, import, export, marketing, sale, promotion and distribution of our products are subject to comprehensive government regulations that govern and influence the design, development, testing, manufacturing, processing, packaging, holding, record keeping, safety, efficacy, approval, advertising, promotion, sale, distribution and import/export of our products.
34
Under these laws and regulations, we are subject to periodic inspection of our facilities, procedures and operations and/or the testing of our products by the FDA, the DEA and similar authorities within and outside the U.S., which conduct periodic inspections to confirm that we are in compliance with all applicable requirements. We are also required to monitor, track and report adverse events and product quality problems associated with our products to the FDA and other regulatory authorities. Failure to comply with the requirements of the FDA or other regulatory authorities, including a failed inspection, a failure to comply with product quality reporting requirements, such as submission of field alert reports, or a failure in our adverse event reporting system or any other unexpected or serious health or safety concerns associated with our products, including our opioid pain products and Acthar Gel, could result in adverse inspection reports, warning letters, product recalls or seizures, product liability claims, labeling changes, monetary sanctions, injunctions to halt the manufacture and distribution of products, civil or criminal sanctions, refusal of a government to grant approvals or licenses, restrictions on operations or withdrawal of existing approvals and licenses. In addition, the requirements of regulatory authorities, including interpretative guidance, are subject to change and compliance with additional or changing requirements or interpretative guidance may subject us to further review, result in product delays or otherwise increase our costs, and thus have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
Furthermore, the FDA and various foreign regulatory authorities approve or clear drugs and medical devices for the treatment of specific indications, and products may be promoted or marketed only for the indications for which they have been approved or cleared. See Item 1. Business - Regulatory Matters for additional information. The laws and regulations relating to the promotion of products for unapproved uses are complex and subject to substantial interpretation by the FDA and other governmental agencies. Promotion of a product for unapproved use is prohibited; however, certain activities related to unapproved uses that we and others in the pharmaceutical industry engage in are permitted by the FDA. We have compliance programs in place, including policies, training and various forms of monitoring, designed to address these risks. Nonetheless, these programs and policies may not always protect us from conduct by individual employees that violate these laws. If the FDA or any other governmental agency initiates an enforcement action against us and it is determined that we violated prohibitions relating to the promotion of products for unapproved uses in connection with past or future activities, we could be subject to substantial civil or criminal fines or damage awards and other sanctions such as consent decrees and corporate integrity agreements pursuant to which our activities would be subject to ongoing scrutiny and monitoring to ensure compliance with applicable laws and regulations. See Item 1. Business included within this Annual Report for additional information regarding the CIA related to our settlement with governmental entities regarding Acthar Gel. Any such fines, awards or other sanctions could have an adverse effect on our business, financial condition, results of operations and cash flows.
In addition, we face significant risks relating to the implementation of the FDA’s new QMSR, which became effective on February 2, 2026. The transition to the QMSR may require changes to our internal procedures, quality documentation architecture, electronic quality management systems and training programs. These changes may increase our compliance costs, divert management and operational resources and create delays or inefficiencies during implementation. Moreover, because FDA inspections under the QMSR will evaluate the implementation and effectiveness of risk‑based processes, deficiencies in risk management documentation, supplier controls, complaint handling or design and development records may be subject to heightened scrutiny. Failure to complete this transition effectively, or to remediate gaps identified during internal assessments or FDA inspections, could result in adverse regulatory findings, including Form 483 observations, warning letters, or other enforcement actions, any of which could disrupt manufacturing or product distribution, necessitate costly corrective actions, or delay approvals of pending submissions.
Our approved products and investigational products, if successfully developed and approved, may cause undesirable side effects that limit their commercial profile, delay or prevent further development or regulatory approval; cause regulatory authorities to require labeling statements, such as boxed warnings or a REMS, or result in other negative consequences.
We may observe undesirable side effects or other potential safety issues in nonclinical studies, in clinical trials at any stage of development of our product candidates, as part of an expanded access program or in commercial use or post-approval studies of any approved product. Clinical trials by their nature utilize a sample of the potential patient population. With a limited number of patients and limited duration of exposure, certain side effects of our product candidates, if successfully developed and approved, may be uncovered only with a larger number of patients exposed to the product. Those side effects could be serious or life-threatening. If we or others identify undesirable side effects caused by our products:
•regulatory authorities may withdraw or limit their approval of such products;
•the FDA or regulatory authorities outside the U.S. may impose a clinical hold or partial clinical hold prior to the initiation of development or during development of our product candidates which could cause us or our collaborators to have to stop, delay or restrict further development; or we or our collaborators may, even without a clinical hold, decide to interrupt, delay or halt existing non-clinical studies and clinical trials or stop development;
•we may have difficulty enrolling patients in our clinical trials and completing such trials on the timelines we expect or at all, or we may have to conduct additional non-clinical studies or clinical trials as part of a development program;
•we ultimately may not be able to demonstrate, to the satisfaction of the FDA or other regulatory authorities, that our product candidates are safe and/or that the benefits outweigh the safety risks, and the FDA or applicable foreign regulatory authorities may not approve the product candidate;
35
•regulatory authorities may require the addition of labeling statements, such as a boxed warning or additions to an existing boxed warning, or a contraindication, including as a result of inclusion in a class of drugs for a particular disease, or may require a REMS, or modifications to an existing REMS;
•we may be required to change the way such products are distributed or administered, conduct post-approval studies or change the labeling of the products;
•we may be subject to regulatory investigations and government enforcement actions;
•we may decide to remove such products from the marketplace;
•we could be sued and held liable for injury caused to individuals exposed to or taking our products or product candidates; and
•our reputation may suffer.
We believe that any of these events could prevent us from achieving or maintaining market acceptance of the affected products, could substantially increase the risks and costs of developing our product candidates or commercializing our approved products, and could significantly adversely impact our ability to successfully develop products, gain regulatory approval for product candidates and commercialize our approved products, which could significantly adversely impact our ability to generate revenues.
We have limited resources, and may not be successful in our efforts to acquire additional products or product candidates at the rate we expect. In addition, we may expend our limited resources to pursue a particular product candidate or indication and fail to capitalize on other product candidates or indications that may be more profitable or for which there is a greater likelihood of success.
The long-term success and growth of our business depends upon our ability to successfully develop, gain approval of and commercialize our products and on our ability to acquire compounds for development and commercialization in the future and to successfully pursue clinical development of such new compounds. Our business development efforts may fail to identify new compounds that meet our standards for development and commercialization, and, even if we are successful in acquiring such compounds, we may not be able to produce the data necessary to support a regulatory approval.
Because we have limited financial and management resources, we focus on a limited number of commercial and R&D programs. As a result, we may forego or delay pursuit of opportunities with other products or product candidates that later prove to have greater commercial potential. We may focus our efforts and resources on potential programs or product candidates that ultimately prove to be unsuccessful and may not yield any commercially viable products. Our resource allocation decisions may cause us to fail to capitalize on other viable opportunities. If we do not accurately evaluate the commercial potential or target market for a particular product candidate, we may relinquish valuable rights through future collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain such sole development and commercialization rights. If any of these events occur, it may have a material adverse effect on our business.
We may be unable to successfully develop, commercialize or launch new products or expand commercial opportunities for existing products or adapt to a changing technology and, as a result, our business may suffer.
Our future results of operations will depend, to a significant extent, upon our ability to successfully develop, commercialize and launch new products or expand commercial opportunities for existing products in a timely manner. There are numerous difficulties in developing, commercializing and launching new products or expanding commercial opportunities for existing products, including:
•developing, testing and manufacturing products in compliance with regulatory and quality standards in a timely manner;
•our ability to successfully engage with the FDA or other regulatory authorities as part of the development and approval or clearance process and to receive requisite regulatory approvals or clearances for such products in a timely manner, or at all;
•agreement on acceptable terms with prospective clinical research organizations (“CROs”) and clinical trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among CROs and trial sites;
•the availability, on commercially reasonable terms, of raw materials or components, including API and other key ingredients for our products;
•delay or failure in obtaining IRB approval or the approval of other reviewing entities, including comparable foreign regulatory authorities, to conduct a clinical trial at each site;
•withdrawal of clinical trial sites from our clinical trials as a result of changing standards of care or the ineligibility of a site to participate in our clinical trials;
•delay or failure in recruiting and enrolling suitable trial patients to participate in a trial;
•clinical sites and investigators deviating from a trial protocol, failing to conduct a trial in accordance with regulatory requirements, or dropping out of a trial;
36
•inability to identify and maintain a sufficient number of trial sites, many of which may already be engaged in other clinical trial programs, including some that may be for competing product candidates with the same indication;
•failure of our third-party clinical trial sites to satisfy their contractual duties or meet expected deadlines;
•ambiguous or negative interim results or results that are inconsistent with earlier results;
•feedback from the FDA or a comparable regulatory authority outside the U.S., IRBs or data safety monitoring boards or results from earlier stage or concurrent preclinical studies and clinical trials, that might require modification to the protocol for the trial;
•decision by the FDA or a comparable regulatory authority outside the U.S., an IRB or us or a recommendation by a data safety monitoring board to suspend or terminate clinical trials at any time for safety issues or for any other reason;
•unacceptable risk-benefit profile, unforeseen safety issues or adverse side effects or adverse reactions associated with a product candidate;
•failure of a product candidate to demonstrate any or enough of a benefit;
•difficulties in manufacturing, or obtaining from third parties, sufficient quantities of a product candidate for use in clinical trials or commercial use that meet internal and regulatory standards;
•lack of adequate funding to continue the clinical trial, including the incurrence of unforeseen costs due to enrollment delays, requirements to conduct additional clinical trials or increased expenses associated with the services of our CROs and other third parties;
•developing, commercializing and launching a new product is time-consuming, costly and subject to numerous factors, including legal actions brought by our competitors, that may delay or prevent the development, commercialization and/or launch of new products;
•multiple product launches in a short period of time may be challenging, particularly for an organization that has had a limited number of launches of new products in many years, and may result in strained resources that could lead to launch delays and cost;
•other unanticipated costs;
•payment of prescription drug or medical device user fees to the FDA to defray the costs of review and approval of marketing applications for branded drugs or devices;
•experiencing delays as a result of limited resources at the FDA or other regulatory authorities;
•changing review and approval policies and standards at the FDA or other regulatory authorities;
•changing standards of care;
•achieving timely, cost-effective and effective execution of the product launches, challenges to which can include having sufficient quantities of product, appropriate marketing materials and resources and a trained sales force;
•identifying appropriate partners for distribution of our products, including any future over-the-counter commercialization opportunities, and negotiating contractual arrangements in a timely manner with commercially reasonable terms; and
•changes in governmental regulations or administrative actions.
As a result of these and other difficulties, products currently in development by us may or may not receive timely regulatory approvals, or approvals at all. This risk is heightened with respect to the development of proprietary branded products due to the uncertainties associated with the results of clinical trials, the costs and length of time associated with R&D of such products and the uncertainty of market acceptance. Moreover, the FDA regulates the facilities, processes and procedures used to manufacture and market pharmaceutical products and medical devices in the U.S. Manufacturing facilities must be registered with the FDA and all products made in such facilities must be manufactured in accordance with quality (cGMP and/or QMSR) regulations enforced by the FDA. Compliance with quality regulations requires significant expenditures and the dedication of substantial resources. Prior to approval of any product, the FDA typically inspects both our facilities and procedures and the manufacturing facilities for our products to ensure compliance with regulatory standards, and those inspections are also conducted periodically after a product is approved for marketing. The FDA may also cause a suspension or withdrawal of product approvals if regulatory standards are not maintained. In the event an approved manufacturing facility for a particular drug or device is required by the FDA to curtail or cease operations, or otherwise becomes inoperable, obtaining the required FDA authorization to manufacture at the same or a different manufacturing site could result in production delays, which could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows. If we or the manufacturing facilities for our products fail to comply with applicable regulatory requirements, a regulatory authority may, among other things:
•issue warning letters or untitled letters;
37
•mandate modifications to promotional materials or require us to provide corrective information to healthcare practitioners;
•require us to enter into a consent decree, which can include imposition of various fines, reimbursements for inspection costs, required due dates for specific actions and penalties for noncompliance;
•seek an injunction or impose civil or criminal penalties or monetary fines;
•suspend or withdraw regulatory approval;
•suspend any ongoing clinical trials;
•refuse to approve or clear pending applications or supplements to applications filed by us;
•suspend or impose restrictions on operations, including costly new manufacturing requirements; or
•seize or detain products, refuse to permit the import or export of products or require us to initiate a product recall.
The occurrence of any event or penalty described above may inhibit or preclude our ability to commercialize our products and generate revenue.
Advertising and promotion of our products is heavily scrutinized by, among others, the FDA, the DOJ, the OIG within the HHS, state Attorneys General, the U.S. Congress and the public. Failure to comply with applicable FDA requirements and restrictions in this area may subject a company to adverse publicity and enforcement action, including enforcement letters, inquiries and investigations and civil and criminal sanctions by the FDA or other government agencies.
Furthermore, the market perception and reputation of us and our products are important to our business and the continued market acceptance of our products. Any negative press reports or other commentary about us or our products, whether accurate or not, could have a material adverse effect on our business, reputation, financial condition, results of operations or cash flows or could cause the market value of our ordinary shares and/or debt securities to decline.
If any of our drug or device applications are not approved or cleared timely, or if we are unable to obtain and realize the full benefits of the respective market exclusivity period for our products, or if our products cannot be successfully manufactured or commercialized timely, our results of operations could be materially adversely affected. In addition, we cannot guarantee that any investment we make in developing products will be recouped, even if we are successful in commercializing those products. Finally, once developed and approved or cleared, new products may fail to achieve commercial acceptance due to various factors, including the price of the product, third-party reimbursement of the product, and the ineffectiveness of sales, marketing and distribution efforts to support the product.
We may not achieve the anticipated benefits of price increases for our products, which may adversely affect our business.
From time to time, we may initiate price increases on certain of our products. There is no guarantee that our customers will be receptive to these price increases and continue to purchase the products at historical quantities. In addition, it is unclear how market participants will react to price increases, particularly in light of the scrutiny being paid to drug pricing in the U.S. If customers do not maintain or increase existing sales volumes, we may be unable to replace lost sales with orders from other customers, and it could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
Our customer concentration may materially adversely affect our business.
We sell a significant amount of our products to a limited number of independent wholesale distributors, large pharmacy chains and specialty distributors. In turn, these wholesale distributors, large pharmacy chains and specialty distributors supply products to pharmacies, hospitals, governmental agencies and physicians. As further discussed in Item 1. Business included within this Annual Report, sales to some of our distributors that supply our products to many end user customers accounted for 10.0% or more of our total net sales. If we were to lose the business of this distributor, or if this distributor failed to fulfill its obligations, experienced difficulty in paying us on a timely basis or negotiated lower pricing terms, it could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
Our product concentration may materially adversely affect our business.
We sell a wide variety of branded pharmaceutical products as well as some related devices. However, a small number of relatively significant products, most notably Acthar Gel, Xiaflex and INOmax, represent a significant percentage of our net sales. Our ability to maintain and increase net sales from these products depends on several factors, including:
•our ability to continue to maintain or increase market demand through our own marketing and support of our sales force;
•our ability to successfully communicate the benefits of our products;
•our ability to implement and maintain pricing;
•our ability to achieve hospital and other third-party payer formulary acceptance and maintain reimbursement levels by third-party payers;
38
•our ability to maintain confidentiality of the proprietary know-how and trade secrets relating to Acthar Gel;
•our ability to continue to procure raw materials or finished goods, as applicable, for Acthar Gel and INOmax from internal and third-party manufacturers in sufficient quantities and at acceptable quality and pricing levels in order to meet commercial demand;
•our ability to maintain fees and discounts payable to the wholesalers and distributors and GPOs, at commercially reasonable levels;
•whether the DOJ or other third parties seek to challenge and are successful in challenging patents or patent-related settlement agreements or our sales and marketing practices;
•warnings or limitations that may be required to be added to FDA-approved labeling; and
•the occurrence of adverse side effects related to or emergence of new information related to the therapeutic efficacy of these products, and any resulting product liability claims or product recalls.
Moreover, net sales of Acthar Gel may also be materially impacted by the decrease in the relatively small number of prescriptions written for Acthar Gel as compared to other products in our portfolio, given Acthar Gel's use in treating rare diseases. Any disruption in our ability to generate net sales from Acthar Gel could have an adverse impact on our business, financial condition, results of operations and cash flows.
We may be unable to protect our intellectual property rights, intellectual property rights may be limited or we may be subject to claims that we infringe on the intellectual property rights of others.
We rely on a combination of patents, trademarks, copyrights, trade secrets and proprietary information, including confidential business information, show-how and know-how, in addition to any market exclusivity gained from the regulatory approval process to support our business strategy. However, our efforts to protect our intellectual property rights may not be sufficient. If we do not obtain sufficient protection for our intellectual property, or if we are unable to effectively enforce our intellectual property rights, or if there is a change in the way courts and regulators interpret the laws, rules and regulations applicable to our intellectual property, our competitiveness could be impacted, which could adversely affect our competitive position, business, financial condition, results of operations and cash flows.
Our pending patent applications may not result in the issuance of patents or the patents issued to or licensed by us in the past or in the future may be challenged or circumvented by competitors. Existing patents may be found to be invalid or found not to cover our competitors’ products or the methods of using, making, or selling of our competitors’ products. Regulatory agencies may refuse to grant us the market exclusivity that we are anticipating, or may unexpectedly grant market exclusivity rights to other parties. In addition, our ability to obtain and enforce intellectual property rights is limited by the unique laws of each country. In some countries, it may be particularly difficult to adequately obtain or enforce intellectual property rights, which could make it easier for competitors to capture market share in such countries by utilizing technologies and product features that are similar or identical to those developed or licensed by us. Competitors also may harm our sales by designing products that mirror the capabilities of our products or technology without infringing our patents, including by coupling separate technologies to replicate what our products accomplish through a single system. Competitors may diminish the value of our trade secrets by reverse engineering or through independent invention. Additionally, current or former employees, partners or other parties in similar positions of trust with us may improperly disclose or otherwise misappropriate such trade secrets or other confidential information to competitors or other third parties. We may not become aware of any such improper disclosure or use, and, in the event we do become aware, we may not have an adequate remedy available to us. We operate in an industry characterized by extensive patent litigation and proceedings, whether in the courts, the ITC or the USPTO, and we may from time to time be a party to such litigation or proceedings.
The pursuit of, or defense against, patent infringement litigation, as well as involvement in USPTO proceedings and other actions, are costly and time-consuming and we may not be able to reasonably anticipate or predict the outcomes of such litigation or proceedings for protracted periods of time. We may be unsuccessful in our efforts to enforce our patent or other intellectual property rights. In addition, patent litigation can result in significant damage awards, including the possibility of treble damages and injunctions. Additionally, we could be forced to stop manufacturing and selling certain products, or we may need to enter into license agreements that require us to make significant royalty or up-front payments in order to continue selling the affected products. Given the nature of our industry, we are likely to face additional claims of patent infringement in the future. A successful claim of patent or other intellectual property infringement against us could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
Specifically, we believe that the following risks could impact our existing product portfolio:
•Acthar Gel - The product has only one patent listed in the Orange Book, which is set to expire on February 25, 2041. There is a risk that a competitor may develop a generic or other competitive product that does not infringe the Orange Book listed patent. We also rely on trade secrets and proprietary know-how to protect the commercial viability and value of Acthar Gel. We currently obtain such protection, in part, through confidentiality and proprietary information agreements. These agreements may not provide meaningful protection or adequate remedies for proprietary technology in the event of
39
unauthorized use or disclosure of confidential and proprietary information. The parties may not comply with or may breach these agreements. Furthermore, our trade secrets may otherwise become known to, or be independently developed by, competitors.
•Xiaflex - We own or have licensed rights to patents and patent applications related to Xiaflex, including U.S. drug product and methods of manufacture patents and patent applications that expire between 2028 and 2038. A competitor may develop a biosimilar or other competitive collagenase product that does not infringe any of the patents we have in our portfolio. Xiaflex is also protected by trade secrets and proprietary know-how. These trade secrets and know-how may otherwise become known to, or be independently developed, by competitors.
•INOmax - The Company has numerous patents and patent applications in support of INOmax and related technologies. Certain patents relating to this product and related technologies have been challenged. A broad-scale launch of competitive nitric oxide products has taken place in the market which has adversely impacted our business, may continue to adversely affect our ability to successfully maximize the value of INOmax and could have an adverse effect on our competitive position, business, financial condition, results of operations and cash flows. In addition, as further discussed in Note 20 Commitments and Contingencies to the Notes to Consolidated Financial Statements, the Company initiated litigation against Airgas Therapeutics LLC (“Airgas”) with respect of certain Company patents. Although the Company obtained a favorable jury verdict in the District Court of Delaware finding that Airgas infringes the Company’s asserted patents, the Court’s ultimate remedies may not be adequate for the Company’s business needs.
•Supprelin LA - Supprelin LA has one Orange Book patent that expires in June 2026. Our inability to maintain market exclusivity could have an adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
•Terlivaz - FDA has granted Terlivaz New Chemical Entity Exclusivity, which expires on September 14, 2027, and Orphan Drug Exclusivity, which expires on September 14, 2029. A generic competitor can file an ANDA referencing our Terlivaz NDA starting September 14, 2026. Terlivaz has one patent listed in the Orange Book that is set to expire on April 5, 2037. In addition, we have additional patent applications pending with the U.S. Patent and Trademark Office. Our inability to maintain market exclusivity could have an adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
See Note 20 to the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for more information about our patent disputes.
Clinical trials demonstrating the efficacy of Acthar Gel are limited, which could cause physicians not to prescribe Acthar Gel, or payers not to reimburse the drug, which could negatively impact our business.
Our net sales of Acthar Gel, which comprise a significant portion of our overall product portfolio, could be negatively impacted by the level of clinical data available on the product. Acthar Gel was originally approved by the FDA in 1952, prior to the enactment of the 1962 Kefauver Harris Amendment, or the “Drug Efficacy Amendment,” to the FFDCA. This amendment introduced the requirement that drug manufacturers provide proof of the effectiveness (in addition to the previously required proof of safety) of their drugs in order to obtain FDA approval. As such, the FDA's original approval in 1952 was based on safety data as clinical trials evaluating efficacy were not then required. In the 1970s, the FDA reviewed the safety and efficacy of Acthar Gel during its approval of Acthar Gel for the treatment of acute exacerbations in multiple sclerosis and evaluated all other previous indications on the label through the DESI process. In this process, the medical and scientific merits of the label and each indication on the label were evaluated based on publications, information from sponsors and the judgment of the FDA. The label obtained after the DESI review and the addition of the multiple sclerosis indication is the Acthar Gel label that was used until the changes in 2010.
In 2010, in connection with its review of a supplemental NDA for use of Acthar Gel in treatment of infantile spasms (“IS”), the FDA again reviewed evidence of safety and efficacy of Acthar Gel, and added the IS indication to the label of approved indications while maintaining approval of Acthar Gel for treatment of acute exacerbations in multiple sclerosis and 17 other indications. In conjunction with its decision to retain these 19 indications on a modernized Acthar Gel label, the FDA eliminated approximately 30 other indications from the label. The FDA review included a medical and scientific review of Acthar Gel and each indication and an evaluation of available clinical and non-clinical literature as of the date of the review. The FDA did not require additional clinical trials for Acthar Gel.
Accordingly, evidence of efficacy is largely based on physicians’ clinical experience with Acthar Gel and does not include clinical trials except for the MS and IS indications. We conducted several Phase 4 clinical trials to supplement the non-clinical evidence supporting the use of Acthar Gel. The completion of future trials to provide further evidence on the efficacy of Acthar Gel in the treatment of its approved indications could take several years to complete and will require the expenditure of significant time and financial and management resources. Such trials may not result in data that supports the use of Acthar Gel to treat any of its approved indications. In addition, a trial to evaluate the use of Acthar Gel to treat indications not on the current Acthar Gel label may not provide a basis to pursue adding such indications to the current Acthar Gel label. Furthermore, even if prescribed by a physician, third-
40
party payers may implement restrictions on reimbursement of Acthar Gel due, in part, to the limited clinical data of efficacy, which may negatively impact our business, financial condition, results of operations and cash flows.
Clinical studies required for our product candidates and new indications of our marketed products are expensive and time-consuming, and their outcome is highly uncertain. If any such studies are delayed or yield unfavorable results, regulatory approval for our product candidates or new indications of our marketed products may be delayed or become unobtainable or, even if approved, physicians might not prescribe our marketed products.
We must conduct extensive testing of our product candidates and new indications of our marketed products before we can obtain regulatory approval to market and sell them. For example, INOmax is approved for sale in the U.S. only for the treatment of HRF associated with pulmonary hypertension in term and near-term infants. In order to market these products in the U.S. for any other indications, we will need to conduct appropriate clinical trials, obtain positive results from those trials and obtain regulatory approval for such proposed indications. Conducting such studies is a lengthy, time-consuming and expensive process and obtaining regulatory approval is uncertain. Even well-conducted studies of effective drugs will sometimes appear negative in either safety or efficacy results, or otherwise may not achieve approval. The regulatory review and approval process to obtain marketing approval for a new indication can take many years, often requires multiple clinical trials and requires the expenditure of substantial resources. This process can vary substantially based on the type, complexity, novelty and indication of the product candidate involved. Success in early clinical trials does not ensure that later clinical trials will be successful, and interim results of a clinical trial do not necessarily predict final results.
These tests and trials may not achieve favorable results for many reasons, including, among others, failure of the product candidate to demonstrate safety or efficacy, the development of serious or life-threatening adverse events (or side effects) caused by or connected with exposure to the product candidate (or prior or concurrent exposure to other products or product candidates), difficulty in enrolling and maintaining subjects in a clinical trial, lack of sufficient supplies of the product candidate or comparator drug, lack of sufficient funding to support a trial through its conclusion and the failure of clinical investigators, trial monitors, contractors, consultants or trial subjects to comply with the trial plan, protocol or applicable regulations related to GLPs or GCPs. A clinical trial may fail because it did not include and retain a sufficient number of patients to detect the endpoint being measured or reach statistical significance. A clinical trial may also fail because the dose(s) of the investigational drug included in the trial were either too low or too high to determine the optimal effect of the investigational drug in the disease setting. The FDA and other regulatory authorities have substantial discretion in the approval process and may refuse to accept any application or may decide that any data submitted is insufficient for approval and require additional studies or clinical trials or varying interpretations of the data obtained from pre-clinical and clinical testing could delay, limit or prevent regulatory approval of product candidate or a new indication for a product candidate. Even if a product is approved, physicians may not prescribe our products if they do not believe the relevant data are persuasive.
We will need to reevaluate any drug candidate that does not test favorably and either conduct new studies, which are expensive and time-consuming, or abandon that drug development program. The failure of clinical trials to demonstrate the safety and effectiveness of our product candidates for the desired indication(s) would preclude the successful development of those candidates for such indication(s), which would have a material adverse effect on our business, financial condition, results of operations and cash flows.
We may incur litigation liability, including but not limited to product liability losses.
We are or may become involved in various legal proceedings and government inquiries and investigations, including with respect to, but not limited to, patent infringement, product liability, personal injury, antitrust matters, securities class action lawsuits, disclosure matters, breach of contract, Medicare and Medicaid reimbursement claims, opioid related matters, promotional practices, compliance with laws relating to the manufacture and sale of products including controlled substances and any challenges to orders issued in our or Endo’s bankruptcy proceedings. Such proceedings, inquiries and investigations may involve claims for, or the possibility of, fines and penalties involving substantial amounts of money or other relief, including, but not limited to, civil or criminal fines and penalties, changes in business practices and exclusion from participation in various government healthcare-related programs. Some of our existing legal proceedings, inquiries and investigations and related matters are described in Note 20 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report. If existing or future legal proceedings, inquiries or investigations were to result in an adverse outcome, the impact could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows. Even if one or more of these matters do not result in a direct adverse outcome, they could lead to distraction of management, the incurrence of additional costs and damage to our reputation, among other potential results that could have a material adverse effect on our business.
With respect to product liability and clinical trial risks, in the ordinary course of business we are subject to liability claims and lawsuits, including potential class actions, alleging that our marketed products or products in development have caused, or could cause, serious adverse events or other injury. Side effects or adverse events known or reported to be associated with, or manufacturing defects in, the products sold by us could exacerbate a patient’s condition, or could result in serious injury or impairment or even death. This could result in product liability claims against us and/or recalls of one or more of our products. In many countries, including in E.U. member states, national laws provide for strict (no-fault) liability that applies even where damages are caused both by a defect in a product and by the act or omission of a third party. Any such claim brought against us, with or without merit, could be costly to
41
defend and could result in an increase in our insurance premiums. We believe our current coverage level is adequate to address our current risk exposure related to product liability claims and lawsuits. However, some claims, such as those brought against us related to our sale of opioids, might not be covered by our insurance policies. Moreover, where the claim is covered by our insurance, if our insurance coverage is inadequate, we would have to pay the amount of any settlement or judgment that is in excess of our policy limits. We may not be able to obtain insurance on terms acceptable to us or at all since insurance varies in cost and can be difficult to obtain. Our failure to maintain adequate insurance coverage or successfully defend against product liability claims could have a material adverse effect on our business, financial condition, results of operations and cash flows.
Our operations expose us to the risk of violations of applicable health, safety and environmental laws and regulations and related liabilities and litigation.
We are subject to numerous federal, state, local and non-U.S. environmental protection and health and safety laws and regulations governing, among other things:
•the generation, storage, use and transportation of hazardous materials;
•emissions or discharges of substances into the environment;
•investigation and remediation of hazardous substances or materials at various sites;
•chemical constituents in products and end-of-life disposal, mandatory recycling and take-back programs; and
•the health and safety of our employees.
We may not have been, or we may not at all times be, in full compliance with environmental and health and safety laws and regulations. In the event a regulatory authority concludes that we are not in full compliance with these laws, we could be fined, criminally charged or otherwise sanctioned. Environmental laws are becoming more stringent, including outside the U.S., resulting in increased costs and compliance burdens.
Certain environmental laws assess liability on current or previous owners of real property and current or previous owners or operators of facilities for the costs of investigation, removal or remediation of hazardous substances or materials at such properties or at properties at which parties have disposed of hazardous substances. Liability for investigative, removal and remediation costs under certain federal and state laws is retroactive, strict (i.e., can be imposed regardless of fault) and joint and several. In addition to cleanup actions brought by governmental authorities, private parties could bring personal injury or other claims due to the presence of, or exposure to, hazardous substances. We have, from time to time, received notification from the EPA and similar state environmental agencies that conditions at a number of sites where the disposal of hazardous substances has taken place requires investigation, cleanup and other possible remedial action. These agencies may require that we reimburse the government for its costs incurred at these sites or otherwise pay for the costs of investigation and cleanup of these sites, including by providing compensation for natural resource damage claims arising from such sites.
In the ordinary course of our business planning process, we take into account our known environmental matters as we plan for our future capital requirements and operating expenditures. The ultimate cost of site cleanup and timing of future cash outflows is difficult to predict, given the uncertainties regarding the extent of the required cleanup, the interpretation of applicable laws and regulations and alternative cleanup methods. Based upon information known to date, we believe our current capital and operating plans are adequate to address costs associated with the investigation, cleanup and potential remedial action for our known environmental matters.
While we have planned for future capital and operating expenditures to comply with environmental laws, our costs of complying with current or future environmental protection and health and safety laws and regulations, or our liabilities arising from past or future releases of, or exposures to, hazardous substances may exceed our estimates or could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows. We may also be subject to additional environmental claims for personal injury or cost recovery actions for remediation of facilities in the future based on our past, present or future business activities.
If our business development activities or other transactions are unsuccessful, it may adversely affect us.
Our business strategy focuses on developing, manufacturing, and commercializing branded therapeutics that address key areas of significant unmet need, including rheumatology, ophthalmology, nephrology, neurology, pulmonology, orthopedics, urology, and neonatal respiratory critical care. Consistent with this strategy, and at the direction of our Board of Directors, we continue to explore a variety of transactions intended to maximize shareholder value, including potential acquisitions, divestitures, financings and other strategic transactions. In connection with this process, we intend to exit from our remaining opioid business and are currently pursuing the divestiture of our Percocet business. The process to evaluate potential business development or other transactions may be complex, time-consuming and expensive. Once a potential opportunity is identified, we may not be able to conclude negotiations of a potential transaction on terms that are satisfactory to us, which could result in a significant diversion of management and other employee time, as well as substantial out-of-pocket costs. In addition, there are a number of risks and uncertainties relating to our ability to close a potential transaction.
42
If an acquisition or licensing transaction is consummated, there are further potential risks related to integration activities, including with regard to operations, personnel, technologies and products. If we are not able to successfully integrate our acquisitions in the expected time frame, we may not obtain the advantages and synergies that such acquisitions were intended to create, which may have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
In addition, we intend to continue to explore opportunities to enter into strategic collaborations with other parties, which may include other pharmaceutical companies, academic and research institutions, government agencies and other public and private research organizations. These third-party collaborators are often directly responsible for certain obligations under these types of arrangements, and we may not have the same level of decision-making capabilities for the prioritization and management of development-related activities as we would for our internal research and development activities. Failures by these partners to meet their contractual, regulatory or other obligations to us, or any disruption in the relationships with these partners, could have a material adverse effect on our pipeline and business. In addition, these collaborative relationships for research and development could extend for many years and may give rise to disputes regarding the relative rights, obligations and revenues of us versus our partners, including the ownership of intellectual property and associated rights and obligations. These could result in the loss of intellectual property rights or other intellectual property protections, delay the development and sale of potential products and lead to lengthy and expensive litigation or arbitration.
Furthermore, the due diligence that we conduct in conjunction with an acquisition or other strategic collaboration may not sufficiently discover risks and contingent liabilities associated with the other party and, consequently, we may consummate an acquisition or otherwise enter into a strategic collaboration for which the risks and contingent liabilities are greater than were projected. In addition, in connection with acquisitions or other strategic collaborations, we could experience disruption in our business, technology and information systems and our customers, licensors, suppliers and employees and may face difficulties in managing the expanded operations of a larger and more complex company. There is also a risk that key employees of companies that we acquire or key employees necessary to successfully commercialize technologies and products that we acquire or otherwise collaborate on may seek employment elsewhere, including with our competitors. Furthermore, there may be overlap between our products or customers and the companies which we acquire or enter into strategic collaborations with that may create conflicts in relationships or other commitments detrimental to the integrated businesses or impacted products. Additionally, the time between our expenditures to acquire new products, technologies or businesses and the subsequent generation of revenues from those acquired products, technologies or businesses, or the timing of revenue recognition related to licensing agreements and/or strategic collaborations, could cause fluctuations in our financial performance from period to period. Finally, if we are unable to successfully integrate products, technologies, businesses or personnel that we acquire, we could incur significant impairment charges or other adverse financial consequences. Many of these factors are outside of our control and any one of them could result in increased costs, decreases in the amount of expected revenues and diversion of management's time and energy, which could materially impact our business, financial condition, results of operations and cash flows.
Divestitures or proposed divestitures may involve the loss of revenue, and the market for the associated assets may dictate that we sell such assets for less than what we paid. In connection with divestitures, we could also reduce the benefit of shared costs across our enterprise, reduce diversification of our portfolio, as well as risk the departure of key employees. Divestitures could also lead to disruption in our business, technology and information systems and the possibility of divestitures could impact the relationships we have with our customers, licensors, suppliers and employees. In addition, in connection with any asset sales or divestitures, we may be required to provide certain representations, warranties and covenants to buyers. While we would seek to ensure the accuracy of such representations and warranties and fulfillment of any ongoing obligations, we may not be completely successful and consequently may be subject to claims by a purchaser of such assets.
If we are unable to attract and retain qualified personnel in key fields (including scientific, technical, manufacturing, regulatory, compliance and commercial), we may be unable to maintain or expand our business.
Because of the specialized scientific nature of our business, our ability to develop products and to compete with our current and future competitors will remain highly dependent, in large part, upon our ability to attract and retain qualified personnel in fields such as scientific, technical, manufacturing, regulatory, compliance and commercial. The loss of such personnel, or the failure to recruit additional personnel in fields that are important to our business, could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows. There is intense competition for qualified personnel in our industry, and we may not be able to continue to attract and retain the qualified personnel necessary for the development or operation of our business.
Our business depends on the continued effectiveness and availability of our information technology infrastructure, and failures of this infrastructure could harm our operations.
Significant disruptions to our information technology systems or other cybersecurity incidents affecting information security could adversely affect our business. To remain competitive in our industry, we must employ information technologies to support manufacturing processes, quality processes, distribution, financial reporting, as well as R&D and regulatory applications that capture, manage and analyze the large streams of data generated in our clinical trials, and it is critical that we do so in a secure manner to maintain the confidentiality, integrity and availability of such information systems, operational technologies and data. We also rely
43
extensively on technology to allow concurrent work sharing around the world. As with all information technology, our systems are vulnerable to potential damage or interruptions from natural disasters, power outages, telecommunications failures and other unexpected events, as well as physical and cyber intrusions, sabotage, piracy or intentional acts of vandalism and the potential risks associated with the deployment and use of artificial intelligence (“AI”) systems. Our cybersecurity policies, standards, and controls are applied to newly acquired businesses as they are integrated into our environment. Until integration is complete, acquired entities may operate legacy systems or processes that do not fully align with our cybersecurity standards, which could increase our exposure to cybersecurity incidents. In addition, as a result of the Business Combination and subsequent Separation, completion of integration of relevant systems, processes and policies presents additional complexities and requires additional resources. Given the extensive reliance of our business on technology, any substantial disruption or resulting loss of data that is not avoided or corrected by our backup measures could harm our business, financial condition, results of operations and cash flows.
We also have outsourced significant elements of our operations to third parties, some of which are outside the U.S. As a result, we are managing many independent vendor relationships with third parties who may or could have access to our confidential information and systems. Furthermore, pursuant to the Par Health TSA (as defined below), we share information technology infrastructure and applications support with Par Health, including privacy and security safeguards, following the closing of the Separation. The size and complexity of our information technology systems, and those of the third parties with whom we contract, make such systems and data potentially vulnerable to service interruptions and other cybersecurity incidents. We and such third parties could be susceptible to third-party attacks on our information security systems, which attacks are of ever-increasing levels of sophistication and are made by groups and individuals with a wide range of motives and expertise, including insiders, hacktivists, criminal groups, nation states and others.
Maintaining the secrecy of all our confidential, proprietary and/or trade secret information is important to our competitive business position. However, such information can be difficult to protect. While we have taken steps to protect such information and have made considerable investments in information technology, there can be no assurance that our efforts will prevent service interruptions or other cybersecurity incidents affecting our systems or the unauthorized or inadvertent wrongful use or disclosure of confidential information, including those caused by our own employees or others to whom we have granted access to our systems, that could adversely affect our business operations or result in the loss, dissemination or misuse of critical or sensitive information. As part of risk management processes, we maintain cybersecurity insurance that provides coverage for certain costs related to cybersecurity incidents. However, the amount or type of coverage may not be sufficient to address costs for handling an incident, or future changes may occur to insurance coverage. A cybersecurity incident such as the accidental loss, inadvertent disclosure, unapproved dissemination, misappropriation or misuse of trade secrets, proprietary information or other confidential information, whether as a result of theft, hacking, human error, sabotage, industrial espionage, fraud, trickery or other forms of deception, AI control failure, or for any other cause, could enable others to produce competing products, use our proprietary technology or information and/or adversely affect our business position. Further, any such event could result in financial, legal, business and reputational harm to us and could have a material adverse effect on our business, financial condition, results of operations and cash flows. In addition, if we are not successful in effectively utilizing technology solutions, including AI, and our competitors are, our business will be adversely affected.
We face risks related to our collection and use of data, which could result in investigations, inquiries, litigation, fines, legislative and regulatory action.
We are subject to laws and regulations governing the privacy and security of health related and other personal data we collect and maintain, including the GDPR, E.U. AI Act, Section 5 of the FTC Act, HIPAA, the CCPA as amended by the CPRA and other state comprehensive privacy laws, consumer protection laws and consumer health privacy laws. Any failure by us or any of our third-party service providers to follow such laws could result in significant liability or reputational harm under such state, federal and international privacy, data protection and other laws. The landscape of federal and state laws regulating personal data is constantly evolving and compliance with these laws requires a flexible privacy framework and substantial resources and compliance efforts will likely be an increasing and substantial cost in the future.
Governmental investigations, inquiries and regulatory actions and lawsuits brought against us by government agencies and private parties, in addition to future legislative actions, with respect to our manufacture or sale of opioids could adversely affect our reputation, business, financial condition, results of operations and cash flows.
We continue to be subject to legal and regulatory requirements as an opioid manufacturer, notably, with respect to Percocet, and our business may be impacted as further explained below. We are required to have systems in place that are designed to identify suspicious orders of controlled substances and report those orders to the DEA. We, along with other opioid manufacturers, have been the subject of federal and state government investigations and enforcement actions, focused on the misuse and abuse of opioid medications in the U.S. Similar investigations may be initiated in the future.
In addition, a significant number of lawsuits were filed against us, other opioid manufacturers, distributors and others in the supply chain by cities, counties, state Attorneys General and private persons seeking to hold us and others accountable for opioid misuse and abuse. While past, present and future opioid claims against us, with certain narrow exceptions, were deemed discharged, in
44
connection with our prior bankruptcies, we may face new opioid claims in the future, which could have a material adverse effect on our reputation, competitive position, business, financial condition and results of operations.
Some of our products are regulated as controlled substances, the manufacture, sale, importation, exportation, distribution and dispensing and administration of which are subject to significant regulation by the DEA and other regulatory agencies and authorities.
Some of our products are considered controlled substances under the CSA. Schedule II controlled substances include oxycodone, used in our opioid product Percocet. Schedule III controlled substances include testosterone, used in Testopel, Testim and Aveed. The manufacturing, distribution, import, export, packaging, storing, prescribing, dispensing, selling and use of controlled substances are subject to additional regulations, including under the CSA and DEA regulations and state requirements. These regulations increase the personnel needs and the expense associated with commercialization of products. Because of their restrictive nature, these laws and regulations could also limit commercialization of our controlled substance products. Failure by us, or any of the third-party manufacturers we rely on, to comply with these laws and regulations could also result in loss of DEA or state registrations, disruption in manufacturing and distribution activities, consent decrees, criminal and civil penalties and state actions, among other consequences.
In addition, we must periodically apply to the DEA for manufacturing quota to manufacture API and for procurement quota to manufacture our opioid product, Percocet, and the quota the DEA grants may be insufficient to meet our customers’ needs.
In addition, the DEA conducts periodic inspections of registered establishments that handle controlled substances and has stringent regulations on those establishments to prevent loss and diversion, among other obligations. Failure by us, or any of the third-party entity we rely on, to maintain compliance with these regulations, particularly as manifested in loss or diversion, can result in regulatory action that could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows. The DEA may seek civil penalties, refuse to renew necessary registrations or initiate proceedings to revoke those registrations. In certain circumstances, violations could lead to criminal proceedings.
Many states require separate state registrations in order to be able to obtain, manufacture, handle, distribute and dispense controlled substances for clinical trials or commercial sale, and failure by us or any of the third-party manufacturers we rely on, to meet applicable regulatory requirements could lead to enforcement and sanctions from the states in addition to those from the DEA or otherwise arising under federal law.
The manufacture of our products is highly exacting and complex, and our business could suffer if we, or our suppliers, manufacturers, distributors or collaboration partners encounter manufacturing, supply or other problems.
The manufacture of our products is highly exacting and complex, due in part to strict regulatory and manufacturing requirements, as well as due to the biologic nature of at least one of our products, which are inherently more difficult to manufacture than chemical-based products. Many of our manufacturing sites are large in scale and complex in operations and therefore require continued investments in quality management and maintenance. These manufacturing sites have multi-year capital plans and risk assessments, but unplanned maintenance activities can have an adverse impact on cash flow projections and production plans. In addition, we rely on third-party suppliers, manufacturers, distributors and collaboration partners to provide services for certain core aspects of our business, including supply and manufacture of key starting materials, components and APIs used in our products and in our product development activities, packaging, shipping, warehousing and distribution.
Manufacturing complex pharmaceutical and medical device products carries inherent risk. Problems may arise during manufacturing for a variety of reasons including equipment malfunction, failure to follow specific protocols and procedures, defective raw materials and environmental factors. If a batch of finished product fails to meet quality standards during a production run, then that entire batch of product may have to be discarded. These problems could lead to launch delays, product shortages, backorders, increased costs (including contractual damages for failure to meet supply requirements), lost revenue, damage to our reputation and customer relationships, time and expense spent investigating, correcting and preventing the root causes and, depending on the root causes, similar losses with respect to other products. If manufacturing problems are not discovered before the product is released to the market, we also could incur product recall and product liability costs. If we incur a product recall or product liability costs involving one of our products, such product could receive reduced market acceptance, thereby reducing product demand, and could harm our reputation and our ability to market our products in the future. Significant manufacturing problems could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
Additionally, we and our third-party manufacturers are subject to FDA, DEA, state and foreign regulatory and legal requirements. Any failure by us or our third-party manufacturers to comply with cGMP or the QMSR or failure to scale up manufacturing processes for any investigational product candidates, including any failure to deliver sufficient quantities of our investigational product candidates in a timely manner, could lead to a delay in, or failure to obtain, regulatory approval of our investigational products. In addition, such failure, or failures by our third-party manufacturers, to comply with cGMP or the QMSR in manufacturing our approved or cleared products could be the basis for the FDA or other regulatory authorities to issue a warning letter, withdraw approvals or take other regulatory or legal action, including recall or seizure of outside supplies of our products, total or partial suspension of production, suspension of ongoing clinical trials, refusal to approve pending applications or supplemental applications, detention of product, refusal to permit the import or export of products, injunction or imposing civil and criminal penalties.
45
Any interruption, delay, inability, dispute, mistake or failure by our suppliers, manufacturers, distributors and collaboration partners to meet our projected timelines or their contractual obligations with us on schedule or in accordance with our expectations or any termination by these third parties of their arrangements with us, which, in each case, could be the result of one or many factors outside of our control, could delay or prevent the development, approval, manufacture, launch or commercialization of our products, result in non-compliance with applicable laws and regulations, cause us to incur failure-to-supply penalties, disrupt our operations or cause reputational harm to us, any or all of which could have a material adverse effect on our business, financial condition, results of operations and cash flows. We may also be unsuccessful in resolving any underlying issues with such suppliers, manufacturers, distributors and partners or replacing them within a reasonable time and on commercially reasonable terms.
Several of our products (including our key products) and their components are manufactured by a single source, at a single manufacturing facility or stored at a single storage site. Loss or damage to a manufacturing facility or storage site due to a natural disaster or otherwise could adversely affect our ability to manufacture sufficient quantities of key products or otherwise deliver products to meet customer demand or contractual requirements which may result in a loss of revenue and other adverse business consequences. Furthermore, while we work closely with our suppliers to ensure the continuity of supply and to diversify our sources of components and materials, in certain instances (including for our key products) we do acquire components and materials from a sole supplier.
Although we do carry strategic inventory and maintain insurance to mitigate the potential risk related to any related supply disruption, there can be no assurance that such measures will be effective. Because of the time required to obtain regulatory approval and licensing of a manufacturing facility, an alternate third-party manufacturer may not be available on a timely basis to replace production capacity in the event we lose manufacturing capacity, experience supply challenges or products are otherwise not available due to natural disaster, regulatory action or otherwise. The occurrence of any of these events could delay or prevent our ability to achieve sales expectations, cause interruptions in our supply of products to customers, cause us to incur failure-to-supply penalties, disrupt our operations or cause reputational harm, any or all of which could have a material adverse effect on our business, financial condition, results of operations and cash flows.
Our operations expose us to risks and challenges associated with conducting business internationally and potential impacts of geopolitical uncertainty.
Our offices and operations are located in many countries. We face several risks inherent in conducting business internationally, including compliance with international and U.S. laws and regulations that apply to our international operations. These laws and regulations include data privacy requirements, labor relations laws, tax laws, anti-competition regulations, import and trade restrictions, export requirements, anti-bribery and anti-corruption laws such as the FCPA and similar local laws, regulations, industry self-regulation codes of conduct and physicians' codes of professional conduct, which also prohibit corrupt payments to governmental officials or certain payments or remunerations to customers. Given the high level of complexity of these laws, there is a risk that some provisions may be violated, inadvertently or through fraudulent or negligent behavior of individual employees or through our failure to comply with certain formal documentation requirements or otherwise. Violations of these laws and regulations could result in fines or criminal sanctions against us, our officers or our employees and prohibitions on the conduct of our business. Any such violations could include prohibitions on our ability to offer our products in one or more countries and could materially damage our reputation, our international expansion efforts and our ability to attract and retain employees.
In addition to the foregoing, engaging in international business inherently involves a number of other difficulties and risks, including:
•potentially longer payment cycles and difficulties in enforcing agreements and collecting receivables through certain non-U.S. legal systems;
•potential inability to sell products into certain countries given the delay of foreign governments in responding to changes in our U.S. business licensing;
•political and economic instability, acts of war or threats of war;
•the unpredictability of U.S. trade policy, including Section 301 tariffs and U.S. trade relations with other countries, that may increase raw material cost or impact our ability to obtain the raw materials we need to manufacture our products and impact our ability to sell our products outside of the U.S.;
•potentially adverse tax consequences, tariffs, customs charges, bureaucratic requirements and trade barriers;
•difficulties and costs of staffing and managing our operations, including our manufacturing and supply chain processes;
•exposure to global economic conditions;
•exposure to potentially unfavorable movements in foreign currency exchange rates associated with international net sales and operating expense and intercompany debt financings; and
•potential negative impact of public health epidemics and geopolitical uncertainty on employees, our business and supply chain and the global economy.
46
These or other factors or any combination of them may have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
New or increased tariffs and evolving trade relations between the United States and other countries, as well as changes in U.S. international trade and taxation policy, could have a material adverse effect on our business, financial condition, results of operations and cash flows.
The U.S. government may seek to impose additional restrictions on international trade, such as increased tariffs on goods imported into the United States. Such tariffs could potentially disrupt our existing supply chains and impose additional costs on our business, including costs with respect to raw materials upon which our business depends. Furthermore, if tariffs, trade restrictions or trade barriers are placed on products such as ours by foreign governments, it could cause us to raise prices for our products, which may result in the loss of customers. If we are unable to pass along increased costs to its customers, our margins could be adversely affected. Additionally, it is possible that further tariffs may be imposed that could affect imports of APIs and other materials used in our products, or our business may be adversely impacted by retaliatory trade measures taken by other countries, including restricted access to APIs or other materials used in our products, causing us to raise prices or make changes to our products. Further, the continued threats of new or increased tariffs, sanctions, trade restrictions and trade barriers could have a generally more disruptive impact on the global economy and, therefore, negatively impact our sales. Given the volatility and uncertainty regarding the scope and duration of these tariffs and other aspects of U.S. international trade and taxation policy, under the current U.S. administration, the impact on our operations and results is uncertain and could be significant. Further governmental action related to tariffs, additional taxes, regulatory changes or other retaliatory trade measures could occur in the future. Any of these factors could have a material adverse effect on our business, financial condition, results of operations and cash flows.
We have significant levels of intangible assets, including those acquired in the Business Combination, which rely on projections of future cash flows in impairment testing. Should we experience unfavorable variances from these projections these assets may have an increased risk of future loss of value and impairment.
At least annually, we review the carrying value of our non-amortizing intangible assets and for amortizing intangible assets when indicators of impairment are present. Events giving rise to asset impairments are an inherent risk in the pharmaceutical industry and often cannot be predicted. Conditions that could indicate impairment and necessitate an evaluation of intangible assets include, but are not limited to, a significant adverse change in the business climate or the legal or regulatory environment.
In performing our impairment tests, we utilize projections of future cash flows. Projections of future cash flows are inherently subjective and reflect assumptions that may or may not ultimately be realized. Significant assumptions utilized in our projections include, but are not limited to, our evaluation of the market opportunity for our products, the current and future competitive landscape and resulting impacts to product pricing, future legislative and regulatory actions or the lack thereof, planned strategic initiatives, the ability to achieve cost synergies from acquisitions, and the realization of benefits associated with our existing and anticipated patents and regulatory approvals. Given the inherent subjectivity and uncertainty in projections, we could experience significant unfavorable variances in future periods or revise our projections downward. Such circumstances could increase the risk that our intangible assets may be impaired. If an impairment were recognized, this could have a material adverse effect on our financial condition, results of operations or cash flows.
We are subject to labor and employment laws and regulations, which could increase our costs and restrict our operations in the future.
Some of our employees are represented by labor organizations and national works councils. Our management believes that our employee relations are satisfactory. However, further organizing activities or collective bargaining may increase our employment-related costs and we may be subject to work stoppages and other labor disruptions. Moreover, if we are subject to employment-related claims, such as individual and class actions relating to alleged employment discrimination, wage-hour and labor standards issues, and such actions are successful in whole or in part, this may affect our ability to compete or have a material adverse effect on our business, financial condition, results of operations and cash flows.
We have implemented changes to our Acthar Gel patient assistance program, which may receive additional review from governmental regulators and, if challenged, could have a material adverse effect on future net sales of Acthar Gel.
We currently offer a patient assistance program (“PAP”) that provides free Acthar Gel vials to certain eligible patients. Beginning January 1, 2024, we implemented changes to expand our program to eligible Medicaid beneficiaries who have been prescribed Acthar Gel for an on-label indication and meet all other PAP eligibility criteria. Our decision to expand PAP eligibility was made in response to changes in the Medicaid Drug Rebate Program’s (“MDRP”) unit rebate amount calculation that became effective in 2024 and is designed to ensure that Medicaid patients retain timely and affordable access to Acthar Gel. We provided CMS and OIG within the HHS with advance notice of these changes. While we believe these changes comply with existing statutory and regulatory requirements and related guidance, including based on consultation with external advisors, it is possible that CMS, OIG within the HHS or other governmental agencies could take issue with such changes. If we are unable to either expand our PAP as currently planned or find an alternative solution, we will incur additional expenses under the 2024 changes to the MDRP unit rebate amount
47
calculation, which could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
Our actual financial results are not comparable to our historical financial statements.
We adopted fresh-start accounting in accordance with the provisions of ASC 852 at the time of our emergence from Chapter 11 bankruptcy in 2023. Fresh-start accounting requires that new fair values be established for assets, liabilities and equity as of the fresh-start effective date, which may differ materially from the recorded values of the assets, liabilities and equity on historical consolidated balance sheets prior to the fresh-start effective date. This, as well as the Business Combination in July 2025 and the Separation of our historical generic pharmaceuticals and sterile injectables businesses in November 2025, make it difficult for our shareholders to assess our performance in relation to prior periods. See Notes 3, 5, and 6 to the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for further information on Fresh-start, the Business Combination, and the Separation, respectively.
Risks Related to Our Indebtedness and Settlement Obligation
Our substantial indebtedness and settlement obligation could adversely affect our financial condition and prevent us from fulfilling our obligations.
We have substantial indebtedness and we have a substantial obligation in respect of the Acthar Gel-Related Litigation Settlement. As of December 31, 2025, total debt principal outstanding was $2,481.3 million, of which $15.0 million was classified as current. In addition, we have a remaining undiscounted cash obligation of $190.0 million in respect of the Acthar Gel-Related Litigation Settlement, inclusive of interest (as defined in Item 7. Management's Discussion and Analysis of Financial Condition and Results of Operations of this Annual Report). Our substantial indebtedness could adversely affect our ability to fulfill our financial obligations (including our ability to service our indebtedness and our obligation in respect of the Acthar Gel-Related Litigation Settlement) and have a negative impact on our financing options and liquidity positions.
Our degree of debt leverage, which has increased as a result of the consummation of the Business Combination, and our significant settlement obligation, as well as restrictions in the agreements governing our indebtedness and settlement obligation, have significant consequences, including the following:
•making it more difficult for us to satisfy our obligations with respect to our debt, including making applicable scheduled principal and interest payments on our indebtedness, and our ongoing obligation in respect of the Acthar Gel-Related Litigation Settlement;
•limiting our ability to refinance our going-forward debt obligations (certain of which are subject to a customary prepayment premium), make prepayments in respect of the Acthar Gel-Related Litigation Settlement obligation, or to obtain additional financing in the future for working capital, capital expenditures, research and development, acquisitions or other general corporate purposes;
•requiring us to sell assets or restructure or refinance our indebtedness and Acthar Gel-Related Litigation Settlement obligation;
•requiring a substantial portion of our cash flows to be dedicated to debt service payments instead of other purposes, thereby reducing the amount of cash flows available for working capital, capital expenditures, research and development, acquisitions and other general corporate purposes;
•placing us at a competitive disadvantage to other less leveraged competitors;
•making us more vulnerable to economic downturns and limiting our ability to withstand competitive pressures; and
•increasing our costs of borrowing.
The operating and financial restrictions imposed on us by our indebtedness and settlement obligation could limit our flexibility in planning for and reacting to changes, opportunities and challenges in our business, including changes in the industry in which we compete, changes in our business and strategic opportunities and adverse developments in our operations. See the risk factor captioned “The terms of the agreements that govern our indebtedness and Acthar Gel-Related Litigation Settlement restrict our current and future operations, particularly our ability to respond to changes or to pursue our business strategies.” for additional information regarding such restrictions.
We may not be able to generate sufficient cash to service all of our indebtedness and settlement obligation and we may be forced to take other actions to satisfy our obligations under our indebtedness and settlement obligation, which may not be successful.
Our ability to make scheduled payments on or to refinance our going-forward debt obligations and Acthar Gel-Related Litigation Settlement obligation depends on our financial condition and operating performance, which are subject to prevailing economic and competitive conditions and to certain financial, business, legislative, regulatory and other factors beyond our control. We may be unable to maintain a level of cash flows from operating activities sufficient to permit us to fund our day-to-day operations or to pay the principal, premium, if any, and interest on our indebtedness and satisfy our Acthar Gel-Related Litigation Settlement obligation.
48
If our cash flows and capital resources are insufficient to fund our debt service obligations and other cash requirements (including our Acthar Gel-Related Litigation Settlement obligation), we could face substantial liquidity problems and could be forced to reduce or delay investments and capital expenditures or, subject to the restrictions from our existing indebtedness, sell assets or operations, seek additional capital or restructure or refinance our indebtedness and Acthar Gel-Related Litigation Settlement obligation. We may not be able to effect any such alternative measures, if necessary, on commercially reasonable terms or at all and, even if successful, we may not be able to obtain proceeds in amount sufficient to meet our scheduled debt service obligations and Acthar Gel-Related Litigation Settlement obligation.
Our inability to generate sufficient cash flows to satisfy our debt obligations and Acthar Gel-Related Litigation Settlement obligation, or to refinance our indebtedness on commercially reasonable terms or at all, would materially and adversely affect our financial position and results of operations.
If we cannot make scheduled payments on our debt or the Acthar Gel-Related Litigation Settlement obligation, we will be in default thereunder and, as a result, creditors under such indebtedness could declare the principal, interest and other obligations thereunder to be due and payable, such creditors, if secured, could foreclose against the assets securing such borrowings, and/or beneficiaries of our then-outstanding Acthar Gel-Related Litigation Settlement obligation could declare such obligations to be due and payable, as applicable, and we could be forced to return to bankruptcy or into liquidation.
The terms of the agreements that govern our indebtedness and Acthar Gel-Related Litigation Settlement restrict our current and future operations, particularly our ability to respond to changes or to pursue our business strategies.
The agreements that govern the terms of our existing indebtedness, including the agreements that govern the indebtedness of certain of our subsidiaries that remains outstanding following consummation of the Business Combination, and Acthar Gel-Related Litigation Settlement obligation, contain a number of restrictive covenants that impose significant operating and financial restrictions on us and may limit our ability to engage in acts that may be in our long-term best interest, including limitations or restrictions on our ability to:
•incur, assume or guarantee additional indebtedness;
•refinance our indebtedness, certain of which are subject to a customary prepayment premium requirements;
•issue redeemable stock and preferred stock;
•declare or pay dividends, make other distributions with respect to equity interests, or purchase or otherwise acquire or retire equity interests;
•make any principal payment on, or redeem or repurchase, subordinated, junior secured or unsecured debt and, with respect to certain of our indebtedness and the Acthar Gel-Related Litigation Settlement obligation;
•make loans, advances or other investments;
•sell or otherwise dispose of assets, including capital stock of subsidiaries;
•use the proceeds from dispositions of assets, including capital stock of subsidiaries;
•incur liens;
•enter into transactions with affiliates;
•enter into sale and lease-back transactions;
•permit the occurrence of certain change of control transactions;
•consolidate or merge with or into or sell all or substantially all of our assets to, another person or entity; and
•enter into swap agreements.
In addition, our existing senior secured credit facilities requires us to comply with a financial maintenance covenant in certain circumstances. Our ability to satisfy this financial maintenance covenant can be affected by events beyond our control and we cannot assure you that we will be able to comply.
A breach of the covenants under the agreements that govern the terms of any of our indebtedness or settlement obligation (including the obligation to make payments thereunder in accordance with the terms thereof) could result in an event of default under the applicable indebtedness or settlement obligation. Any such default may allow the applicable creditors or beneficiaries to accelerate the related debt or settlement obligation and, in the case of our debt, may result in the acceleration of any other debt to which a cross-acceleration or cross-default provision applies. In addition, an event of default under our existing senior secured credit facilities, would permit the lenders under such facility to terminate all commitments to extend further credit thereunder. Furthermore, if we are unable to repay the amounts due and payable under our secured indebtedness, those creditors will be able to proceed against the collateral granted to them to secure that secured indebtedness. Additionally, if a change in control transaction were to occur, such a transaction may accelerate the maturity dates on our indebtedness. If the holders of our debt or settlement obligation accelerate the repayment of
49
our borrowings or the payment of our settlement obligation for the above reasons, or any other, we may not have sufficient assets to repay such indebtedness or settlement obligation.
These restrictions may affect our ability to operate in accordance with our plans, otherwise achieve our operational and financial objectives in a timely manner or at all, and have an adverse effect on our business, financial condition, results of operations and cash flows.
Our variable-rate indebtedness exposes us to interest rate risk, which could cause our debt service obligations to increase significantly.
Certain of our secured indebtedness, including our existing senior secured credit facilities, is subject to variable rates of interest and exposes us to interest rate risk. Any future indebtedness could also be at variable rates. If interest rates increase, our debt service obligations on the variable-rate indebtedness would increase and our net loss would increase, even though the amount borrowed under the facilities remained the same. As of December 31, 2025, we had variable-rate debt consisting of $1,481.3 million outstanding principal amount on our senior secured credit facilities. An unfavorable movement in interest rates, primarily the Secured Overnight Financing Rate (“SOFR”), could result in higher interest expense and cash payments for us.
We may incur additional debt in the future. This could further exacerbate the risks described above.
We may incur substantial additional indebtedness in the future. Although agreements governing our existing indebtedness (including the agreements that govern the indebtedness of certain of our subsidiaries that remains outstanding after the Business Combination) and settlement obligation restrict the incurrence of additional indebtedness, these restrictions are and will be subject to a number of qualifications and exceptions and the additional indebtedness incurred in compliance with these restrictions could be substantial. If new debt is added to our current debt levels, the related risks that we now face could intensify.
We may need additional financing in the future to meet our capital needs or to make acquisitions, and such financing may not be available on favorable or acceptable terms, and may be dilutive to existing shareholders. The use of proceeds from future financings will be subject to the restrictions from our existing indebtedness.
We may need to seek additional financing in the future. For example, we may need to seek additional financing to increase our investment in new product acquisitions, refinance our existing indebtedness or settlement obligation or for other general corporate purposes. Subject to the restrictions from our existing indebtedness, adequate funds may not be available to us on favorable or acceptable terms or at all, including if there is a material decline in the demand for our products or in the solvency of our customers or suppliers or in the event of other significantly unfavorable changes in economic conditions, and we may be unable to fund our expansion, successfully develop or enhance products, respond to competitive pressures or otherwise operate our business or satisfy our obligations now or in the future. In addition, volatility in the world financial markets could increase borrowing costs or affect our ability to access the capital markets. Any of these factors could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows. If we raise additional funds through the issuance of equity securities, our shareholders will experience dilution of their ownership interest. In addition, even if we are able to raise such additional funds, the use of proceeds therefrom will be subject to the limitations imposed by our existing indebtedness.
Risks Related to Tax Matters
The United States could treat Keenova Therapeutics plc as a U.S. taxpayer or otherwise subject Keenova Therapeutics plc to certain adverse U.S. federal income tax consequences under Internal Revenue Code Section 7874.
Following the emergence from the 2023 Bankruptcy Proceedings and the Business Combination, Keenova Therapeutics plc continues to be an Irish tax resident. The Internal Revenue Service (“IRS”) may, however, assert that Keenova Therapeutics plc should be treated as a U.S. corporation for U.S. federal income tax purposes pursuant to Internal Revenue Code (“IRC”) Section 7874. For U.S. federal income tax purposes, a corporation is generally considered to be tax resident in the jurisdiction of its organization or incorporation. Because Keenova Therapeutics plc is an Irish incorporated entity, it would generally be classified as a foreign corporation under these rules. IRC Section 7874 provides an exception to this general rule under which a foreign corporation may, in certain circumstances, be treated as a U.S. corporation for U.S. federal income tax purposes if the following requirements are met: (i) the foreign corporation completes the direct or indirect acquisition of substantially all of the assets held directly or indirectly by a U.S. corporation (including the indirect acquisition of assets of the U.S. corporation by acquiring the outstanding shares of the U.S. corporation), (ii) the former shareholders of the acquired U.S. corporation hold (or are treated as holding) at least 80% of the shares of the foreign acquiring corporation after applying certain adjustments required under Section 7874 (the “ownership percentage”) and (iii) the foreign corporation's “expanded affiliated group” does not have substantial business activities in the foreign corporation's country of organization or incorporation compared to the expanded affiliated group's worldwide activities. Even if a corporation is not treated as a U.S. corporation under the above rule, if the ownership percentage is at least 60% (but less than 80%) and the rest of the requirements described above are met, IRC Section 7874 would cause a foreign corporation to become subject to certain unfavorable U.S. federal income tax rules, including restrictions on the use of tax attributes with respect to “inversion gain” recognized over a 10-year period following the transaction, and potentially additional tax liabilities under the so-called “base erosion and anti-abuse” minimum tax rules. Further, the U.S. shareholders of such a foreign corporation could be subject to a higher rate of tax on any dividends received.
50
Although it is not free from doubt, we believe that as a result of the implementation of the 2023 Plan, Keenova Therapeutics plc should not be treated as acquiring directly or indirectly substantially all of the properties of a U.S. corporation and, as a result, Keenova Therapeutics plc is not expected to be treated as a U.S. corporation or otherwise subject to the adverse tax consequences of IRC Section 7874 as a result of the implementation of the 2023 Plan. Similarly with respect to the Business Combination, based on current law and the percentage of Keenova Therapeutics plc ordinary shares received by Endo, Inc. stockholders in the Business Combination, and taking into account certain adjustments required under IRC Section 7874 in determining the ownership percentage, we do not expect IRC Section 7874 to apply so as to cause Keenova Therapeutics plc to be treated as a U.S. corporation for U.S. federal income tax purposes or to otherwise be subject to IRC Section 7874 as a result of the Business Combination. However, the rules under IRC Section 7874 are highly complex, unclear and subject to change. Accordingly, there can be no assurance that the IRS will agree with and not challenge this conclusion or that a court would not sustain any such challenge.
If it is determined that IRC Section 7874 is applicable, Keenova Therapeutics plc could be treated as a U.S. corporation for U.S. federal income tax purposes or otherwise become subject to certain unfavorable U.S. federal income tax rules as described above, which could cause Keenova Therapeutics plc to become subject to significant additional U.S. tax liability. In addition, if IRS Section 7874 were to apply such that Keenova Therapeutics plc is treated as a U.S. corporation for U.S. federal income tax purposes, it would also be considered an Irish tax resident for Irish tax and other non-U.S. tax purposes.
The IRS may interpret IRC Section 382 limitation and cancellation of debt income attribution rules differently.
In general, IRC Section 382, provides an annual limitation with respect to the ability of a corporation to utilize its tax attributes, as well as certain built-in-losses (“BILs”), against future taxable income in the event of a change in ownership. Emergence from the Chapter 11 proceedings and Irish examinership proceedings on June 16, 2022 (together, the “2020 Bankruptcy Proceedings”) and the 2023 Bankruptcy Proceedings resulted in a change in ownership for purposes of IRC Section 382. Any discharge of our external or internal debt obligations as a result of the bankruptcy proceedings for an amount less than the adjusted issue price may give rise to cancellation of debt income, which must either be included in our taxable income or result in a reduction to our tax attributes. U.S. tax attributes subject to reduction include: (i) net operating loss (“NOL(s)”) and NOL carryforwards; (ii) credit carryforwards (iii) capital losses and capital loss carryforwards; and (iv) the tax basis of our depreciable, amortizable and other assets. The amount of our post-ownership change annual U.S. taxable income that can be offset by the pre-ownership change U.S. NOLs and BILs generally cannot exceed an amount equal to the product of (a) the applicable federal long-term tax exempt rate in effect on the date of the ownership change and (b) the value of our U.S. affiliate stock immediately prior to implementation of each respective plan of reorganization (“Annual Limitation”) (a separate Annual Limitation must be computed for both the plan of reorganization for the 2020 Bankruptcy Proceedings (“2020 Plan”) and the 2023 Plan). The Annual Limitation may also be increased or decreased during the first five years post-ownership change for certain realized built-in-gains or realized BILs, respectively. Our interpretation of the impact of the IRC's limitations on the utilization of tax attributes after the ownership change caused by the emergence from bankruptcy may differ from the IRS's interpretation. Any additional limitations on our ability to prospectively use these tax attributes may have an adverse effect on our prospective cash flow.
Our ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
Under IRC Sections 382 and 383, if a corporation undergoes an “ownership change,” generally defined as a greater than 50 percent change, determined by value in its equity ownership by certain stockholders over a rolling three-year period, the corporation’s ability to use its pre-ownership change NOLs and other pre-ownership change tax attributes to offset its post-ownership change taxable income or tax liability may be limited. We may experience ownership changes in the future due to shifts in our stock ownership, some of which is outside of our control. Additionally, similar laws at the state level may apply.
A loss of a major tax dispute, if one were to arise, or a challenge to our operating structure or intercompany pricing policies could result in a higher tax rate on our worldwide earnings, which could result in a material adverse effect on our financial condition, results of operations and cash flows.
Income tax returns that we file are subject to review and examination. We recognize the benefit of income tax positions we believe are more likely than not to be sustained upon challenge by a tax authority. If any tax authority successfully challenges our operational structure, intercompany pricing or financing policies; if the terms of certain income tax treaties are interpreted in a manner that is adverse to our structure; or if we have a disagreement with a tax authority that results in the loss of a material tax dispute in any country; our effective tax rate on our worldwide earnings could increase substantially and result in a material adverse effect on our financial condition.
Our status as a foreign corporation for U.S. federal tax purposes could be affected by a change in law.
We believe that, under current law, we are treated as a foreign corporation for U.S. federal tax purposes. However, changes in tax law, such as additional changes to the rules under IRC Section 7874 or the U.S. Treasury Regulations promulgated thereunder or other IRS guidance, could adversely affect our status as a foreign corporation for U.S. federal tax purposes, and any such changes could have prospective or retroactive application to us and our shareholders and affiliates. In addition, legislative proposals issued by the U.S. Department of the Treasury and Congress have aimed to expand the scope of U.S. corporate tax residence, and such proposals, if
51
passed, could have an adverse effect on us. Although the proposals would generally apply to prospective transactions, no assurance can be given that such proposals will not be changed to apply retroactively.
Future changes to U.S. and foreign tax laws could adversely affect us.
The European Commission, U.S. Congress and Treasury Department, the Organization for Economic Co-operation and Development (“OECD”), and other government agencies in jurisdictions where we and our affiliates do business have had an extended focus on issues related to the taxation of multinational corporations, particularly payments made between affiliates from a jurisdiction with high tax rates to a jurisdiction with lower tax rates. As a result, the tax laws in the U.K., Ireland, E.U., Switzerland, Japan, U.S. and other countries in which we and our affiliates do business could change on a prospective or retroactive basis, and any such changes could adversely affect us and our affiliates.
Recent examples include the European Commission's Anti-Tax Avoidance Directives (ATAD I and ATAD II), the Multilateral Convention to Implement Tax Treaty Related Measures to Prevent Base Erosion and Profit Shifting (Multilateral Instrument) and the new corporate alternative minimum tax created in the U.S. by the Inflation Reduction Act.
Additionally, on December 20, 2021, the OECD released the Global Anti-Base Erosion (“GloBE”) Model Rules (“Pillar Two”) providing a legislative framework for the Income Inclusion Rule and the Under-Taxed Payment Rule (“UTPR”). Pillar Two is designed to ensure that large multinational enterprise groups pay a minimum level of tax on the income arising in each of the jurisdictions where they operate, principally creating a 15% minimum global effective tax rate. On December 15, 2022, the E.U. member states adopted a directive implementing the Pillar Two global minimum tax rules. A number of jurisdictions have transposed the directive into national legislation with the rules applicable for fiscal years beginning on or after December 31, 2023, with the exception of the UTPR which is applicable for fiscal years beginning on or after December 31, 2024. For the fiscal year beginning December 28, 2024, the Company was in scope of the enacted or substantively enacted legislation, and an assessment of the potential exposure to Pillar Two income taxes was performed for the fiscal year ended December 31, 2025. Based on the assessment of Pillar Two, certain transitional safe harbor relief applied for most jurisdictions, and where the transitional safe harbor relief did not apply, the impact to income tax expense was not material for the fiscal year ended December 31, 2025. Because Pillar Two rules are complex and their implementation across jurisdictions remains uncertain, we cannot predict the impact that the Pillar Two rules will have on our effective tax rate and cash tax obligations in future periods, which could be material.
Future changes to U.S. and foreign tax laws, including changes to the Pillar Two rules, could adversely affect us and our affiliates by increasing our effective tax rate and cash tax obligations, which could have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows.
We may not be able to maintain a competitive worldwide effective corporate tax rate.
We cannot give any assurance as to what our effective tax rate will be in the future, because of, among other things, uncertainty regarding the tax policies of the jurisdictions where we operate. Our actual effective tax rate may vary from our expectation and that variance may be material. Additionally, the tax laws of Ireland and other jurisdictions could change in the future, and such changes could cause a material change in our effective tax rate.
Risks Related to Our Jurisdiction of Incorporation
Irish law differs from the laws in effect in the U.S. and may afford less protection to holders of our securities.
It may not be possible to enforce court judgments obtained in the U.S. against us in Ireland based on the civil liability provisions of the U.S. federal or state securities laws. In addition, there is some uncertainty as to whether the courts of Ireland would recognize or enforce judgments of U.S. courts obtained against us or our directors or officers based on the civil liabilities provisions of the U.S. federal or state securities laws or hear actions against us or those persons based on those laws. We have been advised the U.S. currently does not have a treaty with Ireland providing for the reciprocal recognition and enforcement of judgments in civil and commercial matters. Therefore, a final judgment for the payment of money rendered by any U.S. federal or state court based on civil liability, whether or not based solely on U.S. federal or state securities laws, would not automatically be enforceable in Ireland.
A judgment obtained against us will be enforced by the courts of Ireland if the following general requirements are met: (i) U.S. courts must have had jurisdiction in relation to the particular defendant according to Irish conflict of law rules (the submission to jurisdiction by the defendant would satisfy this rule) and (ii) the judgment must be final and conclusive and the decree must be final and unalterable in the court which pronounces it. A judgment can be final and conclusive even if it is subject to appeal or even if an appeal is pending. Where however the effect of lodging an appeal under the applicable law is to stay execution of the judgment, it is possible that in the meantime the judgment may not be actionable in Ireland. It remains to be determined whether final judgment given in default of appearance is final and conclusive. However, Irish courts may refuse to enforce a judgment of the U.S. courts which meets the above requirements for one of the following reasons: (i) if the judgment is not for a definite sum of money; (ii) if the judgment was obtained by fraud; (iii) the enforcement of the judgment in Ireland would be contrary to natural or constitutional justice; (iv) the judgment is contrary to Irish public policy or involves certain U.S. laws which will not be enforced in Ireland; or (v) jurisdiction cannot be obtained by the Irish courts over the judgment debtors in the enforcement proceedings by personal service in Ireland or outside Ireland under Order 11 of the Ireland Superior Courts Rules.
52
As an Irish company, we are governed by the Irish Companies Act 2014, which differs in some material respects from laws generally applicable to U.S. corporations and shareholders, including, among others, differences relating to interested director and officer transactions and shareholder lawsuits. Likewise, the duties of directors and officers of an Irish company generally are owed to the company only. Shareholders of Irish companies generally do not have a personal right of action against directors or officers of the company and may exercise such rights of action on behalf of the company only in limited circumstances. Accordingly, holders of our securities may have more difficulty protecting their interests than would holders of securities of a corporation incorporated in a jurisdiction of the U.S.
Irish law imposes restrictions on certain aspects of capital management.
Irish law allows our shareholders to pre-authorize shares to be issued by our Board of Directors without further shareholder approval for up to a maximum of five years. Additionally, subject to specified exceptions, including as opt-out approved by a shareholder vote, Irish law grants statutory pre-emptive rights to existing shareholders to subscribe for new issuances of shares for cash. Our Memorandum and Articles of Association, effective from November 11, 2025 and amended effective July 31, 2025, (“Articles of Association”), contain a five-year pre-authorization of the Board of Directors to issue shares up to the amount of Keenova’s authorized share capital and opt-out of pre-emption rights. We cannot guarantee that renewal of the pre-authorization or opt-out from pre-emptive rights will always be sought or approved. We cannot provide assurance that these Irish legal restrictions will not interfere with our capital management.
Risks Related to Our Ordinary Shares
Our ordinary shares are not listed on any national securities exchange and our plans to list on an exchange and to conduct a concurrent underwritten public offering are subject to a variety of factors, several of which are outside our control.
Our ordinary shares are not listed on any national securities exchange. We previously announced our intention to pursue a listing on the New York Stock Exchange (the “NYSE”) in 2026, as well as a concurrent underwritten public offering of, our ordinary shares subject to approval of our Board of Directors and other considerations and conditions. The proposed listing and public offering necessarily depend on variety of factors, including market, geopolitical, industry and macroeconomic conditions, the NYSE and SEC review processes, as well as our ability to achieve our business and strategic goals. As a result, we may not be successful in listing our ordinary shares on the NYSE, or consummating a public offering of our ordinary shares, in 2026 or at all.
Our ordinary shares are issued solely through a transfer agent because they are not listed on a national securities exchange and are therefore not eligible for settlement through The Depository Trust Company (“DTC”), which ordinarily facilitates trades in listed securities in the U.S. As a result, our ordinary shares can only be held in registered form, which could be either directly or beneficially through a banker, broker or other nominee. This means that trading in our ordinary shares requires additional administrative steps as compared to shares that are listed on a national securities exchange or quoted on the OTC market. Furthermore, because the ordinary shares are not listed on a national securities exchange, additional transfer taxes and administrative steps are necessary to effect the sale, transfer and settlement of shares. So long as the ordinary shares are not listed on a national securities exchange, it will be an offense for a transferee of ordinary shares to fail to comply with requirements to file an Irish stamp duty return and to pay any Irish stamp duty due with the Irish Revenue Commissioners following such transfer, and interest and penalties will accrue. The filing of such returns and payment of the stamp duty requires both the transferee and transferor to have obtained an Irish tax reference number from the Irish Revenue Commissioners and requires payment of the stamp duty from an Irish bank account to the Irish Revenue Commissioners. Until such stamp duty return has been duly filed (or the transfer is exempt) and the related stamp duty duly paid, the transfer will not be registered on the register of members of the company (“Register”). Under Irish law and our Articles of Association, rights in respect of our ordinary shares are exercisable only by the registered shareholder as entered in the Register. For example, the exercise of voting rights and rights related to the appointment or nomination of directors is only effective under Irish law if executed by the registered shareholder. Because administrative steps to transfer our ordinary shares take additional time, there is a delay between the nominal transfer of shares and the recording of such transfer on the Register, and as a result, there is a delay between when a new shareholder purchases the ordinary shares and when that shareholder is able to exercise their rights as a shareholder. Where any transfer of ordinary shares occurs at less than market value, the transferor can be liable for all of the obligations of the transferee in relation to Irish stamp duty.
Because the ordinary shares are not listed and because of the additional administrative steps and tax ramifications related to transferring ordinary shares, there is limited liquidity for our shares, which could have a negative impact on the market price of our ordinary shares. For so long as our ordinary shares are not listed, holders of our ordinary shares may have difficulty selling or transferring any ordinary shares that they hold, and the number of investors willing to hold or acquire ordinary shares may be reduced, the trading price of ordinary shares may be depressed, we may receive decreased news and analyst coverage and we may be limited in our ability to issue additional securities or obtain additional equity financing in the future on terms acceptable to us, or at all.
The absence of an active trading market for our ordinary shares also impacts our ability to access the capital markets and severely limits our ability to use equity to effect acquisitions or recruit employees.
53
Our shareholders may experience dilution in the future.
Our shareholders may be diluted in the future because of equity issuances for acquisitions, capital market transactions, or otherwise, including, without limitation, equity awards that we may grant to our directors, officers, and employees. Such issuances may have a dilutive effect on our earnings per share, which could adversely affect the value of our ordinary shares.
Subject to our shares being listed on a recognized stock exchange (including the NYSE), any attempts to acquire us may be subject to the Irish Takeover Rules and subject to the supervisory jurisdiction of the Irish Takeover Panel and our Board of Directors may be limited by the Irish Takeover Rules in its ability to defend an unsolicited takeover attempt.
In the event that our ordinary shares are listed on a recognized stock exchange (which includes the NYSE), we would be subject to the Irish Takeover Panel Act, 1997 (as amended) and the Irish Takeover Panel Act, 1997, Takeover Rules 2022 (the “Irish Takeover Rules”), which regulate the conduct of takeovers of, and certain other relevant transactions affecting, Irish public limited companies listed on certain stock exchanges, including the NYSE. The Irish Takeover Rules are administered by the Irish Takeover Panel, which has supervisory jurisdiction over such transactions. Among other matters, the Irish Takeover Rules operate to ensure that no offer is frustrated or unfairly prejudiced and, in situations involving multiple bidders, that there is a level playing field.
Under the Irish Takeover Rules, we would not be permitted to take certain actions that might “frustrate” an offer for our ordinary shares once the Board of Directors has received an offer, or has reason to believe an offer is or may be imminent, without the consent of the Irish Takeover Panel and, in some instances, approval of holders of more than 50% of the shares entitled to vote at a general meeting of our shareholders.
This could limit the ability of the Board of Directors to take defensive actions even if it believes that such defensive actions would be in our company’s best interests or the best interests of our shareholders.
The operation of the Irish Takeover Rules in the event that our ordinary shares are listed on a recognized stock exchange (including the NYSE) and/or provisions of our Articles of Association may affect the ability of certain parties to acquire our ordinary shares.
In the event that our ordinary shares are listed on the NYSE (or another recognized stock exchange to which the Irish Takeover Rules apply), the Irish Takeover Rules would apply to us. The operation of the Irish Takeover Rules and/or provisions of our Articles of Association could delay, defer or prevent a third party from acquiring us or otherwise adversely affect the price of our ordinary shares.
Under the Irish Takeover Rules, certain separate persons will be presumed to be acting in concert. The Board of Directors and their relevant family members, related trusts and “controlled companies” are presumed to be acting in concert with any corporate shareholder who holds 20% or more of our company. The application of these presumptions may result in restrictions upon the ability of any of the concert parties and/or members of the Board of Directors to acquire more of our securities, including under the terms of any executive incentive arrangements. Accordingly, the application of the Irish Takeover Rules may frustrate the ability of certain of Keenova’s shareholders and directors to acquire Keenova ordinary shares.
The Irish Takeover Rules provide that if an acquisition of our ordinary shares were to increase the aggregate holding of the acquirer and its concert parties to our ordinary shares that represent 30% or more of the voting rights of our company, the acquirer and, in certain circumstances, its concert parties would be required (except with the consent of the Irish Takeover Panel) to make an offer for our outstanding ordinary shares at a price not less than the highest price paid for the ordinary shares by the acquirer or its concert parties during the previous 12 months.
This requirement would also be triggered by an acquisition of our ordinary shares by any person holding (together with its concert parties) our ordinary shares that represent between 30% and 50% of the voting rights in our company if the effect of such acquisition were to increase that person’s percentage of the voting rights by 0.05% within a 12-month period.
Additionally, our Articles of Association provide (i) that the Board of Directors may issue preference shares without shareholder approval, with such rights and preferences as it may designate; (ii) that the Board of Directors may, subject to applicable law, adopt a shareholder rights plan upon such terms and conditions as it deems expedient and in the best interests of Keenova; (iii) for an advance notice procedure for shareholder proposals to be brought before a general meeting, including proposed nominations of persons for election to the Board of Directors; and (iv) that the Board of Directors may fill vacancies on the Board of Directors in certain circumstances.
These and other provisions may discourage potential takeover attempts, discourage bids for Keenova ordinary shares at a premium over the market price or adversely affect the market price of, and the voting and other rights of the holders of, the Keenova ordinary shares. These provisions could also discourage proxy contests and make it more difficult for Keenova shareholders to elect directors other than the candidates nominated by the Board of Directors.
54
Our ability to pay dividends and fund share repurchases is limited, and Irish law requires that we meet certain financial requirements before we pay dividends or fund repurchases of our ordinary shares
Under Irish law, we may only pay dividends, fund the repurchase of shares, and make other distributions out of distributable reserves. Distributable reserves are the accumulated realized profits that have not previously been utilized in a distribution or capitalization, less accumulated realized losses that have not previously been written off in a reduction or reorganization of capital and may include reserves created through a capital reduction process under Irish law as described below. In addition, no dividend may be paid by us unless our net assets are equal to, or exceed, the aggregate of our called-up share capital plus undistributable reserves and the dividend does not reduce our net assets below such aggregate. The Irish financial statements for the fiscal year ended December 31, 2025 will set out the amount of available distributable reserves, and our ability to pay dividends, fund repurchases of shares or make other distributions is therefore limited to this amount currently, and may be further limited if this amount reduces (for example as a result of losses being incurred by Keenova Pharmaceuticals plc at entity level, including as a result of management expenses or impairment reviews). We have not historically paid dividends. We have not made any share repurchases during the period ended December 31, 2025.
Since our generation of realized profits alone may not result increased (or maintained) levels of distributable reserves, the creation of increased distributable reserves under Irish law would require a capital reduction process involving the identification of non-distributable reserves convertible to capital, followed by the conversion of such capital. Such process would require a special shareholder resolution, approved by greater than 75% of votes cast by our shareholders, followed by the approval of the Irish High Court. The duration and outcome of such a process is uncertain, and there is no guarantee of Keenova having the increased capacity to pay dividends, repurchase shares or carry out other forms of distribution to shareholders. that would result from such a process. Alternatively, the creation of distributable reserves could result from the reversal of previous impairment of asset values as part of an impairment review; there is no guarantee that such a review will occur and that the outcome of such review would be the creation of distributable reserves.
Any determination to pay dividends in the future will be at the sole discretion of our Board of Directors after considering our financial condition, results of operations, capital requirements, general business conditions and other factors our Board of Directors may deem relevant, and subject to compliance with contractual restrictions (such as debt covenants) and applicable laws including Irish law restrictions summarized above.
| Item 1B. | Unresolved Staff Comments. | ||||
Not applicable.
Item 1C. | Cybersecurity. | ||||
Cybersecurity Program & Strategy
Risk Management Processes and Controls
Our Company has implemented safeguards that include documented standards, procedures, policies, tools supporting network and endpoint detection and protection, access management, security monitoring, and patch and vulnerability management. Applicable personnel are provided with cybersecurity awareness training and receive periodic awareness updates through ad hoc communications on security topics, such as how to spot phishing emails, or how to report suspicious activity or potential incidents. We engage qualified third parties for independent assessments and managed detection and response services to supplement internal controls and monitoring. Notwithstanding these measures, vulnerabilities or threats identified through our cybersecurity program may nonetheless take time to remediate or mitigate.
Third-Party Risk Oversight
55
Governance
Management Oversight
We acknowledge the potential risks associated with the deployment and use of AI systems. We provide training for employees on AI use cases and have an enterprise AI risk governance program that enables leaders from IT, Cybersecurity, Legal, Audit, and Commercial Compliance to review AI project controls.
Board Oversight
Process for Assessing, Identifying and Managing Material Risks from Cybersecurity Threats
To assess, identify, and manage potential cybersecurity threats, our Security Operations Center team monitors systems and threats, including those on systems managed by third parties, such as cloud platforms.
We maintain cybersecurity insurance that provides coverage for certain costs related to cybersecurity-related incidents. However, the amount or type of coverage may not be sufficient to address costs for handling an incident, or future changes may occur to insurance coverage.
| Item 2. | Properties. | ||||
As of December 31, 2025, we owned or leased building space in 24 locations globally. Our principal executive offices are located at College Business & Technology Park, Cruiserath, Blanchardstown, Dublin 15, Ireland, with additional office facilities in New Jersey, Pennsylvania, Missouri, D.C., and Japan, among others. We have a strong U.S. manufacturing footprint, with five facilities in Louisiana, New Jersey, New York, Pennsylvania and Wisconsin. Our sites consist of 123,000 and 569,000 square feet of owned and leased property, respectively. We believe our facilities are in good condition and suitable for their current use. We may reduce, add or replace facilities as considered appropriate to meet the needs of our business. See Note 13 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for additional information regarding our leased facilities.
56
| Item 3. | Legal Proceedings. | ||||
We are subject to various legal proceedings and claims, including government investigations, environmental matters, product liability matters, patent infringement claims, antitrust matters, securities class action lawsuits, personal injury claims, employment disputes, contractual and other commercial disputes, and other legal proceedings, sometimes in the ordinary course of business. Although it is not feasible to predict the outcome of these matters, we believe, unless otherwise indicated, given the information currently available, that the ultimate resolution of any particular matter, or matters that have the same legal or factual issues, would not have a material adverse effect on our financial condition, results of operations and cash flows.
For further information regarding our material pending legal proceedings, refer to Note 20 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report, which is incorporated by reference into this Item 3. Legal Proceedings.
| Item 4. | Mine Safety Disclosures. | ||||
Not applicable.
57
PART II
| Item 5. | Market for Registrant's Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities. | ||||
On November 14, 2023, upon emergence from the 2023 Bankruptcy Proceedings, all outstanding ordinary shares of our Predecessor were cancelled, and we issued a total of 19,696,335 new ordinary shares. There is currently no established public trading market for our ordinary shares.
As of April 8, 2026, we had 39,581,987 ordinary shares outstanding, held by 513 shareholders of record. The number of record holders does not necessarily bear any relationship to the number of beneficial owners of our ordinary shares.
Under Irish law, we can only pay dividends and repurchase shares out of distributable reserves. We did not declare or pay any dividends and we do not currently intend to pay dividends in the foreseeable future.
Item 6. | Reserved. | ||||
Item 7. | Management's Discussion and Analysis of Financial Condition and Results of Operations. | ||||
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our consolidated financial statements and the accompanying notes included within this Annual Report. The following discussion may contain forward-looking statements that reflect our plans, estimates and beliefs and involve risks, uncertainties and assumptions. Our actual results could differ materially from those discussed in these forward-looking statements. Factors that could cause or contribute to these differences include those discussed in Item 1A. Risk Factors and “Forward-Looking Statements” included within this Annual Report.
Fiscal Year
In the fourth quarter of fiscal year 2025, we approved a change in our fiscal year end from a 52-53-week year ending on the last Friday of December to a calendar year ending on December 31, 2025. As a result of this change, fiscal 2025 includes five additional operating days. The change in fiscal year did not result in a material impact to the fiscal 2025 results of operations. Beginning with fiscal 2026, our fiscal year will correspond to the calendar year from January 1 through December 31.
The number of operating days in the quarterly periods in fiscal year 2026 under a calendar year end will change as compared to the 52-53 weeks periods in fiscal year 2025 and prior periods, which will have an impact on the Company’s quarterly financial results as well as the comparative presentation of period over period information.
Overview of Business
We are a leading U.S.-focused branded therapeutics company that strives to help patients with rare or unaddressed conditions live happier and healthier lives. Our rare disease capabilities underpin our diversified brands portfolio, which is focused across a wide range of therapeutics areas of significant unmet need. These include rheumatology, ophthalmology, nephrology, neurology, pulmonology, orthopedics, urology, and neonatal respiratory critical care.
Our operating results for the year ended December 31, 2025 reflect the operating results of Endo following the closing of the Business Combination on July 31, 2025. Further, following the Separation on November 10, 2025, our financial statements and accompanying notes have been recast to reflect Par Health’s assets, liabilities, results of operations and cash flows as discontinued operations for all periods presented. Refer to Note 5 and Note 6 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for further information on the Business Combination and the Separation, respectively.
For further information on our business and products, refer to Item 1. Business included within this Annual Report.
Financial Highlights
The sections that follow summarize the results of our operations for the periods presented. The financial effects of certain significant transactions, including most notably our emergence from the 2023 Bankruptcy Proceedings and application of fresh-start accounting in 2023 and the Business Combination and the Separation in 2025, significantly affect the comparability of our operating results and make it difficult to compare our underlying business performance during the periods presented in this Annual Report. Our results of operations as reported in our consolidated financial statements for the Successor and Predecessor periods are in accordance with GAAP. Due to the application of fresh-start accounting in November 2023, the results for the year ended December 27, 2024, are not comparable to the results for the Successor period from November 15, 2023, through December 29, 2023, and a comparison of the results in those periods would not be meaningful to a reader of our financial statements. Accordingly, in the discussion that follows, we have arithmetically combined the financial information of the Predecessor and Successor periods in fiscal 2023 and provided commentary regarding the significant items affecting the results in those periods. While this presentation is not in accordance with GAAP, we believe that for purposes of discussion and analysis in this Annual Report, the combined financial information is useful for
58
management and investors to assess our ongoing financial and operational performance and trends. Unless otherwise indicated, all dollar amounts are presented in millions.
We operate our business in one operating and reportable segment. Accordingly, the discussion that follows focuses on our results of operations, our liquidity and our cash flows on a consolidated basis, with appropriate disaggregation where necessary to provide better insight into our operating performance, such as net sales.
Net sales and (loss) income from continuing operations are as follows:
| GAAP | GAAP | GAAP | GAAP | Non-GAAP | GAAP | Non-GAAP | ||||||||||||||||||||||||||||||||||||||
| Successor | Predecessor | Percentage Change | ||||||||||||||||||||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | Combined Fiscal Year Ended December 29 2023 | Fiscal Year Ended December 31, 2025 vs. Fiscal Year Ended December 27, 2024 | Fiscal Year Ended December 27, 2024 vs. Combined Fiscal Year Ended December 29, 2023 | ||||||||||||||||||||||||||||||||||||||
| Net sales | $ | 1,430.6 | $ | 1,083.4 | $ | 139.8 | $ | 949.2 | $ | 1,089.0 | 32.0 | % | (0.5) | % | ||||||||||||||||||||||||||||||
| Net (loss) income from continuing operations | $ | (457.5) | $ | 344.3 | $ | (39.0) | $ | (2,086.4) | $ | (2,125.4) | NM | NM | ||||||||||||||||||||||||||||||||
NM indicates that the percentage change is not meaningful or is greater than 100%.
Net sales of $1,430.6 million for the year ended December 31, 2025 increased $347.2 million, or 32% compared to net sales of $1,083.4 million for the year ended December 27, 2024. The increase is driven primarily by the net sales of products acquired from Endo of $397.0 million, coupled with growth in Acthar Gel net sales of $191.8 million, reflecting increased patient demand, partially offset by the reduction of $241.6 million in Therakos net sales in 2025 following the Therakos Divestiture in November 2024.
Net sales of $1,083.4 million for the year ended December 27, 2024 decreased $5.6 million, or 0.5%, compared to net sales of $1,089.0 million for the non-GAAP combined year ended December 29, 2023. The decrease is driven primarily by declines in net sales of INOmax, Therakos, which was sold in November 2024, and Amitiza partially offset by growth in net sales of Acthar Gel and Terlivaz.
Loss from continuing operations of $457.5 million for the year ended December 31, 2025 changed by $801.8 million when compared to income from continuing operations of $344.3 million for the year ended December 27, 2024. This change is driven primarily by the non-recurring gain of $754.4 million on the divestiture of our Therakos business in the prior period and increased selling, general and administrative (“SG&A”) expense of $299.8 million, combination, integration and other related costs related to the Business Combination of $141.2 million and R&D expenses of $1.4 million. These were partially offset by changes in income taxes of $137.1 million, liabilities management and separation costs of $43.9 million, debt extinguishment of $35.6 million and restructuring of $12.7 million, higher gross profit of $103.5 million and lower interest expense of $59.0 million.
Income from continuing operations of $344.3 million for the year ended December 27, 2024 changed by $2,469.7 million when compared to loss from continuing operations of $2,125.4 million for the non-GAAP combined year ended December 29, 2023. This change is driven primarily by $1,542.7 million of reorganization items in the prior period, the non-recurring gain of $754.4 million on the divestiture of our Therakos business and lower cost of sales of $388.1 million, decrease in interest expense of $211.2 million and liabilities management and separation costs of $110.3 million. These were partially offset by higher income taxes of $518.2 million.
See Results of Operations below for further details.
Significant Events
Separation of Par Health. On November 10, 2025, we completed the separation of our generic pharmaceuticals and sterile injectables businesses into an independent, private company named Par Health. Following the Separation, the results of Par Health are reported as discontinued operations. As a result of the Separation, we and our subsidiaries are no longer borrowers or guarantors under the Par Health Credit Agreement (as defined in Note 15 to Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report). Separation‑related costs were recorded within liabilities management and separation costs in the consolidated statements of operations. We also entered into certain agreements with Par Health, including a separation agreement, an amended and restated multi-tenant lease agreement, a manufacturing and supply agreement, a tax matters agreement and a transition services agreement to provide and receive certain services following the separation (the “Par Health TSA”). Refer to Note 6 to Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for additional details.
59
Business Combination. On July 31, 2025, we completed the acquisition of Endo through a merger transaction in which Endo shareholders received a combination of our ordinary shares and cash. Costs related to the Business Combination were recorded within combination, integration and other related costs in the consolidated statements of operations. Refer to Note 5 to the Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for additional details.
Therakos Divestiture. On November 29, 2024, we completed the Therakos Divestiture for $887.6 million and used the proceeds to mandatorily prepay and redeem senior secured debt, resulting in makewhole payments and a net loss on extinguishment of debt. Final working capital settlement impacts were recognized in 2025. In connection with the sale, we entered into a transition services agreement to provide certain business support services to the buyer. Refer to Note 6 to Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for additional details.
Transaction Incentive Plan. The Therakos Divestiture and the Business Combination each met certain criteria defined in our transaction incentive plan (as amended and restated on August 4, 2024 and December 2, 2024, the “Transaction Incentive Plan”), resulting in the recognition of compensation expense, which is reflected in SG&A expense in the consolidated statements of operations, of approximately $91.5 million during the year ended December 31, 2025, in relation to the Business Combination and approximately $11.9 million and $15.4 million during the years ended December 31, 2025, and December 27, 2024, respectively, related to the Therakos Divestiture. Refer to Note 5 and Note 6 to Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for additional details.
Emergence from Voluntary Reorganization. We emerged from the 2023 Bankruptcy Proceedings in November 2023 and adopted fresh-start accounting, resulting in successor and predecessor financial statements that are not comparable. Refer to Note 3 to the Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for additional details.
Results of Operations
Net Sales
The table below sets forth a disaggregation of our net sales. A majority of our net sales were generated in the United States. Refer to Note 22 to Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report for additional details.
Net sales of key products are as follows:
| GAAP | GAAP | GAAP | GAAP | Non-GAAP | GAAP | Non-GAAP | ||||||||||||||||||||||||||||||||||||||
| Successor | Predecessor | Percentage Change | ||||||||||||||||||||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | Combined Fiscal Year Ended December 29 2023 | Fiscal Year Ended December 31, 2025 vs. Fiscal Year Ended December 27, 2024 | Fiscal Year Ended December 27, 2024 vs. Combined Fiscal Year Ended December 29, 2023 | ||||||||||||||||||||||||||||||||||||||
| Acthar Gel | $ | 677.5 | $ | 485.7 | $ | 57.0 | $ | 368.3 | $ | 425.3 | 39.5 | % | 14.2 | % | ||||||||||||||||||||||||||||||
Xiaflex(1) | 246.6 | — | — | — | — | NM | NM | |||||||||||||||||||||||||||||||||||||
| INOmax | 244.8 | 261.4 | 35.3 | 267.9 | 303.2 | (6.4) | (13.8) | |||||||||||||||||||||||||||||||||||||
| Therakos | — | 241.6 | 39.1 | 220.0 | 259.1 | (100.0) | (6.8) | |||||||||||||||||||||||||||||||||||||
| Amitiza | 70.6 | 62.8 | 4.9 | 67.7 | 72.6 | 12.4 | (13.5) | |||||||||||||||||||||||||||||||||||||
Other Products(1) | 160.4 | 31.8 | 3.4 | 21.0 | 24.4 | NM | 30.3 | |||||||||||||||||||||||||||||||||||||
License Revenue (1) | 30.7 | 0.1 | 0.1 | 4.3 | 4.4 | NM | (97.7) | |||||||||||||||||||||||||||||||||||||
| Net sales | $ | 1,430.6 | $ | 1,083.4 | $ | 139.8 | $ | 949.2 | $ | 1,089.0 | 32.0 | % | (0.5) | % | ||||||||||||||||||||||||||||||
NM indicates that the percentage change is not meaningful or is greater than 100%.
(1)Is or contains products acquired in the Business Combination. Net sales during the year ended December 31, 2025 include five months of sales totaling $397.0 million from products acquired in the Business Combination. Accordingly, there are no comparable net sales for these products in prior periods.
Acthar Gel net sales increased $191.8 million, or 39.5%, for the year ended December 31, 2025, compared to the year ended December 27, 2024, driven primarily by increased patient demand and continued momentum in SelfJect uptake due to commercial investments and strong execution that drove category awareness and expansion. Acthar Gel net sales increased by $60.4 million, or 14.2% for the year ended December 27, 2024, compared to the non-GAAP combined year ended December 29, 2023, driven primarily by patient demand and the successful launch of SelfJect on August 6, 2024, with favorable physician and patient feedback, providing patients with an important new option to manage challenging chronic and acute inflammatory and autoimmune conditions.
60
Xiaflex net sales during the five-month period following the Business Combination were $246.6 million, driven by increased demand stemming from Peyronie’s disease and price increases.
INOmax net sales decreased $16.6 million, or 6.4%, for the year ended December 31, 2025, compared to the year ended December 27, 2024, driven primarily by continued competition in the U.S. from alternative nitric oxide products, which could continue to adversely affect our ability to successfully maximize the value of INOmax and have an adverse effect on our financial condition, results of operations, and cash flows. Following the successful introduction of the INOmax EVOLVE DS device pilot program in 2024, we remain focused on expanding the multi-year rollout of EVOLVE to U.S. hospitals nationwide in order to help meet the needs of neonatal intensive care patients and healthcare professionals by offering improved automation, which enhances safety features, and a streamlined design that elevates the user experience. Net sales decreased $41.8 million, or 13.8%, for the year ended December 27, 2024, compared to the non-GAAP combined year ended December 29, 2023, driven primarily by competition in the U.S. from alternative nitric oxide products.
We completed the Therakos Divestiture on November 29, 2024. Accordingly, there were no sales during the year ended December 31, 2025. Net sales decreased $17.5 million, or 6.8%, for the year ended December 27, 2024, compared to the non-GAAP combined year ended December 29, 2023, reflecting eleven months of sales in the year ended December 27, 2024 compared to a full year in the non-GAAP combined year ended December 29, 2023.
Amitiza net sales increased $7.8 million, or 12.4%, for the year ended December 31, 2025, compared to the year ended December 27, 2024, driven primarily by increased volume in Japan and, to a lesser extent, in the U.S. Net sales decreased $9.8 million, or 13.5%, for the year ended December 27, 2024, compared to the non-GAAP combined year ended December 29, 2023, driven primarily by price declines as a result of additional generic competitors in the market.
Other Products net sales increased $128.6 million for the year ended December 31, 2025, compared to the year ended December 27, 2024, driven by five months of sales from products acquired in the Business Combination, including primarily Supprelin LA of $29.8 million, Percocet of $28.7 million, Aveed of $22.0 million, Edex of $17.0 million and Testopel of $16.8 million, coupled with an increase in Terlivaz net sales of $8.2 million, or 33.2% compared to the year ended December 27, 2024, driven by continued improvements in hospital adoptions resulting from ongoing engagement with healthcare providers emphasizing the importance of early patient identification and treatment initiation. Other Products net sales increased by $7.4 million, or 30.3%, for the year ended December 27, 2024 compared to the non-GAAP combined year-ended December 29, 2023, as we continued to expand adoption of Terlivaz during fiscal 2024 through outreach to healthcare providers emphasizing the importance of early patient identification and treatment initiation. Net sales from certain of our Other Products have been and will continue to be negatively impacted by competitive pressures and other factors, which could unfavorably impact future net sales of these products.
License revenue for the year ended December 31, 2025, primarily represents five months of royalties on net sales under certain license arrangements acquired in the Business Combination, including royalties associated with a sales-based milestone earned by our license partner during the fourth quarter of 2025. Such milestones are not expected to recur going forward. License revenues decreased $4.3 million for the year ended December 27, 2024 compared to the non-GAAP combined year-ended December 29, 2023 due to the elimination of U.S. royalties under certain prior license agreements.
61
Cost of Sales and Operating Expenses
The table below sets forth a comparison of cost of sales and operating expenses for the relevant periods presented. Amounts for the year-ended December 31, 2025 reflect the inclusion of costs for the five-month period following the Business Combination.
| GAAP | GAAP | GAAP | GAAP | Non-GAAP | GAAP | Non-GAAP | |||||||||||||||||||||||||||||||||||||||||
| Successor | Predecessor | Percentage Change | |||||||||||||||||||||||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | Combined Fiscal Year Ended December 29 2023 | Fiscal Year Ended December 31, 2025 vs. Fiscal Year Ended December 27, 2024 | Fiscal Year Ended December 27, 2024 vs. Combined Fiscal Year Ended December 29, 2023 | |||||||||||||||||||||||||||||||||||||||||
| Cost of sales | $ | 787.8 | $ | 544.1 | $ | 84.9 | $ | 847.3 | $ | 932.2 | 44.8 | % | (41.6) | % | |||||||||||||||||||||||||||||||||
| Selling, general and administrative expenses | $ | 761.3 | $ | 461.5 | $ | 56.0 | $ | 365.6 | $ | 421.6 | 65.0 | % | 9.5 | % | |||||||||||||||||||||||||||||||||
| Combination, integration, and other related expenses | $ | 141.2 | $ | — | $ | — | $ | — | $ | — | NM | NM | |||||||||||||||||||||||||||||||||||
| Research and development expenses | $ | 90.7 | $ | 89.3 | $ | 11.8 | $ | 74.5 | $ | 86.3 | 1.6 | % | 3.5 | % | |||||||||||||||||||||||||||||||||
| Restructuring (benefit) charges, net | $ | (2.2) | $ | 10.5 | $ | — | $ | 0.9 | $ | 0.9 | NM | NM | |||||||||||||||||||||||||||||||||||
| Non-restructuring impairment charges | $ | — | $ | — | $ | 2.6 | $ | 50.1 | $ | 52.7 | NM | (100.0) | % | ||||||||||||||||||||||||||||||||||
| Liabilities management and separation costs | $ | — | $ | 43.9 | $ | 1.4 | $ | 152.8 | $ | 154.2 | (100.0) | % | (71.5) | % | |||||||||||||||||||||||||||||||||
__________
NM indicates that the percentage change is not meaningful or is greater than 100%.
Cost of sales. Cost of sales for the year ended December 31, 2025, increased $243.7 million, or 44.8% compared to the year ended December 27, 2024, driven primarily by higher intangible asset amortization of $49.2 million and higher inventory fair value step up amortization of $141.9 million arising from the application of fresh-start accounting in 2023 and the Business Combination in 2025, and higher inventory provisions of approximately $22.8 million, coupled with increased costs of sales relating to products acquired in the Business Combination. Cost of sales for the year ended December 27, 2024, decreased $388.1 million, or 41.6% compared to the non-GAAP combined year ended December 29, 2023, driven by lower intangible asset amortization of $381.6 million and $44.0 million related to the non-recurrence of an Acthar Gel inventory write-down to net realizable value which occurred during the non-GAAP combined year ended December 29, 2023, partially offset by higher inventory fair value step-up amortization of $45.4 million.
Selling, general and administrative expenses. SG&A expenses for the year ended December 31, 2025, increased $299.8 million, or 65.0% compared to the year ended December 27, 2024. The increase was driven primarily by incremental compensation costs of $184.0 million, including an increase of $88.0 million related to the Transaction Incentive Plan, $30.4 million of additional share-based compensation costs, and increased salaries and other employee compensation costs of $65.6 million, which includes the impact of increased headcount and related compensation costs following the Business Combination, coupled with increased advertising costs of $38.8 million, increased third-party professional services costs of $24.9 million, increases in the estimated fair value of contingent consideration of $11.5 million, litigation settlement costs of $5.2 million, and the non-recurrence of the recovery of bad debt expense of $6.4 million and a $2.5 million gain during the year ended December 27, 2024, as described further below. The remaining increase reflects higher operating costs across a wide range of spend categories following the Business Combination, including information technology, utilities, insurance, payroll and other taxes, and employee benefits costs, among others. SG&A expenses for the year ended December 27, 2024, increased by $39.9 million, or 9.5%, compared to the non-GAAP combined year ended December 29, 2023. The increase is attributable to incremental compensation costs of $25.0 million, including $15.4 million associated with the Transaction Incentive Plan as a result of the Therakos Divestiture, coupled with an unfavorable change in the fair value adjustment in contingent consideration liabilities of the Terlivaz CVR of $10.5 million, partially offset by a recovery of bad debt expense of $6.4 million related to a customer’s emergence from bankruptcy and a $2.5 million gain related to the ceased commercialization and wind down of production of StrataGraft® (allogeneic cultured keratinocytes and dermal fibroblasts in murine collagen - dsat) (“StrataGraft”) during the year ended December 27, 2024.
Combination, integration and other related expenses. Transaction expenses associated with the Business Combination are included in combination, integration, and other related expenses in the consolidated statements of operations. During the year ended December 31, 2025, the Company recorded $141.2 million of costs, which includes legal, financial, other advisory and consulting costs, which primarily relate to shareholder matters, integration planning and execution, and regulatory matters associated with the Business Combination, as well as severance costs of approximately $44.1 million.
62
Research and development expenses. R&D expenses for the year ended December 31, 2025, increased $1.4 million, or 1.6% compared to the year ended December 27, 2024. The increase is primarily driven by the inclusion of five months of costs associated with ongoing Xiaflex development programs following the Business Combination, offset by reductions due to the Therakos Divestiture. R&D expenses for the year ended December 27, 2024, increased $3.0 million, or 3.5% compared to the non-GAAP combined year ended December 29, 2023, driven by costs associated with clinical studies and publishing clinical and non-clinical experiences and evidence that support health economic activities and patient outcomes.
Restructuring (benefit) charges, net. During the year ended December 31, 2025, we recognized $2.2 million of income related to a vendor refund associated with the wind down of production of StrataGraft. During the year ended December 27, 2024, we incurred $10.5 million of restructuring and related charges, net, related to one-time termination benefits and contract termination costs for ceased commercialization and clinical development and wind down of production of StrataGraft. During the non-GAAP combined year ended December 29, 2023, we incurred $0.9 million of restructuring and related charges, net, primarily related to employee severance and benefits.
Non-restructuring impairment charges. During the non-GAAP combined year ended December 29, 2023, we recorded non-cash, non-restructuring impairment charges of $52.7 million related to the full impairment of our StrataGraft long-lived assets of $2.6 million and intangible asset of $50.1 million.
Liabilities management and separation costs. During the year ended December 27, 2024, we incurred $43.9 million of liabilities management and separation costs primarily related to professional fees and similar costs as we explored potential sales of non-core assets to enable further deleveraging post-emergence from the 2023 Bankruptcy Proceedings. During the non-GAAP combined year ended December 29, 2023, we incurred $154.2 million of professional fees (including where we were responsible for the fees of third parties) in connection with the evaluation of our then-existing financial situation and related discussions with our stakeholders.
Non-Operating Items
| GAAP | GAAP | GAAP | GAAP | Non-GAAP | GAAP | Non-GAAP | |||||||||||||||||||||||||||||||||||||||||
| Successor | Predecessor | Percentage Change | |||||||||||||||||||||||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | Combined Fiscal Year Ended December 29 2023 | Fiscal Year Ended December 31, 2025 vs. Fiscal Year Ended December 27, 2024 | Fiscal Year Ended December 27, 2024 vs. Combined Fiscal Year Ended December 29, 2023 | |||||||||||||||||||||||||||||||||||||||||
| Interest expense | $ | (168.8) | $ | (227.8) | $ | (28.3) | $ | (410.7) | $ | (439.0) | (25.9) | % | (48.1) | % | |||||||||||||||||||||||||||||||||
| Interest income | $ | 19.6 | $ | 23.1 | $ | 0.4 | $ | 11.2 | $ | 11.6 | (15.2) | % | 99.1 | % | |||||||||||||||||||||||||||||||||
| (Loss) gain on divestiture | $ | (5.9) | $ | 754.4 | $ | — | $ | — | $ | — | NM | NM | |||||||||||||||||||||||||||||||||||
| Gain (loss) on debt extinguishment, net | $ | 15.9 | $ | (19.7) | $ | — | $ | — | $ | — | NM | NM | |||||||||||||||||||||||||||||||||||
| Other income (expense), net | $ | 6.1 | $ | (6.5) | $ | 5.4 | $ | (6.7) | $ | (1.3) | NM | NM | |||||||||||||||||||||||||||||||||||
| Reorganization items, net | $ | — | $ | — | $ | (3.5) | $ | (1,539.2) | $ | (1,542.7) | NM | (100.0) | % | ||||||||||||||||||||||||||||||||||
| Income tax benefit (expense) | $ | 23.8 | $ | (113.3) | $ | 3.9 | $ | 401.0 | $ | 404.9 | NM | NM | |||||||||||||||||||||||||||||||||||
__________
NM indicates that the percentage change is not meaningful or is greater than 100%.
Interest expense. Interest expense during the year ended December 31, 2025, decreased $59.0 million, or 25.9% compared to the year ended December 27, 2024, driven by lower coupon interests rates on the debt obligations assumed in connection with the Business Combination as compared to the Second-out Takeback Term Loan (as defined below) due November 2028 and the 14.75% Second-out Takeback Notes (as defined below) due November 2028, partially offset by higher average outstanding debt balances reflecting the assumption of Endo’s outstanding debt obligations. Interest expense for the year ended December 27, 2024 decreased, $211.2 million, or 48.1%, compared to the non-GAAP combined year ended December 29, 2023, driven by a significant decrease in the average outstanding debt balances in connection with our emergence from the 2023 Bankruptcy Proceedings.
Interest income. Interest income for the years ended December 31, 2025, December 27, 2024, and for the non-GAAP combined year ended December 29, 2023 was primarily related to interest received on money market accounts as well as interest income on our interest rate cap agreement.
(Loss) gain on divestiture. During the year ended December 27, 2024, we completed the Therakos Divestiture for total cash consideration of $887.6 million, net of preliminary purchase price adjustments, including an adjustment based on estimated net working capital at close, and recognized a gain on sale of $754.4 million. We paid $6.2 million for the final working capital settlement during the year ended December 31, 2025.
63
Gain (loss) on debt extinguishment, net. During the year ended December 31, 2025, as a result of the mandatory prepayment of our Second-Out Takeback Term Loans and Takeback Notes, we recorded $15.9 million as a net gain on debt extinguishment, comprised of $40.2 million to write-off certain unamortized premiums, net of debt issuance costs, offset by the $24.3 million payment of the makewhole premium. During the year ended December 27, 2024, after the sale of the Therakos business, we made mandatory prepayments on our First-Out Takeback Term Loans (as defined below), Second-Out Takeback Term Loans and our Takeback Notes, as previously discussed, resulting in a loss on debt extinguishment, net, of $19.7 million primarily driven by the makewhole premium of $63.7 million offset by the gain on unamortized premium write off of $44.0 million.
Other income (expense), net. During the years ended December 31, 2025, and December 27, 2024, we recorded other income of $6.1 million and other expense of $6.5 million, respectively. Other income during 2025 primarily reflects income of $9.5 million associated with the Therakos TSA and the Par Health TSA, partially offset by $5.3 million of unrealized losses related to the changes in fair value of derivative assets and liabilities and $1.7 million of unrealized losses related to our investment in Silence Therapeutics. Other expense in 2024 primarily relates to $17.4 million of unrealized losses on equity securities related to our investments in Silence Therapeutics plc, offset by a $7.6 million unrealized gain related to the changes in fair value of derivative assets and liabilities as discussed further in Note 21 to the Consolidated Financial Statements included in Item 8. Financial Statements and Supplementary Data of this Annual Report. During the non-GAAP combined year ended December 29, 2023, we incurred other expense of $1.3 million.
Reorganization items, net. During the period November 15, 2023, through December 29, 2023 (Successor), we incurred expenses of $3.5 million from reorganization items, net, comprised entirely of professional fees associated with the implementation of the 2023 Plan. Beginning December 29, 2023, professional fees directly related to the 2023 Bankruptcy Proceedings that were previously reflected as reorganization items, net, are classified within SG&A expenses. During the period December 31, 2022, through November 14, 2023 (Predecessor) we recognized $1,539.2 million of reorganization items, net. These expenses were primarily driven by the loss on application of fresh-start accounting of $1,452.7 million, $1,139.5 million related to adjustments of other claims, $154.6 million of debt financing costs, approximately $61.7 million related to professional and other service provider fees, among other costs. These costs were partially offset by a $1,966.0 million gain on settlement of liabilities subject to compromise in accordance with the 2023 Plan.
Income tax (benefit) expense. The table below sets forth the (loss) income from continuing operations before income taxes, income tax (benefit) expense and the effective tax rate for each corresponding period:
| GAAP | GAAP | GAAP | GAAP | Non-GAAP | |||||||||||||||||||||||||||||||
| Successor | Predecessor | ||||||||||||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | Combined Fiscal Year Ended December 29 2023 | |||||||||||||||||||||||||||||||
| (Loss) income from continuing operations before income taxes | $ | (481.3) | $ | 457.6 | $ | (42.9) | $ | (2,487.4) | $ | (2,530.3) | |||||||||||||||||||||||||
| Income tax (benefit) expense | $ | (23.8) | $ | 113.3 | $ | (3.9) | $ | (401.0) | $ | (404.9) | |||||||||||||||||||||||||
| Effective tax rate | 4.9 | % | 24.8 | % | 9.1 | % | 16.1 | % | 16.0 | % | |||||||||||||||||||||||||
The table below sets forth the components of income tax expense (benefit) for each corresponding period:
| GAAP | GAAP | GAAP | GAAP | Non-GAAP | GAAP | Non-GAAP | |||||||||||||||||||||||||||||||||||||||||
| Successor | Predecessor | Percentage Change | |||||||||||||||||||||||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | Combined Fiscal Year Ended December 29 2023 | Fiscal Year Ended December 31, 2025 vs. Fiscal Year Ended December 27, 2024 | Fiscal Year Ended December 27, 2024 vs. Combined Fiscal Year Ended December 29, 2023 | |||||||||||||||||||||||||||||||||||||||||
| Current tax expense (benefit) | $ | 30.4 | $ | (23.4) | $ | (0.4) | $ | 41.1 | $ | 40.7 | NM | NM | |||||||||||||||||||||||||||||||||||
| Deferred tax (benefit) expense | (54.2) | 136.7 | (3.5) | (442.1) | (445.6) | NM | NM | ||||||||||||||||||||||||||||||||||||||||
| Total tax (benefit) expense from continuing operations | $ | (23.8) | $ | 113.3 | $ | (3.9) | $ | (401.0) | $ | (404.9) | NM | NM | |||||||||||||||||||||||||||||||||||
__________
NM indicates that the percentage change is not meaningful or is greater than 100%.
For the year ended December 31, 2025 (Successor), the effective tax rate of 4.9% differs from the Irish statutory tax rate of 12.5% predominately due to statutory rate differences in our operating jurisdictions, net of valuation allowances, on pretax earnings which include the impacts of inventory step-up and intangible asset amortization expenses. Additional factors that influence our effective tax
64
rate include non-deductible combination, integration, and other related expenses, non-deductible compensation, and changes in uncertain tax positions. See Note 9. Income Taxes to our audited consolidated financial statements for further details.
Our income tax expense of $113.3 million for the year ended December 27, 2024 (Successor), was impacted by $27.2 million tax benefit associated with $246.7 million of impacts of fresh-start accounting expense which is partially offset by accretion expense related to debt and settlement obligations, $9.2 million tax benefit associated with $66.2 million of intangible asset amortization expense, $7.0 million tax benefit associated with $43.9 million of liabilities management and separation costs, $2.4 million of tax benefit associated with $10.5 million of restructuring and related charges, net, $0.7 million tax benefit associated with $19.7 million of loss on debt extinguishment, net, offset by $104.4 million of tax expense associated with $754.4 million of gain associated with the Therakos Divestiture, $18.1 million of tax expense primarily related to valuation allowance adjustments on interest within the United States, and $37.3 million tax expense on income of $90.2 million predominately associated with pretax earnings in various jurisdictions net of valuation allowances.
Our income tax benefit of $3.9 million for the period November 15, 2023, through December 29, 2023 (Successor), was impacted by $2.2 million of tax benefit associated with $14.4 million of intangible asset amortization expense, $1.1 million of tax benefit associated with impacts of fresh-start accounting and $40.5 million of inventory step-up amortization expense, $0.5 million of tax benefit associated with $2.4 million of accretion expense related to our settlement obligation, and $0.4 million of tax benefit on income of $11.4 million predominately associated with pretax earnings in various jurisdictions, net of valuation allowances, offset by $0.3 million of tax expense associated with $3.0 million of amortization related to our debt.
Our income tax benefit of $401.0 million for the period December 31, 2022, through November 14, 2023 (Predecessor), was impacted by $304.2 million of tax benefit associated with impacts on emergence and $1,539.2 million of loss on reorganization items, net, $44.7 million of tax benefit related to legal entity reorganizations, $11.5 million of tax benefit associated with $50.1 million of non-restructuring charges related to the full impairment of our StrataGraft intangible asset, $21.8 million of tax benefit associated with $152.8 million of liabilities management and separation costs, and $18.8 million of tax benefit on the loss of $745.3 million predominately associated with pretax earnings in various jurisdictions net of valuation allowances.
Liquidity and Capital Resources
Our principal sources of liquidity are cash generated from operations and access to our $400 million revolving credit facility, net of $4.0 million of outstanding standby letters of credit, which remains undrawn at December 31, 2025. Cash and cash equivalents, which primarily consisted of bank deposits and money market accounts, totaled $812.8 million at December 31, 2025, compared to $319.0 million at December 27, 2024. Our principal liquidity requirements include working capital, capital expenditures, principal and interest payments associated with our indebtedness, lease obligations and purchase obligations, and our obligation related to the Acthar Gel-Related Litigation Settlement. From time to time, we have also completed acquisitions, including licensing agreements, and divestitures, which have significantly affected our liquidity and financial position. For example, during the year ended December 31, 2025, we completed the Business Combination and the Separation, described further below. The net effect of these transactions is expected to result in an increase in future cash flows from operations, which will be used to fund our operations and to make future debt principal and interest payments associated with our increased indebtedness. We have historically generated, and expect to continue to generate, positive cash flows from operations. We expect foreseeable liquidity and capital resource requirements to be met through existing cash and cash equivalents and anticipated cash flows from operations, as well as long-term borrowings if needed. We believe that our sources of liquidity are adequate to fund our operations for the next twelve months and beyond the next twelve months. Our ability to fund our capital needs is impacted by our ongoing ability to generate cash from operations and access to capital markets.
Business Combination and the Separation
We have incurred significant costs in connection with the Business Combination. Transaction expenses associated with the Business Combination are included in combination, integration, and other related expenses in the consolidated statements of operations. During the year ended December 31, 2025, the Company recorded $141.2 million of costs, which includes legal, financial, other advisory and consulting costs, which primarily relate to shareholder matters, integration planning and execution, and regulatory matters associated with the Business Combination, as well as severance costs of approximately $44.1 million. We expect to continue to incur costs related to the Business Combination as we seek to further integrate the acquired Endo business and to achieve anticipated synergies.
There can be no assurances that the anticipated benefits of the Business Combination and the Separation will be realized fully within the expected timeframe or at all or such benefits may take longer to realize or cost more than expected, which could materially impact our business, cash flows, financial condition, and results of operations.
Cash Requirements and Sources From Existing Contractual Arrangements
Our material cash requirements from known contractual obligations include debt obligations, legal settlements, lease obligations, purchase obligations, and other liabilities reflected on our balance sheet, as presented and discussed below.
65
The following table summarizes our contractual obligations as of December 31, 2025:
| Payments Due By Period | |||||||||||||||||||||||||||||
| Total | Less than 1 year | 1 - 3 years | 3 - 5 years | More than 5 years | |||||||||||||||||||||||||
Long-term debt obligations (1) | $ | 2,481.3 | $ | 15.0 | $ | 30.0 | $ | 30.0 | $ | 2,406.3 | |||||||||||||||||||
Interest on long-term debt obligations (2) | 1,047.8 | 196.7 | 389.7 | 386.0 | 75.4 | ||||||||||||||||||||||||
Acthar Gel-Related Litigation Settlement (3) | 193.3 | 33.7 | 96.8 | 62.8 | — | ||||||||||||||||||||||||
Operating lease obligations (4) | 102.4 | 18.5 | 37.0 | 21.7 | 25.2 | ||||||||||||||||||||||||
Purchase obligations (5) | 21.9 | 9.1 | 12.6 | 0.2 | — | ||||||||||||||||||||||||
| Total contractual obligations | $ | 3,846.7 | $ | 273.0 | $ | 566.1 | $ | 500.7 | $ | 2,506.9 | |||||||||||||||||||
(1)For further details on our debt obligations, refer to Note 15 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report.
(2)Interest on long-term debt obligations are projected for future periods using interest rates in effect as of December 31, 2025. Contractual obligations under the long-term debt agreements have been shown in the table above. Certain of these projected interest payments may differ in the future based on changes in market interest rates.
For further information regarding the fixed and variable rates of our debt obligations, refer to Note 15 of the Notes to the Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report.
(3)Acthar Gel-Related Litigation Settlement includes cash interest obligation of $1.2 million, $1.8 million, and $0.4 million due within one year, one to three years and three to five years, respectively.
(4)Includes obligations for leases with an initial term of 12 months or less and not recorded on the consolidated balance sheet. Refer to Note 13 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for further information on our lease liabilities.
(5)Purchase obligations consist of commitments for purchases of goods and services made in the ordinary course of business to meet operational requirements.
Indebtedness
As of December 31, 2025, total debt principal was $2,481.3 million compared to $865.6 million as of December 27, 2024. As of December 31, 2025, such total debt principal outstanding consists of the following Endo legacy indebtedness that remained outstanding following the Business Combination and is an obligation of certain of our subsidiaries: (i) an undrawn (net of outstanding letters of credit) revolving credit facility with a maturity date of April 23, 2029 and commitments equal to $400.0 million governed by a credit agreement (“Credit Agreement”) (ii) a term facility with a maturity date of April 23, 2031 with an outstanding principal balance of $1,481.3 million governed by the Credit Agreement and (iii) senior secured notes due April 2031 with an outstanding principal balance of $1,000.0 million issued pursuant to an Indenture (“Indenture”).
We did not file this Annual Report by its initial due date of March 31, 2026, which resulted in our failure to comply with the covenants in the Credit Agreement and in the Indenture that required us to deliver audited annual financial statements by that date. Our delivery of the audited financial statements that are contained in this Annual Report will cure the aforementioned covenant defaults.
Concurrent with the Business Combination and in contemplation of the Separation, certain of our then-existing subsidiaries entered into a credit agreement (as amended, modified or supplemented, the “Par Health Credit Agreement”) providing for a $1.2 billion senior secured term loan facility (“Par Health Term Facility”), and $150.0 million senior secured revolving credit facility (“Par Health Revolving Credit Facility” and together with the Par Health Term Facility, the “Facilities”). One of our then-existing subsidiaries borrowed $1.2 billion under the Par Health Term Facility on August 1, 2025 and the Par Health Revolving Credit Facility was never drawn.
The proceeds from the issuance of the Par Health Term Facility were used, in part, to (i) prepay in full approximately $385.5 million in outstanding aggregate principal amount of “second-out” senior secured takeback term loans (“Second-Out Takeback Term Loans”), constituting all of the remaining indebtedness then-outstanding under our then-existing credit agreement, together with a payment of approximately $10.6 million in required makewhole premium and (ii) redeem in full approximately $477.2 million in outstanding principal amount of “second-out” 14.75% senior secured first lien notes due 2028 (“Takeback Notes”), constituting all of the then-outstanding Takeback Notes, together with a payment of approximately $13.7 million in required makewhole premium.
Following the Separation, neither we nor our remaining subsidiaries continue to be borrowers or guarantors of the indebtedness under the Par Health Credit Agreement. All borrowers and guarantors in respect of the Par Health Credit Agreement are subsidiaries of Par Health.
We were required to use net proceeds from the Therakos Divestiture to prepay our Takeback Term Loans and redeem a portion of the Takeback Notes. On December 6, 2024, we (i) mandatorily prepaid a portion of our takeback term loans in an aggregate principal amount of approximately $474.1 million (of which approximately $227.1 million consisted of our “first-out” takeback term loans (“First-Out Takeback Term Loans” and, together with the Second-Out Takeback Term Loans, “Takeback Term Loans”) and approximately $247.0 million consisted of our Second-Out Takeback Term Loans) together with a payment of approximately $36.4 million in required makewhole premium and (ii) mandatorily redeemed $301.4 million in aggregate principal amount of
66
Takeback Notes together with a payment of approximately $27.3 million in required makewhole premium.
During the period November 15, 2023, through December 29, 2023 (Successor), we made a $100.0 million payment on December 22, 2023 to repay in full our receivables securitization financing facility.
See Notes 5 and 15 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for a further discussion of our debt instruments, including a discussion of the impacts of the Business Combination and the Separation.
Acthar Gel-Related Settlement
Pursuant to the 2020 Plan and the scheme of arrangement confirmed by the Irish High Court, based on and consistent in all respects with the 2020 Plan, on June 22, 2023 (the “2020 Effective Date”), all claims of the DOJ and other governmental parties against us relating to Acthar Gel were deemed to have been settled, discharged, waived, released, and extinguished in full, and we ceased to have any liability or obligation with respect to such claims, which were treated in accordance with the 2020 Plan and the terms of the settlement. We entered into an agreement with the DOJ and other governmental parties to settle a range of litigation matters and disputes relating to Acthar Gel (“Acthar Gel-Related Settlement”), including a Medicaid lawsuit with the Centers for Medicare and Medicaid Services, a related FCA lawsuit in Boston, and an Eastern District of Pennsylvania (“EDPA”) FCA lawsuit principally relating to interactions of Acthar Gel's previous owner (Questcor Pharmaceuticals Inc.) with an independent charitable foundation. To implement the Acthar Gel-Related Settlement, we entered into two settlement agreements with the U.S. and certain relators. Under the Acthar Gel-Related Settlement, which was conditioned upon us commencing Chapter 11 cases for the 2020 Bankruptcy Proceedings and provided for the distributions the applicable claimants received under the 2020 Plan, we agreed to pay $260.0 million to the DOJ and other parties over seven years and reset Acthar Gel’s Medicaid rebate calculation as of July 1, 2020, such that state Medicaid programs would receive 100% rebates on Acthar Gel Medicaid sales, based on then-current Acthar Gel pricing. The $260.0 million in payments consists of (i) a $15.0 million payment upon the 2020 Effective Date; (ii) a $15.0 million payment upon the first anniversary of the 2020 Effective Date; (iii) a $20.0 million payment upon each of the second and third anniversaries of the 2020 Effective Date; (iv) a $32.5 million payment upon each of the fourth and fifth anniversaries of the 2020 Effective Date; and (v) a $62.5 million payment upon the sixth and seventh anniversaries of the 2020 Effective Date. Also in connection with the Acthar Gel-Related Settlement, we entered into (a) separate settlement agreements with certain states, the Commonwealth of Puerto Rico, the District of Columbia and the above-noted relators, which further implement the Acthar Gel-Related Settlement, and (b) a five-year corporate integrity agreement with the Office of Inspector General of the U.S. Department of Health and Human Services in March 2022. As a result of these agreements, upon effectiveness of the Acthar Gel-Related Settlement in connection with the effectiveness of the 2020 Plan, the U.S. Government dropped its demand for approximately $640 million in retrospective Medicaid rebates for Acthar Gel and agreed to dismiss the FCA lawsuit in Boston and the EDPA FCA lawsuit. Similarly, state and territory Attorneys General also dropped related lawsuits. In turn, we dismissed our appeal of the U.S. District Court for the District of Columbia's adverse decision in the Medicaid lawsuit, which was filed in the U.S. Court of Appeals for the District of Columbia Circuit.
During the years ended December 31, 2025, and December 27, 2024, respectively, we made payments of $21.3 million and $21.4 million, inclusive of interest, related to our Acthar Gel Related Settlement and are required to make a $33.7 million payment, inclusive of interest, upon the four-year anniversary in 2026.
Other Matters
As of December 31, 2025, we had $30.4 million of unrecognized tax benefits, including interest and penalties. The timing of when the unrecognized tax benefits will be settled remains uncertain. For further information regarding unrecognized tax benefits, refer to Note 9 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for further information.
During the period December 31, 2022, through November 14, 2023 (Predecessor), we received $141.6 million of tax refunds as a result of provisions in the Coronavirus Aid, Relief and Economic Security (“CARES”) Act.
We are exposed to interest rate risk on our variable-rate debt. On March 14, 2023, we entered into an interest rate cap agreement by converting a portion of our variable-rate debt to a fixed rate through the expiration date of the interest rate cap, which served to reduce the volatility on future interest expense cash outflows. The interest rate cap agreement had a total notional value of $860.0 million with an upfront premium of $20.0 million and provides us with interest rate protection (i) for the period March 16, 2023, through July 19, 2023, to the extent that one-month London Interbank Offered Rate exceeded 4.65%, and (ii) for the period July 20, 2023, through March 26, 2026, to the extent that one-month SOFR exceeds 3.84%. The interest rate cap expired in accordance with its terms on March 26, 2026. Refer to Note 21 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for further information.
In general, we intend to fund capital expenditures with cash generated from operations.
67
Cash Flows
A summary of our cash flows from continuing operations is provided in the following table and described in further detail below:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
Net cash from: | ||||||||||||||||||||||||||
Operating activities - continuing operations | $ | 125.0 | $ | (48.0) | $ | 73.4 | $ | (187.6) | ||||||||||||||||||
Investing activities - continuing operations | 372.6 | 825.1 | (1.3) | (13.0) | ||||||||||||||||||||||
Financing activities - continuing operations | (1,178.6) | (846.4) | (102.2) | 273.2 | ||||||||||||||||||||||
Operating Activities - Continuing Operations
Net cash provided by operating activities of $125.0 million for the year ended December 31, 2025, was attributable to a loss from continuing operations of $457.5 million, adjusted for non-cash items, including depreciation and amortization of $130.5 million, amortization of inventory fair value step-up from the Business Combination of $209.0 million, share-based compensation costs of $43.7 million, inventory provisions of $31.3 million, changes in fair value of contingent consideration of $14.3 million, and loss on divestiture of $5.9 million driven by the final working capital true up for the Therakos Divestiture, partially offset by changes in deferred income taxes of $54.2 million and the gain on debt extinguishment of $15.9 million. Net cash provided by operating activities was also impacted by a change in working capital, driven by a $187.4 million inflow related to a decrease in inventory, of which $183.8 million relates to the inventory step-up expenses as a result of fresh-start accounting in 2023, a $21.3 million outflow related to a decrease in the Acthar Gel-Related Settlement and a $24.5 million net inflow in income taxes.
Net cash used in operating activities of $48.0 million for the year ended December 27, 2024, was attributable to income from continuing operations of $344.3 million, adjusted for non-cash items of $506.5 million driven by the gain on Therakos Divestiture of $754.4 million and partially offset by a decrease in deferred income taxes of $136.7 million, depreciation and amortization of $73.6 million, the loss on debt extinguishment, net of $19.7 million, inventory provisions of $8.5 million, and changes in fair value of contingent consideration of $2.8 million. The change in working capital was driven by a $210.9 million inflow related to a decrease in inventory, of which $250.9 million relates to the inventory step-up expenses as a result of fresh-start accounting in 2023, partially offset by a $46.0 million net outflow in income taxes, a $8.6 million outflow related to a decrease in accounts payable, a $21.4 million outflow related to a decrease in the Acthar Gel-Related Settlement, and a $21.4 million outflow related to a decrease in accrued consulting fees.
Net cash provided by operating activities of $73.4 million for the period November 15, 2023, through December 29, 2023 (Successor), was attributable to a loss from continuing operations of $39.0 million, adjusted for non-cash items of $22.5 million, driven by depreciation and amortization of $15.3 million, inventory provisions of $2.4 million, non-cash impairment charges of $3.8 million, and other non-cash items of $5.5 million, partially offset by deferred income taxes of $3.5 million and non-cash amortization expense on our debt obligation of $0.7 million. The change in working capital was primarily driven by an $21.3 million cash inflow related to a decrease in accounts receivable, a $36.1 million cash inflow related to a decrease in inventories, which includes $40.5 million related to the inventory step-up expenses as a result of fresh-start accounting in 2023, a $33.4 million cash inflow related to an increase in other net working capital, and a $7.6 million cash inflow related to an increase in accounts payable, partially offset by $6.8 million cash outflow related to a decrease in accrued consulting fees.
Net cash used in operating activities of $187.6 million for the period December 31, 2022, through November 14, 2023, (Predecessor), was attributable to a loss from continuing operations of $2,086.4 million, adjusted for non-cash items of $1,686.5 million, driven by reorganization items, net, of $1,477.5 million, depreciation and amortization of $457.8 million, non-cash impairment charges of $94.1 million, non-cash accretion expense of $80.0 million, inventory provisions of $9.6 million, other non-cash items of $8.2 million and share-based compensation of $8.6 million, partially offset by deferred income taxes of $442.1 million and changes in fair value of contingent consideration of $7.2 million. The change in working capital, was primarily driven by a $101.9 million net cash outflow in other working capital and a $13.9 million cash outflow related to a decrease in accounts payable, partially offset by a $169.3 million cash inflow related to an increase in income taxes payable, a $111.7 million net cash inflow related to a decrease in inventories of which $30.1 million relates to the fresh-start inventory step-up expenses as a result of fresh-start accounting in 2023 and a $37.7 million cash inflow related to a decrease in accounts receivable, net.
68
Investing Activities - Continuing Operations
Net cash provided by investing activities was $372.6 million for the year ended December 31, 2025, primarily driven by a net inflow related to unrestricted and restricted cash of $333.4 million and $93.4 million, respectively, acquired in the Business Combination, partially offset by capital expenditures of $50.4 and the payment of $6.2 million for the final working capital settlement related to the Therakos Divestiture.
Net cash provided by investing activities was $825.1 million for the year ended December 27, 2024, primarily driven by $876.2 million of proceeds related to the Therakos Divestiture partially offset by $54.8 million in capital expenditures.
Net cash used in investing activities was $1.3 million for the period November 15, 2023, through December 29, 2023 (Successor), primarily driven by $2.2 million in capital expenditures.
Net cash used in investing activities was $13.0 million for the period December 31, 2022, through November 14, 2023 (Predecessor), primarily driven by $14.2 million in capital expenditures.
Financing Activities - Continuing Operations
Net cash used in financing activities was $1,178.6 million for the year ended December 31, 2025, driven primarily by $873.2 million of debt repayments and related makewhole premiums of $24.3 million, $244.8 million divested cash from the Separation and a $30.0 million payment in connection with the CVR Termination Agreement.
Net cash used in financing activities was $846.4 million for the year ended December 27, 2024, driven primarily by debt repayments, including a $775.5 million mandatory repayment of debt principal on our Takeback Term Loans and Takeback Notes and related makewhole premium payment of $63.7 million as a result of the receipt of proceeds from the Therakos Divestiture.
Net cash used in financing activities was $102.2 million for the period November 15, 2023, through December 29, 2023 (Successor), entirely driven by debt repayments, including a $100.0 million payment to repay in full our receivables securitization financing facility.
Net cash provided by financing activities was $273.2 million for the period December 31, 2022, through November 14, 2023 (Predecessor), primarily attributable to proceeds from the issuance of $380.0 million of debt driven by the issuance of $250.0 million from the debtor-in-possession financing coupled with the draw on our receivables financing facility of $130.0 million. This was partially offset by $102.6 million in debt repayments driven by the $50.6 million cash sweep prior to our emergence from the 2020 Bankruptcy Proceedings, $30.0 million repayment on our receivables financing facility and $22.0 million repayment on our variable-rate term loans. We also incurred $4.1 million of debt issuance costs associated with our receivables financing facility.
Concentration of Credit and Other Risks
Financial instruments that potentially subject us to concentrations of credit risk primarily consist of accounts receivable. We generally do not require collateral from customers. A portion of our accounts receivable outside the U.S. includes sales to government-owned or supported healthcare systems in several countries, which are subject to payment delays. Payment is dependent upon the financial stability and creditworthiness of those countries' national economies.
Capitalization
On July 31, 2025, prior to the completion of the Business Combination, we adopted new Articles of Association, which among other things, provided that the authorized share capital of our company was $10,000,000 and €25,000, divided into 500,000,000 ordinary shares, par value $0.01 per share, 500,000,000 preferred shares, par value $0.01 per share, and 25,000 ordinary A shares, par value €1.00 per share. The preferred shares may be issued with such rights as the Board may determine.
On July 31, 2025, pursuant to the terms of the Transaction Agreement, at the effective time of the Business Combination (“Merger Effective Time”), each share of common stock of Endo issued and outstanding as of immediately prior to the Merger Effective Time, other than the shares of Endo common stock owned by Endo, us, Merger Sub, or any of our or their respective subsidiaries, was cancelled and converted into the right to receive 0.2575 of an ordinary share and approximately $1.31 in cash, without interest and subject to applicable withholding. Former holders of Endo common stock received cash in lieu of any fractional ordinary shares they would otherwise have been entitled to receive. The issuance of ordinary shares in connection with the Business Combination was registered under the Securities Act, pursuant to our registration statement on Form S-4 filed with the SEC on April 23, 2025, as amended.
Endo’s common stock outstanding immediately prior to the Business Combination was 76,313,462 shares, which resulted in the issuance of 19,650,663 ordinary shares to former holders of Endo common stock.
On October 8, 2025, our shareholders approved an ordinary resolution to subdivide and increase our authorized share capital to $3,005.0 million and €25,000 divided into 500 million ordinary shares of $0.01 each, 3 trillion preferred shares of $0.001 each (“Preferred Shares”) and 25,000 ordinary A shares of €1.00 each. On October 10, 2025, we declared the issuance of 45,564 Preferred Shares for each outstanding ordinary share to our shareholders of record as of the close of business on October 8, 2025. Under the Irish law, the preferred shares were credited as paid up pursuant to a capitalization of a merger reserve account.
69
On November 10, 2025, at 12:01 a.m. (Eastern Time in the United States), we completed the Separation, which was implemented by way of a Redemption of all of our issued and outstanding Preferred Shares, upon which the Preferred Shares were automatically cancelled and as such are no longer outstanding. In connection with the Redemption and pursuant to Irish law, we allocated the right to receive 39,421,398 shares of Par Health Common Stock to certain holders of record of Preferred Shares as of October 27, 2025. Refer to Note 17 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for additional information on the Redemption.
Shareholders' equity was $2,065.2 million as of December 31, 2025, compared to $1,645.8 million as of December 27, 2024. The increase in shareholders' equity during the year ended December 31, 2025 is primarily attributed to ordinary shares issued in connection with the Business Combination of $1,778.4 million, partially offset by the distribution of Par Health’s net assets in connection with the Separation of $879.7 million, coupled with net loss of $489.5 million and $30.0 million related to the CVR Termination Agreement.
Dividends
Historically, we have not made any cash dividend payments and we do not currently intend to pay dividends in the foreseeable future.
Commitments and Contingencies
Legal Proceedings
We are subject to various legal proceedings and claims, including governmental investigations, environmental matters, product liability matters, patent infringement claims, antitrust matters, securities class action lawsuits, personal injury claims, employment disputes, contractual and other commercial disputes, and other legal proceedings, sometimes in the ordinary course of business. Although it is not feasible to predict the outcome of these matters, we believe, unless otherwise indicated, given the information currently available, that the ultimate resolution of any particular matter, or matters that have the same legal or factual issues, would not have a material adverse effect on our financial condition, results of operations, and cash flows.
For further information regarding our material pending legal proceedings, refer to Note 20 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report.
Guarantees
In disposing of assets or businesses, we have from time to time provided representations, warranties and indemnities to cover various risks and liabilities, including unknown damage to the assets, environmental risks involved in the sale of real estate, liability to investigate and remediate environmental contamination at waste disposal sites and manufacturing facilities, and unidentified tax liabilities related to periods prior to disposition. We assess the probability of potential liabilities related to such representations, warranties and indemnities and adjust potential liabilities as a result of changes in facts and circumstances. As of December 31, 2025, we believe the likelihood of payment is remote and the fair value of such guarantees is not material. These representations, warranties and indemnities are discussed in Note 19 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report.
Off-Balance Sheet Arrangements
As of December 31, 2025, we had various letters of credit, guarantees, and surety bonds totaling $132.3 million, including approximately $37.9 million related to Par Health. In connection with the Separation, we and Par Health entered into agreements pursuant to which each party is required to use commercially reasonable efforts to cause the removal of the other party and its respective subsidiaries as guarantor of, or obligor for, certain indebtedness and other obligations following the separation. To the extent we cannot be released from any such guarantee or obligation, Par Health is required to indemnify us for any losses, costs, or exposure arising from such guarantee. Certain of these guarantees require that we maintain cash collateral, which is classified as restricted cash and is included in Prepaid expenses and other current assets and Other assets on our consolidated balance sheets. There are no off-balance sheet arrangements that are material or reasonably likely to become material to our financial condition or results of operations.
Critical Accounting Estimates
The consolidated financial statements have been prepared in U.S. dollars and in accordance with GAAP. The preparation of the consolidated financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amount of assets and liabilities, disclosure of contingent assets and liabilities and the reported amounts of revenues and expenses. The following critical accounting estimates are based on, among other things, judgments and assumptions made by management that include inherent risks and uncertainties. Management's estimates are based on the relevant information available at the end of each period.
70
Revenue Recognition
Product Sales Revenue
We sell our products through independent channels which are considered our customers, including direct to retail pharmacies, direct to hospitals and other institutions and through distributors. We also enter into arrangements with health care providers and payers, wholesalers, government agencies, institutions, managed care organizations and GPOs to establish contract pricing for certain products that provide for government (Medicare and Medicaid) and/or privately-negotiated (Managed Care) rebates, sales incentives, chargebacks, distribution service agreements fees, fees for services and administration fees and discounts with respect to the purchase of our products.
Reserve for Variable Considerations
Product sales are recorded at the net sales price (transaction price), which includes estimates of variable consideration for which reserves are established. These reserves result from estimated government (Medicare and Medicaid) and/or privately-negotiated (Managed Care) rebates, sales incentives, chargebacks, distribution service agreements fees, fees for services and administration fees and discounts that are offered within contracts between the Company and our customers and health care providers and payers, government agencies, institutions, managed care organizations and group purchasing organizations health care providers and payers relating to the sale of our products. These reserves are based on the expected value. These estimates take into consideration a range of possible outcomes for relevant factors such as our historical experience, estimated future trends, estimated customer inventory levels, contractual agreements, and the level of utilization of our products. Overall, these reserves reflect our best estimate of the amount of consideration to which we are entitled based on the terms of the contract. The amount of variable consideration that is included in the transaction price may be constrained (reduced) and is included in the net sales price only to the extent that it is probable that a significant reversal in the amount of the cumulative revenue recognized will not occur in a future period. We adjust reserves for chargebacks, government (Medicare and Medicaid) rebates, privately negotiated (Managed Care) rebates, product returns and other sales deductions to reflect differences between estimated and actual experience either on a monthly or quarterly basis (dependent on the deduction type). Such adjustments impact the amount of net sales recognized in the period of adjustment.
The following table reflects activity in our sales reserve accounts:
| Rebates and Chargebacks | Product Returns | Other Sales Deductions | Total | ||||||||||||||||||||
| Balance as of December 29, 2023 | $ | 94.6 | $ | 3.3 | $ | 0.1 | $ | 98.0 | |||||||||||||||
| Provisions | 176.8 | 8.9 | 0.6 | 186.3 | |||||||||||||||||||
| Payments or credits | (206.4) | (8.0) | (0.6) | (215.0) | |||||||||||||||||||
| Balance as of December 27, 2024 | $ | 65.0 | $ | 4.2 | $ | 0.1 | $ | 69.3 | |||||||||||||||
| Acquisitions | 117.3 | 36.0 | 2.0 | 155.3 | |||||||||||||||||||
| Provisions | 492.9 | 18.5 | 9.5 | 520.9 | |||||||||||||||||||
| Payments or credits | (440.8) | (10.4) | (9.8) | (461.0) | |||||||||||||||||||
| Balance as of December 31, 2025 | $ | 234.4 | $ | 48.3 | $ | 1.8 | $ | 284.5 | |||||||||||||||
Provisions presented in the table above are recorded as reductions to net sales. As of December 31, 2025, a five percent change in our sales reserve accounts would have led to an approximately $14.2 million impact on our loss from continuing operations before income taxes. For our disaggregation of net sales by product family, refer to Note 22 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report.
Total provisions for the years ended December 31, 2025, and December 27, 2024, were $520.9 million and $186.3 million, respectively. The increase of $334.6 million was driven primarily by the inclusion of gross-to-net provisions for products obtained through the Business Combination of $236.7 million coupled with an increase of $97.9 million primarily as a result of an increase in Acthar Gel net sales.
Product sales are recognized when the customer obtains control of our product. Control is transferred either at a point in time, generally upon delivery to the customer site, or in the case of certain of our products, over the period in which the customer has access to the product and related services. Revenue recognized over time is based upon either consumption of the product or passage of time based upon our determination of the measure that best aligns with how the obligation is satisfied. Our considerations of why such measures provide a faithful depiction of the transfer of our products are as follows:
•For those contracts whereby revenue is recognized over time based upon consumption of the product, we either have:
1.the right to invoice the customer in an amount that directly corresponds with the value to the customer of our performance to date, for which the practical expedient to recognize in proportion to the amount it has the right to invoice has been applied, or
2.the remaining goods and services to which the customer is entitled is diminished upon consumption.
71
•For those contracts whereby revenue is recognized over time based upon the passage of time, the benefit that the customer receives from unlimited access to our product does not vary, regardless of consumption. As a result, our obligation diminishes with the passage of time; therefore, ratable recognition of the transaction price over the contract period is the measure that best aligns with how the obligation is satisfied.
For additional information, refer to Note 4 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report.
Intangible Assets
Our intangible assets include developed technology. Intangible assets acquired in a business combination are recorded at fair value, while intangible assets acquired in other transactions are recorded at cost. Intangible assets with finite useful lives are amortized according to the pattern in which the economic benefit of the asset is used up over their estimated useful lives. We assess the remaining useful life and the recoverability of finite-lived intangible assets whenever events or circumstances indicate that the carrying value of an asset may not be recoverable. When a triggering event occurs, we evaluate potential impairment of finite-lived intangible assets by first comparing undiscounted cash flows associated with the asset, or the asset group they are a part of, to its carrying value. If the carrying value is greater than the undiscounted cash flows, the amount of potential impairment is measured by comparing the fair value of the assets, or asset group, with their carrying value. The fair value of the intangible asset, or asset group, is estimated using an income approach. If the fair value is less than the carrying value of the intangible asset, or asset group, the amount recognized for impairment is equal to the difference between the carrying value of the asset and the fair value of the asset. We annually test indefinite-lived intangible assets for impairment, or whenever events or changes in circumstances indicate that the carrying value may not be recoverable by either a qualitative or income approach. We compare the fair value of the assets with their carrying value and record an impairment when the carrying value exceeds the fair value. Changes in economic and operating conditions impacting these assumptions could result in intangible asset impairment in future periods.
During the fourth quarter 2025, notification of changes in third-party reimbursement coding constituted a triggering event requiring us to evaluate the recoverability of a developed technology intangible asset with a carrying value of $173.8 million as of December 31, 2025. We evaluated the recoverability of the asset by comparing the carrying value to the sum of the associated undiscounted future cash flows. Based on this analysis, we concluded that the carrying value was recoverable, and no impairment charge was recorded. While the asset was determined to be recoverable, the excess of projected undiscounted cash flows over its carrying value was limited. As a result, adverse changes in key assumptions, including future demand, pricing or operating costs, could result in future impairment charges that may be material to our results of operations.
As part of our strategic planning process, we periodically evaluate alternatives to enhance shareholder value, including the potential sale of certain assets or businesses. No assets met the criteria for held for sale reporting as of December 31, 2025; however, we are currently pursuing the potential divestiture of the Percocet business. While no assurance can be given that a transaction will be completed, or as to the timing or terms of any such transaction, these strategic initiatives may represent changes in circumstances that could impact the estimated fair values of the related assets. If the estimated fair value of these assets is less than their carrying value, we may be required to record an impairment charge in the future. Any such charge could be material and could adversely affect our results of operations and financial condition.
For more information on our intangible impairment analyses and the results thereof, refer to Note 14 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report.
Acquisitions
For acquisitions that meet the criteria for business combination accounting, the amounts paid are allocated to the tangible assets acquired and liabilities assumed based on their estimated fair values at the date of acquisition. We then allocate the purchase price in excess of net tangible assets acquired to identifiable intangible assets, including purchased research and development. The fair value of identifiable intangible assets is based on detailed valuations. These valuations rely on a number of factors including operating results, business plans, economic projections, anticipated future cash flows, transactions and market place data. There are inherent uncertainties related to these factors and judgment in applying them to estimate the fair value of individual assets acquired in a business combination. Due to these inherent uncertainties, there is risk that the carrying value of our recorded intangible assets may be overstated, which may result in an increased risk of impairment in future periods. We perform our intangible asset valuations using an income approach based on the present value of future cash flows. This approach incorporates many assumptions including future growth rates, discount factors and income tax rates. Changes in economic and operating conditions impacting these assumptions could result in impairment in future periods.
Certain asset acquisitions or license agreements may not meet the criteria for a business combination. We account for these transactions as an asset acquisition and recognize the identifiable assets acquired, the liabilities assumed and any noncontrolling interest in the acquired entity. Any initial up-front payments incurred in connection with the acquisition or licensing of our in-process research and development (“IPR&D”) product candidates that do not meet the definition of a business are treated as research and development expense.
72
Contingent Consideration
As part of certain acquisitions, we are subject to contractual arrangements to pay contingent consideration to former owners of these businesses. The payment of obligations under these arrangements are uncertain, and even if payments are expected to be made the timing of these payments may be uncertain as well. These contingent consideration obligations are required to be recorded at fair value within the consolidated balance sheet and adjusted at each respective balance sheet date, with changes in the fair value being recognized in the consolidated statement of operations. The determination of fair value is dependent upon a number of factors, which include projections of future revenues, the probability of successfully achieving certain regulatory milestones, competitive entrants into the marketplace, the timing associated with the aforementioned criteria and marketplace data (e.g., interest rates). Several of these assumptions require projections several years into the future. Due to these inherent uncertainties, there is risk that the contingent consideration liabilities may be overstated or understated. Changes in economic and operating conditions impacting these assumptions are expected to impact future operating results, with the magnitude of the impact tied to the significance in the change in assumptions. For additional information, refer to Note 20 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report.
Contingencies
We are involved, either as a plaintiff or a defendant, in various legal proceedings that arise in the ordinary course of business, including, without limitation, government investigations, product liability matters, patent infringement claims, antitrust matters, securities class action lawsuits, personal injury claims, employment disputes, contractual and other commercial disputes, and other legal proceedings, as further discussed in Note 20 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report. Accruals recorded for various contingencies, including legal proceedings, self-insurance and other claims, are based on judgment, the probability of losses and, where applicable, the consideration of opinions of internal and/or external legal counsel, internal and/or external technical consultants and actuarially determined estimates. When a range is established but a best estimate cannot be made, we record the minimum loss contingency amount. These estimates are often initially developed substantially earlier than the ultimate loss is known, and the estimates are reevaluated each accounting period as additional information becomes available. When we are initially unable to develop a best estimate of loss, we record the minimum amount of loss, which could be zero. As information becomes known, additional loss provisions are recorded when either a best estimate can be made or the minimum loss amount is increased. When events result in an expectation of a more favorable outcome than previously expected, our best estimate is changed to a lower amount. We record receivables from third-party insurers up to the amount of the related liability when we have determined that existing insurance policies will provide reimbursement. In making this determination, we consider applicable deductibles, policy limits and the historical payment experience of the insurance carriers. Receivables are not netted against the related liabilities for financial statement presentation.
Income Taxes
In determining income for financial statement purposes, we must make certain estimates and judgments. These estimates and judgments affect the calculation of certain tax liabilities and the determination of the recoverability of certain of the deferred tax assets, which arise from temporary differences between the tax and financial statement recognition of revenue and expense.
Deferred tax assets are reduced by a valuation allowance if, based on the weight of available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized. In evaluating our ability to recover our deferred tax assets, we consider all available positive and negative evidence including our past operating results, the existence of cumulative losses in the most recent years and our forecast of future taxable income. In estimating future taxable income, we develop assumptions including the amount of future state, federal and international pre-tax operating income, the reversal of temporary differences, and the implementation of feasible and prudent tax planning strategies. These assumptions require significant judgment about the forecasts of future taxable income and are consistent with the plans and estimates we use to manage the underlying businesses.
We determine whether it is more likely than not that a tax position will be sustained upon examination. The tax benefit of any tax position that meets the more-likely-than-not recognition threshold is calculated as the largest amount that is more than 50.0% likely of being realized upon resolution of the uncertainty. To the extent a full benefit is not realized on the uncertain tax position, an income tax liability or a reduction to a deferred tax asset (“contra-DTA”), is established. We adjust these liabilities and contra-DTAs as a result of changing facts and circumstances; however, due to the complexity of some of these uncertainties, the ultimate resolution may result in a payment that is materially different from our current estimate of the tax liabilities.
The calculation of our tax liabilities involves dealing with uncertainties in the application of complex tax regulations in a multitude of jurisdictions across our global operations. Changes in tax laws and rates could affect recorded deferred tax assets and liabilities in the future. Management is not aware of any such changes, however, which would have a material adverse effect on our competitive position, business, financial condition, results of operations and cash flows. Refer to Note 9 of the Notes to Consolidated Financial Statements included within Item 8. Financial Statements and Supplementary Data of this Annual Report for further information.
73
Recently Issued Accounting Standards
See Note 4 of the Notes to Consolidated Financial Statements in Item 8. Financial Statements and Supplementary Data of this Annual Report for a discussion regarding recently issued accounting standards.
Quarterly Information
As a result of the retrospective changes associated with the Separation, which is reflected as discontinued operations for all periods presented, the following table sets forth selected consolidated quarterly financial information that reflects these presentation changes.
| Three Months Ended (Unaudited) | ||||||||||||||||||||||||||
| Dollars and shares in millions, except per share amounts | December 31, 2025 | September 26, 2025 | June 27, 2025 | March 28, 2025 | ||||||||||||||||||||||
| Net sales | $ | 543.0 | $ | 416.1 | $ | 264.3 | $ | 207.2 | ||||||||||||||||||
| Gross profit | $ | 207.6 | $ | 172.4 | $ | 150.7 | $ | 112.1 | ||||||||||||||||||
| Loss from continuing operations | $ | (109.8) | $ | (241.9) | $ | (30.5) | $ | (75.3) | ||||||||||||||||||
| (Loss) income from discontinued operations, net of tax | $ | (63.3) | $ | (49.2) | $ | 32.9 | $ | 47.6 | ||||||||||||||||||
| Net (loss) income | $ | (173.1) | $ | (291.1) | $ | 2.4 | $ | (27.7) | ||||||||||||||||||
| Basic (loss) income per share: | ||||||||||||||||||||||||||
| Basic loss from continuing operations | $ | (2.78) | $ | (7.49) | $ | (1.55) | $ | (3.82) | ||||||||||||||||||
| Basic (loss) income from discontinued operations, net of tax | (1.60) | (1.52) | 1.67 | 2.42 | ||||||||||||||||||||||
| Net (loss) income | $ | (4.38) | $ | (9.01) | $ | 0.12 | $ | (1.40) | ||||||||||||||||||
| Basic weighted-average shares outstanding | 39.5 | 32.3 | 19.7 | 19.7 | ||||||||||||||||||||||
| Diluted (loss) income per share: | ||||||||||||||||||||||||||
| Diluted loss from continuing operations | $ | (2.78) | $ | (7.49) | $ | (1.55) | $ | (3.82) | ||||||||||||||||||
| Diluted (loss) income from discontinued operations, net of tax | (1.60) | (1.52) | 1.67 | 2.42 | ||||||||||||||||||||||
| Net (loss) income | $ | (4.38) | $ | (9.01) | $ | 0.12 | $ | (1.40) | ||||||||||||||||||
| Diluted weighted average shares outstanding | 39.5 | 32.3 | 19.7 | 19.7 | ||||||||||||||||||||||
| Three Months Ended (Unaudited) | ||||||||||||||||||||||||||
| Dollars and shares in millions, except per share amounts | December 27, 2024 | September 27, 2024 | June 28, 2024 | March 29, 2024 | ||||||||||||||||||||||
| Net sales | $ | 265.6 | $ | 285.9 | $ | 274.5 | $ | 257.4 | ||||||||||||||||||
| Gross profit | $ | 160.0 | $ | 148.7 | $ | 117.4 | $ | 113.2 | ||||||||||||||||||
| Income (loss) from continuing operations | $ | 566.4 | $ | (62.4) | $ | (74.8) | $ | (84.9) | ||||||||||||||||||
| Income from discontinued operations, net of tax | $ | 46.4 | $ | 36.2 | $ | 31.5 | $ | 19.5 | ||||||||||||||||||
| Net income (loss) | $ | 612.8 | $ | (26.2) | $ | (43.3) | $ | (65.4) | ||||||||||||||||||
| Basic income (loss) per share: | ||||||||||||||||||||||||||
| Basic income (loss) from continuing operations | $ | 28.75 | $ | (3.17) | $ | (3.80) | $ | (4.31) | ||||||||||||||||||
| Basic income from discontinued operations, net of tax | 2.36 | 1.84 | 1.60 | 0.99 | ||||||||||||||||||||||
| Net income (loss) | $ | 31.11 | $ | (1.33) | $ | (2.20) | $ | (3.32) | ||||||||||||||||||
| Basic weighted-average shares outstanding | 19.7 | 19.7 | 19.7 | 19.7 | ||||||||||||||||||||||
| Diluted income (loss) per share: | ||||||||||||||||||||||||||
| Diluted income (loss) from continuing operations | $ | 28.61 | $ | (3.17) | $ | (3.80) | $ | (4.31) | ||||||||||||||||||
| Diluted income from discontinued operations, net of tax | 2.34 | 1.84 | 1.60 | 0.99 | ||||||||||||||||||||||
| Net income (loss) | $ | 30.95 | $ | (1.33) | $ | (2.20) | $ | (3.32) | ||||||||||||||||||
| Diluted weighted average shares outstanding | 19.8 | 19.7 | 19.7 | 19.7 | ||||||||||||||||||||||
Item 7A. | Quantitative and Qualitative Disclosures About Market Risk. | ||||
Our operations include activities in the U.S. and countries outside of the U.S. These operations expose us to a variety of market risks, including the effects of changes in interest rates and currency exchange rates. We monitor and manage these financial exposures as an integral part of our overall risk management program. We do not utilize derivative instruments for trading or speculative purposes.
74
Interest Rate Risk
Our exposure to interest rate risk relates primarily to our variable-rate debt instruments, which bear interest based on SOFR plus a margin. As of December 31, 2025, our outstanding debt included $1,481.3 million of variable-rate debt on our senior secured term loans. Assuming a one percent increase in the applicable interest rates, in excess of applicable minimum floors, annual interest expense on existing variable-rate debt would be expected to increase by approximately $13.6 million.
The remaining outstanding debt as of December 31, 2025, is fixed-rate debt. Changes in market interest rates generally affect the fair value of fixed-rate debt, but do not impact earnings or cash flows.
Currency Risk
Certain net sales and costs of our international operations are denominated in the local currency of the respective countries. As such, profits from these subsidiaries may be impacted by fluctuations in the value of these local currencies relative to the U.S. dollar. We also have significant intercompany financing arrangements that may result in gains and losses in our results of operations. In an effort to mitigate the impact of currency exchange rate effects we may hedge certain operational and intercompany transactions; however, our hedging strategies may not fully offset gains and losses recognized in our results of operations.
The consolidated statement of operations is exposed to currency risk from intercompany financing arrangements, which primarily consist of intercompany debt and intercompany cash pooling, where the denominated currency of the transaction differs from the functional currency of one or more of our subsidiaries. The aggregate potential unfavorable impact from a hypothetical 10.0% adverse change in foreign exchange rates was $0.5 million as of December 31, 2025, with all other variables held constant. This hypothetical loss does not reflect any hypothetical benefits that would be derived from hedging activities, including cash holdings in similar foreign currencies, that we have historically utilized to mitigate our exposure to movements in foreign exchange rates.
Item 8. | Financial Statements and Supplementary Data. | ||||
INDEX TO FINANCIAL STATEMENTS
| Consolidated Financial Statements | Page | |||||||
Report of Independent Registered Public Accounting Firm (PCAOB ID No. | ||||||||
Report of Independent Registered Public Accounting Firm (PCAOB ID No. 34). | ||||||||
Consolidated Statements of Operations for the fiscal year ended December 31, 2025 (Successor), the fiscal year ended December 27, 2024 (Successor), the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor). | ||||||||
Consolidated Statements of Comprehensive Operations for the fiscal year ended December 31, 2025 (Successor), the fiscal year ended December 27, 2024 (Successor), the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor). | ||||||||
Consolidated Balance Sheets as of December 31, 2025 and December 27, 2024. | ||||||||
Consolidated Statements of Cash Flows for the fiscal year ended December 31, 2025 (Successor), the fiscal year ended December 27, 2024 (Successor), the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor). | ||||||||
Consolidated Statement of Changes in Shareholders' Equity for the fiscal year ended December 31, 2025 (Successor), the fiscal year ended December 27, 2024 (Successor), the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor). | ||||||||
| Notes to Consolidated Financial Statements. | ||||||||
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Shareholders of Keenova Therapeutics plc
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Keenova Therapeutics plc and its subsidiaries (the "Company") as of December 31, 2025 and December 27, 2024, and the related consolidated statements of operations, of comprehensive operations, of changes in shareholders' equity and of cash flows for the years then ended, including the related notes (collectively referred to as the "consolidated financial statements"). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and December 27, 2024, and the results of its operations and its cash flows for the years then ended in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits of these consolidated financial statements in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company's internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
Critical Audit Matters
The critical audit matters communicated below are matters arising from the current period audit of the consolidated financial statements that were communicated or required to be communicated to the audit committee and that (i) relate to accounts or disclosures that are material to the consolidated financial statements and (ii) involved our especially challenging, subjective, or complex judgments. The communication of critical audit matters does not alter in any way our opinion on the consolidated financial statements, taken as a whole, and we are not, by communicating the critical audit matters below, providing separate opinions on the critical audit matters or on the accounts or disclosures to which they relate.
Acquisition of Endo, Inc. – Valuation of Certain Developed Technology Intangible Assets
As described in Note 5 to the consolidated financial statements, on July 31, 2025, the Company completed the business combination of Endo, Inc. for total consideration transferred of $1,884.5 million. Of the acquired intangible assets, $2,226.7 million of developed technology intangible assets were recorded. A majority of the acquired intangible assets related to certain developed technology intangible assets. The fair value of the developed technology assets was determined by management using the excess earnings method, which involves the use of assumptions including future sales, cost of sales, operating expenses, and discount rates.
The principal considerations for our determination that performing procedures relating to the valuation of certain developed technology intangible assets acquired in the acquisition of Endo, Inc. is a critical audit matter are (i) the significant judgment by management when developing the fair value estimate of certain developed technology intangible assets acquired; (ii) a high degree of auditor judgment, subjectivity, and effort in performing procedures and evaluating management’s significant assumptions related to future sales, cost of sales, operating expenses, and discount rates, as applicable to the certain developed technology intangible assets; and (iii) the audit effort involved the use of professionals with specialized skill and knowledge.
76
Addressing the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated financial statements. These procedures included, among others (i) reading the transaction agreement; (ii) testing management’s process for developing the fair value estimate of certain developed technology intangible assets acquired; (iii) evaluating the appropriateness of the excess earnings method used by management; (iv) testing the completeness and accuracy of the underlying data used in the excess earnings method; and (v) evaluating the reasonableness of the significant assumptions used by management related to future sales, cost of sales, operating expenses, and discount rates, as applicable to the certain developed technology intangible assets. Evaluating management’s assumptions related to future sales, cost of sales, and operating expenses, as applicable to the certain developed technology intangible assets, involved considering (i) the current and past performance of the Endo, Inc. business; (ii) the consistency with external market and industry data; and (iii) whether the assumptions were consistent with evidence obtained in other areas of the audit. Professionals with specialized skill and knowledge were used to assist in evaluating (i) the appropriateness of the excess earnings method and (ii) the reasonableness of the discount rate assumption, as applicable to the certain developed technology intangible assets.
Certain Rebate Reserves – Medicare, Medicaid and Managed Care
As described in Notes 4 and 7 to the consolidated financial statements, product sales are recorded at the net sales price (transaction price), which includes estimates of variable consideration for which reserves are established. These reserves result from estimated government (Medicare and Medicaid) and/or privately negotiated (Managed Care) rebates. These reserves are based on the expected value method. These estimates take into consideration relevant factors such as the Company's historical experience, estimated future trends, estimated customer inventory levels, contractual agreements, and the level of utilization of the Company's products. Management adjusts these reserves to reflect differences between estimated and actual experience. The accrued rebates reserve balance related to Medicare, Medicaid and Managed Care was $155.9 million as of December 31, 2025, of which a majority relates to certain Medicare, Medicaid and Managed Care rebate reserves.
The principal considerations for our determination that performing procedures relating to certain rebate reserves for Medicare, Medicaid and Managed Care is a critical audit matter are (i) the significant judgment by management when developing the estimate of certain rebate reserves for Medicare, Medicaid and Managed Care and (ii) a high degree of auditor judgment, subjectivity, and effort in performing procedures and evaluating management’s significant assumptions related to historical experience and the level of utilization of the Company's products.
Addressing the matter involved performing procedures and evaluating audit evidence in connection with forming our overall opinion on the consolidated financial statements. These procedures included, among others (i) developing an independent estimate of certain rebate reserves for Medicare, Medicaid and Managed Care by utilizing third-party information on the level of utilization of the Company’s products, price of the Company’s products, the terms of the contractual agreements, and the historical experience of actual rebate claims paid; (ii) comparing the independent estimate to management’s estimate to evaluate the reasonableness of management’s estimate; and (iii) testing, on a sample basis, certain rebate claims paid by the Company for Medicare, Medicaid and Managed Care, including evaluating those claims for consistency with the terms of the contractual agreements.
/s/ PricewaterhouseCoopers LLP
April 15, 2026
We have served as the Company’s auditor since 2024.
77
Report of Independent Registered Public Accounting Firm
To the shareholders and the Board of Directors of Keenova Therapeutics plc
Opinion on the Financial Statements
We have audited the accompanying consolidated statements of operations, comprehensive operations, changes in shareholders' equity, and cash flows for the period from November 15, 2023 through December 29, 2023 (Successor Company operations) and for the period from December 31, 2022 through November 14, 2023 (Predecessor Company operations) of Keenova Therapeutics plc (formerly known as Mallinckrodt plc) (the “Company”), and the related notes (collectively referred to as the "financial statements"). In our opinion, the Successor Company financial statements present fairly, in all material respects, the results of its operations and its cash flows for the period from November 15, 2023 through December 29, 2023, in conformity with accounting principles generally accepted in the United States of America. Further, in our opinion, the Predecessor Company financial statements present fairly, in all material respects, the results of its operations and its cash flows for the period from December 31, 2022 through November 14, 2023, in conformity with accounting principles generally accepted in the United States of America.
Fresh-Start Accounting
As discussed in Note 2 to the financial statements, on October 10, 2023, and November 10, 2023, the United States Bankruptcy Court for the District of Delaware and the High Court of Ireland, respectively, entered an order confirming the plan of reorganization and the scheme of arrangement, respectively, related to the 2023 bankruptcy proceedings, which became effective on November 14, 2023. Accordingly, the accompanying financial statements have been prepared in conformity with FASB Accounting Standard Codification (ASC) Topic 852, Reorganizations, for the Successor Company as a new entity with assets, liabilities, and a capital structure having carrying values not comparable with prior periods as described in Note 3 to the financial statements.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company's financial statements based on our audit. We are a public accounting firm registered with the PCAOB and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. Our audit included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audit also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audit provides a reasonable basis for our opinion.
/s/ Deloitte & Touche LLP
St. Louis, Missouri
March 26, 2024, except for Note 22, as to which the date is March 12, 2025, and except for Note 6, as to which the date is April 15, 2026
We began serving as the Company's auditor in 2011. In 2024 we became the predecessor auditor.
78
KEENOVA THERAPEUTICS PLC
CONSOLIDATED STATEMENTS OF OPERATIONS
(in millions, except per share data)
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
Net sales | $ | $ | $ | $ | ||||||||||||||||||||||
Cost of sales | ||||||||||||||||||||||||||
Gross profit | ||||||||||||||||||||||||||
Selling, general and administrative expenses | ||||||||||||||||||||||||||
| Combination, integration, and other related expenses | ||||||||||||||||||||||||||
Research and development expenses | ||||||||||||||||||||||||||
| Restructuring (credits) charges, net | ( | |||||||||||||||||||||||||
Non-restructuring impairment charges | ||||||||||||||||||||||||||
Liabilities management and separation costs | ||||||||||||||||||||||||||
| Operating loss | ( | ( | ( | ( | ||||||||||||||||||||||
| Interest expense | ( | ( | ( | ( | ||||||||||||||||||||||
Interest income | ||||||||||||||||||||||||||
| (Loss) gain on divestiture (Note 6) | ( | |||||||||||||||||||||||||
| Gain (loss) on debt extinguishment, net | ( | |||||||||||||||||||||||||
| Other income (expense), net | ( | ( | ||||||||||||||||||||||||
| Reorganization items, net | ( | ( | ||||||||||||||||||||||||
| (Loss) income from continuing operations before income taxes | ( | ( | ( | |||||||||||||||||||||||
| Income tax (benefit) expense | ( | ( | ( | |||||||||||||||||||||||
| (Loss) income from continuing operations | ( | ( | ( | |||||||||||||||||||||||
| (Loss) income from discontinued operations, net of tax | ( | |||||||||||||||||||||||||
| Net (loss) income | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
Basic (loss) income per share (Note 10): | ||||||||||||||||||||||||||
| (Loss) income from continuing operations | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
| (Loss) income from discontinued operations, net of tax | ( | |||||||||||||||||||||||||
| Net (loss) income | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
Basic weighted-average shares outstanding | ||||||||||||||||||||||||||
Diluted (loss) income per share (Note 10): | ||||||||||||||||||||||||||
| (Loss) income from continuing operations | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
| (Loss) income from discontinued operations, net of tax | ( | |||||||||||||||||||||||||
| Net (loss) income | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
| Diluted weighted-average shares outstanding | ||||||||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
79
KEENOVA THERAPEUTICS PLC
CONSOLIDATED STATEMENTS OF COMPREHENSIVE OPERATIONS
(in millions)
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Net (loss) income | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
| Other comprehensive income (loss), net of tax | ||||||||||||||||||||||||||
| Currency translation adjustments | ( | ( | ||||||||||||||||||||||||
| Derivatives | ||||||||||||||||||||||||||
| Benefit plans | ( | ( | ||||||||||||||||||||||||
| Total other comprehensive income (loss), net of tax | ( | |||||||||||||||||||||||||
| Comprehensive (loss) income | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
80
KEENOVA THERAPEUTICS PLC
CONSOLIDATED BALANCE SHEETS
(in millions, except share data)
| December 31, 2025 | December 27, 2024 | ||||||||||
| Assets | |||||||||||
| Current Assets: | |||||||||||
| Cash and cash equivalents | $ | $ | |||||||||
Accounts receivable, less allowance for doubtful accounts of $ | |||||||||||
| Inventories | |||||||||||
| Prepaid expenses and other current assets | |||||||||||
| Current assets of disposed business | |||||||||||
| Total current assets | |||||||||||
| Property, plant and equipment, net | |||||||||||
| Goodwill | |||||||||||
| Intangible assets, net | |||||||||||
| Deferred income taxes | |||||||||||
| Other assets | |||||||||||
| Long-term assets of disposed business | |||||||||||
| Total Assets | $ | $ | |||||||||
| Liabilities and Shareholders' Equity | |||||||||||
| Current Liabilities: | |||||||||||
| Current maturities of long-term debt | $ | $ | |||||||||
| Accounts payable | |||||||||||
| Accrued payroll and payroll-related costs | |||||||||||
| Accrued interest | |||||||||||
| Acthar Gel-Related Settlement liability | |||||||||||
| Accrued rebates and returns | |||||||||||
| Accrued and other current liabilities | |||||||||||
| Current liabilities of disposed business | |||||||||||
| Total current liabilities | |||||||||||
| Long-term debt | |||||||||||
| Acthar Gel-Related Settlement liability | |||||||||||
| Deferred income taxes | |||||||||||
| Other income tax liabilities | |||||||||||
| Other liabilities | |||||||||||
| Long-term liabilities of disposed business | |||||||||||
| Total Liabilities | |||||||||||
| Commitments and contingencies (Note 20) | |||||||||||
| Shareholders' Equity: | |||||||||||
Preferred Shares, $ | |||||||||||
Ordinary A shares, € | |||||||||||
Ordinary shares, $ | |||||||||||
| Additional paid-in capital | |||||||||||
| Accumulated other comprehensive (loss) income | |||||||||||
| (Accumulated deficit) retained earnings | ( | ||||||||||
| Total Shareholders' Equity | |||||||||||
| Total Liabilities and Shareholders' Equity | $ | $ | |||||||||
The accompanying notes are an integral part of these consolidated financial statements.
81
KEENOVA THERAPEUTICS PLC
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in millions)
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Cash Flows From Operating Activities - Continuing Operations: | ||||||||||||||||||||||||||
| (Loss) income from continuing operations | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
| Adjustments to reconcile net cash from operating activities: | ||||||||||||||||||||||||||
| Depreciation and amortization | ||||||||||||||||||||||||||
| Share-based compensation | ||||||||||||||||||||||||||
| Deferred income taxes | ( | ( | ( | |||||||||||||||||||||||
| Non-cash impairment charges | ||||||||||||||||||||||||||
Loss (gain) on divestiture (Note 6) | ( | |||||||||||||||||||||||||
Loss (gain) on debt extinguishment (Note 15) | ( | |||||||||||||||||||||||||
| Inventory step-up amortization from acquisitions | ||||||||||||||||||||||||||
| Inventory provisions | ||||||||||||||||||||||||||
| Fair value adjustment in contingent consideration liabilities | ( | ( | ||||||||||||||||||||||||
| Reorganization items, net | ||||||||||||||||||||||||||
Non-cash (amortization) accretion expense | ( | ( | ||||||||||||||||||||||||
| Other non-cash items | ||||||||||||||||||||||||||
Changes in assets and liabilities: | ||||||||||||||||||||||||||
| Accounts receivable, net | ( | |||||||||||||||||||||||||
| Inventories | ||||||||||||||||||||||||||
| Accounts payable | ( | ( | ||||||||||||||||||||||||
| Accrued consulting fees | ( | ( | ||||||||||||||||||||||||
| Income taxes | ( | ( | ||||||||||||||||||||||||
Acthar Gel-Related Litigation Settlement liability | ( | ( | ( | |||||||||||||||||||||||
| Other | ( | ( | ||||||||||||||||||||||||
| Net cash provided by (used in) operating activities - continuing operations | $ | $ | ( | $ | $ | ( | ||||||||||||||||||||
| Cash Flows From Investing Activities - Continuing Operations: | ||||||||||||||||||||||||||
| Capital expenditures | $ | ( | $ | ( | $ | ( | $ | ( | ||||||||||||||||||
| Receipts of unrestricted cash, net of payments related to the Business Combination (Note 5) | ||||||||||||||||||||||||||
| Receipts of restricted cash related to the Business Combination (Note 5) | ||||||||||||||||||||||||||
(Payments) proceeds from divestiture, net of divested cash (Note 6) | ( | |||||||||||||||||||||||||
| Other | ||||||||||||||||||||||||||
| Net cash provided by (used in) investing activities - continuing operations | $ | $ | $ | ( | $ | ( | ||||||||||||||||||||
| Cash Flows From Financing Activities - Continuing Operations: | ||||||||||||||||||||||||||
| Issuance of external debt | $ | $ | $ | $ | ||||||||||||||||||||||
| Repayment of external debt | ( | ( | ( | ( | ||||||||||||||||||||||
| Divested cash from spin of Par Health | ( | |||||||||||||||||||||||||
Makewhole premium (Note 15) | ( | ( | ||||||||||||||||||||||||
| Debt financing costs | ( | |||||||||||||||||||||||||
| Settlement of Opioid Contingent Value Rights | ( | |||||||||||||||||||||||||
| Other | ( | ( | ( | |||||||||||||||||||||||
| Net cash (used in) provided by financing activities - continuing operations | $ | ( | $ | ( | $ | ( | $ | |||||||||||||||||||
| Discontinued Operations: | ||||||||||||||||||||||||||
| Net cash provided by (used in) operating activities - discontinued operations | $ | $ | $ | $ | ( | |||||||||||||||||||||
| Net cash used in investing activities - discontinued operations | ( | ( | ( | ( | ||||||||||||||||||||||
| Net cash provided by financing activities - discontinued operations | ||||||||||||||||||||||||||
| Net cash provided by (used in) discontinued operations | $ | $ | $ | $ | ( | |||||||||||||||||||||
| Effect of currency rate changes on cash | ( | ( | ||||||||||||||||||||||||
| Net change in cash, cash equivalents and restricted cash | $ | $ | $ | $ | ( | |||||||||||||||||||||
| Cash, cash equivalents and restricted cash at beginning of period | ||||||||||||||||||||||||||
| Cash, cash equivalents and restricted cash at end of period | $ | $ | $ | $ | ||||||||||||||||||||||
82
KEENOVA THERAPEUTICS PLC
CONSOLIDATED STATEMENTS OF CASH FLOWS
(in millions)
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Cash and cash equivalents at end of period | $ | $ | $ | $ | ||||||||||||||||||||||
Restricted cash included in prepaid expenses and other assets at end of period (Note 21) | ||||||||||||||||||||||||||
Restricted cash included in other long-term assets at end of period (Note 21) | ||||||||||||||||||||||||||
| Cash, cash equivalents and restricted cash at end of period | $ | $ | $ | $ | ||||||||||||||||||||||
| Less: Cash and cash equivalents at end of period - discontinued operations | $ | $ | $ | $ | ||||||||||||||||||||||
| Less: Restricted cash included in other long-term assets at end of period - discontinued operations | ||||||||||||||||||||||||||
| Cash, cash equivalents and restricted cash at end of period - continuing operations | $ | $ | $ | $ | ||||||||||||||||||||||
| Cash and cash equivalents at end of period - continuing operations | $ | $ | $ | $ | ||||||||||||||||||||||
| Restricted cash included in prepaid expenses and other assets at end of period - continuing operations | ||||||||||||||||||||||||||
| Restricted cash included in other long-term assets at end of period - continuing operations | ||||||||||||||||||||||||||
| Cash, cash equivalents and restricted cash at end of period - continuing operations | $ | $ | $ | $ | ||||||||||||||||||||||
| Supplemental Disclosures of Cash Flow Information: | ||||||||||||||||||||||||||
Cash paid for interest, net | $ | $ | $ | $ | ||||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
83
KEENOVA THERAPEUTICS PLC
CONSOLIDATED STATEMENT OF CHANGES IN SHAREHOLDERS' EQUITY
(in millions)
| Ordinary Shares | Preferred Shares | Treasury Shares | Additional Paid-In Capital | (Accumulated Deficit) Retained Earnings | Accumulated Other Comprehensive Income | Total Shareholders' Equity | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Number | Par Value | Number | Par Value | Number | Amount | |||||||||||||||||||||||||||||||||||||||||||||||||||
Balance as of December 30, 2022 (Predecessor) | $ | $ | $ | $ | $ | ( | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | ( | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||
Other comprehensive loss | — | — | — | — | — | — | — | — | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
Vesting of restricted shares | — | — | — | ( | — | — | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||||
| Cancellation of Predecessor equity | ( | ( | — | — | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
Issuance of common stock | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
Issuance of Opioid Contingent Value Rights | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
Balance as of November 14, 2023 (Predecessor) | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | ( | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||
Other comprehensive income | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
Balance as of December 29, 2023 (Successor) | $ | $ | $ | $ | $ | ( | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||
| Net income | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
Other comprehensive income | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
Share-based compensation | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
| Balance as of December 27, 2024 (Successor) | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||||
| Net loss | — | — | — | — | — | — | — | ( | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
| Preferred share issuance | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||
Divestiture of Par Health (1) (Note 6) | — | — | ( | — | — | — | ( | — | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||
| Settlement of Opioid Contingent Value Rights | — | — | — | — | — | — | ( | — | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
| Ordinary shares issued in Business Combination (Note 5) | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Ordinary shares issued (Other) | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
| Share cancellation | — | — | — | — | — | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||
| Pre-combination value of converted Endo awards (Note 5) | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||
| Vesting of restricted share units, net of tax withholdings | — | — | — | — | ( | ( | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||
| Treasury share cancellation | — | — | — | — | — | — | ( | — | ||||||||||||||||||||||||||||||||||||||||||||||||
| Balance as of December 31, 2025 (Successor) | $ | $ | $ | $ | $ | ( | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||
1.Includes certain amounts paid to non-qualified shareholders. Refer to Note 6 Divestitures for further detail.
The accompanying notes are an integral part of these consolidated financial statements.
84
KEENOVA THERAPEUTICS PLC
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
(dollars in millions, except share data and where indicated)
| 1. | Background and Basis of Presentation | ||||
Background
Keenova Therapeutics plc, formerly Mallinckrodt plc, and its consolidated subsidiaries (collectively, “Keenova” or “the Company”) is a leading U.S.-focused branded therapeutics company that strives to help patients with rare or unaddressed conditions live happier and healthier lives.
On July 31, 2025, the Company completed the business combination with Endo Inc. (which has since been converted to Endo LP, (“Endo”) (the “Business Combination”), which resulted in increased scale and enhanced capabilities to develop, manufacture and commercialize branded therapeutics, generic pharmaceuticals, and sterile injectables. The Company’s operating results for the year ended December 31, 2025 reflect the consolidated results of five months of operations following the closing of the Business Combination. On November 10, 2025, the Company completed the separation of its generic pharmaceuticals and sterile injectables businesses into an independent, private company named Par Health, Inc. (“Par Health”) (the “Separation”). As a result of the Separation, the Company’s operations are now centered on its branded therapeutics portfolio. Following the Separation, the results of Par Health are reported as discontinued operations. As a result of the Separation, the Company and its subsidiaries are no longer borrowers or guarantors under the Par Health Credit Agreement (as defined below). The Company also entered into certain agreements with Par Health, including a lease agreement, a manufacturing and supply agreement, and a transition services agreement to provide and receive certain services following the Separation. For additional information related to the Business Combination and the Separation, see Note 5 and Note 6, respectively. Separation‑related costs were recorded within liabilities management and separation costs.
As a result of the Business Combination, for the period from July 31, 2025, to November 10, 2025, the Company operated in three reportable segments: Specialty Brands, Generics, and Sterile Injectables. Following the Separation, the Company operates its business in one operating and reportable segment with a clear and focused strategy centered on its branded therapeutics. Keenova’s rare disease capabilities underpin its diversified brands portfolio, which is focused across a wide range of therapeutics areas of significant unmet need. These include rheumatology, ophthalmology, nephrology, neurology, pulmonology, orthopedics, urology, and neonatal respiratory critical care. For additional information relating to our single reportable segment, see Note 22.
The Company’s principal executive offices are located at College Business & Technology Park, Cruiserath, Blanchardstown, Dublin 15, Ireland, where certain manufacturing operations are also located. In addition, the Company has locations in the United States (“U.S.”), including manufacturing facilities in Horsham, Pennsylvania; Port Allen, Louisiana; Rye, New York; Cranbury, New Jersey; and Madison, Wisconsin, as well as office facilities in Bridgewater, New Jersey; Malvern, Pennsylvania; Hazelwood; Missouri; Washington, D.C.; and in Tokyo, Japan, among others. The Company also has seven regional service centers in the U.S.
Basis of Presentation
In the fourth quarter of fiscal year 2025, the Company approved a change in its fiscal year end from a 52-53-week year ending on the last Friday of December to a calendar year ending on December 31, 2025. As a result of this change, fiscal 2025 includes five additional operating days. Beginning with fiscal 2026, the Company’s fiscal year will correspond to the calendar year from January 1 through December 31.
The Company’s operating results for the year ended December 31, 2025, include five months of operations following the Business Combination. The results of Par Health are presented as discontinued operations. The divestiture of any product lines and businesses that do not meet the criteria for discontinued operations, such as the Therakos Divestiture, are reflected within continuing operations.
Use of Estimates
In preparing the Company’s Consolidated Financial Statements, management is required to make estimates and assumptions. This includes estimates and assumptions regarding the nature, timing and extent of the impacts that certain global macroeconomic conditions, including, but not limited to, those related to inflation and supply chain, will have on the Company's operations and cash flows. The estimates and assumptions used by the Company affect the reported amounts of assets and liabilities, the disclosure of contingent liabilities at the date of the financial statements and the reported amounts of revenues and expenses during the reporting periods. Significant estimates made by management include: provisions for product returns, rebates, chargebacks, discounts and
85
allowances and distribution fees paid to certain wholesalers; useful lives of finite-lived intangible assets and property, plant and equipment; expected future cash flows used in evaluating intangible assets for impairment, assessing compliance with debt covenants, reporting unit fair values for testing goodwill for impairment; acquisition-related contingent consideration liabilities; provisions for loss contingencies; provisions for income taxes, uncertain tax positions and realizability of deferred tax assets; and the recognition of the fair value of assets and liabilities in connection with fresh start accounting and those acquired in a business combination or asset acquisition.
All estimates in these Consolidated Financial Statements are based on assumptions that management believes are reasonable. On an ongoing basis, management reviews its estimates to ensure that these estimates appropriately reflect changes in the Company's business and new information as it becomes available. If historical experience and other factors used by management to make these estimates do not reasonably reflect future activity, the Company's business, financial condition, cash flows and results of operations could be materially impacted.
The extent to which certain global macroeconomic conditions, including, but not limited to, those related to inflation and supply chain, may continue to impact the Company’s business, financial condition, cash flows and results of operations, in particular, will depend on future developments which are highly uncertain and many of which are outside the Company’s control. The Company has assessed the possible effects and outcomes of these macroeconomic conditions on, among other things, its supply chain, customers and distributors, discounts and rebates, employee base, product sustainability, research and development efforts, product pipeline and consumer demand and currently believes that its estimates are reasonable.
Adoption of Fresh-Start Accounting
Upon emergence from the 2023 Bankruptcy Proceedings, as defined below, on November 14, 2023, the Company adopted fresh-start accounting in accordance with the provisions of Financial Accounting Standards Board Accounting Standards Codification (“ASC”) Topic 852 - Reorganizations (“ASC 852”), and became a new entity for financial reporting purposes as of the 2023 Effective Date. References to "Successor" relate to the financial position as of December 31, 2025 and December 27, 2024 and results of operations of the reorganized Company subsequent to November 14, 2023, while references to “Predecessor” relate to the results of operations of the Company for the period December 31, 2022 through November 14, 2023. All emergence-related transactions related to the 2023 Effective Date were recorded as of November 14, 2023. Accordingly, the consolidated financial statements for the Successor are not comparable to the consolidated financial statements for the Predecessor. See Note 3 for further information.
| 2. | Emergence from Voluntary Reorganization | ||||
On August 28, 2023 (“2023 Petition Date”), the Company voluntarily initiated Chapter 11 proceedings (“2023 Chapter 11 Cases”) under chapter 11 of title 11 (“Chapter 11”) of the U.S. Code (“Bankruptcy Code”) in the U.S. Bankruptcy Court for the District of Delaware (“Bankruptcy Court”). On September 20, 2023, the directors of the Company initiated examinership proceedings with respect to Mallinckrodt plc by presenting a petition to the High Court of Ireland pursuant to Section 510(1)(b) of the Companies Act 2014 seeking the appointment of an examiner to Mallinckrodt plc. On October 10, 2023, the Bankruptcy Court entered an order confirming a plan of reorganization (“2023 Plan”). Subsequent to the Bankruptcy Court’s order confirming the 2023 Plan, the High Court of Ireland made an order confirming a scheme of arrangement on November 10, 2023, which is based on and consistent in all respects with the 2023 Plan (“2023 Scheme of Arrangement”). The 2023 Plan and the 2023 Scheme of Arrangement became effective on November 14, 2023, (“2023 Effective Date”), and the Company emerged from the 2023 Chapter 11 Cases and the Irish examinership proceedings (together, the “2023 Bankruptcy Proceedings”) on that date. See Note 3 for further information on the 2023 Plan and emergence from the 2023 Bankruptcy Proceedings.
During the pendency of the 2023 Bankruptcy Proceedings, the Company and each of the respective debtors and debtors in-possession in the 2023 Chapter 11 Cases (“2023 Debtors”) operated their businesses as debtors-in-possession under the jurisdiction of the Bankruptcy Court and in accordance with the applicable provisions of the Bankruptcy Code and orders of the Bankruptcy Court. As debtors-in-possession, the 2023 Debtors were authorized to continue to operate as ongoing businesses and were allowed to pay all debts and honor all obligations arising in the ordinary course of their businesses after the 2023 Petition Date. However, the 2023 Debtors were not allowed to pay third-party claims or creditors on account of obligations arising before the 2023 Petition Date or engage in transactions outside the ordinary course of business without approval of the Bankruptcy Court.
Under the Bankruptcy Code, third-party actions to collect pre-petition indebtedness owed by the 2023 Debtors, as well as most litigation pending against the Company as of the 2023 Petition Date, were subject to an automatic stay. See “Plan of Reorganization” below for the distributions to creditors and interest holders.
Plan of Reorganization
2023 Plan
In accordance with the 2023 Plan, the following significant transactions occurred upon the Company's emergence from the 2023 Bankruptcy Proceedings on the 2023 Effective Date:
•DIP Claims (as defined below) were converted on a dollar-for-dollar basis into First-Out Takeback Term Loans (as defined below);
86
•Pre-petition first lien term debt was reduced from $2,861.8 million to $1,650.0 million, which was in the form of Takeback Debt (as defined below) distributed to post-petition term lenders and pre-petition first lien creditors;
•The pre-petition first lien creditors also received 92.3 % of the 2023 Debtors’ reorganized equity (subject to dilution from equity reserved under the management incentive program (“MIP”) and the Opioid CVRs (as defined below) if equity settled), plus cash in an amount sufficient to repay in full accrued and unpaid interest on the pre-petition first lien debt, and Second-Out Takeback Debt (as defined below);
•Pre-petition second lien debt was eliminated in its entirety, with pre-petition second lien creditors receiving 7.7 % of the 2023 Debtors’ reorganized equity (subject to dilution from equity reserved under the MIP and the Opioid CVRs, if equity settled);
•The 2023 Debtors’ remaining opioid-related litigation settlement payment obligations (including the $200.0 million installment payment originally due on June 16, 2023) were permanently eliminated, subject to the Company (a) making a $250.0 million payment to the Opioid Master Disbursement Trust II (“Trust”) prior to the commencement of the 2023 Chapter 11 Cases (which was made on August 24, 2023) and (b) entering into the CVR Agreement (as defined below);
•The 2023 Debtors’ non-monetary obligations to the Trust were generally preserved, including the compliance-related operating injunction;
•All other claims against the 2023 Debtors (with the exception of subordinated securities claims) were treated as unimpaired, including settlements under the Chapter 11 plan of reorganization for the 2020 Bankruptcy Proceedings (“2020 Plan”), with governmental entities regarding Acthar® Gel (repository corticotropin injection) (“Acthar Gel”), and the associated Corporate Integrity Agreement, and also trade liabilities; and
•All of the Company’s then-existing ordinary shares were cancelled for no consideration.
Contingent Value Right Agreement
On the 2023 Effective Date and pursuant to the 2023 Plan, the Company entered into a contingent value right agreement (“CVR Agreement”) with the Trust. Pursuant to the terms of the CVR Agreement, the Company issued 1,036,649 contingent value rights (“Opioid CVRs”) to the Trust, which Opioid CVRs entitle the Trust to receive from the Company, when exercised, an amount in cash equal to (a) the Market Price (as defined in the CVR Agreement) of one new ordinary share of the Company (subject to adjustment as described in the CVR Agreement) at the time of exercise less (b) $99.36 (subject to adjustment as described in the CVR Agreement) (“Cash Payment”), subject to the right of the Company to, at its option but subject to certain conditions, issue new ordinary shares to the Trust in lieu of making some or all of the Cash Payment due upon exercise in accordance with the terms of the CVR Agreement. The Opioid CVRs are exercisable at any time for four years after the 2023 Effective Date.
Upon entering into the CVR Agreement the terms of the final amendment to the opioid-related litigation settlement (“Opioid-Related Litigation Settlement”) obligation agreement (“Opioid Deferred Cash Payment Agreement”) and the Company's prior obligation to pay all remaining Opioid-Related Litigation Settlement payment obligations (“Opioid Deferred Cash Payment”) were permanently eliminated.
As described above, on November 10, 2025, in connection with the Separation, the Company entered into the CVR Termination Agreement (as defined below) to cancel the Opioid CVRs issued under the CVR Agreement and terminate the CVR Agreement in exchange for a payment by the Company of $35.0 million, resulting in an adjustment of the Opioid CVRs to fair value through additional-paid-in-capital of $4.9 million and the recognition of $5 million of expense during the year ended December 31, 2025, which is recorded within loss from discontinued operations. The CVR Termination Agreement also included customary representations and warranties and a waiver of certain claims.
Registration Rights Agreement
On the 2023 Effective Date and pursuant to the 2023 Plan, the Company entered into a registration rights agreement (“Registration Rights Agreement”) with certain owners of new ordinary shares (any owner of new ordinary shares, a “Company Shareholder”). Pursuant to the terms of the Registration Rights Agreement, following an initial public offering, any Company Shareholder that owns 1 % or more of the new ordinary shares (calculated in accordance with the Registration Rights Agreement) shall have customary “piggyback” registration rights. In addition, 180 days following an initial public offering, any Company Shareholder owning at least 15 % of the new ordinary shares (calculated in accordance with the Registration Rights Agreement) shall have the right to initiate up to three (3 ) demand registrations each, subject to customary exceptions.
Takeback Debt
On the 2023 Effective Date and pursuant to the 2023 Plan, Mallinckrodt International Finance S.A. (“MIFSA”) and Mallinckrodt CB LLC (“MCB” and, together with MIFSA, the “Issuers”), each of which is a subsidiary of the Company, (i) entered into a new senior secured first lien term loan facility with an aggregate principal amount of approximately $871.4 million (“Takeback Term Loans”), consisting of approximately $229.4 million of “first-out” Takeback Term Loans (“First-Out Takeback Term Loans”) and approximately $642.0 million of “second-out” Takeback Term Loans (“Second-Out Takeback Term Loans”) and (ii) issued approximately $778.6 million in aggregate principal amount of “second-out” 14.75 % senior secured first lien notes due 2028
87
(“Takeback Notes” and, together with the Second-Out Takeback Term Loans, “Second-Out Takeback Debt” and, together with the Takeback Term Loans, “Takeback Debt”).
All allowed claims (“DIP Claims”) under the Senior Secured Debtor-In-Possession Credit Agreement, dated as of September 8, 2023 (“DIP Credit Agreement”), by and among the Company, MIFSA and MCB, as debtors and debtors-in-possession, the lenders from time to time party thereto, Acquiom Agency Services LLC and Seaport Loan Products LLC, as co-administrative agents, and Acquiom Agency Services LLC, as collateral agent, not otherwise satisfied in cash were converted on a dollar-for-dollar basis into First-Out Takeback Term Loans.
| 3. | Fresh-start Accounting | ||||
The Company qualified for and adopted fresh-start accounting as of the 2023 Effective Date in accordance with ASC 852 as (i) the reorganization value of the assets of the Company immediately prior to the date of effectuation of the 2023 Plan was less than the post-petition liabilities and allowed claims and (ii) the holders of the voting shares of the Predecessor immediately before effectuation of the 2023 Plan received less than 50 % of the voting shares of the Successor.
Reorganization Value
Reorganization value represents the fair value of the Successor's total assets and is intended to approximate the amount a willing buyer would pay for the assets immediately after restructuring. Upon the application of fresh-start accounting, the Company allocated the reorganization value to its identified tangible and intangible assets and liabilities based on their estimated fair values in accordance with ASC Topic 805 - Business Combinations. Deferred income tax amounts were determined in accordance with ASC Topic 740 - Income Taxes.
As set forth in the disclosure statement approved by the Bankruptcy Court, the enterprise value of the Successor was estimated to be between $2,700.0 million and $3,200.0 million, with a midpoint of $2,950.0 million, which was estimated with the assistance of third-party valuation advisors using various valuation methods, including (i) discounted cash flow analysis, a calculation of the present value of the future cash flows to be generated by the business based on its projection, and (ii) comparable public company analysis, a method to estimate the value of a company relative to other publicly traded companies with similar operation and financial characteristics. The estimated enterprise value per the disclosure statement included estimated equity value in a range between $1,110.0 million and $1,610.0 million, with a midpoint of $1,360.0 million.
The basis of the discounted cash flow analysis used in developing the enterprise value was based on Company prepared projections that included a variety of estimates and assumptions. While the Company considers such estimates and assumptions reasonable, they are inherently subject to significant business, economic and competitive uncertainties, many of which are beyond the Company’s control and, therefore, may not be realized. Changes in these estimates and assumptions may have had a significant effect on the determination of the Company’s enterprise value.
The following table reconciles the enterprise value to the implied fair value of the Successor's equity as of the 2023 Effective Date:
| Enterprise value | $ | |||||||
Plus: Non-operating assets, net (1) | ||||||||
Less: Fair value of debt | ( | |||||||
Less: Fair value of Acthar Gel-Related Settlement and Terlivaz contingent value rights | ( | |||||||
| Successor equity value | $ | |||||||
(1)Represents non-operating assets and liabilities which were excluded from the enterprise value as put forth in the disclosure statement as there were no cash projections associated with these net assets.
Upon the application of fresh-start accounting, the Company allocated the reorganization value to its individual assets based on their estimated fair values. Reorganization value represents the fair value of the Successor’s assets before considering liabilities.
88
The following table reconciles the Company's enterprise value to its reorganization value as of the 2023 Effective Date:
Enterprise value | $ | |||||||
Plus: Non-operating assets, net | ||||||||
Plus: Current liabilities (excluding debt or debt-like items) (1) | ||||||||
Plus: Other non-current liabilities (excluding debt or debt-like items) (2) | ||||||||
| Reorganization value of Successor assets | $ | |||||||
(1)Excludes $7.6 million related to the of the embedded derivative.
(2)Excludes $15.0 million and $7.5 million related to the Terlivaz CVR (as defined below) and the non-current portion of the embedded derivative, respectively.
Consolidated Balance Sheet
The four-column consolidated balance sheet as of the 2023 Effective Date included herein, applies effects of the 2023 Plan (reflected in the column “Reorganization Adjustments”) and fresh-start accounting (reflected in the column “Fresh-Start Adjustments”) to the carrying values and classifications of assets or liabilities. Upon adoption of fresh-start accounting, the recorded amounts of assets and liabilities were adjusted to reflect their estimated fair values. Accordingly, the reported historical financial statements of the Predecessor prior to the adoption of fresh-start accounting for periods ended on or prior to the 2023 Effective Date are not comparable to those of the Successor. The explanatory notes highlight methods used to determine fair values or other amounts of the assets and liabilities as well as significant assumptions. The amounts reflected in the table have been retrospectively adjusted to reflect the separation of Par Health’s assets and liabilities as discontinued operations.
89
The four-column consolidated balance sheet as of November 14, 2023, is as follows:
| Predecessor | Reorganization Adjustments | Fresh-Start Adjustments | Successor | ||||||||||||||||||||||||||
| Assets | |||||||||||||||||||||||||||||
| Current Assets: | |||||||||||||||||||||||||||||
| Cash and cash equivalents | $ | $ | ( | (a) | $ | $ | |||||||||||||||||||||||
| Accounts receivable, less allowance for doubtful accounts | |||||||||||||||||||||||||||||
| Inventories | (o) | ||||||||||||||||||||||||||||
| Prepaid expenses and other current assets | (b) | ( | (p) | ||||||||||||||||||||||||||
| Current assets of disposed business | (aa) | ||||||||||||||||||||||||||||
| Total current assets | ( | ||||||||||||||||||||||||||||
| Property, plant and equipment, net | ( | (q) | |||||||||||||||||||||||||||
| Intangible assets, net | ( | (r) | |||||||||||||||||||||||||||
Deferred income taxes | (c) | (s) | |||||||||||||||||||||||||||
| Other assets | ( | (d) | (t) | ||||||||||||||||||||||||||
| Long-term assets of disposed business | (aa) | (aa) | |||||||||||||||||||||||||||
| Total Assets | $ | $ | $ | ( | $ | ||||||||||||||||||||||||
| Liabilities and Shareholders' Equity | |||||||||||||||||||||||||||||
| Current Liabilities: | |||||||||||||||||||||||||||||
| Current maturities of long-term debt | $ | $ | ( | (e) | $ | $ | |||||||||||||||||||||||
| Accounts payable | ( | (f) | |||||||||||||||||||||||||||
| Accrued payroll and payroll-related costs | |||||||||||||||||||||||||||||
Accrued interest | ( | (g) | |||||||||||||||||||||||||||
Income taxes payable | |||||||||||||||||||||||||||||
| Accrued and other current liabilities | (h) | (u) | |||||||||||||||||||||||||||
| Acthar Gel-Related Settlement | (i) | ||||||||||||||||||||||||||||
| Current liabilities of disposed business | ( | (aa) | |||||||||||||||||||||||||||
Total current liabilities | ( | ( | |||||||||||||||||||||||||||
| Long-term debt | (e) | ||||||||||||||||||||||||||||
| Pension and postretirement benefits | |||||||||||||||||||||||||||||
| Deferred income taxes | ( | (v) | |||||||||||||||||||||||||||
| Other income tax liabilities | |||||||||||||||||||||||||||||
| Other liabilities | (j) | (w) | |||||||||||||||||||||||||||
| Acthar Gel-Related Settlement | (i) | ( | (x) | ||||||||||||||||||||||||||
| Liabilities subject to compromise | ( | (k) | |||||||||||||||||||||||||||
| Liabilities subject to compromise of discontinued operations | ( | (aa) | |||||||||||||||||||||||||||
| Long-term liabilities of disposed business | (aa) | ||||||||||||||||||||||||||||
| Total Liabilities | ( | ( | |||||||||||||||||||||||||||
| Shareholders' Equity: | |||||||||||||||||||||||||||||
Predecessor ordinary shares | ( | (l) | |||||||||||||||||||||||||||
Successor ordinary shares | (l) | ||||||||||||||||||||||||||||
Predecessor ordinary shares held in treasury | ( | (l) | |||||||||||||||||||||||||||
Predecessor additional paid-in capital | ( | (l) | |||||||||||||||||||||||||||
Successor additional paid-in capital | (l) | ||||||||||||||||||||||||||||
Predecessor accumulated other comprehensive income | (m) | ( | (y) | ||||||||||||||||||||||||||
| Retained (deficit) earnings | ( | (n) | ( | (z) | |||||||||||||||||||||||||
| Total Shareholders' Equity | ( | ( | |||||||||||||||||||||||||||
| Total Liabilities and Shareholders' Equity | $ | $ | $ | ( | $ | ||||||||||||||||||||||||
90
Reorganization Adjustments
(a)The table below reflects the sources and uses of cash on the 2023 Effective Date:
| Uses: | ||||||||
| Payment of professional fees | $ | |||||||
| Payment to fund professional fees escrow (prepaid and other current assets restricted cash) | ||||||||
Payment of costs, fees and expenses related to exit-financing activities and accrued and unpaid interest on certain pre-emergence debt | ||||||||
Payment of cash sweep | ||||||||
Total Uses of Cash | $ | |||||||
(b)Represents the transfer of funds to a restricted cash account for purposes of funding the $24.0 million professional fee reserve offset by the net write-off of $17.2 million and $0.4 million of prepaid expenses related to premiums for the Predecessor's directors' and officers' insurance policy and the Predecessor's directors' compensation, respectively.
(c)Reflects adjustments primarily consisting of the reduction in the valuation allowance on the Company's deferred tax assets, and the net increase on the Company’s deferred tax assets as a result of Reorganization Adjustments.
(d)Represents the write-off of $2.4 million of the non-current portion of premiums related to the Predecessor's directors' and officers' insurance policy.
(e)Impacts to long-term debt, net of current maturities, pursuant to the 2023 Plan, include the following:
•Conversion of all DIP Claims (i) under the DIP Credit Agreement of $280.0 million and (ii) related to the 2025 First Lien Notes, the 2028 First Lien Notes and the First Lien Term Loans into $871.4 million of Takeback Term Loans, and $778.6 million in aggregate principal amount of Takeback Notes;
•Elimination of the Second Lien Notes; and
•Capitalization of an additional $1.7 million of deferred financing fees associated with the receivables financing facility due December 2027.
All Predecessor debt was classified as liabilities subject to compromise (“LSTC”) as of the 2023 Effective Date except for the DIP Credit Agreement and the receivables financing facility. The receivables financing facility, with an outstanding balance of $98.7 million, net of deferred financing fees, was reclassified to long-term debt as the maturity date was amended to December 2027 upon the effectuation of the 2023 Plan.
Reflects the fair value adjustments to the carrying value of debt instruments impacted by the 2023 Plan as determined by the Black-Derman-Toy model as follows:
First-Out Takeback Term Loans | $ | |||||||
Second-Out Takeback Term Loans | ||||||||
Takeback Notes | ||||||||
| Total fair value adjustment to debt instruments | $ | |||||||
(f)Represents $19.4 million of professional fees paid to the Company’s restructuring advisors upon the Company's emergence from the 2023 Bankruptcy Proceedings.
(g)Represents payments of accrued interest on the Company's predecessor DIP Credit Agreement, the 2025 First Lien Notes, the 2028 First Lien Notes and the First Lien Term Loans, in accordance with the cash collateral order on the 2023 Effective Date.
(h)Represents the reserve for $24.0 million related to the professional fees coupled with the current portion of the embedded derivative of $7.6 million related to certain of the Company's debt obligations. Refer to Note 21 for further information on the valuation of the embedded derivative.
(i)Represents the reinstatement of the Acthar Gel-Related Settlement liability from LSTC.
(j)Represents the non-current portion of the -related embedded derivative of $7.5 million as further described in Note 21.
91
(k)LSTC were settled as follows in accordance with the 2023 Plan:
| Liabilities subject to compromise | ||||||||
| Accrued interest | $ | |||||||
Debt (1) | ||||||||
Acthar Gel-Related Settlement liability (1) | ||||||||
Opioid-Related Litigation Settlement liability (1) | ||||||||
Other non-current liabilities | ||||||||
| Total liabilities subject to compromise | $ | |||||||
| Attributable to: | ||||||||
| Continuing operations | $ | |||||||
| Discontinued operations | $ | |||||||
To be reinstated on the 2023 Effective Date: | ||||||||
| Acthar Gel-Related Settlement liability | $ | ( | ||||||
Other non-current liabilities | ( | |||||||
| Total liabilities reinstated | $ | ( | ||||||
Consideration provided to settle amounts per the 2023 Plan | ||||||||
Issuance of Successor ordinary shares | $ | ( | ||||||
Issuance of Opioid CVRs | ( | |||||||
Issuance of Second-Out Takeback Term Loans and Second-Out Takeback Notes | ( | |||||||
Total consideration provided to settle amounts per the 2023 Plan | $ | ( | ||||||
| Gain on settlement of liabilities subject to compromise | $ | |||||||
| Attributable to: | ||||||||
| Continuing operations | $ | |||||||
| Discontinued operations | $ | |||||||
(1)Excluded from the calculation of gain on settlement of LSTC is the accretion acceleration of $377.6 million, $145.0 million and $598.4 million on the Company's debt obligations, Acthar Gel-Related Settlement liability and Opioid-Related Litigation Settlement liability, respectively, to the estimated allowed claim amount. Also excluded is $18.5 million of deferred financing fee write-offs in order to reflect the carrying value of debt at its estimated allowed claim amount.
(l)Pursuant to the 2023 Plan, as of the 2023 Effective Date, all Predecessor preferred and ordinary shares were cancelled without any distribution. The following table reconciles Reorganization Adjustments made to Successor ordinary shares, Opioid CVRs and additional paid in capital:
Par value of (par valued at $ | $ | |||||||
Fair value of Opioid CVRs issued to the Trust (1) | ||||||||
Additional paid in capital - Successor ordinary shares | ||||||||
| Successor equity | $ | |||||||
(1)The fair value of the Opioid CVRs were estimated using a Black-Scholes model with the following assumptions: $60.28 implied share price of the Successor; exercise price per share of $99.36 ; expected volatility of 65.0 %; risk free interest rate of 4.49 %, continuously compounded; and a holding period of four years . The expected volatility assumption is based on the historical and implied volatility of the Company's peer group with similar business models.
92
(m)Represents adjustments primarily consisting of the reduction in the valuation allowance on the Company's accumulated other comprehensive income.
(n)Retained (deficit) earnings - The cumulative effect of the consummation of the 2023 Plan on the Predecessor's retained deficit is as follows:
| Gain on settlement of LSTC | $ | |||||||
Professional and exit fees | ( | |||||||
Release of prepaid insurance (1) and directors fees | ( | |||||||
Fair value of First-Out Takeback Term Loans and related embedded derivative | ( | |||||||
Income tax benefit on plan adjustments(2) | ||||||||
| Cancellation of Predecessor equity | ||||||||
Net impact on retained earnings | $ | |||||||
(1)Write off of prepaid expenses related to premiums for the Predecessor’s directors’ and officers’ insurance policy.
Fresh-Start Adjustments
(o)Reflects the fair value adjustment related to the Company's inventory. Both the bottom-up and top-down approach were used. The bottom-up approach considers the inventory value that had been created by the Company including the costs incurred, profit realized, and tangible and intangible assets used pre-2023 Effective Date. The top-down approach measures the incremental inventory value created by the market participant buyer as part of its selling effort to an end customer and considers the costs that will be incurred, the profit that will be realized, and the tangible and intangible assets that will be used post-2023 Effective Date.
(p)Reflects the reduction of prepaid income taxes due to remeasurement as a result of fresh-start accounting.
(q)Reflects the fair value adjustment related to the Company's property, plant and equipment. Both the market and cost approaches were utilized to fair value land and buildings. The cost approach was utilized to fair value capitalized software and machinery and equipment. Construction in process was reported at its cost. The results from all approaches were adjusted for the impact of economic obsolescence.
(r)Reflects the fair value adjustment related to the Company's intangible assets. The fair value of the developed technology intangible assets were determined using the income approach. The cash flows were discounted commensurate with the level of risk associated with each asset within its projected cash flows. The discount rates applied to the intangible assets consider the overall risk of the business, which reflects a level of risk commensurate with the Company having emerged from bankruptcy twice in the most recent two fiscal years. In addition, the intangible asset discount rates reflect differences in risk within each business segment, as well as the impact of certain tax attributes recorded on the balance sheet. The valuation used discount rates ranging from 13.5 % through 52.5 %, depending on the asset. See Note 14 for further information on intangible assets.
(s)Reflects the net increase on the Company's deferred tax assets as a result of fresh-start accounting, primarily driven by the fair value adjustment on the Company's intangible assets.
(t)The Company’s lease obligations were revalued using the incremental borrowing rate applicable to the Company upon emergence from the 2023 Bankruptcy Proceedings and commensurate with its new capital structure. The incremental borrowing rate used in the revaluation of the lease obligations decreased from 13.2 % in the Predecessor period to 8.1 % in the Successor period. The revaluation of lease obligations includes the adjustment for contract-based off-market intangibles for favorable or unfavorable terms to the right-of-use assets as well as the removal of right-of-use assets (and affiliated lease liabilities) associated with the Company’s leases with a remaining contract term of less than one year as of the 2023 Effective Date. The revaluation resulted in an increase in the right-of-use asset of $10.8 million.
(u)Reflects an adjustment of $1.2 million to decrease the Company's current lease liabilities as a result of the revaluation of the lease obligations as described in footnote (t) above.
(v)Reflects the reduction of the Company's deferred tax liabilities as a result of fresh-start accounting.
(w)Reflects the (i) fair value adjustment to the contingent value rights associated with Terlivaz (“Terlivaz CVR”) utilizing a net present value of a probability-weighted assessment estimated using a Monte Carlo simulation. The Company determined the fair value adjustment to be $14.9 million; and (ii) an increase to the Company's non-current lease liabilities of $9.2 million as described in footnote (t) above.
(x)Reflects the fair value adjustment to the Acthar Gel-Related Settlement liability utilizing a discounted cash flow model with an average credit-adjusted discount rate of 13.3 %.
93
(y)Reflects the fair value adjustment to eliminate the accumulated other comprehensive income of $1.0 million related to pension benefits and $5.7 million of cash flow hedges, partially offset by the elimination of $2.0 million of currency translation adjustment and $2.5 million of income tax effects, which resulted in income tax benefit of zero . Fair value adjustments for discontinued operations were $9.0 million related to pension benefits, partially offset by the elimination of $0.1 million of currency translation adjustment.
| AOCI adjustment | ||||||||
| Pension benefits | $ | |||||||
| Cash flow hedges | ||||||||
| CTA | ( | |||||||
| Income tax effect | ( | |||||||
| Total AOCI Impact | $ | |||||||
(z)The cumulative effect of the fresh-start accounting on the Successor's accumulated deficit is as follows:
| Fresh-start adjustment: | ||||||||
| Inventories | $ | |||||||
Property, plant and equipment | ( | |||||||
Intangible assets | ( | |||||||
Acthar Gel-Related Settlement | ||||||||
| Other assets and liabilities | ( | |||||||
| Total fresh-start adjustments impacting reorganization items, net | ( | |||||||
Fresh-start adjustments to accumulated other comprehensive income, net of | ||||||||
| Total fresh-start adjustments recorded to income tax benefit | ||||||||
| Net fresh-start impact to accumulated deficit | $ | ( | ||||||
| Attributable to: | ||||||||
| Continuing operations | $ | ( | ||||||
| Discontinued operations | $ | |||||||
(aa)Reflects the fresh-start accounting and Reorganization Adjustments related to the discontinued operations:
| Discontinued Operations | |||||||||||
| Reorganization | Fresh-Start Accounting | ||||||||||
| Inventories | $ | $ | |||||||||
| Current assets of disposed business | $ | $ | |||||||||
| Property, plant and equipment, net | $ | $ | |||||||||
| Intangible assets, net | |||||||||||
| Deferred income taxes | ( | ||||||||||
| Other assets | |||||||||||
| Long-term assets of disposed business | $ | $ | |||||||||
| Accrued and other current liabilities | $ | $ | |||||||||
| Current liabilities of disposed business | $ | $ | |||||||||
| Other liabilities | $ | $ | ( | ||||||||
| Liabilities subject to compromise | ( | ||||||||||
| Long-term liabilities of disposed business | $ | ( | $ | ( | |||||||
Reorganization items, net
Reorganization items, net, include amounts incurred after a petition date but prior to emergence from bankruptcy as a direct result of the 2023 Chapter 11 Cases, as well as gains and losses associated with emergence from the 2023 Chapter 11 Cases. These amounts include gains and losses associated with the reorganization, primarily the loss on Fresh-Start Adjustments, gain on settlement of LSTC, bankruptcy-related professional fees, debt financing fees, write-off of debt issuance costs and related unamortized premiums and discounts and other items.
94
The period November 15, 2023 through December 29, 2023 (Successor), included professional fees associated with the implementation of the 2023 Plan incurred after the 2023 Effective Date that are directly related to the restructuring and reorganization of the Company. The period December 31, 2022, through November 14, 2023 (Predecessor), primarily included a gain on the settlement of LSTC of $1,966.0 million partially offset by a loss of $1,452.7 million on Fresh-Start Adjustment and $1,139.5 million of adjustments of claims to their estimated allowed claim amount as a result of the emergence from the 2023 Bankruptcy Proceedings. Of the total reorganization items, net of $892.7 million, $1,539.2 million is attributable to continuing operations and $(646.5 ) million is attributable to discontinued operations.
Cash paid for reorganization items, net for the period November 15, 2023, through December 29, 2023 (Successor), and December 31, 2022, through November 14, 2023 (Predecessor) were $0.8 million and $22.6 million, respectively.
Reorganization items, net, were comprised of the following:
| Successor | Predecessor | |||||||||||||
| Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||
| Gain on settlements of LSTC | $ | $ | ( | |||||||||||
| Loss on fresh-start adjustments | ||||||||||||||
| Adjustments of other claims | ||||||||||||||
| Professional and other service provider fees | ||||||||||||||
Debt financing | ||||||||||||||
| Debt valuation adjustments | ||||||||||||||
Write off of prepaid premium for directors and officers' insurance policies, net, and directors fees | ||||||||||||||
Acceleration of the vesting of Predecessor equity awards upon the 2023 Effective Date | ||||||||||||||
| Total reorganization items, net | $ | $ | ||||||||||||
| Attributable to: | ||||||||||||||
| Continuing operations | $ | $ | ||||||||||||
| Discontinued operations | $ | $ | ( | |||||||||||
| 4. | Summary of Significant Accounting Policies | ||||
Product Sales Revenue
The Company sells its products through independent channels which are considered its customers, including direct to retail pharmacies, direct to hospitals and other institutions and through distributors. The Company also enters into arrangements with health care providers and payers, wholesalers, government agencies, institutions, managed care organizations and group purchasing organizations to establish contract pricing for certain products that provide for government (Medicare and Medicaid) and/or privately-negotiated (Managed Care) rebates, sales incentives, chargebacks, distribution service agreements fees, fees for services and administration fees and discounts with respect to the purchase of the Company’s products.
Reserve for Variable Considerations
Product sales are recorded at the net sales price (transaction price), which includes estimates of variable consideration for which reserves are established. These reserves result from estimated government (Medicare and Medicaid) and/or privately-negotiated (Managed Care) rebates, sales incentives, chargebacks, distribution service agreements fees, fees for services and administration fees and discounts that are offered within contracts between the Company and its customers, health care providers and payers, government agencies, institutions, managed care organizations and group purchasing organizations relating to the sale of the Company's products. These reserves are based on the expected value method. These estimates take into consideration a range of possible outcomes for relevant factors such as the Company’s historical experience, estimated future trends, estimated customer inventory levels, contractual agreements, and the level of utilization of the Company’s products. Overall, these reserves reflect the Company’s best estimate of the amount of consideration to which it is entitled based on the terms of the contract. The amount of variable consideration that is included in the transaction price may be constrained (reduced) and is included in the net sales price only to the extent that it is probable that a significant reversal in the amount of the cumulative revenue recognized will not occur in a future period. The Company adjusts reserves for chargebacks, government (Medicare and Medicaid) rebates, privately negotiated (Managed Care) rebates, product returns and other sales deductions to reflect differences between estimated and actual experience either on a monthly or quarterly basis (dependent on the deduction type). Such adjustments impact the amount of net sales recognized in the period of adjustment.
Product sales are recognized when the customer obtains control of the Company's product. Control is transferred either at a point in time, generally upon delivery to the customer site, or in the case of certain of the Company's products, over the period in which the
95
customer has access to the product and related services. Revenue recognized over time is based upon either consumption of the product or passage of time based upon the Company's determination of the measure that best aligns with how the obligation is satisfied. The Company's considerations of why such measures provide a faithful depiction of the transfer of its products are as follows:
•For those contracts whereby revenue is recognized over time based upon consumption of the product, the Company either has:
1.the right to invoice the customer in an amount that directly corresponds with the value to the customer of the Company's performance to date, for which the practical expedient to recognize in proportion to the amount it has the right to invoice has been applied, or
2.the remaining goods and services to which the customer is entitled is diminished upon consumption.
•For those contracts whereby revenue is recognized over time based upon the passage of time, the benefit that the customer receives from unlimited access to the Company's product does not vary, regardless of consumption. As a result, the Company's obligation diminishes with the passage of time; therefore, ratable recognition of the transaction price over the contract period is the measure that best aligns with how the obligation is satisfied.
Transaction price allocated to the remaining performance obligations
The majority of the Company's contracts have a term of less than one year; and the amount of transaction price allocated to the performance obligations that are unsatisfied at period end is generally expected to be satisfied within one year.
Cost to obtain a contract
As the majority of the Company's contracts are short-term in nature, sales commissions are generally expensed when incurred as the amortization period would have been less than one year. These costs are recorded within SG&A in the consolidated statements of operations. For contracts that extend beyond one year, the incremental expense recognition matches the recognition of related revenue.
Costs to fulfill a contract
The Company capitalizes the costs associated with the devices used in the Company's portfolio of drug-device combination products, which are used in satisfaction of future performance obligations. Capital expenditures for these devices represent cash outflows for the Company's cost to produce the asset, which is classified in property, plant and equipment, net on the consolidated balance sheets and expensed to cost of sales over the useful life of the equipment.
License Revenues
The Company licensed certain product rights to third parties in exchange for royalties on net sales of the respective products and, in certain limited circumstances, payments based on the achievement of specified sales-based milestones. The Company generally recognizes such royalty and milestone revenue as the related sales occur or milestones are achieved.
Contract Balances
Accounts receivable are recorded when the right to consideration becomes unconditional. Payments received from customers are typically based upon payment terms between 30 and 120 days. The Company does not maintain significant contract asset balances. Contract liabilities are recorded when cash payments are received in advance of the Company's performance, including amounts that are refundable.
Amounts collected from customers and remitted to third parties
Amounts collected from customers and remitted to third parties, such as sales taxes collected from customers and remitted to governmental authorities, are accounted for on a net basis. Accordingly, such amounts are excluded from both net sales and expenses.
Shipping and Handling Costs
Shipping costs, which are costs incurred to physically move product from the Company's premises to the customer's premises, are classified as SG&A. Handling costs, which are costs incurred to store, move and prepare product for shipment, are classified as cost of sales. Shipping costs amounted to $6.0 million, $9.2 million, $1.3 million, and $7.3 million for the years ended December 31, 2025 and December 27, 2024 (Successor), the period November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor), respectively.
Advertising Costs
Advertising costs are expensed as incurred and classified as SG&A. Advertising costs amounted to $74.7 million, $35.9 million, $5.8 million, $30.4 million for the years ended December 31, 2025 and December 27, 2024 (Successor), the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor), respectively.
96
Research and Development
Internal research and development (“R&D”) costs are expensed as incurred. R&D expenses include salary and benefits, allocated overhead and occupancy costs, clinical trial and related clinical manufacturing costs, contract services, medical affairs and other costs.
From time to time, the Company has entered into licensing or collaborative agreements with third parties to develop a new drug candidate or intellectual property asset. These agreements may include R&D, marketing, promotion and selling activities to be performed by one or all parties involved. These collaborations generally include upfront, milestone and royalty or profit sharing payments contingent upon future events tied to the developmental and commercial success of the asset. In general, upfront and milestone payments made to third parties under these agreements are expensed as incurred up to the point of regulatory approval of the product. Milestone payments made to third parties upon regulatory approval are capitalized as an intangible asset and amortized to cost of sales over the estimated useful life of the related product.
Restructuring
Share-Based Compensation
The Company recognizes the cost of employee services received in exchange for awards of equity or liability-based instruments based on the grant-date fair value of those awards. That cost is recognized over the requisite service period, which is the period an employee is required to provide service in exchange for the award (generally the vesting period). The cost for liability-based instruments is remeasured each reporting period throughout the requisite service period.
As of the 2023 Effective Date, the Company’s ordinary shares were no longer traded on an active market. Accordingly, the fair value of share-based awards granted after the 2023 Effective Date requires the valuation of the Company’s equity. With the assistance of a third-party valuation advisor, the Company estimates the fair value of share-based awards using an income approach, which reflects a calculation of the present value of the Company’s projected future cash flows. The preparation of projected future cash flows involves a variety of estimates and assumptions, including but not limited to expected future revenue and expenses, discount rates, and the probability of possible future events. While the Company considers such estimates and assumptions reasonable, they are inherently subject to significant business, economic and competitive uncertainties, many of which are beyond the Company’s control and, therefore, may not be realized. The use of different estimates and assumptions could have a significant effect on the determination of the Company’s equity value. Beginning in 2025, the grant-date fair value of these awards is recognized generally as expense on a graded basis over the service period. The Company accounts for forfeitures as they occur for service condition aspects of certain share-based awards.
The Company is not listed on a national securities exchange or quoted on the automated quotation system of a national securities association, and as such, the Company used an estimated fair value per ordinary share as of July 31, 2025, in accordance with the U.S. Internal Revenue Code Section 409A and Accounting Standards Codification 820, Fair Value Measurement, to determine fair value of consideration transferred in connection with the Business Combination. See Note 5 for additional information.
Discontinued Operations
The Company classifies a component as discontinued operations when it has been disposed of or classified as held for sale and the disposal represents a strategic shift with a major effect on the Company’s operations and financial results, in accordance with ASC 205‑20, Presentation of Financial Statements - Discontinued Operations (“ASC 205-20”).
The results of discontinued operations, including any gain or loss on disposal (if applicable), are presented as a single line item, net of tax, in the consolidated statements of operations for all periods presented. Assets and liabilities of discontinued operations are presented separately on the consolidated balance sheets when the criteria triggering discontinued operations are met. Prior‑period amounts are reclassified to conform to current‑period presentation.
Assets and Liabilities Held for Sale and Gain on Divestitures
Assets and liabilities to be disposed of together as a group in a single transaction (“disposal groups”) are classified as held for sale if their carrying amounts are principally expected to be recovered through a sale transaction rather than through continuing use. Held for sale classification is required when the six criteria in ASC 360 - Property, Plant and Equipment are met. This generally occurs when an agreement to sell exists, or management has committed to a plan to sell the assets within one year.
97
The long-lived assets included in a disposal group are reported at the lower of their carrying value or fair value less cost to sell, beginning in the period the held for sale criteria are met. Fair value is determined using acceptable valuation principles, such as the excess earnings, relief from royalty, lost profit or cost method, or a market-based measurement, as applicable. Prior to disposal, losses are recognized for any initial or subsequent write-down to fair value less cost to sell, while gains are recognized for any subsequent increase in fair value less cost to sell, but not in excess of any cumulative losses previously recognized. Any gains or losses not previously recognized that result from the sale of a disposal group shall be recognized at the date of sale. Depreciation and amortization expense are not recorded on long-lived assets included within the disposal group. Gains or losses on the sale of businesses are recognized upon disposition.
(Loss) Income Per Share
(Loss) income per share is computed by dividing net (loss) income by the number of weighted-average shares outstanding during the period. Diluted income per share is computed using the weighted-average shares outstanding and, if dilutive, potential ordinary shares outstanding during the period. Potential ordinary shares represent the incremental ordinary shares issuable for restricted share units. The Company calculates the dilutive effect of outstanding restricted share units on income per share by application of the treasury stock method. Dilutive securities, including participating securities, are not included in the computation of income per share if the impact would have been anti-dilutive.
Currency Translation
For the Company's non-U.S. subsidiaries that transact in a functional currency other than U.S. dollars, assets and liabilities are translated into U.S. dollars using period-end exchange rates. Revenues and expenses are translated at the average exchange rates in effect during the related month. The net effect of these translation adjustments is shown in the consolidated financial statements as a component of accumulated other comprehensive income (loss). From time to time, the Company has entered into derivative instruments to mitigate the exposure of movements in certain of these foreign currency transactions. Gains and losses resulting from foreign currency transactions are included in net income (loss).
Cash and Cash Equivalents
The Company classifies cash on hand and deposits in banks, including commercial paper, money market accounts and other investments it may hold from time to time, with an original maturity to the Company of three months or less, as cash and cash equivalents.
Restricted Cash and Cash Equivalents
Cash and cash equivalents that are legally restricted as to withdrawal or use are excluded from Cash and cash equivalents. As of December 31, 2025, $111.4 million and $29.8 million is included in Prepaid expenses and other current assets and Other assets, in the consolidated balance sheet, respectively. Refer to Note 21 for additional information.
Accounts Receivable and Allowance for Doubtful Accounts
Trade accounts receivable are presented net of an allowance for doubtful accounts. The allowance for doubtful accounts reflects an estimate of losses inherent in the Company's accounts receivable portfolio determined on the basis of historical experience, current facts and circumstances, reasonable and supportable forecasts and other available evidence. Accounts receivable are written off when management determines they are uncollectible. Trade accounts receivable are also presented net of reserves related to chargebacks and rebates payable to customers with whom the Company has trade accounts receivable and the right of offset exists.
Inventories
Inventories are recorded at the lower of cost or net realizable value, using the first-in, first-out convention. The Company reduces the carrying value of inventories for those items that are determined, in the judgment of management, to be excess, obsolete or slow-moving, taking into consideration factors such as changes in customer demand, technology developments or other economic factors. Inventory that is in excess of the amount expected to be sold within one year is classified as long-term inventory and is recorded in Other assets in the consolidated balance sheets. The Company capitalizes inventory costs associated with certain products prior to regulatory approval and product launch when it is reasonably certain, based on management’s judgment of future commercial use and net realizable value, that the pre-launch inventories will be saleable. The determination to capitalize is made on a case-by-case basis. The Company could be required to write down previously capitalized costs related to pre-launch inventories upon a change in such judgment, a denial or delay of approval by regulatory bodies, a delay in commercialization or other potential factors.
98
Property, Plant and Equipment
Property, plant and equipment is stated at cost less accumulated depreciation and impairment. Major renewals and improvements are capitalized, while routine maintenance and repairs are expensed as incurred. Depreciation for property, plant and equipment, other than land and construction in process, is generally based upon the following estimated useful lives, using the straight-line method:
| Buildings | to | ||||||||||
| Leasehold improvements | Remaining term of lease | ||||||||||
| Capitalized software | to | ||||||||||
| Machinery and equipment | to | ||||||||||
The Company capitalizes certain computer software and development costs incurred in connection with developing or obtaining software for internal use.
Upon retirement or other disposal of property, plant and equipment, the cost and related amount of accumulated depreciation are eliminated from the asset and accumulated depreciation accounts, respectively. The difference, if any, between the net asset value and the proceeds is included in net income (loss).
The Company assesses the recoverability of assets or asset groups using undiscounted cash flows whenever events or circumstances indicate that the carrying value of an asset or asset group may not be recoverable. If an asset or asset group is found to be impaired, the amount recognized for impairment is equal to the difference between the carrying value of the asset or asset group and its fair value.
Leases
The Company assesses all contracts at inception to determine whether a lease exists. The Company leases office space, manufacturing and warehousing facilities, equipment and vehicles, which are generally operating leases. Leases with an initial term of 12 months or less are not recorded on the consolidated balance sheet; the Company recognizes operating lease expense for these leases on a straight-line basis over the lease term. The Company has lease agreements with lease and non-lease components, which are accounted for separately. The Company's lease agreements generally do not contain variable lease payments or any material residual value guarantees.
Lease assets and liabilities are recognized based on the present value of the future minimum lease payments over the lease term as of the commencement date. As the Company's leases do not generally provide an implicit rate, the Company utilizes its incremental borrowing rate based on the information available at commencement date in determining the present value of future lease payments. Most leases include one or more options to terminate or renew, with renewal terms that can extend the lease term from to five years . The exercise of lease renewal options is at the Company's sole discretion. Termination and renewal options are included within the lease assets and liabilities only to the extent they are reasonably certain.
Goodwill
Goodwill represents the excess of consideration transferred over the fair value of identifiable net assets acquired in a business combination. Goodwill is tested for impairment at least annually, or more frequently if events or changes in circumstances indicate potential impairment. The Company first assesses qualitative factors to determine whether it is more likely than not that the fair value of the reporting unit is less than its carrying amount. If this threshold is met, or if the qualitative assessment is bypassed, a quantitative impairment test is performed. An impairment loss is recognized for the amount by which the carrying amount of the reporting unit exceeds its fair value and cannot be reversed in future periods.
Acquisitions
For acquisitions that meet the criteria for business combination accounting, the amounts paid are allocated to the tangible assets acquired and liabilities assumed based on their estimated fair values at the date of acquisition. The Company then allocates the purchase price in excess of net tangible assets acquired to identifiable intangible assets, including purchased research and development. The fair value of identifiable intangible assets is based on detailed valuations. These valuations rely on a number of factors including operating results, business plans, economic projections, anticipated future cash flows, transactions and market place data. There are inherent uncertainties related to these factors and judgment in applying them to estimate the fair value of individual assets acquired in a business combination. Due to these inherent uncertainties, there is risk that the carrying value of the Company’s recorded intangible assets may be overstated, which may result in an increased risk of impairment in future periods. The Company performs its intangible asset valuations using an income approach based on the present value of future cash flows. This approach incorporates many assumptions including future growth rates, discount factors and income tax rates. Changes in economic and operating conditions impacting these assumptions could result in impairment in future periods.
99
Certain asset acquisitions or license agreements may not meet the criteria for a business combination. The Company accounts for these transactions as an asset acquisition and recognizes the identifiable assets acquired, the liabilities assumed and any noncontrolling interest in the acquired entity. Any initial up-front payments incurred in connection with the acquisition or licensing of our in-process research and development (“IPR&D”) product candidates that do not meet the definition of a business are treated as research and development expense.
Intangible Assets
Intangible assets acquired in a business combination are recorded at fair value, while intangible assets acquired in other transactions are recorded at cost. Intangible assets with finite useful lives are amortized over their useful lives on a straight-line basis or utilizing an accelerated method of amortization if that method better reflects the pattern in which the economic benefit of the assets are used. Useful lives range from 5.9 to 13 years. Amortization expense is included in cost of sales.
Income Taxes
Deferred tax assets and liabilities are recognized for the expected future tax consequences of events that have been reflected in the consolidated financial statements. Deferred tax assets and liabilities are determined based on the differences between the financial reporting and tax bases of assets and liabilities and operating loss carryforwards, using tax rates expected to be in effect for the years in which the differences are expected to reverse. A valuation allowance is provided to reduce net deferred tax assets if, based upon the available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized.
The Company determines whether it is more likely than not that a tax position will be sustained upon examination. The tax benefit of any tax position that meets the more-likely-than-not recognition threshold is calculated as the largest amount that is more than 50.0% likely of being realized upon resolution of the uncertainty. To the extent a full benefit is not realized on the uncertain tax position, an income tax liability or a reduction to a deferred tax asset (“contra-DTA”) is established. Interest and penalties on income tax obligations, associated with uncertain tax positions, are included in the provision for income taxes.
The calculation of the Company's tax liabilities involves dealing with uncertainties in the application of complex tax regulations in a multitude of jurisdictions across the Company's global operations. The Company adjusts these liabilities and contra-DTAs as a result of changing facts and circumstances; however, due to the complexity of some of these uncertainties, the ultimate resolution may result in a payment that is materially different from current estimates of the tax liabilities. If the Company's estimate of tax liabilities proves to be less than the ultimate assessment, an additional charge to expense would result. If payment of these amounts ultimately proves to be less than the recorded amounts, the reversal of the liabilities may result in income tax benefits being recognized in the period when it is determined that the liabilities are no longer necessary. Refer to Note 9 for further information regarding the classification of such amounts in the consolidated balance sheets.
Contingencies
The Company may from time to time be subject to various legal proceedings and claims, including government investigations, environmental matters, product liability matters, patent infringement claims, antitrust matters, securities class action lawsuits, personal injury claims, employment disputes, contractual and other commercial disputes, and all other legal proceedings, all in the ordinary course of business as further discussed in Note 20. The Company records accruals for loss contingencies when it is probable that a liability has been incurred and the amount can be reasonably estimated. Legal fees are expensed as incurred. Insurance recoveries related to potential claims are recognized up to the amount of the recorded liability when coverage is confirmed and the estimated recoveries are probable of payment. Assets and liabilities are not netted for financial statement presentation.
Liabilities Subject to Compromise
As a result of the commencement of the 2023 Bankruptcy Proceedings, the payment of pre-petition liabilities was subject to compromise or other treatment pursuant to the 2023 Plan. The determination of how liabilities would ultimately be settled or treated could not be made until the confirmed 2023 Plan became effective. Accordingly, the ultimate amount of such liabilities was not determinable prior to the 2023 Effective Date. Pre-petition liabilities that were subject to compromise were reported at the amounts that were expected to be allowed by the Bankruptcy Court, even if they may be settled for different amounts. The amounts classified as LSTC prior to the 2023 Effective Date were preliminary and were subject to future adjustments dependent upon Bankruptcy Court actions, further developments with respect to disputed claims, determinations of the secured status of certain claims, the values of any collateral securing such claims, rejection of executory contracts, continued reconciliation or other events. Refer to Note 3.
100
Contingent Consideration
Recently Issued Accounting Pronouncements
Recently Issued Accounting Standards Adopted
The FASB issued ASU 2023-09, Income Taxes (Topic 740): Improvements to Income Tax Disclosures in December 2023. This ASU requires public business entities to disclose additional information in specified categories with respect to the reconciliation of the effective tax rate to the statutory rate (the “rate reconciliation”) for federal, state, and foreign income taxes. It also requires greater detail about individual reconciling items in the rate reconciliation to the extent the impact of those items exceeds a specified threshold. ASU 2023-09 is effective for the Company for the fiscal year ending December 31, 2025. The Company adopted this accounting standards update prospectively, effective for the fiscal year ended December 31, 2025, resulting in incremental disclosures. Refer to Note 9 for additional information.
Recently Issued Accounting Standards Not Yet Adopted
In November 2024, the FASB issued ASU 2024-03, Income Statement - Reporting Comprehensive Income - Expense Disaggregation Disclosures (Subtopic 220-40): Disaggregation of Income Statement Expenses. This ASU requires new financial statement disclosures about the nature, amount, and timing of relevant expense categories underlying income statement expense, including purchases of inventory, employee compensation, depreciation, and amortization in commonly presented expense captions such as cost of revenue and selling, general and administrative expenses. ASU 2024-03 is effective for fiscal years beginning after December 15, 2026, and interim periods within fiscal years beginning after December 15, 2027, with early adoption permitted. The disclosure updates are required to be applied prospectively with the option for retrospective application. The Company is currently evaluating the disclosure requirements of this standard and the impact on its consolidated financial statements.
In September 2025, the FASB issued ASU 2025‑06, Intangibles—Goodwill and Other—Internal‑Use Software (Subtopic 350‑40): Targeted Improvements to the Accounting for Internal‑Use Software. This ASU modernizes the accounting guidance for internal‑use software costs by eliminating references to software development project stages and introducing a principles‑based model that requires capitalization of costs when management has authorized and committed to funding the project and it is probable that the project will be completed and the software will be used for its intended purpose. The ASU also supersedes existing guidance on website development costs and incorporates that guidance into Subtopic 350‑40. ASU 2025‑06 is effective for annual reporting periods beginning after December 15, 2027, including interim periods within those fiscal years, with early adoption permitted. The standard may be applied prospectively, modified retrospectively, or retrospectively. The Company is currently evaluating the impact of this standard on its accounting for internal‑use software costs and related disclosures.
No other new accounting pronouncement issued or effective during the fiscal year has had, or is expected to have, a material impact on the Company’s consolidated financial statements.
| 5. | Business Combination | ||||
On March 13, 2025, the Company entered into a Transaction Agreement (as amended on April 23, 2025) (“Transaction Agreement”), with Endo and Salvare Merger Sub LLC, the Company’s wholly owned subsidiary (“Merger Sub”), bringing together two highly complementary companies with durable, on-market products and best-in-class capabilities across the value chain. On July 31, 2025, the Company completed the Business Combination, whereby the Company acquired all of the issued and outstanding shares of common stock of Endo in exchange for a combination of cash and the Company’s ordinary shares in accordance with the Transaction Agreement. Outstanding shares of common stock of Endo were cancelled and converted into the right to receive 0.2575 of a Company ordinary share (“Per Share Stock Consideration”) and approximately $1.31 in cash (“Per Share Cash Consideration”), without interest and subject to applicable withholding.
The Company acquired Endo by means of the merger of Merger Sub with and into Endo, with Endo continuing as the surviving entity in the merger and a wholly-owned subsidiary of the Company (“Business Combination”). On July 31, 2025, prior to the completion of the Business Combination, the memorandum and articles of association of the Company were amended by means of a scheme of arrangement (“Scheme”) under the Companies Act 2014 of Ireland (as amended) and certain other amendments that had been previously approved by the Company’s shareholders (the “constitution amendment,” and, together with the Scheme and the Business Combination, the “Transactions”).
101
The Business Combination resulted in increased scale and enhanced capabilities to develop, manufacture, and commercialize branded therapeutics, generic pharmaceuticals and sterile injectables.
The Company’s operating results for the year ended December 31, 2025, reflect the consolidated results of five months of Endo’s branded products. Net sales and loss from continuing operations before income taxes of Endo subsequent to closing of the Business Combination included in the Company’s consolidated statements of operation were $397.0 million and $253.9 million, respectively. Endo’s contribution to the loss from continuing operations before income taxes is significantly affected by the amortization of inventory fair value step-up and of intangible assets recognized in connection with the Business Combination.
The Company is the acquiring entity for accounting purposes. In identifying the Company as the acquiring entity for accounting purposes, management took into account the voting rights of all equity instruments, the composition of the corporate governing body and senior management, the size of each of the companies, and the terms of the exchange of equity interests. The Company accounted for the Business Combination under the acquisition method of accounting. This method requires the recording of acquired assets, including separately identifiable intangible assets and liabilities assumed at their fair value on the acquisition date. Any excess of the purchase price over the estimated fair value of the identifiable net assets acquired is recorded as goodwill.
The determination of the estimated fair value of assets acquired and liabilities assumed requires management’s judgment and often involves the use of significant estimates and assumptions, including assumptions with respect to future sales, cost of sales, operating expenses, discount rates, asset lives and market multiples, among other items. Fair values were determined by management using a variety of methodologies and resources, including external independent valuation experts. The valuation methods consisted of physical appraisals, discounted cash flow analyses, excess earnings, relief from royalty, and other appropriate valuation techniques to determine the fair value of assets acquired and liabilities assumed. The fair value of the developed technology assets was determined using the excess earnings method, which involves the use of assumptions including future sales, cost of sales, operating expenses and discount rates.
Consideration Transferred
The consideration for the Business Combination is calculated as follows:
Endo shares of common stock outstanding as of July 31, 2025 | ||||||||
| Exchange Ratio | ||||||||
| Company ordinary shares issued in exchange | ||||||||
Company closing stock price (1) | ||||||||
| Estimated fair value of Company ordinary shares issued | $ | |||||||
Other cash consideration (2) | ||||||||
| Payment to Endo stockholders | ||||||||
| Other merger consideration attributable to Endo stock-based awards | ||||||||
| Obligation to cash settle shares underlying certain Endo stock-based awards | ||||||||
| Total consideration transferred | $ | |||||||
(1)The Company is not listed on a national securities exchange or quoted on the automated quotation system of a national securities association, and as such, used an estimated fair value per ordinary share as of July 31, 2025, in accordance with Accounting Standards Codification 820, Fair Value Measurement, to determine fair value of consideration transferred.
(2)Other cash consideration represents less than $0.1 million of aggregate cash payments to Endo stockholders in lieu of any fractional shares.
102
Estimated Fair Values
The table below represents the allocation of the consideration to Endo’s tangible and intangible assets acquired and liabilities assumed based on management’s estimate of their respective fair values.
| Estimated fair value | Measurement Period Adjustments | Adjusted fair value | ||||||||||||||||||
| Total consideration (in millions) | $ | $ | — | $ | ||||||||||||||||
| Cash and cash equivalents | $ | $ | — | $ | ||||||||||||||||
| Restricted cash and cash equivalents | — | |||||||||||||||||||
| Accounts receivable, net | — | |||||||||||||||||||
| Inventories | ( | |||||||||||||||||||
| Prepaid expenses and other current assets | ||||||||||||||||||||
| Income taxes receivable | ||||||||||||||||||||
| Property, plant and equipment, net | — | |||||||||||||||||||
| Inventories, long-term | — | |||||||||||||||||||
| Operating lease assets | — | |||||||||||||||||||
| Intangible assets, net | ||||||||||||||||||||
| Deferred income tax assets | ||||||||||||||||||||
| Other assets | — | |||||||||||||||||||
| Total assets, excluding goodwill | $ | $ | ( | $ | ||||||||||||||||
| Current maturities of long-term debt | — | |||||||||||||||||||
| Accounts payable | — | |||||||||||||||||||
| Accrued payroll and payroll-related costs | — | |||||||||||||||||||
| Accrued interest | — | |||||||||||||||||||
| Accrued and other current liabilities | ( | |||||||||||||||||||
| Long-term debt | — | |||||||||||||||||||
| Operating lease liabilities | — | |||||||||||||||||||
| Deferred tax liabilities | ( | |||||||||||||||||||
| Other liabilities | ||||||||||||||||||||
| Net assets acquired | $ | $ | ( | $ | ||||||||||||||||
| Goodwill | $ | $ | $ | |||||||||||||||||
During the measurement period, the Company adjusted the provisional amounts recognized at the acquisition date to reflect new information obtained about facts and circumstances that existed at the acquisition date. The decrease in inventory fair value reflects the removal of certain components that were concluded to be non-saleable on a stand-alone basis and have not been valued at the opening balance sheet date. The increase in intangible assets relates to the impact of lower inventory fair value step up amortization as a result of the aforementioned decrease in inventory fair value. The increase in prepaid expenses and other current assets reflects the recognition of a non-trade receivable associated with the resolution of an acquired contingency during the measurement period. Measurement period adjustments also reflect the corresponding deferred tax impacts associated with the changes in inventory and intangible assets, and certain insignificant changes in income taxes payable and other liabilities. As a result of the adjustments noted above, goodwill increased by $24.5 million.
The amounts assigned to the identifiable intangible assets, the weighted average useful lives, and the amortization method are as follows:
| Net Book Value | Amortization Method | Weighted Average Useful Lives (in years) | ||||||||||||||||||
| Developed technology - Branded | $ | Straight-line | ||||||||||||||||||
Intangible assets associated with discontinued operations (1) | $ | Straight-line | ||||||||||||||||||
| Total intangible assets | $ | |||||||||||||||||||
As summarized above, the Company recorded $31.8 million of goodwill, none of which will be tax deductible. Goodwill reflects the value of the assembled workforce, expanded product capabilities and future growth opportunities that are not separately identifiable as intangible assets under ASC 805.
103
As of December 31, 2025, the measurement period has not ended. Further adjustments may be necessary as a result of the Company’s on-going assessment of additional information related to the fair value of assets acquired and liabilities assumed.
Receipts of unrestricted cash, net of payments related to the Business Combination
Cash receipts related to the Business Combination, net of cash paid is as follows:
| Cash acquired | ||||||||
| Less: | ||||||||
| Payment to stockholders | ( | |||||||
| Payment to cash settle shares underlying certain Endo stock-based awards | ( | |||||||
Receipts of unrestricted cash, net of payments related to the Business Combination | $ | |||||||
Combination, Integration, and Other Related Expenses
Transaction expenses associated with the Business Combination are included in combination, integration, and other related expenses in the consolidated statements of operations. During the year ended December 31, 2025, the Company recorded $141.2 million of costs, which includes legal, financial, other advisory and consulting costs, which primarily relate to shareholder matters, integration planning and execution, and regulatory matters associated with the Business Combination, as well as severance costs of approximately $44.1 million.
Unaudited Pro Forma Financial Information
The following unaudited pro forma consolidated financial information has been prepared as if the Business Combination had taken place on December 30, 2023, which is the beginning of the Company’s fiscal year for 2024. The unaudited pro forma financial information includes certain adjustments to each company’s historical actual financial results, including, but not limited to:
(i)Alignment of related accounting policies;
(ii)Certain eliminations of intercompany transactions between the two companies made to the Company’s and Endo’s historical standalone financial statements;
(iii)Adjustments to amortization expense based on the initial estimates of fair value inventory step up and finite-lived intangible assets. The adjustments include an expense recorded in costs of goods sold (“COGS”) associated with selling inventory acquired in the Business Combination which was adjusted to fair value as part of purchase accounting;
(iv)A decrease in depreciation expense based on the initial estimates of the fair value of acquired tangible assets, including real and personal property; and
(v)Adjustments to interest expense to reflect the impact of the financing and capital structure of the combined Company.
The unaudited pro forma financial information is provided for illustrative purposes only and may not provide an indication of results in the future. The unaudited pro forma financial information and underlying pro forma adjustments are based upon currently available information and include certain estimates and assumptions made by management. Accordingly, actual results could differ materially from the unaudited pro forma financial information.
| Year Ended | |||||||||||
| Unaudited Pro Forma Financial Information | December 31, 2025 | December 27, 2024 (1) | |||||||||
| Net Sales | $ | $ | |||||||||
| Net (loss) income | ( | ||||||||||
(1) On April 23, 2024, Endo International plc’s (“Endo’s Predecessor”) plan of reorganization became effective. In accordance with the Endo Plan (as defined below) on the Endo Effective Date (as defined below), Endo acquired substantially all of the assets, as well as certain equity interests of and assumed certain liabilities of Endo’s Predecessor. In accordance with ASC Topic 852, Reorganization, the provisions of fresh-start accounting were applied on the Endo Effective Date and Endo became the Successor entity for financial reporting purposes. Endo’s Predecessor’s reorganization items, net were $6,125.1 million.
Transaction Incentive Plan
On February 2, 2024, the Company’s then-existing Board of Directors (“Board”) adopted a Transaction Incentive Plan (as amended on August 4, 2024 and December 2, 2024, the “Transaction Incentive Plan”), which was intended to compensate designated executive officers and directors with bonus payments to be made upon the consummation of qualifying strategic transactions and dispositions (each, a “Qualifying Transaction”). Each bonus payment earned under the Transaction Incentive Plan was to be generally delivered 50 % in connection with closing of the applicable Qualifying Transaction and 50 % on the earlier of (a) December 31, 2026 or a qualifying significant event, as defined in the Transaction Incentive Plan, and (b) a significant asset transaction, as defined in the Transaction Incentive Plan (“Final Payment Date”); provided, however that in the event that a Qualifying Transaction closes following a qualifying significant event or significant asset transaction, 100 % of the applicable bonus payment earned with respect to such
104
Qualifying Transaction generally will be paid in connection with closing of such Qualifying Transaction or, if later, when the associated proceeds are received. The Therakos Divestiture qualified as a Qualifying Transaction and the Business Combination qualified as a Qualifying Transaction and a qualifying significant event under the Transaction Incentive Plan, and as a result, the Final Payment Date was within 30 days of the closing of the Business Combination, which occurred on July 31, 2025. The Transaction Incentive Plan terminated in accordance with its terms as a result of the closing of the Business Combination.
Transaction Incentive Plan Payments associated with the Business Combination
Because the Business Combination was not considered probable under U.S. GAAP until it closed, the Company recognized $91.5 million in expense related to the Transaction Incentive Plan payments associated with the Business Combination during the third quarter of 2025, which was recorded within SG&A expenses in the consolidated statement of operations.
| 6. | Divestitures | ||||
During the year ended December 31, 2025, the Company recorded $76.8 million of costs related to the Separation (described below), including legal, financial, other advisory and consulting costs, as well as $5 million related to the CVR Termination Agreement (described below). The Separation met the criteria for discontinued operations reporting; accordingly, such costs are included in Discontinued Operations, net of tax in the Company’s consolidated statements of operations. During the year ended December 27, 2024, the Company recorded $43.9 million of legal, financial, other advisory and consulting costs related to the sales and potential sales of non-core assets, including the Therakos Divestiture (as defined below), within liabilities management and separation costs on the consolidated statements of operations.
Par Health Separation
On November 10, 2025, the Company completed the Separation. This transaction was executed through a redemption of all issued and outstanding 2025 Preferred Shares, par value $0.001 per share, comprising 1,796,196,578,472 preferred shares. The Separation resulted in Par Health becoming an independent, private company not listed on any securities exchange.
The Separation was structured to allocate shares of Par Health Common Stock to certain holders of 2025 Preferred Shares, with a total of 39,421,398 shares distributed as part of the transaction. As a result of the Separation, the Company no longer retains any ownership interest in Par Health, and the associated assets and liabilities have been classified as discontinued operations in the consolidated financial statements.
In connection with the consummation of the Separation, the Company and the Trust entered into an agreement to cancel the Opioid CVRs and terminate the CVR Agreement in exchange for a payment by the Company of $35.0 million to the Trust (the “CVR Termination Agreement”). Pursuant to the CVR Termination Agreement, on November 10, 2025, the Opioid CVRs were cancelled and the CVR Agreement was terminated, resulting in an adjustment of the Opioid CVRs to fair value through additional-paid-in-capital of approximately $4.9 million and the recognition of approximately $5 million of expense during the year ended December 31, 2025, which is recorded within loss from discontinued operations. The CVR Termination Agreement also included customary representations and warranties and a waiver of certain claims.
In connection with the Separation, the Company entered into several agreements with Par Health that govern the relationship of the parties following the Separation, including a separation agreement, a transition services agreement to provide and receive certain services following the separation (the “Par Health TSA”), a tax matters agreement, an employee matters agreement, a manufacturing and supply agreement and an amended and restated multi-tenant lease agreement. For the period from November 10, 2025 to December 31, 2025, there were no cash inflows or outflows associated with these arrangements and the financial statement impacts of are otherwise not material. As of December 31, 2025, under the provisions of these certain agreements, the Company was owed approximately $21.3 million from Par Health and the Company owed approximately $7.9 million to Par Health, which primarily reflect amounts owed between the parties for certain pass-through costs paid or received by one party on behalf of the other party.
Transition Services Agreement: Costs incurred under the Par Health TSA are primarily related to certain core business services and operational support to be provided by Par Health, including supply chain and manufacturing support, quality assurance, commercial, R&D, human resources (“HR”), finance, and information technology (“IT”) services. Costs associated with the Par Health TSA are reflected within cost of sales, selling, general and administrative expenses or research and development expenses, based on the nature of services provided. Par Health also receives transitional support from the Company under the Par Health TSA for up to 24 months. The Company provides Par Health with a broad range of transitional services, including supply chain, manufacturing, quality, commercial, R&D, HR, procurement, compliance, environmental health and safety, facilities and security, finance, IT, and suspicious order monitoring and controlled substance compliance, to ensure business continuity and operational support post transaction. Income generated from these services are reflected in other income (expense), net. A committee with representatives from both parties to the Par Health TSA will oversee service delivery and dispute resolution. Charges are billed monthly.
105
Multi-Tenant Lease Agreement: Under the amended and restated multi-tenant lease agreement, the Company leases designated portions of Par Health’s corporate campus in Hazelwood, Missouri. The lease term extends through October 31, 2027, and includes provisions allowing the Company to reduce or terminate certain leased space or renew the arrangement for additional one‑year periods. Under the agreement, Par Health continues to provide access to building systems, common‑area maintenance, utilities, and related landlord services for the duration of the term. Lease expense associated with this arrangement is recognized within continuing operations and represents a form of continuing involvement with the discontinued Par Health business.
Manufacturing and Supply Agreement: The Company also maintains an ongoing commercial relationship with Par Health under the manufacturing and supply agreement dated November 10, 2025, which has an initial five-year term and automatically renews for additional 24 month periods unless terminated earlier. This agreement includes provisions for the obligations of Par Health to continue to supply certain finished products and provides associated technical and regulatory support as needed, while the Company supplies the required active pharmaceutical ingredient.
The financial results of Par Health are classified as discontinued operations in accordance with ASC 205-20, for all relevant periods presented. The following table summarizes the financial performance of Par Health for the fiscal years ended December 31, 2025, December 27, 2024, the period from November 15, 2023, through December 29, 2023 (Successor), and the period from December 31, 2022, through November 14, 2023 (Predecessor):
| Successor | Predecessor | |||||||||||||||||||||||||
| Major line items constituting income (loss) from discontinued operations | Year Ended December 31, 2025(1) | Year Ended December 27, 2024(1) | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | ||||||||||||||||||||||
| Net sales | $ | $ | $ | $ | ||||||||||||||||||||||
| Cost of sales | ||||||||||||||||||||||||||
| Gross profit | ||||||||||||||||||||||||||
| Selling, general and administrative expenses | ||||||||||||||||||||||||||
| Research and development expenses | ||||||||||||||||||||||||||
| Non-restructuring impairments | ||||||||||||||||||||||||||
| Liabilities management & Separation Costs | ||||||||||||||||||||||||||
| Combination, integration, and other related expenses | ||||||||||||||||||||||||||
| Operating income (loss) | ( | |||||||||||||||||||||||||
| Interest expense | ( | ( | ( | |||||||||||||||||||||||
| Interest income | ||||||||||||||||||||||||||
| Loss on debt extinguishment | ( | |||||||||||||||||||||||||
| Other (expense) income, net | ( | ( | ||||||||||||||||||||||||
| Reorganization items | ( | |||||||||||||||||||||||||
| (Loss) income from discontinued operations before income taxes | ( | ( | ||||||||||||||||||||||||
| Provision for (benefit) from income taxes | ( | |||||||||||||||||||||||||
| (Loss) income from discontinued operations, net of tax | $ | ( | $ | $ | $ | |||||||||||||||||||||
(1) Results do not include $0.3 million and $1.3 million of income from discontinued operations, net of tax, for the years ended December 31, 2025 and December 27, 2024, respectively, relating to the Company’s prior divestiture of it’s Nuclear Imaging business.
The Separation was structured as a pro rata spin-off resulting in the distribution of economic value among shareholders, with the value of shares received by qualified shareholders commensurate with the cash amount received by non-qualified shareholders. It is estimated that non-qualified shareholders represent an insignificant portion of the Company’s shareholder base. Consequently, the Separation was accounted for at the carrying amount of the net assets distributed, with no gain or loss recognized.
106
As set forth below, the assets and liabilities associated with Par Health have been classified as assets and liabilities of the discontinued business in the Company’s consolidated balance sheet as of December 27, 2024.
| Carrying amounts of major classes of assets included as part of discontinued operations | December 27, 2024 | ||||
| Current assets | |||||
| Cash and cash equivalents | $ | ||||
| Accounts receivable, less allowance for doubtful accounts | |||||
| Inventories | |||||
| Prepaid expenses and other current assets | |||||
| Total current assets of the discontinued business | |||||
| Non-current assets | |||||
| Property, plant and equipment, net | |||||
| Intangible assets, net | |||||
| Deferred income taxes | |||||
| Other assets | |||||
| Total non-current assets of the discontinued business | |||||
| Total assets of the discontinued business | $ | ||||
| Carrying amounts of major classes of liabilities included as part of discontinued operations | |||||
| Current liabilities | |||||
| Accounts payable | $ | ||||
| Accrued payroll and payroll-related costs | |||||
| Accrued rebates and returns | |||||
| Accrued and other current liabilities | |||||
| Total current liabilities of the discontinued business | |||||
| Non-current liabilities | |||||
| Pension and postretirement benefits | |||||
| Environmental liabilities | |||||
| Other income tax liabilities | |||||
| Other liabilities | |||||
| Total non-current liabilities of the discontinued business | |||||
| Total liabilities of the discontinued business | $ | ||||
Endo Divestiture of the International Pharmaceutical Business
Prior to the Business Combination, on March 10, 2025, Endo entered into a definitive agreement to divest its International Pharmaceuticals business to Knight Therapeutics Inc. (“Knight”). The sale closed on June 17, 2025, prior to the Business Combination, and Endo received net cash consideration of approximately $78.6 million, consisting of $89.9 million upfront, less approximately $11.3 million related to certain permitted holdbacks. During the remainder of 2025, the Company received additional cash consideration from Knight of $2.4 million, which includes $0.5 million representing a final true-up payment relating to inventory on-hand as of the sale date and $1.9 million representing costs associated with certain employee termination costs initially paid by Endo and subject to reimbursement by Knight upon the satisfaction of certain conditions defined in the purchase and sale agreement. As of December 31, 2025, the Company remained eligible to receive up to an additional $7.2 million related to certain permitted holdbacks and up to $15 million in potential future payments contingent upon the achievement of certain milestones. In March 2026, the Company was informed by Knight that the conditions necessary for the resolution of the certain permitted holdbacks had been satisfied and that the Company was entitled to receive approximately $6.1 million in full and final resolution of the permitted holdbacks, subject to the Company’s execution of a customary release. The Company concluded that this represents the resolution of an acquired contingency during the measurement period following the Business Combination and therefore made an adjustment to its purchase accounting estimates. Refer to Note 5 for additional information. The Company remains eligible to receive the aforementioned potential future milestone payments.
Therakos Divestiture
On August 3, 2024, the Company entered into a Purchase and Sale Agreement for the Company’s Therakos business for a base purchase price of $925.0 million. On November 29, 2024, the Company completed the divestiture of its Therakos business (the “Therakos Divestiture”) for total cash consideration of $887.6 million, net of preliminary purchase price adjustments. The Company recorded a gain on sale of $754.4 million, comprised of the $887.6 million of initial net proceeds less the elimination of $125.5 million of net assets divested and $7.7 million in success-based professional fees. The Company was required to use the proceeds to make a mandatory prepayment on certain portions of its debt, which is further described in Note 15. The Company paid $6.2 million for the final working capital settlement during the year ended December 31, 2025.
107
In connection with the Therakos Divestiture, the Company entered into a transition services agreement (the “Therakos TSA”) effective upon closing to provide certain business support services generally for up to 18 months after the closing date or a longer period for certain services. These services include, but are not limited to, information technology, procurement, distribution, logistics and order to delivery, compliance, accounting, finance, and administrative activities. Revenue associated with the Therakos TSA is recorded within other income (expense), net, and expenses associated with servicing the Therakos TSA are recorded within their natural expense classification, respectively, on the audited condensed consolidated statement of operations. During the years ended December 31, 2025 and December 27, 2024, income under the TSA was $9.5 million and $1.0 million, respectively.
The Therakos Divestiture did not qualify as discontinued operations as it did not represent a strategic shift that will have a major effect on the Company’s operations and financial results. The following table summarizes income (loss) from continuing operations before income taxes for the Therakos business, through the divestiture date. Certain amounts that the Company considers to be non-operational are excluded from income (loss) from continuing operations before income taxes for the Therakos business. These items may include, but are not limited to, corporate and unallocated expenses and liabilities management and separation costs.
| Dollars in millions | Successor | Predecessor | |||||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | ||||||||||||||||||||||||||
Income (loss) from continuing operations before income taxes (1) | $ | $ | $ | $ | ( | ||||||||||||||||||||||||
(1)Includes inventory step-up expense of $0 million, $66.3 million, $13.0 million and $30.1 million, during the year ended December 31, 2025, the year ended December 27, 2024, the period November 15, 2023 through December 29, 2023 (Successor), and the period December 31, 2022 through November 14, 2023 (Predecessor). Also includes intangible asset amortization of $0 million, $16.1 million, $4.6 million and $142.7 million, during the year ended December 31, 2025, the year ended December 27, 2024, the period November 15, 2023 through December 29, 2023 (Successor), the period December 31, 2022 through November 14, 2023 (Predecessor), respectively.
Transaction Incentive Plan Payments associated with the Therakos Divestiture
During the years ended December 31, 2025 and December 27, 2024, the Company recognized $11.9 million and $15.4 million, respectively, in expense related to the Transaction Incentive Plan payments associated with the Therakos Divestiture, which were recorded within SG&A expenses in the consolidated statements of operations.
| 7. | Revenue from Contracts with Customers | ||||
Product Sales Revenue
See Note 22 for disaggregation of the Company's net sales by product.
Reserves for variable consideration
The following table reflects activity in the Company's sales reserve accounts:
Rebates and Chargebacks(1) | Product Returns | Other Sales Deductions | Total | ||||||||||||||||||||
| Balance as of December 30, 2022 (Predecessor) | $ | $ | $ | $ | |||||||||||||||||||
| Provisions | |||||||||||||||||||||||
| Payments or credits | ( | ( | ( | ( | |||||||||||||||||||
Balance as of November 14, 2023 (Predecessor) | $ | $ | $ | $ | |||||||||||||||||||
Balance as of November 15, 2023 (Successor) | $ | $ | $ | $ | |||||||||||||||||||
| Provisions | |||||||||||||||||||||||
| Payments or credits | ( | ( | ( | ( | |||||||||||||||||||
Balance as of December 29, 2023 (Successor) | $ | $ | $ | $ | |||||||||||||||||||
| Provisions | |||||||||||||||||||||||
| Payments or credits | ( | ( | ( | ( | |||||||||||||||||||
| Balance as of Balance as of December 27, 2024 (Successor) | $ | $ | $ | $ | |||||||||||||||||||
| Acquisitions | |||||||||||||||||||||||
| Provisions | |||||||||||||||||||||||
| Payments or credits | ( | ( | ( | ( | |||||||||||||||||||
| Balance as of December 31, 2025 (Successor) | $ | $ | $ | $ | |||||||||||||||||||
(1) Amounts classified within accrued and other current liabilities in the consolidated balance sheets are comprised of $155.9 million and $59.0 million of accrued Medicare, Medicaid, and Managed Care rebates, as of December 31, 2025, and December 27, 2024, respectively.
108
Product sales transferred to customers at a point in time and over time were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Product sales transferred at a point in time | % | % | % | % | ||||||||||||||||||||||
| Product sales transferred over time | ||||||||||||||||||||||||||
Transaction price allocated to the remaining performance obligations
The following table includes estimated revenue from contracts extending greater than one year for certain of the Company's hospital products that are expected to be recognized in the future related to performance obligations that were unsatisfied or partially unsatisfied as of December 31, 2025:
| Fiscal 2026 | $ | ||||
| Fiscal 2027 | $ | ||||
| Thereafter | $ | ||||
Costs to fulfill a contract
As of December 31, 2025, and December 27, 2024, the total net book value of the devices used in the Company's portfolio of drug-device combination products, which are used in satisfying future performance obligations and reflected in property, plant and equipment, net, on the consolidated balance sheets was $62.0 million and $37.8 million, respectively. The associated depreciation expense recognized during the year ended December 31, 2025, the year ended December 27, 2024, the period November 15, 2023, through December 29, 2023 (Successor), and the period December 31, 2022, through November 14, 2023 (Predecessor), was $6.5 million, $2.0 million, zero , and $1.9 million, respectively.
License Revenues
The Company licensed certain rights to third parties in exchange for royalties on net sales of certain of its products, including in certain limited circumstances, payments based on the achievement of specified sales-based milestones. The Company estimates and recognizes such royalty revenue as the related sales occur or as the milestones are achieved, and the amount is reasonably estimable. License revenues recognized were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| License revenues | $ | $ | $ | $ | ||||||||||||||||||||||
| 8. | Restructuring and Related Charges | ||||
The Company, from time to time, seeks more cost-effective means to improve profitability and to respond to changes in its markets. The Company has incurred certain restructuring costs under previously approved restructuring plans, including the 2018 restructuring program (the “2018 Program”) and the 2021 restructuring program (the “2021 Program”).
109
During the year ended December 31, 2025, the company recognized $2.2 million of income from a vendor refund related to the previous wind down of production of StrataGraft® (allogeneic cultured keratinocytes and dermal fibroblasts in murine collagen - dsat) (“StrataGraft”). No future charges are expected to be incurred under the previously approved restructuring plans. Restructuring costs and related credits under these restructuring plans were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Restructuring (credits) charges, net | $ | ( | $ | $ | ||||||||||||||||||||||
| Less: accelerated depreciation | ( | |||||||||||||||||||||||||
| Restructuring (credits) charges, net | $ | ( | $ | $ | $ | |||||||||||||||||||||
Restructuring (credits) charges, net by program from continuing operations were comprised of the following:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
2021 Program | $ | ( | $ | $ | $ | |||||||||||||||||||||
2018 Program | ||||||||||||||||||||||||||
| Total programs | ( | |||||||||||||||||||||||||
Less: non-cash charges, including accelerated depreciation | ( | |||||||||||||||||||||||||
| Restructuring charges, net | $ | ( | $ | $ | $ | |||||||||||||||||||||
The following table summarizes the restructuring reserves, which are included in accrued and other current liabilities on the Company's consolidated balance sheets.
| Dollars in millions | Severance | Contract Costs | Total | |||||||||||||||||
Balance as of December 30, 2022 (Predecessor) | $ | $ | $ | |||||||||||||||||
| Changes in estimate | ( | ( | ||||||||||||||||||
| Cash payments | ( | ( | ||||||||||||||||||
| Balance as of November 14, 2023 (Predecessor) | $ | $ | $ | |||||||||||||||||
Balance as of November 15, 2023 (Successor) and December 29, 2023 (Successor) | $ | $ | $ | |||||||||||||||||
Cash payments | ( | ( | ( | |||||||||||||||||
| Balance as of December 27, 2024 (Successor) | $ | $ | $ | |||||||||||||||||
| Changes in estimate | ( | ( | ||||||||||||||||||
Cash (payments)/receipts, net | ( | |||||||||||||||||||
| Balance as of December 31, 2025 (Successor) | $ | $ | $ | |||||||||||||||||
Cumulative restructuring charges, net of credits, for the 2021 Program were as follows as of December 31, 2025:
| 2021 Program | ||||||||
| Restructuring charges, net | $ | |||||||
110
| 9. | Income Taxes | ||||
The domestic and international components (1) of (Loss) income from continuing operations before income taxes were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Domestic | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
| International | ( | ( | ||||||||||||||||||||||||
| Total | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
(1) Domestic reflects Ireland.
Significant components(1) of income taxes related to continuing operations were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Current: | ||||||||||||||||||||||||||
| Domestic | $ | $ | ( | $ | ( | $ | ||||||||||||||||||||
| International | ||||||||||||||||||||||||||
| Current income tax (benefit) provision | $ | $ | ( | $ | ( | $ | ||||||||||||||||||||
| Deferred: | ||||||||||||||||||||||||||
| Domestic | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
| International | ( | ( | ||||||||||||||||||||||||
| Deferred income tax (benefit) provision | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
| Total | $ | ( | $ | $ | ( | $ | ( | |||||||||||||||||||
(1) Domestic reflects Ireland.
Refer to Note 6 for additional information of income taxes related to discontinued operations.
The domestic current income tax provision reflects a tax benefit of $3.7 million, zero , zero , and $2.9 million from using net operating loss (“NOL”) carryforwards for the year ended December 31, 2025, the year ended December 27, 2024, the period from November 15, 2023, through December 29, 2023 (Successor), and the period from December 31, 2022, through November 14, 2023 (Predecessor), respectively. The international current income tax provision reflects a tax benefit of $127.4 million, $55.1 million, $6.9 million, and $259.6 million from using NOL carryforwards for the year ended December 31, 2025, the year ended December 27, 2024, the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022, through November 14, 2023 (Predecessor), respectively.
The following table presents income taxes paid (net of refunds received) for the year ended December 31, 2025:
| Income Taxes Paid | |||||
Domestic (1) | $ | ||||
| International | |||||
| US state and local | |||||
| Japan | |||||
| Other | |||||
| Total | $ | ||||
(1) Domestic reflects Ireland.
During the year ended December 27, 2024, net cash payments for income taxes were $25.7 million. During the period November 15, 2023, through December 29, 2023 (Successor), and the period December 31, 2022, through November 14, 2023 (Predecessor), net cash payments and net cash refunds for income taxes were $0.3 million and $128.0 million, respectively. Included within the net cash refunds of $128.0 million were refunds of $141.6 million received as a result of the provisions in the Coronavirus Aid, Relief and Economic Security (“CARES”) Act.
111
The Company adopted ASU 2023-09 "Income Taxes (Topic 740): Improvements to Income Tax Disclosures" on a prospective basis beginning with the year ended December 31, 2025. The reconciliation between domestic income taxes at the statutory rate and the Company's provision for income taxes on continuing operations is as follows:
| Year Ended December 31, 2025 | |||||||||||
| Amount | Percent | ||||||||||
| Irish statutory tax rate | $ | ( | % | ||||||||
| Statutory tax rate differences within Ireland (1) | ( | % | |||||||||
| Foreign tax effects: | |||||||||||
| United States: | |||||||||||
| Foreign rate variance (Federal) | ( | % | |||||||||
| State and local income taxes, net of federal tax effect | ( | % | |||||||||
| Changes in valuation allowances | ( | % | |||||||||
| Nontaxable or nondeductible items: | |||||||||||
| Non-deductible compensation | ( | % | |||||||||
| Other nontaxable or nondeductible items | ( | % | |||||||||
| Other adjustments | ( | % | |||||||||
| Luxembourg: | |||||||||||
| Foreign rate variance | ( | % | |||||||||
| Changes in valuation allowances | ( | % | |||||||||
| Effects of the Business Combination and Par Health Separation | ( | % | |||||||||
| Other adjustments (2) | ( | % | |||||||||
| Other foreign jurisdictions | ( | % | |||||||||
| Changes in valuation allowance | ( | % | |||||||||
| Nontaxable or nondeductible items: | |||||||||||
| Non-deductible combination, integration, and other related expenses | ( | % | |||||||||
| Other nontaxable or nondeductible items | ( | % | |||||||||
| Changes in unrecognized tax benefits | ( | % | |||||||||
| Other adjustments, net | ( | % | |||||||||
| Effective tax rate | $ | ( | % | ||||||||
(1)Reflects the difference between the Irish statutory tax rate and the applicable Irish tax rates of 25 % for non-trading activity and 33 % for capital gains on certain types of income.
(2)The $40.5 million of expense is primarily related to Luxembourg impairment, recapture, and related expenses.
The reconciliation between domestic income taxes at the statutory rate and the Company's provision for income taxes on continuing operations, in accordance with the required disclosures prior to adoption of ASU 2023-09, is as follows:
| Successor | Predecessor | |||||||||||||||||||
| Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | ||||||||||||||||||
Provision (benefit) for income taxes at domestic statutory income tax rate (1) | $ | $ | ( | $ | ( | |||||||||||||||
| Adjustments to reconcile to income tax provision: | ||||||||||||||||||||
| Rate difference between domestic and international jurisdictions | ( | ( | ||||||||||||||||||
Permanently nondeductible and nontaxable items (2) | ( | |||||||||||||||||||
| Emergence | ( | |||||||||||||||||||
Legal entity reorganization (3) | ( | |||||||||||||||||||
| Reorganization items, net | ||||||||||||||||||||
| Other | ( | ( | ||||||||||||||||||
| Valuation allowances | ||||||||||||||||||||
| Provision (benefit) for income taxes | $ | $ | ( | $ | ( | |||||||||||||||
(1)The statutory tax rate reflects the Irish statutory tax rate of 12.5 %.
(2)For the year ended December 27, 2024 (Successor), the permanently nondeductible and nontaxable items of $18.3 million includes $6.1 million of expense related to nondeductible compensation.
(3)Associated unrecognized tax expense is netted within this line.
During the year ended December 27, 2024 (Successor), the rate difference between domestic and international jurisdictions was $21.3 million of tax expense, which was primarily related to $14.5 million of tax expense predominately related to pretax earnings in
112
various jurisdictions and $10.4 million of tax expense related to the Therakos Divestiture, partially offset by $3.6 million of tax benefit related to the remaining effects of adoption of fresh-start accounting as a result of emergence from the 2023 Bankruptcy Proceedings.
During the period November 15, 2023 through December 29, 2023 (Successor), the rate difference between domestic and international jurisdictions was $0.9 million of tax benefit, which was primarily related to $2.2 million of tax benefit attributable to inventory step-up amortization expense and $0.2 million of tax benefit attributable to accretion expense associated with the Company's settlement obligation, offset by $1.2 million of tax expense predominately attributable to the pretax earnings in various jurisdictions, and $0.3 million of tax expense attributable to amortization associated with the Company's debt.
During the period December 31, 2022 through November 14, 2023 (Predecessor), the rate difference between domestic and international jurisdictions was $5.2 million of a tax benefit, which was primarily related to $24.6 million of tax benefit predominately related to pretax earnings in various jurisdictions, $15.7 million of tax benefit related to liabilities management and separation costs, and $2.6 million of tax benefit related to non-restructuring impairment charges, offset by $37.7 million of tax expense attributable to reorganization items, net.
During the period December 31, 2022, through November 14, 2023 (Predecessor), the Company recognized a tax benefit of $95.0 million upon emergence from the 2023 Bankruptcy Proceedings. These impacts of emergence consist of a $241.2 million tax benefit related to the revaluation of net deferred tax assets as a result of fresh-start accounting, offset by $146.2 million of tax expense related to permanently nondeductible impacts on fair value adjustments.
As a result of the 2023 Plan, the Company recognized cancellation of debt income (“CODI”) on its indebtedness, resulting in the utilization of, and reduction to, certain of its tax losses and tax credits in the U.S. and Luxembourg. The emergence from the 2023 Bankruptcy Proceedings resulted in a change in ownership for purposes of IRC Section 382, causing the remaining U.S. tax losses, credits, and certain built in losses (“BILs”) to be limited under IRC Sections 382 and 383. The amount of the Company’s pre-ownership change U.S. NOLs and BILs that can be utilized generally cannot exceed an amount equal to the product of (a) the applicable federal long-term tax-exempt rate in effect on the date of the ownership change and (b) the value of its U.S. affiliate stock immediately prior to the implementation of each respective plan. The portion of deferred tax assets associated with the tax losses and credits that are limited under IRC Section 382 or 383, and that have a remote possibility of being utilized, have been written off. Refer to Note 4 for further information regarding the Company's income tax accounting policies.
On December 20, 2021, the Organization for Economic Co-operation and Development (“OECD”) released the Global Anti-Base Erosion (“GloBE”) Model Rules (“Pillar Two”) providing a legislative framework for the Income Inclusion Rule and the Under-Taxed Payment Rule (“UTPR”). Pillar Two is designed to ensure that large multinational enterprise groups pay a minimum level of tax on the income arising in each of the jurisdictions where they operate, principally creating a 15% minimum global effective tax rate. On December 15, 2022, the E.U. member states unanimously adopted a directive implementing the Pillar Two global minimum tax rules. On December 20, 2022, the OECD released three guidance documents related to Pillar Two. These documents included guidance on safe harbors and penalty relief and consultation papers on the GloBE Information Return and Tax Certainty for the GloBE rules. On January 15, 2025, the OECD issued additional administrative guidance aimed at streamlining the administration of the global minimum tax. A number of jurisdictions have transposed the directive into national legislation with the rules to be applicable for fiscal years beginning on or after December 31, 2023, with the exception of the UTPR which is to be applicable for fiscal years beginning on or after December 31, 2024. The Company's fiscal year end of December 29, 2023 allowed the Company to postpone the effective date of these law changes by one year. Based on the assessment for the period ending December 31, 2025 (Successor), certain transitional safe harbor relief applied to all jurisdictions and resulted in zero impact to income taxes.
The following table summarizes the activity related to the Company's unrecognized tax benefits, excluding interest:
| Year Ended, | |||||||||||
| December 31, 2025 | December 27, 2024 | ||||||||||
| Balance at beginning of period | $ | $ | |||||||||
| Additions related to current year tax positions | |||||||||||
| Additions related to prior period tax positions | |||||||||||
| Reductions related to prior period tax positions | ( | ||||||||||
| Additions or reductions related to business combinations, acquisitions, or divestitures | ( | ||||||||||
| Settlements | |||||||||||
| Lapse of statute of limitations | ( | ||||||||||
| Balance at end of period | $ | $ | |||||||||
113
Unrecognized tax benefits, excluding interest, were reported in the following consolidated balance sheet captions in the amounts shown:
| December 31, 2025 | December 27, 2024 | ||||||||||
| Deferred income tax asset | $ | $ | |||||||||
| Other income tax liabilities | |||||||||||
| $ | $ | ||||||||||
Total unrecognized tax benefits of $26.6 million as of the year ended December 31, 2025 (Successor), $30.9 million as of December 27, 2024 (Successor), and $30.1
The Company recorded a decrease to accrued interest and penalties of $2.1 million for the year ended December 31, 2025 (Successor). The Company recorded an increase to accrued interest and penalties of $1.7 million for the year ended December 27, 2024 (Successor). The Company recorded zero accrued interest and penalties for the period November 15, 2023, through December 29, 2023 (Successor), and an increase to accrued interest and penalties of $1.4 million during the period December 31, 2022, through November 14, 2023 (Predecessor). The total amount of accrued interest and penalties related to uncertain tax positions was $3.8 million and $5.9 million as of December 31, 2025 (Successor), and December 27, 2024 (Successor), respectively.
Certain of the Company's subsidiaries continue to be subject to examination by taxing authorities. The earliest open year subject to examination for U.S. federal and U.S. state purposes is 2018 and 2011, respectively. The earliest open year subject to examination in other jurisdictions, including Ireland, Japan, Luxembourg, Switzerland and the U.K. is 2014.
Income taxes payable, including uncertain tax positions and related interest accruals, were reported in the following consolidated balance sheet captions in the amounts shown:
| December 31, 2025 | December 27, 2024 | ||||||||||
| Accrued and other current liabilities | $ | $ | |||||||||
| Other income tax liabilities | |||||||||||
| $ | $ | ||||||||||
Tax receivables were included in the following consolidated balance sheet captions in the amounts shown:
| December 31, 2025 | December 27, 2024 | ||||||||||
| Prepaid expenses and other current assets | $ | $ | |||||||||
Deferred income taxes result from temporary differences between the amount of assets and liabilities recognized for financial reporting and tax purposes. The components of the net deferred tax asset (liability) at the end of each fiscal year were as follows:
| December 31, 2025 | December 27, 2024 | ||||||||||
| Deferred tax assets: | |||||||||||
| Tax loss and credit carryforward | $ | $ | |||||||||
| Capital tax loss carryforward and related assets | |||||||||||
| Intangible assets | |||||||||||
| Other | |||||||||||
| $ | $ | ||||||||||
| Deferred tax liabilities: | |||||||||||
| Other | $ | ( | $ | ( | |||||||
| $ | ( | $ | ( | ||||||||
| Net deferred tax asset before valuation allowances | $ | $ | |||||||||
| Valuation allowances | ( | ( | |||||||||
| Net deferred tax asset | $ | $ | |||||||||
114
The following table presents a reconciliation of the deferred tax asset valuation allowance:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Balance at beginning of period | $ | $ | $ | $ | ||||||||||||||||||||||
| Charged to costs and expenses | ( | ( | ( | |||||||||||||||||||||||
| Charged to Other Accounts | ( | ( | ( | |||||||||||||||||||||||
| Balance at the end of the period | $ | $ | $ | $ | ||||||||||||||||||||||
The net deferred tax asset before valuation allowances was $4,340.6 million as of December 31, 2025 (Successor), compared to $4,323.1 million as of December 27, 2024 (Successor). This increase consists of $119.1 million related to the Business Combination and the Separation and $25.8 million related to other operational activity, offset by $127.4 million related to tax attribute utilization primarily related to the effects of the Business Combination and Par Health Separation.
The deferred tax asset valuation allowances were $3,801.4 million and $3,756.0 million as of December 31, 2025 (Successor) and December 27, 2024 (Successor), respectively. The valuation allowances as of both December 31, 2025 (Successor) and December 27, 2024 (Successor) relate primarily to the uncertainty of the utilization of certain deferred tax assets, driven by domestic and international net operating and capital losses, credits, and intangible assets. The increase is primarily driven by an increase in capital tax loss carryforwards and related assets.
Deferred taxes were included in the following consolidated balance sheet captions in the amounts shown:
| December 31, 2025 | December 27, 2024 | ||||||||||
| Deferred income tax asset | $ | $ | |||||||||
| Other liabilities | ( | ||||||||||
| Net deferred tax asset | $ | $ | |||||||||
As of December 31, 2025 (Successor), the Company had $57.1 million of domestic NOL carryforwards measured at the applicable statutory rates, which have no expiration date. As of December 31, 2025 (Successor), the Company had approximately $3,345.6 million of NOL carryforwards in certain international jurisdictions measured at the applicable statutory rates, of which $1,230.7 million have no expiration and the remaining $2,114.9 million will expire in future years through 2041.
As of December 31, 2025 (Successor), the Company had approximately $312.9 million of domestic capital loss carryforwards measured at the applicable statutory rates, which have no expiration date. As of December 31, 2025 (Successor), the Company had $14.7 million of capital loss carryforwards in certain international jurisdictions measured at the applicable statutory rates, which will expire in 2030.
As of December 31, 2025 (Successor), the Company had $2.1 million of tax credits available to reduce future income taxes payable, in international jurisdictions, which will expire in future years through 2045.
As of December 31, 2025 (Successor), the Company had cumulative unremitted earnings of $1.9 million. Such amount is indefinitely reinvested and it is not practicable to determine the associated potential tax liability due to the complexity of the Company's legal entity structure as well as the timing, extent, and nature of any hypothetical realization.
| 10. | (Loss) income per Share | ||||
The weighted-average number of shares outstanding used in the computations of both basic and diluted (loss) income from continuing operation per share and discontinued operations per share were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Basic | ||||||||||||||||||||||||||
| Dilutive impact of restricted share units | ||||||||||||||||||||||||||
| Diluted | ||||||||||||||||||||||||||
115
The computation of diluted weighted-average shares outstanding for the years ended December 31, 2025 and December 27, 2024 (Successor), the period November 15, 2023 through December 29, 2023 (Successor), and the period December 31, 2022 through November 14, 2023 (Predecessor) excluded approximately 1.7 million, 1.7 million, 1.0 million and zero shares of Opioid CVRs for the Successor periods only and equity awards for all periods presented, respectively, because the effect would have been anti-dilutive.
As previously noted, pursuant to the CVR Termination Agreement, on November 10, 2025, the CVRs were cancelled and the CVR Agreement was terminated in exchange for a payment by the Company of $35.0 million to the Trust.
| 11. | Inventories | ||||
Inventories were comprised of the following at the end of each period:
| December 31, 2025 | December 27, 2024 | ||||||||||
Raw materials | $ | $ | |||||||||
Work in process | |||||||||||
Finished goods | |||||||||||
Inventories (1) | $ | $ | |||||||||
Inventories, long-term (1) (2) | $ | $ | |||||||||
(1)As of December 31, 2025 inventories and inventories, long-term include approximately $368.0 million and $413.0 million, respectively of remaining unamortized fair value step up. As of December 27, 2024, inventories and inventories, long-term include $176.4 million and $56.5 million respectively, of remaining unamortized fair value step up. The remaining unamortized fair value step up will be expensed as COGS in future periods as the inventory is sold.
(2)Inventories, long-term are included in other assets in the consolidated balance sheets at December 31, 2025, and December 27, 2024.
| 12. | Property, Plant and Equipment | ||||
The gross carrying amount and accumulated depreciation of property, plant and equipment were comprised of the following at the end of each period:
| December 31, 2025 | December 27, 2024 | ||||||||||
| Land | $ | $ | |||||||||
| Buildings | |||||||||||
| Capitalized software | |||||||||||
| Machinery and equipment | |||||||||||
| Construction in process | |||||||||||
| Property, plant and equipment, at cost | |||||||||||
| Less: accumulated depreciation | ( | ( | |||||||||
| Property, plant and equipment, net | $ | $ | |||||||||
Depreciation expense was as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Depreciation expense | $ | $ | $ | $ | ||||||||||||||||||||||
116
| 13. | Leases | ||||
Lease assets and liabilities related to the Company's operating leases are reported in the following consolidated balance sheet captions:
| December 31, 2025 | December 27, 2024 | ||||||||||
| $ | $ | ||||||||||
| Total lease liabilities | $ | $ | |||||||||
Dependent on the nature of the leased asset, lease expense is included within cost of sales or SG&A. The primary components of lease expense were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Lease expense: | ||||||||||||||||||||||||||
| Operating lease | $ | $ | $ | $ | ||||||||||||||||||||||
| Short-term lease | ||||||||||||||||||||||||||
| Variable lease | ||||||||||||||||||||||||||
| Total lease expense | $ | $ | $ | $ | ||||||||||||||||||||||
Lease terms and discount rates were as follows:
| December 31, 2025 | December 27, 2024 | ||||||||||
| Weighted-average remaining lease term (in years) - operating leases | |||||||||||
| Weighted-average discount rate - operating leases | % | % | |||||||||
Contractual maturities of operating lease liabilities as of December 31, 2025 were as follows:
| Fiscal 2026 | $ | ||||
| Fiscal 2027 | |||||
| Fiscal 2028 | |||||
| Fiscal 2029 | |||||
| Fiscal 2030 | |||||
| Thereafter | |||||
| Total lease payments | |||||
| Less: Interest | ( | ||||
| Present value of lease liabilities | $ | ||||
Other supplemental cash flow information related to leases were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Cash paid for amounts included in the measurement of lease liabilities: | ||||||||||||||||||||||||||
| Operating cash flows from operating leases | $ | $ | $ | $ | ||||||||||||||||||||||
| Lease assets obtained in exchange for lease obligations: | ||||||||||||||||||||||||||
| Operating leases | $ | $ | $ | $ | ||||||||||||||||||||||
117
| 14. | Goodwill and Intangible Assets | ||||
Goodwill
The following table presents the changes in the carrying amount of goodwill for the year ended December 31, 2025 (Successor).
| Total | ||||||||
| Balance as of December 27, 2024 (Successor) | $ | |||||||
| Goodwill additions (Note 5) | ||||||||
| Balance as of December 31, 2025 (Successor) | $ | |||||||
Intangible Assets
In connection with the Business Combination, the Company acquired $2,277.7 million of intangible assets. Refer to Note 5 for additional information on the Business Combination.
During the fourth quarter 2025, notification of changes in third-party reimbursement coding constituted a triggering event requiring the Company to evaluate the recoverability of a developed technology intangible asset with a carrying value of $173.8 million as of December 31, 2025 (Successor). The Company evaluated the recoverability of the asset by comparing the carrying value to the sum of the associated undiscounted future cash flows. Based on this analysis, the Company concluded that the carrying value was recoverable and no impairment charge was recorded. Although no impairment was recorded, the recoverability assessment is sensitive to key assumptions, including projected future sales, cost of sales and operating costs, and unfavorable changes in these assumptions or related facts and circumstances could lead to future impairment charges.
In connection with the Therakos Divestiture, during the fiscal year ended December 27, 2024 (Successor), the Company divested intangible assets with a carrying value of $108.7 million, which was comprised of $129.4 million gross carrying amount and $20.7 million of accumulated amortization. Refer to Note 6 for additional information on the Therakos Divestiture.
As part of fresh-start accounting, as of the 2023 Effective Date, the Company wrote-off the existing intangible assets and accumulated amortization of the Predecessor and recorded $361.1 million to reflect the fair value of intangible assets of the Successor. Refer to Note 3 for additional information on fresh-start accounting.
The Company recorded impairment charges totaling $50.1 million during the period December 31, 2022, through November 14, 2023 (Predecessor), related to StrataGraft due to lower than anticipated cash flows.
The gross carrying amount and accumulated amortization of intangible assets were comprised of the following at the end of each period:
| Successor | |||||||||||||||||||||||||||||||||||
| December 31, 2025 | December 27, 2024 | ||||||||||||||||||||||||||||||||||
| Gross Carrying Amount | Accumulated Amortization | Net Book Value | Gross Carrying Amount | Accumulated Amortization | Net Book Value | ||||||||||||||||||||||||||||||
| Amortizable: | |||||||||||||||||||||||||||||||||||
| Developed technology | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||
| Intangible assets, net | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||
Intangible asset amortization expense
The weighted-average amortization period for developed technology is 11.7 years. Intangible asset amortization expense was as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Amortization expense | $ | $ | $ | $ | ||||||||||||||||||||||
118
The estimated aggregate amortization expense on amortizable intangible assets as of December 31, 2025 (Successor) is expected to be as follows:
| Fiscal 2026 | $ | |||||||
| Fiscal 2027 | ||||||||
| Fiscal 2028 | ||||||||
| Fiscal 2029 | ||||||||
| Fiscal 2030 | ||||||||
| 15. | Debt | ||||
Debt was comprised of the following at the end of each period:
| December 31, 2025 | December 27, 2024 | ||||||||||||||||||||||||||||||||||
Principal | Carrying Value (2) | Unamortized Discount and Debt Issuance Costs | Principal | Carrying Value (2) | Unamortized Discount and Debt Issuance Costs | ||||||||||||||||||||||||||||||
| Current maturities of long-term debt: | |||||||||||||||||||||||||||||||||||
Term Loan due April 2031 (1) (2) | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||
Second-Out Takeback Term Loan due November 2028 (2) | |||||||||||||||||||||||||||||||||||
| Total current maturities of long-term debt | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||
| Long-term debt: | |||||||||||||||||||||||||||||||||||
Term Loan due April 2031 (1) (2) | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||
Second-Out Takeback Term Loan due November 2028 (2) | |||||||||||||||||||||||||||||||||||
| Receivables financing facility due December 2027 | |||||||||||||||||||||||||||||||||||
| Total long-term debt | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||
| Total debt | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||
(1)These instruments were assumed in connection with the Business Combination.
(2)Following the initial recognition at fair value, the Company accounted for its debt instruments utilizing the amortized cost method and amortizes the fair value premium to the principal amount over the term of the respective instruments. Such amortization expense is reflected as interest expense on the consolidated statement of operations. As of December 31, 2025, and December 27, 2024, the total unamortized premium within the consolidated balance sheets was $66.0 million and $50.0 million, respectively.
Takeback Debt
On November 14, 2023, the Company entered into Takeback Term Loans, consisting of approximately $229.4 million of First-Out Takeback Term Loans and approximately $642.0 million of Second-Out Takeback Term Loans. The Company also issued approximately $778.6 million in aggregate principal amount of Takeback Notes.
On December 6, 2024, the Company (i) mandatorily prepaid a portion of its Takeback Term Loans in an aggregate principal amount of approximately $474.1 million (of which approximately $227.1 million consisted of its First-Out Takeback Term Loans and approximately $247.0 million consisted of its Second-Out Takeback Loans) together with a payment of approximately $36.4 million in required makewhole premium (of which approximately $15.2 million was in respect of its First-Out Takeback Term Loans and approximately $21.2 million was in respect of its Second-Out Takeback Term Loans) and (ii) mandatorily redeemed $301.4 million in aggregate principal amount of Takeback Notes together with a payment of approximately $27.3 million in required makewhole premium. As a result of the mandatory prepayment, the Company recorded $19.7 million as a net loss on extinguishment of debt, comprised of the $63.7 million payment of the makewhole premium, offset by a $44.0 million gain to write-off certain unamortized premiums.
119
On August 1, 2025, in connection with the consummation of the Business Combination, the Company and its subsidiaries prepaid in full approximately $385.5 million in outstanding aggregate principal amount of the Second-Out Takeback Term Loans, constituting all of the remaining indebtedness then-outstanding under the then-existing credit agreement, together with accrued and unpaid interest thereon, as well as a payment of approximately $10.6 million in required makewhole premium.
Also in connection with the consummation of the Business Combination, on August 1, 2025, the Company and its subsidiaries redeemed in full approximately $477.2 million in outstanding principal amount of Takeback Notes, constituting all of the existing Takeback Notes then-outstanding under the then-existing indenture, for a redemption price equal to such outstanding principal amount, accrued and unpaid interest thereon and approximately $13.7 million in required makewhole premium. As a result of such prepayment, and redemption, the then-existing credit agreement was terminated, the then-existing indenture was discharged and all guarantees of, and liens securing, the obligations thereunder were released.
As a result of the mandatory prepayment, the Company recorded $15.9 million as a net gain on debt extinguishment, comprised of $40.2 million to write-off certain unamortized premiums net of debt issuance costs write-offs, offset by the $24.3 million payment of the makewhole premium.
Par Health Credit Agreement
On July 31, 2025, in connection with the consummation of the Business Combination, ST 2020, Inc. (“Par Health Parent”), a former wholly owned subsidiary of the Company, and MEH, Inc. (“Par Health Borrower”), a wholly owned subsidiary of Par Health Parent, entered into a credit agreement (as amended, modified or supplemented, the “Par Health Credit Agreement”) with the lenders named therein, Wilmington Savings Fund Society, FSB, as administrative agent and collateral agent, and OPY Credit Corp., as trading agent, providing for $1,350.0 million in aggregate principal amount of senior secured credit facilities (“Par Health Facilities”), comprising (i) a $1,200.0 million senior secured term loan facility with a maturity date of July 31, 2030 (“Par Health Term Facility”), and (ii) a $150.0 million senior secured revolving credit facility with a maturity date of July 31, 2030 (“Par Health Revolving Credit Facility”). Par Health Borrower borrowed $1,200.0 million under the Par Health Term Facility on August 1, 2025.
The Company capitalized $28.3 million of certain third-party debt issuance costs in connection with executing the Par Health Credit Agreement. Approximately $26.8 million of the capitalized costs were attributed to the Par Health Credit Agreement and were recorded as a direct reduction of long-term debt on the Company’s Consolidated Balance Sheet. Approximately $1.5 million of the capitalized costs were attributed to the Par Health Revolving Credit Facility and were recorded within other assets on the Company’s Consolidated Balance Sheet. These capitalized costs will be amortized into interest expense over the five-year term of the Par Health Credit Agreement.
The Par Health Credit Agreement was transferred to Par Health in connection with the Separation and all relevant obligations and remaining unamortized capitalized costs were derecognized from the Company’s balance sheet. As a result of the Separation, none of the Company or its subsidiaries continue to be borrowers or guarantors of the indebtedness under the Par Health Credit Agreement. All borrowers and guarantors in respect of such indebtedness are subsidiaries of Par Health.
Endo’s Indebtedness that Remained Outstanding after the Business Combination
After the consummation of the Business Combination, Endo’s existing indebtedness, consisting of its existing senior secured credit facilities and existing senior secured notes, remained outstanding as obligations of certain subsidiaries of the Company.
Endo Credit Facilities
Endo’s existing senior secured credit facilities consisting of (i) a revolving credit facility with a maturity date of April 23, 2029, and commitments equal to $400.0 million (“Revolving Credit Facility due April 2029”) and (ii) a term facility with a maturity date of April 23, 2031 with an outstanding principal balance of $1,489.0 million (“Term Facility due April 2031”, together with the Revolving Credit Facility due April 2029, the “Endo Credit Facilities”).
The Endo Credit Facilities are governed by that certain credit agreement, dated as of April 23, 2024, among Endo Finance Holdings LP (f/k/a Endo Finance Holdings, Inc.) (“Endo Borrower”), as borrower, Endo, as parent, the additional borrowers from time to time party thereto, the lenders from time to time party thereto and Goldman Sachs Bank USA, as administrative agent, collateral agent, issuing bank and swingline lender (as amended, modified or supplemented, the “Endo Credit Agreement”). The Endo Credit Agreement contains mandatory prepayment provisions, representations and warranties, affirmative and negative covenants and events of default (including as to certain change in control events) that, in each case, the Company believes to be customary for senior secured credit facilities of this type. The negative covenants include, among other things, indebtedness, fundamental changes, dispositions of property and assets (including sale-leaseback transactions), investments, restricted payments, restrictive agreements, transactions with affiliates, swap arrangements, amending subordinated debt documents, changes in fiscal year and changes in the nature of business. If the Endo Borrower draws more than 40 % of total available credit under its Revolving Credit Facility due April 2029 (other than (a) undrawn letters of credit in an amount not to exceed $20.0 million and (b) cash collateralized or backstopped letters of credit), Endo will be required to comply with a maximum first lien net leverage ratio not to exceed 6.10 to 1.00.
Borrowings bear interest, at the borrower’s election, based on: (x) under the Revolving Credit Facility due April 2029, (i) the alternate base rate; (ii) the Canadian prime rate; (iii) Term SOFR (as defined in the Endo Credit Agreement); or (iv) Adjusted Term
120
CORRA (as defined in the Endo Credit Agreement) and (y) under the Term Facility due April 2031, (i) the alternate base rate or (ii) Term SOFR (as defined in the Endo Credit Agreement), in each case, plus the applicable margin; provided that Term SOFR and Adjusted Term CORRA shall not be less than, with respect to loans under the Revolving Credit Facility due April 2029, — % per annum, and with respect to loans under the Term Facility due April 2031, 0.50 %. The applicable margins are based upon a first lien net leverage ratio as set forth in the Endo Credit Agreement, which range from: (i) for loans under the Revolving Credit Facility due April 2029 based on (x) Term SOFR or Adjusted Term CORRA, 3.00 % to 3.50 % and (y) alternate base rate or Canadian prime rate, 2.00 % to 2.50 %; and (ii) for loans under the Term Facility due April 2031 based on (x) Term SOFR, 3.75 % to 4.00 % and (y) alternate base rate, 2.75 % to 3.00 %. The Endo Borrower is also required to pay quarterly in arrears a commitment fee on undrawn commitments under the Revolving Credit Facility due 2029 at a per annum rate, based upon a first lien net leverage ratio, of 0.25 % to 0.50 %.
As of December 31, 2025, the aggregate outstanding principal amount of loans under the Term Facility due April 2031 was $1,481.3 million and approximately $396.0 million of capacity under the Revolving Credit Facility due April 2029 was undrawn and available to the Endo Borrower, net of outstanding standby letters of credit.
The obligations under the Endo Credit Agreement are guaranteed by Endo and certain of the Company’s other subsidiaries (“Guarantors”) and secured by a lien on substantially all the assets (with certain exceptions) of the Endo Borrower and the Guarantors in accordance with the terms of the Endo Credit Agreement, the other related security documents and that certain first lien intercreditor agreement, dated as of April 23, 2024, among the notes collateral agent for the 8.50 % Senior Secured Notes due April 2031 (as defined below), the collateral agent for the Endo Credit Agreement, the Endo Borrower, the Guarantors from time to time party thereto and the other agents from time to time party thereto (as amended, modified or supplemented, the “Intercreditor Agreement”).
Pursuant to the Intercreditor Agreement, with respect to any Shared Collateral (as defined in the Intercreditor Agreement) proceeds received after the occurrence, and during the continuance, of an event of default under the applicable secured debt documents and certain other circumstances, holders of the obligations under the Revolving Credit Facility due April 2029 and certain specified cash management and hedging obligations secured in connection therewith (the “Revolving Facility Obligations”), shall be paid prior to the obligations under the Term Facility due April 2031 and the obligations under the Indenture for the 8.50 % Senior Secured Notes due April 2031. Moreover, the collateral agent for the Endo Credit Agreement is the controlling agent under the Intercreditor Agreement and, prior to the discharge of the Revolving Facility Obligations, will take direction from lenders holding a majority of the commitments under the Revolving Credit Facility due April 2029 in respect of the exercise of rights and remedies, including in any insolvency proceeding, consent to debtor-in-possession financing, sale of collateral, use of cash collateral, adequate protection and other customary bankruptcy provisions.
The Company did not file this Annual Report on Form 10-K (“Annual Report”) by its initial due date of March 31, 2026, which resulted in its failure to comply with the covenant in the Endo Credit Agreement that required the Company to deliver audited annual financial statements by that date. The Company’s delivery of the audited financial statements that are contained in this Annual Report will cure the aforementioned covenant default.
Endo’s 8.50 % Senior Secured Notes due April 2031
As of December 31, 2025, Endo’s 8.50 % senior secured notes with a maturity date of April 15, 2031 (the 8.50 % Senior Secured Notes due April 2031”), had an outstanding principal balance of $1,000.0 million. The 8.50 % Senior Secured Notes due April 2031 are obligations of the Endo Borrower (and a subsidiary thereof), are guaranteed by the Guarantors and are secured by a lien on substantially all the assets (with certain exceptions) of the Endo Borrower and the Guarantors in accordance with the terms of the Indenture (as defined below), the other related security documents and the Intercreditor Agreement. The 8.50 % Senior Secured Notes due April 2031 will mature on April 15, 2031, subject to earlier repurchase or redemption in accordance with the terms of the Indenture (as defined below), and bear interest at 8.50 % per annum, payable semi-annually in cash in arrears on April 15 and October 15 of each year, commencing on October 15, 2024.
At any time prior to April 15, 2027, the 8.50 % Senior Secured Notes due April 2031 are redeemable by the Endo Borrower, in whole or in part, at a redemption price equal to 100.00 % of the principal amount of the 8.50 % Senior Secured Notes due April 2031 redeemed, plus the greater of 1.0 % of the principal amount of the 8.50 % Senior Secured Notes due April 2031 redeemed and a makewhole premium, plus accrued and unpaid interest, if any, to, but not including, the date of redemption.
At any time prior to April 15, 2027, the Endo Borrower may redeem up to 10.00 % of the original aggregate principal amount of the 8.50 % Senior Secured Notes due April 2031 during each twelve-month period commencing with April 23, 2024 at a redemption price equal to 103.00 % of the principal amount thereof, plus accrued and unpaid interest, if any, to, but not including, the date of redemption.
At any time prior to April 15, 2027, the Endo Borrower may redeem up to 40.00 % of the aggregate principal amount of the 8.50 % Senior Secured Notes due April 2031 with the net cash proceeds from specified equity offerings at a redemption price equal to 108.50 % of the aggregate principal amount of the 8.50 % Senior Secured Notes due April 2031 redeemed, plus accrued and unpaid interest, if any, to, but not including, the date of redemption.
121
Upon the occurrence of certain change of control events, the Endo Borrower must offer to repurchase the 8.50 % Senior Secured Notes due April 2031 at 101.00 % of their aggregate principal amount, plus accrued and unpaid interest, if any, to, but not including, the date of purchase.
On or after April 15, 2027, the Endo Borrower may on any one or more occasions redeem all or part of the 8.50 % Senior Secured Notes due April 2031 at a redemption price expressed as a percentage of the principal amount thereof, which percentages are 104.25 % for the twelve-month period beginning on April 15, 2027; 102.13 % for the twelve-month period beginning on April 15, 2028; and 100.00 % beginning on April 15, 2029 and thereafter, in each case, plus accrued and unpaid interest, if any, to, but not including, the date of redemption.
The 8.50 % Senior Secured Notes due April 2031 and the guarantees thereof were issued pursuant to an indenture by and among the Endo Borrower, Endo, the subsidiary guarantors from time to time party thereto and Computershare Trust Company, National Association, as trustee and notes collateral agent (as amended, modified or supplemented, the “Indenture”). The Indenture contains customary events of default, as well as covenants that, among other things, restrict Endo’s ability and the ability of its restricted subsidiaries to incur certain additional indebtedness and issue preferred stock, make certain dividend payments, distributions, investments and other restricted payments, sell certain assets, agree to any restrictions on the ability of restricted subsidiaries to make payments to the Endo Borrower, create certain liens, merge, consolidate, or sell all or substantially all of Endo’s or any restricted subsidiary’s assets, or enter into certain transactions with affiliates. These covenants are subject to a number of important exceptions and qualifications, including the suspension of certain of these covenants upon the 8.50 % Senior Secured Notes due April 2031 receiving investment grade credit ratings.
The Company did not file this Annual Report by its initial due date of March 31, 2026, which resulted in its failure to comply with the covenant in the Indenture that required the Company to deliver audited annual financial statements by that date. The Company’s delivery of the audited financial statements that are contained in this Annual Report will cure the aforementioned covenant default.
Applicable interest rate
As of December 31, 2025, the applicable interest rate on the Company's debt instruments were as follows:
| Applicable interest rate | ||||||||
| Term Loan due April 2031 | % | |||||||
The Company's stated long-term debt principal repayment obligations as of December 31, 2025 are as follows:
| 2026 | $ | |||||||
| 2027 | ||||||||
| 2028 | ||||||||
| 2029 | ||||||||
| 2030 | ||||||||
| 16. | Retirement Plans | ||||
Defined Contribution Retirement Plans
122
| 17. | Equity | ||||
On July 31, 2025, prior to the completion of the Business Combination, the Company adopted new Articles of Association, which among other things, provided that the authorized share capital of the Company was $10.0 million and €25,000 , divided into 500,000,000 ordinary shares, par value $0.01 per share, 500,000,000 preferred shares, par value $0.01 per share, and 25,000 ordinary A shares, par value €1.00 per share. The preferred shares may be issued with such rights as the Board may determine.
On July 31, 2025, pursuant to the terms of the Transaction Agreement, at the effective time of the Business Combination (“Merger Effective Time”), each share of common stock of Endo issued and outstanding as of immediately prior to the Merger Effective Time, other than the shares of Endo common stock owned by Endo, any Endo subsidiary, the Company, Merger Sub or any of the Company or their respective subsidiaries, was cancelled and converted into the right to receive 0.2575 of a Company ordinary share and approximately $1.31 in cash, without interest and subject to applicable withholding. Former holders of Endo common stock received cash in lieu of any fractional Company ordinary shares they would otherwise have been entitled to receive. The issuance of Company ordinary shares in connection with the Business Combination was registered under the Securities Act, pursuant to the Company’s registration statement on Form S-4 filed with the SEC on April 23, 2025, as amended.
Endo’s common stock outstanding immediately prior to the Business Combination was 76,313,462 shares, which resulted in the issuance of 19,650,663 Company ordinary shares to former holders of Endo common stock.
On October 8, 2025, the Company’s shareholders approved an ordinary resolution to subdivide and increase the Company’s authorized share capital to $3,005.0 million and €25,000 divided into 500 million ordinary shares of $0.01 each, 3 trillion preferred shares of $0.001 each and 25,000 ordinary A shares of €1.00 each. On October 10, 2025, the Company declared the issuance of 45,564 preferred shares for each outstanding ordinary share to the Company’s shareholders of record as of the close of business on October 8, 2025. As this involves the issuance of equity shares, the Company accounted for this as a dividend-in-kind and was recorded at the fair value of the shares distributed. The issuance was intended to facilitate the distribution of Par Health shares. As such, the Company determined that the fair value of the issued preferred shares is de minimis and thus no distribution was recorded on the issuance date. Under the Irish law, the preferred shares were credited as paid up pursuant to a capitalization of a merger reserve account.
On November 10, 2025, at 12:01 a.m. (Eastern Time in the United States), the Company completed the Separation, which was implemented by way of a redemption of all of the Company’s issued and outstanding preferred shares, upon which the preferred shares were automatically cancelled and as such are no longer outstanding. In connection with the Redemption and pursuant to Irish law, the Company allocated the right to receive 39,421,398 shares of Par Health Common Stock to certain holders of record of preferred shares as of October 27, 2025. Refer to Note 6 for additional information on the Redemption.
| 18. | Share-Based Compensation | ||||
Share-Based Compensation Plans
The Keenova Therapeutics plc 2025 Stock and Incentive Plan, effective August 13, 2025 (the “2025 Stock and Incentive Plan”), provides for the award of stock options, stock appreciation rights, long-term performance awards and other share-based awards (collectively, “Awards”). The maximum number of ordinary shares to be issued as Awards under the 2025 Stock and Incentive Plan as of the effective date of the 2025 Stock and Incentive Plan was 6,936,576 (which reflects the adjustment for the Separation), as may be further adjusted from time to time pursuant to the terms of the 2025 Stock and Incentive Plan.
The 2025 Stock and Incentive Plan supersedes and replaces the Mallinckrodt Pharmaceuticals Stock and Incentive Plan, as amended and restated effective February 2, 2024 (the “2024 Stock and Incentive Plan”). The 2024 Stock and Incentive Plan similarly provided for the grant of Awards; however, following the effectiveness of the 2025 Stock and Incentive Plan, no further Awards will be granted under the 2024 Stock and Incentive Plan.
In connection with the Business Combination, the Company assumed the Endo, Inc. 2024 Stock Incentive Plan (“Endo Plan”). Following the effectiveness of the 2025 Stock and Incentive Plan, no further Awards will be granted under the Endo Plan.
On November 15, 2023, all outstanding equity-based awards under the previous Mallinckrodt Pharmaceuticals Stock and Incentive Plan, as amended and restated effective February 23, 2022, were automatically cancelled without consideration. No awards were granted during the period November 15, 2023, through December 29, 2023 (Successor).
123
Share-Based Compensation Expense
Total share-based compensation cost was as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 (1) | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Share-based compensation expense | $ | $ | $ | $ | ||||||||||||||||||||||
| Discontinued operations (Note 5) | ( | ( | ( | |||||||||||||||||||||||
| Share-based compensation expense - continuing operations | $ | $ | $ | $ | ||||||||||||||||||||||
(1)No awards were granted during the period November 15, 2023 through December 29, 2023 (Successor).
These amounts are generally included within SG&A expenses in the consolidated statements of operations. The Company recognized zero
Awards
Restricted share units (“RSUs”). Recipients of RSUs have no voting rights and receive dividend-equivalent units that vest concurrently with the related shares. RSUs generally vest in equal annual installments over a period of three years . Restrictions on RSUs lapse upon vesting over time, which may be accelerated in certain circumstances pursuant to the terms of the applicable award agreements and the 2025 Stock and Incentive Plan, 2024 Stock and Incentive Plan or the Endo Plan, as applicable. The fair value of RSUs are amortized on a graded basis over the respective vesting period of each award. As of the 2023 Effective Date, the Company’s ordinary shares were no longer traded on an active market. Accordingly, the fair value of share-based awards granted after the 2023 Effective Date requires the valuation of the Company’s equity utilizing the application of significant estimates, assumptions, and judgments, as further described in Note 4.
A portion of the RSUs granted during the fiscal year ended December 27, 2024 (Successor), could have been settled in shares and were classified as equity-based awards, and a portion of the RSUs had the ability to be settled in either shares or cash at the holder’s discretion and were classified as liability-based awards.
RSU activity was as follows:
| Shares | Weighted-Average Grant-Date Fair Value | ||||||||||
Non-vested as of December 30, 2022 (Predecessor) | |||||||||||
| Granted | |||||||||||
| Vested | ( | ||||||||||
| Forfeited/Cancelled | ( | ||||||||||
Non-vested as of November 14, 2023 (Predecessor) | |||||||||||
Non-vested as of December 29, 2023 (Successor) | |||||||||||
| Granted | |||||||||||
| Forfeited/Cancelled | ( | ||||||||||
Non-vested as of December 27, 2024 (Successor) | |||||||||||
| Granted | |||||||||||
PSU Conversions to RSUs | |||||||||||
| Endo RSU Award Conversions | |||||||||||
| Endo PSU Award Conversions | |||||||||||
Accelerated Vestings as a result of the Business Combination | ( | ||||||||||
| Net Separation Impact | N/A | ||||||||||
| Vested | ( | ||||||||||
| Forfeited/Cancelled | ( | ||||||||||
Non-vested as of December 31, 2025 (Successor) | |||||||||||
Granted. During the year-ended December 31, 2025 (Successor), certain executives and non-employee directors were granted awards, resulting in 191,217 RSUs being awarded with a grant date value of 90.50 per share. The total fair value of RSU awards granted during the year ended December 31, 2025 (Successor), was $17.3 million.
124
As of December 31, 2025 (Successor), there was $27.9 million of total unrecognized compensation costs related to non-vested RSUs, which is expected to be recognized over a weighted-average period of 1.2 years.
Performance Stock Unit (“PSU”) Conversions to RSUs. The Business Combination qualified as a “Qualifying Significant Event” that was not also a change of control under the terms of the PSUs granted under the 2024 Stock and Incentive Plan, which were held by certain then-existing employees and non-employee directors of the Company. Therefore, as a result of the Business Combination, such PSUs were converted into RSUs that will fully vest on December 25, 2026. The remaining fair value of such awards will be recognized as expense over the requisite service period of the award.
Endo RSU award Conversions. As a result of the Business Combination, each outstanding RSU award in respect of Endo common stock that was subject only to time-based vesting requirements (an “Endo RSU award”) and that was held by an employee or Endo or a subsidiary of Endo, was assumed by the Company and converted into a RSU award in respect of a number of Company ordinary shares equal to (i) the total number of Endo common stock underlying such Endo RSU award as of immediately prior to the Merger Effective Time multiplied by (ii) the sum of (x) the Per Share Stock Consideration plus (y) the quotient obtained by dividing the Per Share Cash Consideration by the per share price of Company ordinary shares as specified in the Transaction Agreement (“Company Per Share Price”). Each assumed Endo RSU award continues to have, and is subject to, the same terms and conditions (including vesting schedules) that applied to the corresponding Endo RSU award immediately prior to the Merger Effective Time.
Endo PSU award Conversions. As a result of the Business Combination, each RSU award in respect of Endo common stock that was subject, in whole or in part, to performance-based vesting conditions (an “Endo PSU award”) was assumed by the Company and converted into an RSU award in respect of a number of Company ordinary shares equal to the product of (i) the total number of Endo common stock underlying such Endo PSU award as of immediately prior to the Merger Effective Time, assuming performance goals are achieved based on target performance, multiplied by (ii) the sum of (x) the Per Share Stock Consideration plus (y) the quotient obtained by dividing the per share cash consideration by the Company Per Share Price. Each Endo PSU award otherwise continues to be subject to the same terms and conditions (including vesting) that applied to the corresponding Endo PSU award immediately prior to the Merger Effective Time.
Accelerated Vestings as a result of the Business Combination. In accordance with the terms of their respective awards, certain non-employee directors and executives vested following the consummation of the Business Combination.
Net Separation Impact. Pursuant to the terms of the employee matters agreement entered into in connection with the Separation, the outstanding RSUs held by executives whose employment was being transferred to Par Health were assumed by Par Health and converted into RSUs of Par Health. Each RSU award in respect of Company ordinary shares that was outstanding as of immediately prior to the effective time of the Separation was converted into a RSU of Par Health, generally subject to the same terms and conditions (including with respect to vesting and settlement) after the effective time of the Separation as were applicable to such RSU award immediately prior to the effective time of the Separation, provided, however, that from and after the effective time of the Separation, the number of ordinary shares subject to the corresponding RSU award immediately prior to the effective time of the Separation, multiplied by the quotient obtained by dividing (a) the Pre-Separation Mallinckrodt stock value by (b) the post-Separation Keenova stock value. Prior to the Separation, RSU amounts included in the table above include awards granted, vested and forfeited that relate to these executives whose employment was being transferred to Par Health. RSU share counts for previously presented periods include the disclosure of awards granted, vested and forfeited that relate to these Par Health employees that are no longer with the Company. The total amount of shares surrendered upon Separation was 125,876 .
Additionally, pursuant to the terms of the employee matters agreement entered into in connection with the Separation, RSU awards held by executives and non-employee directors who remained with the Company after the Separation that were outstanding immediately prior to the effective time of the Separation remained outstanding. Such RSU awards continued to be subject to the same terms and conditions (including with respect to vesting and settlement) after the effective time of the Separation as were applicable to such RSU award immediately prior to the effective time of the Separation, except that the number of ordinary shares subject to the RSU award was adjusted such that the number of ordinary shares subject to the RSU award was determined by multiplying (a) the number of ordinary shares subject to the RSU award immediately prior to the effective time of the Separation and (b) the quotient obtained by dividing the Pre-Separation Parent Stock Value by the Post-Separation Parent Stock Value (both as defined in the employee matters agreement). The number of additional RSUs issued in connection with such adjustment was 147,339 .
Performance share units. Similar to recipients of RSUs, recipients of PSUs have no voting rights and receive dividend-equivalent units. The accounting policy election to recognize the expense associated with the grant-date fair value of PSUs, which were deemed probable to vest, is further discussed in Note 4.
For the awards granted during the fiscal year ended December 27, 2024 (Successor), the determination of fair value required the valuation of the Company’s equity utilizing the application of significant estimates, assumptions, and judgments, as further described in Note 4.
125
For the Predecessor PSU awards, the Company generally used the Monte Carlo model to estimate the probability of satisfying the performance criteria and the resulting fair value. The assumptions used in the Monte Carlo model for PSUs granted during the period December 31, 2022, through November 14, 2023 (Predecessor), were as follows:
Predecessor | |||||
Period from December 31, 2022 through November 14, 2023 (1) | |||||
| Expected stock price volatility | % | ||||
| Peer group stock price volatility | |||||
| Correlation of returns | |||||
(1)These PSU awards were subsequently cancelled upon the emergence from the 2023 Bankruptcy Proceedings.
A portion of the PSUs granted during the period December 31, 2022 to November 14, 2023 (Predecessor) could have been settled in shares and were classified as equity-based awards, and a portion of the PSUs had the ability to be settled in either shares or cash, at the holder’s discretion and were classified as liability-based awards. The vesting of these PSUs was based on various performance metrics and relative total shareholder return (total shareholder return for the Company as compared to total shareholder return of the PSU peer group). The fair value of the liability-based awards was measured quarterly and based on the Company's performance.
PSU activity was as follows (1):
| Shares | Weighted-Average Grant-Date Fair Value | ||||||||||
Non-vested as of December 30,2022 (Predecessor) | |||||||||||
Granted | |||||||||||
Forfeited | ( | ||||||||||
Non-vested as of November 14, 2023 (Predecessor) | |||||||||||
Non-vested as of December 29, 2023 (Successor) | |||||||||||
Granted | |||||||||||
Non-vested as of December 27, 2024 (Successor) | |||||||||||
| Granted | |||||||||||
PSU Conversions to RSUs | ( | ||||||||||
| Forfeited | |||||||||||
Non-vested as of December 31, 2025 (Successor) | |||||||||||
PSU Conversions. Immediately prior to the Business Combination, the performance metrics for the PSUs granted under the 2024 Stock and Incentive Plan were based on a realized value of the Company, as defined by the applicable award agreements. As discussed above, as a result of the Business Combination, such PSUs were converted into RSUs.
| 19. | Guarantees | ||||
In disposing of assets or businesses, the Company has from time to time provided representations, warranties and indemnities to cover various risks and liabilities, including unknown damage to assets, environmental risks involved in the sale of real estate, liability to investigate and remediate environmental contamination at waste disposal sites and manufacturing facilities, and unidentified tax liabilities related to periods prior to disposition. The Company assesses the probability of potential liabilities related to such representations, warranties and indemnities and adjusts potential liabilities as a result of changes in facts and circumstances. As of December 31, 2025, the Company believes the likelihood of payment is remote and the fair value of such guarantees is not material.
Pursuant to the terms of the separation agreement with Par Health, each of the Company and Par Health are required to use commercially reasonable efforts to cause the removal of the other party and its respective subsidiaries as guarantor of, or obligor for, certain non-indebtedness obligations following the separation. To the extent that the Company cannot be released from any such guarantee or obligation, Par Health is required to indemnify the Company for any losses, costs, or exposure arising from such guarantee. Certain of these guarantees require that the Company maintain cash collateral which totals $37.9 million as of December 31, 2025 and which is classified as restricted cash on the Company’s consolidated balance sheet. As of December 31, 2025, the Company believes the likelihood of payment is remote and the fair value of such guarantees is not material.
126
In connection with the sale of the Specialty Chemical business (formerly known as Mallinckrodt Baker) in fiscal 2010, the Company agreed to indemnify the purchaser with respect to various matters, including certain environmental, health, safety, tax and other matters. The indemnification obligations relating to certain environmental, health and safety matters have a term of 17 years from the sale, while some of the other indemnification obligations have an indefinite term. The liability relating to all of these indemnification obligations was governed by a contract that was rejected as part of the Chapter 11 proceedings and Irish examinership proceedings on June 16, 2022 (“2020 Bankruptcy Proceedings”) and is no longer a liability subsequent to June 16, 2022 (“2020 Effective Date”). The Company was required to pay $30.0 million into an escrow account as collateral to the purchaser. The contract governing the escrow account was assumed in the 2020 Bankruptcy Proceedings. As of December 31, 2025, and December 27, 2024, $22.2 million and $21.3 million, respectively, remained in restricted cash, included in Other assets on the consolidated balance sheets. As of December 31, 2025, the Company does not expect to make future payments related to these indemnification obligations.
| 20. | Commitments and Contingencies | ||||
Legal Proceedings and Investigations
The Company is subject to various legal proceedings and claims, including government investigations, environmental matters, product liability matters, patent infringement claims, antitrust matters, securities class action lawsuits, personal injury claims, employment disputes, contractual and other commercial disputes, and other legal proceedings, all in the ordinary course of business, including those described below. Although it is not feasible to predict the outcome of these matters, the Company believes, unless otherwise indicated below, given the information currently available, that the ultimate resolution of any particular matter, or matters that have the same legal or factual issues, will not have a material adverse effect on its financial condition, results of operations and cash flows.
Government Proceedings
U.S. Attorney's Office Subpoena W.D. Va. In March 2025, Endo USA, Inc. (“Endo USA”) received a subpoena duces tecum issued by the WDVA USAO requesting documents and information from 1996 through the present related to any interactions by Endo USA, its affiliates, predecessors or other related parties with pharmacy benefit managers, including (i) remuneration provided, (ii) negotiation of rebates, (iii) communications regarding the prescription, administration or payment for opioid medications, and (iv) communications regarding the safety or efficacy of opioid medications. Endo USA received two additional subpoenas from WDVA USAO seeking related material in April 2025. Endo USA has responded to the subpoenas and is cooperating with the investigation. The Company cannot predict the eventual scope, duration or outcome of this matter at this time.
U.S. Department of Justice Consumer Protection Branch Subpoena. In April 2025, Endo USA received subpoenas from the U.S. Department of Justice’s Consumer Protection Branch seeking documents and information, if any, related to the marketing and promotion of Supprelin® LA from January 2020 through the present, for certain unapproved uses, including transgender care and gender dysphoria. Endo USA is cooperating with the investigation and is in the process of responding to the subpoenas. The Company cannot predict the eventual scope, duration or outcome of the investigation at this time.
U.S. Department of Justice Civil Investigative Demand. In October 2025, Endo received a Civil Investigative Demand (“CID”) from the U.S. Department of Justice under the False Claims Act seeking documents and information from January 2020 through the present. The CID concerns allegations that (1) Endo violated the False Claims Act by paying kickbacks to induce the purchase of Xiaflex®, in violation of the Anti-Kickback Statute and (2) Endo inflated reimbursement rates for Xiaflex® by excluding applicable price concessions from average sales price reports submitted to the Centers for Medicare & Medicaid Services. Endo is cooperating with the investigation and is in the process of responding to the CID. The Company cannot predict the eventual scope, duration or outcome of this matter at this time.
Securities Litigation
Putative Class Action Securities Litigation (Continental General). On July 7, 2023, a putative class action lawsuit was filed against the Company, its Chief Executive Officer (“CEO”) Sigurdur Olafsson, its former Chief Financial Officer (“CFO”) Bryan Reasons, and the former Chair of the Board, Paul Bisaro, in the U.S. District Court for the District of New Jersey(“DNJ”), captioned Continental General Insurance Company and Percy Rockdale, LLC v. Mallinckrodt plc et al., No. 23-cv-03662. The complaint purports to be brought on behalf of all persons who purchased or otherwise acquired the Company’s securities between June 17, 2022 and June 14, 2023. The lawsuit generally alleges that the defendants made false and misleading statements in violation of Sections 10(b) and 20(a) of the Securities Exchange Act of 1934, as amended (“Exchange Act”) and Rule 10b-5 promulgated thereunder related to the Company’s business, operations, and prospects, including its financial strength, its ability to timely make certain payments related to the Company’s opioid-related litigation settlement and the risk of additional filings for bankruptcy protection. The lawsuit seeks monetary damages in an unspecified amount. A lead plaintiff was designated by the court in September 2023 and in December 2023, an amended complaint was filed by the lead plaintiff against Olafsson, Reasons, and Bisaro (“Continental Individual Defendants”). As to the Company, any liability to the plaintiffs in this matter was discharged upon emergence from the 2023 Bankruptcy Proceedings. The Company assumed the obligation to defend and indemnify the Continental Individual Defendants. In September 2024, the court denied the Continental Individual Defendants' motion to dismiss. The Continental Individual Defendants answered the amended complaint in October 2024. In April 2025, the parties reached an agreement in principle to resolve all claims in
127
this matter for a settlement payment of $5.5 million, which was funded in part by the Company and in part by the Company’s insurance carriers. The Company paid the settlement amount during the three months ended June 27, 2025. The DNJ granted final approval of the settlement in December 2025.
Alta Fundamental. In September 2024, a lawsuit was filed against the Company’s CEO Sigurdur Olafsson, its former CFO Bryan Reasons, the former Chair of the Board Paul Bisaro, its former Chief Strategy and Restructuring Officer Jason Goodson, and its former Global Controller and Chief Investor Relations Officer Daniel Speciale (“Alta Individual Defendants”), in the U.S. District Court for the DNJ, captioned Alta Fundamental Advisors, LLC et al. v. Bisaro et al., No. 24-cv-09245. Plaintiffs allege similar facts to those in the Continental General action, and like in that action, the Alta Fundamental lawsuit generally alleges that the defendants made false and misleading statements related to the Company’s business, operations, and prospects, including its financial strength, its ability to timely make certain payments related to the Company’s opioid-related litigation settlement and the risk of additional filings for bankruptcy protection. The lawsuit alleges claims under Sections 10(b), 18(a), and 20(a) of the Exchange Act, Rule 10b-5 promulgated thereunder, and the New Jersey Uniform Securities Act, as well as common law fraud and negligent misrepresentation. The Company assumed the obligation to defend and indemnify the Alta Individual Defendants. The lawsuit seeks monetary damages in an unspecified amount. In June 2025, the court granted in part and denied in part the Alta Individual Defendants' motion to dismiss. Certain of the Alta Individual Defendants filed a motion for reconsideration as to the court’s partial denial. In February 2026, the court granted the motion for reconsideration and the Individual Defendants Goodson and Speciale were dismissed from this case. The Alta Individual Defendants who did not file a motion for reconsideration answered the complaint in August 2025. The remaining parties are now in discovery and have been engaging in settlement negotiations, with any resulting settlement expected to be funded by the Company’s insurance carriers and not material to the Company’s financial condition, results of operations or cash flows.
Putative Class Action Securities Litigation (Strougo). In July 2019, a putative class action lawsuit was filed against the Company, its former CEO Mark C. Trudeau, its former CFO Bryan M. Reasons, its former Interim CFO George A. Kegler and its former CFO Matthew K. Harbaugh, in the U.S. District Court for the Southern District of New York, captioned Barbara Strougo v. Mallinckrodt plc, et al. The complaint purports to be brought on behalf of all persons who purchased or otherwise acquired the Company’s securities between February 28, 2018 and July 16, 2019. The lawsuit generally alleges that the defendants made false and/or misleading statements in violation of Sections 10(b) and 20(a) of the Exchange Act and Rule 10b-5 promulgated thereunder related to the Company's clinical study designed to assess the efficacy and safety of its Acthar Gel in patients with amyotrophic lateral sclerosis. The lawsuit seeks monetary damages in an unspecified amount. In July 2020, the court approved the transfer of the case to the U.S. District Court for the District of New Jersey. In August 2020, an amended complaint was filed by the lead plaintiff alleging an expanded putative class period of May 3, 2016 through March 18, 2020 against the Company and Mark C. Trudeau, Bryan M. Reasons, George A. Kegler and Matthew K. Harbaugh, as well as newly named defendants Kathleen A. Schaefer, Angus C. Russell, Melvin D. Booth, JoAnn A. Reed, Paul R. Carter, and Mark J. Casey (collectively with Trudeau, Reasons, Kegler and Harbaugh, the “Strougo Defendants”). The amended complaint claims that the defendants made various false and/or misleading statements and/or failed to disclose various material facts regarding Acthar Gel and its results of operations. In October 2020, the defendants filed a motion to dismiss the amended complaint. In March 2022, the Strougo action was administratively closed. In March 2022, the Strougo action was reinstated only with respect to the Strougo Defendants, and the Strougo Defendants filed their reply in support of their motion to dismiss in May 2022. As to the Company, this matter was resolved in the 2020 Bankruptcy Proceedings with no further liability against the Company. However, the Company had indemnification obligations as to the Strougo Defendants. In December 2022, the District Court issued an order denying the Strougo Defendants' motion to dismiss in all respects and the Strougo Defendants answered the complaint. In June 2024, the parties reached an agreement in principle to resolve all claims in this matter for a settlement payment of $46.0 million, which was funded by the Company’s insurance carriers. The district court granted final approval of the settlement in April 2025. The Company released the $46.0 million receivable and payable upon final approval of the settlement and payment by the insurance carriers during the three months ended June 27, 2025.
Endo Bankruptcy
Historically, Endo’s business had been operated by Endo International plc, together with its subsidiaries. On August 16, 2022 (“Endo Petition Date”), Endo International plc, together with certain of its direct and indirect subsidiaries, filed voluntary petitions for relief under the chapter 11 of title 11 of the United States Code (“Bankruptcy Code,” and such cases, the “Endo Chapter 11 Cases”); certain additional Endo entities filed voluntary petitions for relief under the Bankruptcy Code on May 25, 2023 and May 31, 2023 (together the “Endo Debtors”). On December 19, 2023, the Endo Debtors filed a proposed chapter 11 plan of reorganization (as amended, including on January 5, 2024, January 9, 2024 and March 18, 2024, and including any exhibits and supplements filed with respect thereto, the “Endo Plan”) and related disclosure statement with the U.S. Bankruptcy Court for the Southern District of New York (“New York Bankruptcy Court”). The New York Bankruptcy Court confirmed the Endo Plan on March 19, 2024, and the Endo Debtors satisfied all conditions required for the Endo Plan effectiveness on April 23, 2024 (“Endo Effective Date”).
At the Endo Debtors’ request, the New York Bankruptcy Court appointed the Future Claimants' Representative (“FCR”) in the Endo Chapter 11 Cases. As further described in the applicable New York Bankruptcy Court filings, the FCR represents the rights of individuals who may in the future assert one or more personal injury claims against the Endo Debtors or a successor of the Endo Debtors’ businesses relating to the Endo Debtors’ opioid or transvaginal surgical mesh products, but who could not assert such claims in the Endo Chapter 11 Cases because, among other reasons, such individuals were unaware of the alleged injury, had a latent
128
manifestation of the alleged injury or were otherwise unable to assert or incapable of asserting claims based on the alleged injury. Under the Endo Plan and the settlement contemplated thereby, the trust established for the benefit of eligible future claimants assumed liability for all future claims in exchange for Endo’s ongoing obligation to fund such trust. As of December 31, 2025, the Company accrued for loss contingencies of approximately $8.0 million. The liability was assumed in connection with the Business Combination, which is discussed further in Note 5.
Under the Endo Plan, the U.S. Government Economic Settlement provides for payment by Endo of contingent consideration of $25.0 100.0 million in the aggregate) if Endo LP’s annual EBITDA for the corresponding calendar year exceeds defined baselines (the “EBITDA Outperformance Targets”), as set forth in the U.S. Government Economic Settlement. In accordance with the provisions of the U.S. Government Economic Settlement, in the event Endo LP acquires or sells assets, such EBITDA Outperformance Targets shall be adjusted upward or downward dollar for dollar in an amount equal to the EBITDA contribution of such acquired or sold assets. The EBITDA Outperformance Targets for 2024 and 2025 were not met and the Company does not expect to meet the EBITDA Outperformance Targets in any of the fiscal years 2026 through 2028. No payments have been made or accrued for related to the achievement of certain EBITDA Outperformance Targets. Such contingent payments continue to apply after the closing of the Business Combination and the Separation.
Patent Litigation
The Company will continue to vigorously enforce its intellectual property rights relating to its products to prevent the marketing of infringing generic, biosimilar or competing products prior to the expiration of patents covering those products, which, if unsuccessful, could adversely affect the Company's ability to successfully maximize the value of individual products and have an adverse effect on its financial condition, results of operations and cash flows. In the case of litigation filed against potential generic, biosimilar or competing products to Company's products, those litigation matters can either be settled or the litigation pursued through a trial and any potential appeals of the lower court decision.
Mallinckrodt Pharmaceuticals Ireland Limited, et al. v. Airgas Therapeutics LLC et al. On December 30, 2022, the Company initiated litigation against Airgas Therapeutics, LLC, Airgas USA LLC, and Air Liquide S.A. (collectively “Airgas”) in the U.S. District Court for the District of Delaware (“District of Delaware”) following notice from Airgas of its ANDA submission seeking approval from the FDA for a generic version of INOmax® (nitric oxide) gas, for inhalation (“INOmax”). Airgas's ANDA received final approval from the FDA in July 2023, and according to Airgas' counsel, the original ANDA was filed in April 2011. In February 2024, the court entered stipulations of consent for filing of an amended complaint. In March 2024, the court granted Air Liquide S.A.’s motion to dismiss. Airgas Therapeutics, LLC and Airgas USA LLC remain parties to the litigation. In January 2025, the court denied the Company’s motion for preliminary injunction seeking to prevent defendants Airgas Therapeutics LLC and Airgas USA LLC from infringing the Company’s U.S. patents during the pendency of the litigation. The defendants have filed a motion for summary judgment. There was a jury trial in September 2025. At trial, the Company asserted three patents. The Company was seeking monetary and equitable relief. On September 12, 2025, the jury returned a verdict in favor of the Company. The jury found that AirGas willfully infringed the asserted patents. The jury awarded approximately $9.5 million in monetary damages. On October 7, 2025, the Court entered judgment that Airgas infringes the asserted patents under 35 U.S.C. §271(e) and will enter final judgment on remedies after considering motions for judgment as a matter of law.
Amitiza Patent Challenges. The Company was granted numerous Japanese patents related to Amitiza. The Company has received notifications of petitions for invalidation trials described below, each of which was filed with the Japan Patent Office (“JPO”) and relates to Amitiza and its use in Japan. The JPO has the authority to determine the validity of each of these patent grants and each of these patent term extension (“PTE”) registration grants. A party may appeal the JPO’s determination to a court of law.
In October 2023, the Company received notification that Sawai Pharmaceutical Co., Ltd. (“Sawai”) had filed petitions for two invalidation trials against two PTE registrations for JP Patent No. 4332353. In June 2025, the JPO determined that none of the invalidation grounds can stand and concluded that the two PTE registrations for the 24 and 12μg capsules are valid. Sawai has appealed the JPO decision for both PTE registrations. Oral arguments were held in February 2026. A decision was reached on April 9, 2026, dismissing Sawai’s complaints. Sawai has the opportunity to appeal the decision within two weeks from the date Sawai receives the decision.
In December 2023, the Company received notification that Sawai had filed a petition for an invalidation trial against JP Patent No. 4332353. The JPO held a hearing in December 2024 relating to Sawai’s challenge of JP Patent No. 4332353, and in May 2025 the JPO issued a decision finding that all of the asserted claims in respect of JP Patent No. 4332353 are valid and will be maintained. Sawai has appealed the JPO's decision. Initial briefs have been filed by all parties and an oral argument will be held on April 23, 2026.
In April 2024, the Company received notification that Sawai had filed petitions for invalidation trials with respect to only the 12µg strength of Amitiza against PTE registrations of three additional patents (JP Patent No. 4786866, JP Patent No. 4852229, and JP Patent No. 4889219). The JPO held a hearing in August 2025 with respect to the three invalidations trials regarding the 12 µg PTE registrations. The JPO has completed their examination and the Company is awaiting the decision from the JPO.
In April 2024, the Company received notification that Sawai had filed a petition for invalidation trial against JP Patent No. 4786866. In December 2025, the JPO issued a Notice of Completion of Examination in the invalidation trial. In February 2026, the
129
JPO issued a decision finding that all of the asserted claims in respect of JP Patent No. 4786866 as amended during the invalidation trial are valid and will be maintained. Sawai has the opportunity to appeal the decision.
In May 2024, the Company received notification that Sawai had filed petitions for invalidation trials with respect to only the 12µg strength of Amitiza against PTE registrations of two additional patents (JP Patent No. 4332316 and JP Patent No. 4684334). An oral hearing was held on March 2, 2026. A decision is expected later this year.
In December 2025, the Company received notification that Towa Pharmaceutical Co., Ltd (“Towa”) had filed petitions for invalidation trials with respect to only the 12µg strength of Amitiza against PTE registrations for JP Patent Nos. 4786866, 4852229, and 4889219. The petitions have been received by the parties, and answers to the petitions are expected to be filed in May 2026. The Company believes that each of these patents and/or PTE registrations is valid, and the Company will vigorously defend these patents and PTE registrations.
In October 2025, the Company intervened in a patent infringement suit filed by Viatris Pharmaceuticals Japan G.K. (“Viatris”) against Sawai, in Osaka District Court, Civil Division, related to certain Japanese patents. Viatris has filed lawsuits against Sawai alleging that Sawai has infringed JP Patent Nos. 4889219 (“the ‘219 patent”) and 4332353 (“the ‘353 patent”). In the lawsuit, Viatris alleges that Sawai has infringed the ‘219 and ‘353 patents by filing an application for marketing approval of a generic drug of Amitiza 24 mcg capsules. Petitions for preliminary injunction have also been filed against Sawai to enjoin them from continuing to infringe the ‘219 and ‘353 patents. The Company has intervened in both lawsuits to support Viatris in their claims against Sawai and to defend the validity of the ‘219 and ‘353 patents. In February 2026, the Osaka District Court ruled on the preliminary injunction cases in favor of Sawai, finding that Sawai does not infringe the ‘219 patent or the ‘353 patent. The Osaka District Court did not rule on the validity of either the ‘219 patent or the ‘353 patent. Viatris has appealed the decision, and the Company has joined the appeal. A decision in the patent infringement lawsuits was received on March 3, 2026, finding that Sawai does not infringe the ‘219 patent or the ‘353 patent. Viatris has appealed the decision. On February 16, 2026, the Japanese regulatory authority approved both Towa’s and Sawai’s generic 24 mcg Amitiza products. Separately, Viatris filed a number of additional proceedings alleging patent infringement against Towa and Sawai in Japan, including petitions to enjoin them from continuing to infringe a number of patents related to the Amitiza 24 mcg capsules. The Company intends to intervene in the lawsuits to support Viatris in their claims and to defend the validity of the patents.
The outcome of the forgoing proceedings is expected to impact the Company’s sales of Amitiza in Japan.
Other Matters
The Company is a defendant in a number of other pending legal proceedings relating to present and former operations, acquisitions and dispositions. The Company does not expect the outcome of these proceedings, either individually or in the aggregate, to have a material adverse effect on its financial condition, results of operations and cash flows.
SpinCo Liabilities
Pursuant to the terms and conditions of the Separation Agreement by and between the Company and Par Health, at the effective time of its separation from the Company, Par Health or one of its subsidiaries assumed certain liabilities (whether accrued, contingent or otherwise) relating to, arising out of or resulting from the generic pharmaceuticals (including APIs) and sterile injectables businesses of the Company or certain related assets, including all related pending, threatened and unasserted legal matters (collectively, the “SpinCo Liabilities”). These SpinCo Liabilities include, among others, environmental proceedings, governmental investigations, patent proceedings, commercial disputes, and other litigation. The Company or one of its subsidiaries retained all liabilities (including whether accrued, contingent or otherwise) other than SpinCo Liabilities, including all related pending, threatened and unasserted legal matters (the “Parent Liabilities”). Par Health agreed to indemnify the Company for any liability arising out of or resulting from the SpinCo Liabilities, and the Company agreed to indemnify Par Health for any liability arising out of or resulting from the Parent Liabilities. Items described above in this Note 20 are considered to be Parent Liabilities.
Based on the Company’s understanding of the matters to date, the Company does not intend to further report on Spinco Liabilities, except for the matters captioned “U.S. Attorney's Office Subpoena W.D. Va.” and “Generic Pharmaceutical Antitrust Multi-District Litigation” in which a Keenova entity has been named a party.
U.S. Attorney's Office Subpoena W.D. Va. In August 2023, the Company received a grand jury subpoena from the WDVA USAO. Subsequently, the Company and Par Health received additional grand jury subpoenas from the WDVA USAO, most recently, in December 2025. The subpoenas seek production of certain data and information for the time period from July 17, 2012, to the present, including information and data relating to the controlled substances compliance program of the Company’s former subsidiaries, reporting of suspicious orders for controlled substances, chargebacks and other transactions, financial accounts related to these issues, financial transactions involving prescription drug products, and communications with the U.S. Drug Enforcement Administration. The Company cannot predict the eventual scope, duration or outcome of this matter at this time.
Generic Pharmaceutical Antitrust Multi-District Litigation. In August 2016, a multi-district litigation (“MDL”) was established in the EDPA relating to allegations of antitrust violations with respect to generic pharmaceutical pricing (“Generic Pricing MDL”). Plaintiffs in the Generic Pricing MDL, captioned In re: Generic Pharmaceuticals Pricing Antitrust Litigation, allege a conspiracy of price-fixing and customer allocation among generic drug manufacturers beginning in or around July 2009. The Generic Pricing MDL
130
| 21. | Financial Instruments and Fair Value Measurements | ||||
Fair value is defined as the exit price that would be received from the sale of an asset or paid to transfer a liability, using assumptions that market participants would use in pricing an asset or liability. The fair value guidance establishes a three-level fair value hierarchy as follows:
•Level 1 — observable inputs such as quoted prices in active markets for identical assets or liabilities;
•Level 2 — significant other observable inputs that are observable either directly or indirectly; and
•Level 3 — significant unobservable inputs in which there is little or no market data, which requires the Company to develop its own assumptions.
The following tables provide a summary of the significant assets and liabilities that are measured at fair value on a recurring basis at the end of each period:
| Fair Value Measurement Using Fair Value Hierarchy: | |||||||||||||||||||||||
| December 31, 2025 | Level 1 | Level 2 | Level 3 | ||||||||||||||||||||
Assets: | |||||||||||||||||||||||
| Equity securities | $ | $ | $ | $ | |||||||||||||||||||
| Interest rate cap | |||||||||||||||||||||||
| $ | $ | $ | $ | ||||||||||||||||||||
Liabilities: | |||||||||||||||||||||||
| Deferred compensation liabilities | $ | $ | $ | $ | |||||||||||||||||||
| Contingent consideration liabilities | |||||||||||||||||||||||
| $ | $ | $ | $ | ||||||||||||||||||||
| Fair Value Measurement Using Fair Value Hierarchy: | |||||||||||||||||||||||
| December 27, 2024 | Level 1 | Level 2 | Level 3 | ||||||||||||||||||||
Assets: | |||||||||||||||||||||||
| Equity securities | $ | $ | $ | $ | |||||||||||||||||||
| Interest rate cap | |||||||||||||||||||||||
| $ | $ | $ | $ | ||||||||||||||||||||
| Liabilities: | |||||||||||||||||||||||
| Deferred compensation liabilities | $ | $ | $ | $ | |||||||||||||||||||
| Contingent consideration liabilities | |||||||||||||||||||||||
| $ | $ | $ | $ | ||||||||||||||||||||
Equity securities. Equity securities consist primarily of shares in Silence Therapeutics plc. for which quoted prices are available in an active market; therefore, these investments are classified as level 1 and are valued based on quoted market prices reported on internationally recognized securities exchanges.
131
During the years ended December 31, 2025 and December 27, 2024 (Successor), the period November 15, 2023, through December 29, 2023 (Successor) and the period December 31, 2022 through November 14, 2023 (Predecessor), the Company recognized unrealized (losses) gains of $(1.7 ) million, $(17.4 ) million, $13.5 million and $(10.1 ) million, respectively, related to the Company’s investments within other income (expense), net in the consolidated statements of operations.
Interest rate cap. The Company is exposed to interest rate risk on its variable-rate debt. During the three months ended March 31, 2023 (Predecessor), the Company entered into an interest rate cap agreement, which served to reduce the volatility on future interest expense cash outflows. The interest rate cap agreement had a total notional value of $860.0 million with an upfront premium of $20.0 million and provided the Company with interest rate protection (i) for the period March 16, 2023 through July 19, 2023, to the extent that the one-month LIBOR exceeds 4.65 %, and (ii) for the period July 20, 2023, through March 26, 2026, to the extent that the one-month SOFR exceeds 3.84 %. The impact of the interest rate cap on the Company’s applicable interest rates as disclosed in Note 15 was not material. The interest rate cap agreement expired in accordance with its terms on March 26, 2026.
The interest rate cap agreement was not accounted for as a cash flow hedge and the changes in fair value of the interest rate cap were recorded within other (expense) income, net in the consolidated statements of operations. The fair value of the interest rate cap is included in other assets on the Company’s consolidated balance sheet as of December 31, 2025, and December 27, 2024.
The Company elected to use the income approach to value the interest rate cap derivative using observable Level 2 market expectations at the measurement date and standard valuation techniques to convert future amounts to a single present amount (discounted) reflecting current market expectations about those future amounts. Level 2 inputs for derivative valuations are limited to quoted prices for similar assets or liabilities in active markets (specifically futures contracts) and inputs other than quoted prices that are observable such as SOFR rate curves, futures and volatilities. Mid-market pricing is used as a practical expedient in the fair value measurements. During the years ended December 31, 2025, and December 27, 2024, and the period November 15, 2023 to December 29, 2023 (Successor), the Company recognized unrealized losses of $5.2 million, $7.6 million and $8.4 5.7 million within AOCI with a gain of $0.7 million being reclassified into earnings as a component of interest expense, net.
Deferred compensation liabilities. The Company maintains a non-qualified deferred compensation plan in the U.S., which permitted eligible employees of the Company to defer a portion of their compensation. A recordkeeping account is set up for each participant and the participant chooses from a variety of funds for the deemed investment of their accounts. The recordkeeping accounts generally correspond to the funds offered in the Company's U.S. tax-qualified defined contribution retirement plan and the account balance fluctuates with the investment returns on those funds. The plan is currently frozen for employee deferrals.
Terlivaz Contingent Consideration. In accordance with the 2020 Plan and the scheme of arrangement confirmed by the Irish High Court, based on and consistent in all respects with the 2020 Plan, the Company will provide consideration for the Terlivaz CVR primarily in the form of the achievement of a cumulative net sales milestone. The Company assesses the likelihood and timing of making such payments at each balance sheet date. The fair value of the contingent payment was measured based on the net present value of a probability-weighted assessment. The Company determined the fair value of the Terlivaz CVR as of December 31, 2025, and December 27, 2024, to be $20.0 million and $17.5 million, respectively, which is classified within other liabilities in the consolidated balance sheets as of December 31, 2025 and December 27, 2024, respectively.
Edex Contingent Consideration. Endo is a party to an agreement pursuant to which it is obligated to make certain contingent cash consideration payments in the form of royalties on net sales of Edex® indefinitely until pre-determined market conditions are met. The Company assumed the obligation in connection with the Business Combination. The acquisition date fair value was estimated based on a discounted cash flow model (income approach). The Company determined the fair value of the Edex Contingent Consideration as of December 31, 2025, to be $34.8 million, of which $3.0 million is classified as current. The current and non-current portion of the liability is classified within accrued and other current liabilities and in other liabilities, respectively, in the consolidated balance sheet.
132
The following table summarizes activity for contingent consideration:
| Terlivaz CVR | Edex | Total | |||||||||||||||
| Balance as of December 29, 2023 | $ | $ | $ | ||||||||||||||
| Fair value adjustment | |||||||||||||||||
| Balance as of December 27, 2024 | $ | $ | $ | ||||||||||||||
| Additions | |||||||||||||||||
| Fair value adjustment | |||||||||||||||||
| Payments | ( | ( | |||||||||||||||
| Balance as of December 31, 2025 | $ | $ | $ | ||||||||||||||
Financial Instruments Not Measured at Fair Value
The following methods and assumptions were used by the Company in estimating fair values for financial instruments not measured at fair value as of December 31, 2025, and December 27, 2024:
•The carrying amounts of cash and cash equivalents, accounts receivable, accounts payable and the majority of other current assets and liabilities approximate fair value because of their short-term nature. The Company classifies cash on hand and deposits in banks, including commercial paper, money market accounts and other investments it may hold from time to time, with an original maturity of three months or less, as cash and cash equivalents (level 1). The fair value of restricted cash was equivalent to its carrying value of $141.1 million and $49.2 million as of December 31, 2025, and December 27, 2024, (level 1), respectively. Restricted cash as of December 31, 2025 primarily relates to certain self-insurance related matters of $85.9 million, $22.2 million related to the Mallinckrodt Baker escrow discussed in Note 19, and approximately $33.0 million related to certain bank guarantees, letters of credit, surety bonds and other collateral arrangements. Restricted cash as of December 27, 2024 primarily relates to $21.3 million related to the Mallinckrodt Baker escrow discussed in Note 19, and approximately $27.9 million related to certain bank guarantees, letters of credit, surety bonds and other collateral arrangements.
•Successor debt. On December 6, 2024, the Company used the proceeds from the Therakos Divestiture to mandatorily prepay the First-Out Takeback Term Loans in full, partially prepay the Second-Out Takeback Term Loans, and partially redeem the Takeback Notes. On August 1, 2025, in connection with the consummation of the Business Combination, the Company prepaid in full all remaining outstanding Second-Out Takeback Term Loans and redeemed in full all remaining outstanding Takeback Notes, which are each defined and further described in Note 15.
| December 31, 2025 | December 27, 2024 | ||||||||||||||||||||||
| Carrying Value | Fair Value | Carrying Value | Fair Value | ||||||||||||||||||||
| Level 1: | |||||||||||||||||||||||
| $ | $ | $ | $ | ||||||||||||||||||||
| Level 2: | |||||||||||||||||||||||
| Term Loan due April 2031 | |||||||||||||||||||||||
| Second-Out Takeback Term Loan Due November 2028 | |||||||||||||||||||||||
| Total Debt | $ | $ | $ | $ | |||||||||||||||||||
Concentration of Credit and Other Risks
Financial instruments that potentially subject the Company to concentrations of credit risk primarily consist of accounts receivable. The Company generally does not require collateral from customers.
The following table shows net sales attributable to distributors that accounted for 10.0% or more of the Company's total net sales:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| FFF Enterprises, Inc. | % | % | % | % | ||||||||||||||||||||||
| Cencora, Inc. | * | * | * | |||||||||||||||||||||||
* Net sales to this distributor were less than 10.0% of total net sales during the respective periods presented above.
133
The following table shows accounts receivable attributable to distributors that accounted for 10.0% or more of the Company's gross accounts receivable at the end of each period:
| December 31, 2025 | December 27, 2024 | ||||||||||
| Cencora, Inc. | % | * | |||||||||
| FFF Enterprises, Inc. | |||||||||||
* Accounts receivable attributable to this distributor was less than 10.0% of total gross accounts receivable at the end of the respective period presented above.
| 22. | Segment and Geographical Data | ||||
The Company operates its business in one
The Company’s chief operating decision maker (“CODM”) is the Chief Executive Officer. The CODM measures and evaluates the Company's operations on a consolidated basis based on net income (loss). Significant segment expenses include cost of sales, research and development and selling, general and administrative expenses. The CODM considers budget-to-actual variances of consolidated net sales and consolidated net income (loss) on a quarterly basis to assess performance and operating trends and to make decisions about allocating resources
The CODM manages assets on a total company basis. The CODM is not regularly provided any asset information below the consolidated balance sheet.
Net sales by product family were as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
| Acthar Gel | $ | $ | $ | $ | ||||||||||||||||||||||
Xiaflex(1) | ||||||||||||||||||||||||||
| INOmax | ||||||||||||||||||||||||||
| Therakos | ||||||||||||||||||||||||||
| Amitiza | ||||||||||||||||||||||||||
Other Products Sales(1) | ||||||||||||||||||||||||||
License Revenues(1) | ||||||||||||||||||||||||||
| Total Net Sales | $ | $ | $ | $ | ||||||||||||||||||||||
(1)Is or contains products acquired in the Business Combination. Net sales include $397.0 million from products acquired in the Business Combination. Accordingly, there are no comparable net sales for these products in prior periods.
Selected information by geographic area was as follows:
| Successor | Predecessor | |||||||||||||||||||||||||
| Year Ended December 31, 2025 | Year Ended December 27, 2024 | Period from November 15, 2023 through December 29, 2023 | Period from December 31, 2022 through November 14, 2023 | |||||||||||||||||||||||
Net sales(1): | ||||||||||||||||||||||||||
| U.S. | $ | $ | $ | $ | ||||||||||||||||||||||
| Europe, Middle East and Africa | ||||||||||||||||||||||||||
| Other | ||||||||||||||||||||||||||
| Net Sales | $ | $ | $ | $ | ||||||||||||||||||||||
(1)Net sales are attributed to regions based on the location of the entity that records the transaction, none of which relate to the country of Ireland.
134
Item 9. | Changes in and Disagreements With Accountants on Accounting and Financial Disclosure. | ||||
None.
Item 9A. | Controls and Procedures. | ||||
Evaluation of Disclosure Controls and Procedures
We maintain disclosure controls and procedures designed to ensure that information required to be disclosed in reports filed or submitted under the Securities Exchange Act of 1934, as amended (“Exchange Act”), is recorded, processed, summarized and reported within the time periods specified in the Securities and Exchange Commission’s rules and forms, and that such information is accumulated and communicated to management, including our Chief Executive Officer (“CEO”) and Chief Financial Officer (“CFO”), as appropriate, to allow timely decisions regarding required disclosure.
Our management, with the participation of our CEO and CFO, evaluated the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act) as of December 31, 2025. Based on that evaluation, our CEO and CFO concluded that, as of that date, our disclosure controls and procedures were effective.
Management's Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting, as defined under Exchange Act Rules 13a-15(f) and 15d-15(f). Our internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with GAAP. Internal control over financial reporting includes those policies and procedures that:
•pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of the Company's assets;
•provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with GAAP, and that the Company's receipts and expenditures are being made only in accordance with authorizations of the Company's management and directors; and
•provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use, or disposition of the Company's assets that could have a material effect on the financial statements.
Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate.
Management assessed the effectiveness of our internal control over financial reporting as of December 31, 2025. In making this assessment, management used the criteria set forth by the Committee of Sponsoring Organizations of the Treadway Commission (COSO) in Internal Control—Integrated Framework (2013).
On July 31, 2025, the Company completed the business combination with Endo. In accordance with the SEC’s published guidance applicable to newly acquired businesses, management has elected to exclude the acquired business from its assessment of internal control over financial reporting for fiscal year 2025. Endo accounted for approximately 28% of total assets and 28% of total revenues as of and for the year ended December 31, 2025. Management is in the process of integrating the operations, processes and internal controls of Endo into the Company’s internal control framework.
Except for the exclusion of Endo as described above, management has assessed the effectiveness of the Company’s internal control over financial reporting as of December 31, 2025, based on the COSO criteria. Based on its assessment, management concluded that, as of December 31, 2025, our internal control over financial reporting was effective.
This Annual Report does not include an attestation report of the Company’s registered public accounting firm regarding internal control over financial reporting. Management’s report was not subject to attestation by the Company’s registered public accounting firm pursuant to rules of the Securities and Exchange Commission that permit the Company to provide only management’s report in this Annual Report.
Changes in Internal Control over Financial Reporting
On November 10, 2025, we completed the Separation. As a result of the Separation, Management designed and implemented relevant new controls to prevent or detect material misstatements with respect to accounting for the Separation and certain ancillary agreements, as well as operating under the transition services agreement. There were no other changes in our internal control over financial reporting during the three months ended December 31, 2025, that have materially affected, or are likely to materially affect, our internal control over financial reporting.
135
Item 9B. | Other Information. | ||||
During the fourth quarter ended December 31, 2025, none of the Company's directors or officers (as defined in Rule 16a-1(f) of the Exchange Act) adopted terminated
Item 9C. | Disclosure Regarding Foreign Jurisdictions that Prevent Inspections. | ||||
Not applicable.
136
PART III
Item 10. | Directors, Executive Officers and Corporate Governance. | ||||
Information regarding our directors required under this Item 10. Directors, Executive Officers and Corporate Governance will be filed with the SEC within 120 days after December 31, 2025, pursuant to General Instruction G(3) to Form 10-K.
Information regarding our executive officers required under this Item 10. Directors, Executive Officers and Corporate Governance is included in Item 1. Business of this Annual Report.
We have adopted the Keenova Code of Conduct, which meets the requirements of a “code of ethics” as defined in Item 406 of Regulation S-K. Our Code of Conduct applies to all employees, officers and directors of Keenova, including, without limitation, our CEO, CFO and other senior financial officers. Our Code of Conduct is posted on our website at keenova.com under the heading Company - Polices. We will also provide a copy of our Code of Conduct to shareholders upon request. We intend to disclose any amendments to our Code of Conduct, as well as any waivers for executive officers or directors, on our website.
Information regarding our policies on insider trading required under this Item 10. Directors, Executive Officers and Corporate Governance will be filed with the SEC within 120 days after December 31, 2025, pursuant to General Instruction G(3) to Form 10-K.
Item 11. | Executive Compensation. | ||||
Information regarding the compensation of our named executive officers and directors required under this Item 11. Executive Compensation will be filed with the SEC within 120 days after December 31, 2025, pursuant to General Instruction G(3) to Form 10-K.
Item 12. | Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters. | ||||
Information regarding individuals or groups which own more than 5.0% of our ordinary shares, as well as information regarding the security ownership of our executive officers and directors, and other shareholder matters required under this Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters will be filed with the SEC within 120 days after December 31, 2025, pursuant to General Instruction G(3) to Form 10-K.
Item 13. | Certain Relationships and Related Transactions, and Director Independence. | ||||
Information regarding transactions with related parties and director independence required under this Item 13. Certain Relationships and Related Transactions, and Director Independence will be filed with the SEC within 120 days after December 31, 2025, pursuant to General Instruction G(3) to Form 10-K.
Item 14. | Principal Accounting Fees and Services. | ||||
Information regarding the services provided by and the fees paid to PricewaterhouseCoopers LLP, our independent auditors, required under this Item 14. Principal Accounting Fees and Services will be filed with the SEC within 120 days after December 31, 2025, pursuant to General Instruction G(3) to Form 10-K.
137
PART IV
Item 15. | Exhibits, Financial Statement Schedules. | ||||
Documents filed as part of this report:
1) Financial Statements. The following are included within Item 8. Financial Statements and Supplementary Data of this Annual Report.
•Consolidated Statements of Operations for the fiscal years ended December 31, 2025 and December 27, 2024 (Successor), the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor).
•Consolidated Statements of Comprehensive Operations for the fiscal years ended December 31, 2025 and December 27, 2024 (Successor), the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor).
•Consolidated Statements of Cash Flows for the fiscal years ended December 31, 2025 and December 27, 2024 (Successor), the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor).
•Consolidated Statement of Changes in Shareholders' Equity for the fiscal years ended December 31, 2025 and December 27, 2024 (Successor), the period from November 15, 2023 through December 29, 2023 (Successor), and the period from December 31, 2022 through November 14, 2023 (Predecessor).
2) Financial Statement Schedules. All schedules have been omitted because they are not applicable, not required, immaterial for all periods presented or the information is included in the financial statements or notes thereto.
3) Exhibits. The exhibits are included in the Exhibit Index that appears at the end of this Annual Report.
Item 16. | Form 10-K Summary. | ||||
The Company has elected not to include a Form 10-K summary under this Item 16.
138
EXHIBIT INDEX
139
10.2† | ||||||||
| 10.3 | ||||||||
| 10.4 | ||||||||
| 10.5 | ||||||||
| 10.6† | ||||||||
| 10.7† | ||||||||
| 10.8† | ||||||||
| 10.9† | ||||||||
| 10.10†* | ||||||||
| 10.11†* | ||||||||
| 10.12†* | ||||||||
| 10.13†* | ||||||||
| 10.14†* | ||||||||
| 10.15†* | ||||||||
| 10.16†* | ||||||||
| 10.17†* | ||||||||
| 10.18† | ||||||||
| 10.19† | ||||||||
| 10.20† | ||||||||
140
| 10.21† | ||||||||
| 10.22† | ||||||||
| 10.23† | ||||||||
| 10.24† | ||||||||
| 10.25† | ||||||||
| 10.26† | ||||||||
| 10.27† | ||||||||
| 10.28† | ||||||||
| 10.29† | ||||||||
| 10.30† | ||||||||
| 10.31 | ||||||||
| 10.32 | ||||||||
| 10.33† | ||||||||
| 10.34† | ||||||||
| 10.35† | ||||||||
| 10.36† | ||||||||
| 10.37† | ||||||||
| 10.38† | ||||||||
| 10.39† | ||||||||
| 10.40** | ||||||||
141
| 10.41 | ||||||||
| 10.42† | ||||||||
| 10.43 | ||||||||
| 10.44† | ||||||||
| 10.45† | ||||||||
| 10.46 | ||||||||
| 10.47 | ||||||||
| 10.48 | ||||||||
| 10.49 | ||||||||
| 10.50 | ||||||||
| 10.51 | ||||||||
| 10.52† | ||||||||
| 10.53** | ||||||||
| 10.54 | ||||||||
| 10.55 | ||||||||
| 10.56** | ||||||||
| 10.57** | ||||||||
| 19.1 | ||||||||
| 21.1 | ||||||||
| 23.1 | ||||||||
| 23.2 | ||||||||
| 31.1 | ||||||||
| 31.2 | ||||||||
| 32.1 | ||||||||
142
| 99.1 | ||||||||
| 99.2 | ||||||||
| 101.INS | Inline XBRL Instance Document | |||||||
| 101.SCH | Inline XBRL Taxonomy Extension Schema Document | |||||||
| 101.CAL | Inline XBRL Taxonomy Calculation Linkbase Document | |||||||
| 101.DEF | Inline XBRL Taxonomy Definition Linkbase Document | |||||||
| 101.LAB | Inline XBRL Taxonomy Label Linkbase Document | |||||||
| 101.PRE | Inline XBRL Taxonomy Presentation Linkbase Document | |||||||
| 104 | Cover Page Interactive Data File (embedded within the inline XBRL document and contained in Exhibit 101.INS). | |||||||
† Compensation plans or arrangements.
* Portions of this exhibit have been omitted in accordance with Item 601(b)(2) or Item 601(b)(10) of Regulation S-K, as applicable.
** Schedules have been omitted pursuant to Item 601(a)(5) of Regulation S-K. The Company agrees to furnish supplementally a copy of any omitted schedule or exhibit to the SEC upon request.
The agreements and other documents filed as exhibits to this report are not intended to provide factual information or other disclosure other than with respect to the terms of the agreements or other documents themselves, and you should not rely on them for that purpose. In particular, any representations and warranties made by us in these agreements or other documents were made solely within the specific context of the relevant agreement or document and may not describe the actual state of affairs as of the date they were made or at any other time.
143
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| KEENOVA THERAPEUTICS PLC | ||||||||
| April 15, 2026 | By: | /s/ Christiana Stamoulis | ||||||
Christiana Stamoulis President and Chief Financial Officer (principal financial officer) | ||||||||
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
| Signature | Title | Date | ||||||||||||
| /s/ Sigurdur Olafsson | President, Chief Executive Officer and Director | April 15, 2026 | ||||||||||||
Sigurdur Olafsson | (principal executive officer) | |||||||||||||
| /s/ Christiana Stamoulis | President and Chief Financial Officer | April 15, 2026 | ||||||||||||
| Christiana Stamoulis | (principal financial officer) | |||||||||||||
| /s/ Frank Raciti | Controller and Chief Accounting Officer | April 15, 2026 | ||||||||||||
| Frank Raciti | (principal accounting officer) | |||||||||||||
| /s/ Marc Yoskowitz | Chair of the Board of Directors | April 15, 2026 | ||||||||||||
| Marc Yoskowitz | ||||||||||||||
| /s/ Paul Bisaro | Director | April 15, 2026 | ||||||||||||
| Paul Bisaro | ||||||||||||||
| /s/ Leslie Donato | Director | April 15, 2026 | ||||||||||||
| Leslie Donato | ||||||||||||||
| /s/ Katina Dorton | Director | April 15, 2026 | ||||||||||||
| Katina Dorton | ||||||||||||||
| /s/ Paul Efron | Director | April 15, 2026 | ||||||||||||
| Paul Efron | ||||||||||||||
| /s/ Scott Hirsch | Director | April 15, 2026 | ||||||||||||
| Scott Hirsch | ||||||||||||||
| /s/ Sophia Langlois | Director | April 15, 2026 | ||||||||||||
| Sophia Langlois | ||||||||||||||
| /s/ Jonathan Zinman | Director | April 15, 2026 | ||||||||||||
| Jonathan Zinman | ||||||||||||||
144
Exhibit 3.2
Companies Act 2014
A PUBLIC COMPANY LIMITED BY SHARES
MEMORANDUM and ARTICLES OF ASSOCIATION
of
KEENOVA THERAPEUTICS PUBLIC LIMITED COMPANY
(as amended by Special Resolution passed on 8 October 2025 with effect from 11 November 2025)
DUBLIN
Companies Act 2014
A PUBLIC COMPANY LIMITED BY SHARES
MEMORANDUM and ARTICLES OF ASSOCIATION
of
KEENOVA THERAPEUTICS PUBLIC LIMITED COMPANY
(as amended by Special Resolution passed on 8 October 2025)
DUBLIN
Cert. No.: 522227
Companies Act 2014
A PUBLIC COMPANY LIMITED BY SHARES
CONSTITUTION
of
KEENOVA THERAPEUTICS PUBLIC LIMITED COMPANY
MEMORANDUM OF ASSOCIATION
1.The name of the Company is Keenova Therapeutics public limited company.
2.The Company is a public limited company for the purposes of Part 17 of the Companies Act 2014 (the “Act”).
3.The objects for which the Company is established are:
3.1
(a)To carry on the business of a healthcare services development company operating in the healthcare field, and to design, manufacture, produce, supply and provide generic and branded pharmaceuticals, contrast media, radiopharmaceuticals, active pharmaceutical ingredients and dosage pharmaceuticals and other devices or products of a surgical, pharmaceutical, diagnostic, medical imaging or medical character necessary or suitable for the proper treatment of sick or injured persons or patients and to carry on business as merchants of and dealers in all supplies required for use in the treatment and care of the sick and injured and to do all things usually dealt in by persons carrying on the above mentioned businesses or any of them or likely to be required in connection with any of the said businesses.
(b)To carry on the business of a holding company and to co-ordinate the administration, finances and activities of any subsidiary companies or associated companies, to do all lawful acts and things whatever that are necessary or convenient in carrying on the business of such a holding company and in particular to carry on in all its branches the business of a management services company, to act as managers and to direct or coordinate the management of other companies or of the business, property and estates of any company or person and to undertake and carry out all such services in connection therewith as may be deemed expedient by the Company’s board of directors and to exercise its powers as a shareholder of other companies.
(c)To acquire the entire issued share capital of Mallinckrodt International Finance S.A., a Luxembourg registered company and Mallinckrodt Belgium BVBA, a Belgian registered company.
3.2To acquire shares, stocks, debentures, debenture stock, bonds, obligations and securities by original subscription, tender, purchase, exchange or otherwise and to subscribe for the same either conditionally or otherwise, and to guarantee the subscription thereof and to exercise and enforce all rights and powers conferred by or incidental to the ownership thereof.
3.3To facilitate and encourage the creation, issue or conversion of and to offer for public subscription debentures, debenture stocks, bonds, obligations, shares, stocks, and securities and to act as trustees in connection with any such securities and to take part in the conversion of business concerns and undertakings into companies.
2
3.4To purchase or by any other means acquire any freehold, leasehold or other property and in particular lands, tenements and hereditaments of any tenure, whether subject or not to any charges or incumbrances, for any estate or interest whatever, and any rights, privileges or easements over or in respect of any property, and any buildings, factories, mills, works, wharves, roads, machinery, engines, plant, live and dead stock, barges, vessels or things, and any real or personal property or rights whatsoever which may be necessary for, or may conveniently be used with, or may enhance the value or property of the Company, and to hold or to sell, let, alienate, mortgage, charge or otherwise deal with all or any such freehold, leasehold, or other property, lands, tenements or hereditaments, rights, privileges or easements.
3.5To sell or otherwise dispose of any of the property or investments of the Company.
3.6To establish and contribute to any scheme for the purchase of shares in the Company to be held for the benefit of the Company’s employees and to lend or otherwise provide money to such schemes or the Company’s employees or the employees of any of its subsidiary or associated companies to enable them to purchase shares of the Company.
3.7To grant, convey, transfer or otherwise dispose of any property or asset of the Company of whatever nature or tenure for such price, consideration, sum or other return whether equal to or less than the market value thereof and whether by way of gift or otherwise as the Directors shall deem fit and to grant any fee, farm grant or lease or to enter into any agreement for letting or hire of any such property or asset for a rent or return equal to or less than the market or rack rent therefor or at no rent and subject to or free from covenants and restrictions as the Directors shall deem appropriate.
3.8To acquire and undertake the whole or any part of the business, good-will and assets of any person, firm or company carrying on or proposing to carry on any of the businesses which this Company is authorised to carry on, and as part of the consideration for such acquisition to undertake all or any of the liabilities of such person, firm or company, or to acquire an interest in, amalgamate with, or enter into any arrangement for sharing profits, or for co-operation, or for limiting competition or for mutual assistance with any such person, firm or company and to give or accept by way of consideration for any of the acts or things aforesaid or property acquired, any shares, debentures, debenture stock or securities that may be agreed upon, and to hold and retain or sell, mortgage or deal with any shares, debentures, debenture stock or securities so received.
3.9To apply for, purchase or otherwise acquire any patents, brevets d’invention, licences, concessions and the like conferring any exclusive or non-exclusive or limited rights to use or any secret or other information as to any invention which may seem capable of being used for any of the purposes of the Company or the acquisition of which may seem calculated directly or indirectly to benefit the Company, and to use, exercise, develop or grant licences in respect of or otherwise turn to account the property, rights or information so acquired.
3.10To enter into partnership or into any arrangement for sharing profits, union of interests, co-operation, joint venture, reciprocal concession or otherwise with any person or company carrying on or engaged in or about to carry on or engage in any business or transaction which the Company is authorised to carry on or engage in or any business or transaction capable of being conducted so as directly to benefit this Company.
3.11To invest and deal with the moneys of the Company not immediately required upon such securities and in such manner as may from time to time be determined.
3.12To lend money to and guarantee the performance of the contracts or obligations of any company, firm or person, and the repayment of the capital and principal of, and dividends, interest or premiums payable on, any stock, shares and securities of any company, whether having objects similar to those of this Company or not, and to give all kinds of indemnities.
3
3.13To engage in currency exchange and interest rate transactions including, but not limited to, dealings in foreign currency, spot and forward rate exchange contracts, futures, options, forward rate agreements, swaps, caps, floors, collars and any other foreign exchange or interest rate hedging arrangements and such other instruments as are similar to, or derived from, any of the foregoing whether for the purpose of making a profit or avoiding a loss or managing a currency or interest rate exposure or any other exposure or for any other purpose.
3.14To guarantee, support or secure, whether by personal covenant or by mortgaging or charging all or any part of the undertaking, property and assets (both present and future) and uncalled capital of the Company, or by both such methods, the performance of the obligations of, and the repayment or payment of the principal amounts of and premiums, interest and dividends on any securities of, any person, firm or company including (without prejudice to the generality of the foregoing) any company which is for the time being the Company’s holding company as defined by the Act, or a subsidiary, as defined in the Act of any such holding company or otherwise associated with the Company in business.
3.15To borrow or secure the payment of money in such manner as the Company shall think fit, and in particular by the issue of debentures, debenture stocks, bonds, obligations and securities of all kinds, either perpetual or terminable and either redeemable or otherwise and to secure the repayment of any money borrowed, raised or owing by trust deed, mortgage, charge, or lien upon the whole or any part of the Company’s property or assets (whether present or future) including its uncalled capital, and also by a similar trust deed, mortgage, charge or lien to secure and guarantee the performance by the Company of any obligation or liability it may undertake.
3.16To draw, make, accept, endorse, discount, execute, negotiate and issue promissory notes, bills of exchange, bills of lading, warrants, debentures and other negotiable or transferable instruments.
3.17To subscribe for, take, purchase or otherwise acquire and hold shares or other interests in, or securities of any other company having objects altogether or in part similar to those of this Company, or carrying on any business capable of being conducted so as directly or indirectly to benefit this Company.
3.18To hold in trust as trustees or as nominees and to deal with, manage and turn to account, any real or personal property of any kind, and in particular shares, stocks, debentures, securities, policies, book debts, claims and chases in actions, lands, buildings, hereditaments, business concerns and undertakings, mortgages, charges, annuities, patents, licences, and any interest in real or personal property, and any claims against such property or against any person or company.
3.19To constitute any trusts with a view to the issue of preferred and deferred or other special stocks or securities based on or representing any shares, stocks and other assets specifically appropriated for the purpose of any such trust and to settle and regulate and if thought fit to undertake and execute any such trusts and to issue, dispose of or hold any such preferred, deferred or other special stocks or securities.
3.20To give any guarantee in relation to the payment of any debentures, debenture stock, bonds, obligations or securities and to guarantee the payment of interest thereon or of dividends on any stocks or shares of any company.
3.21To construct, erect and maintain buildings, houses, flats, shops and all other works, erections, and things of any description whatsoever either upon the lands acquired by the Company or upon other lands and to hold, retain as investments or to sell, let, alienate, mortgage, charge or deal with all or any of the same and generally to alter, develop and improve the lands and other property of the Company.
3.22To provide for the welfare of persons in the employment of or holding office under or formerly in the employment of or holding office under the Company including Directors and ex-Directors of the Company and the wives, widows and families,
4
dependants or connections of such persons by grants of money, pensions or other payments and by forming and contributing to pension, provident or benefit funds or profit sharing or co-partnership schemes for the benefit of such persons and to form, subscribe to or otherwise aid charitable, benevolent, religious, scientific, national or other institutions, exhibitions or objects which shall have any moral or other claims to support or aid by the Company by reason of the locality of its operation or otherwise.
3.23To remunerate by cash payments or allotment of shares or securities of the Company credited as fully paid up or otherwise any person or company for services rendered or to be rendered to the Company whether in the conduct or management of its business, or in placing or assisting to place or guaranteeing the placing of any of the shares of the Company’s capital, or any debentures or other securities of the Company or in or about the formation or promotion of the Company.
3.24To enter into and carry into effect any arrangement for joint working in business or for sharing of profits or for amalgamation with any other company or association or any partnership or person carrying on any business within the objects of the Company.
3.25To distribute in specie or otherwise as may be resolved, any assets of the Company among its members and in particular the shares, debentures or other securities of any other company belonging to this Company or of which this Company may have the power of disposing.
3.26To vest any real or personal property, rights or interest acquired or belonging to the Company in any person or company on behalf of or for the benefit of the Company, and with or without any declared trust in favour of the Company.
3.27To transact or carry on any business which may seem to be capable of being conveniently carried on in connection with any of these objects or calculated directly or indirectly to enhance the value of or facilitate the realisation of or render profitable any of the Company’s property or rights.
3.28To accept stock or shares in or debentures, mortgages or securities of any other company in payment or part payment for any services rendered or for any sale made to or debt owing from any such company, whether such shares shall be wholly or partly paid up.
3.29To pay all costs, charges and expenses incurred or sustained in or about the promotion and establishment of the Company or which the Company shall consider to be preliminary thereto and to issue shares as fully or in part paid up, and to pay out of the funds of the Company all brokerage and charges incidental thereto.
3.30To procure the Company to be registered or recognised in any part of the world.
3.31To do all or any of the matters hereby authorised in any part of the world or in conjunction with or as trustee or agent for any other company or person or by or through any factors, trustees or agents.
3.32To make gifts or grant bonuses to the Directors or any other persons who are or have been in the employment of the Company including substitute directors.
3.33To do all such other things that the Company may consider incidental or conducive to the attainment of the above objects or as are usually carried on in connection therewith.
3.34To carry on any business which the Company may lawfully engage in and to do all such things incidental or conducive to the business of the Company.
3.35To make or receive gifts by way of capital contribution or otherwise.
The objects set forth in any sub-clause of this clause shall be regarded as independent objects and shall not, except where the context expressly so requires, be in any way limited or
5
restricted by reference to or inference from the terms of any other sub-clause, or by the name of the Company. None of such sub-clauses or the objects therein specified or the powers thereby conferred shall be deemed subsidiary or auxiliary merely to the objects mentioned in the first sub-clause of this clause, but the Company shall have full power to exercise all or any of the powers conferred by any part of this clause in any part of the world notwithstanding that the business, property or acts proposed to be transacted, acquired or performed do not fall within the objects of the first sub-clause of this clause.
NOTE: It is hereby declared that the word “company” in this clause, except where used in reference to this Company shall be deemed to include any partnership, body corporate or other body of persons whether incorporated or not incorporated and whether domiciled in Ireland or elsewhere and the intention is that the objects specified in each paragraph of this clause shall except where otherwise expressed in such paragraph be in no way limited or restricted by reference to or inference from the terms of any other paragraph.
4.The share capital of the Company is US$3,005,000,000 and €25,000 divided into 500,000,000 Ordinary Shares of US$0.01, 3,000,000,000,000 Preferred Shares of US$0.001 each and 25,000 Ordinary Α Shares of €1.00 each.
5.The liability of the members is limited.
6.The shares forming the capital, increased or reduced, may be increased or reduced and be divided into such classes and issued with any special rights, privileges and conditions or with such qualifications as regards preference, dividend, capital, voting or other special incidents, and be held upon such terms as may be attached thereto or as may from time to time be provided by the original or any substituted or amended articles of association and regulations of the Company for the time being, but so that where shares are issued with any preferential or special rights attached thereto such rights shall not be alterable otherwise than pursuant to the provisions of the Company’s articles of association for the time being.
6
COMPANIES ACT 2014
A PUBLIC COMPANY LIMITED BY SHARES
ARTICLES OF ASSOCIATION
-of-
KEENOVA THERAPEUTICS PUBLIC LIMITED COMPANY
(as amended by Special Resolution passed on 8 October 2025)
PRELIMINARY
1.
(a)The provisions set out in these articles of association shall constitute the whole of the regulations applicable to the Company and no “optional provision” as defined by section 1007(2) of the Act with the exception of Sections 83 and 84 of the Act shall apply to the Company.
2.
(a)In these articles:
“Act” means the Companies Act 2014 and all other enactments and statutory instruments which are to be read as one with, or construed or read together as one with the Act and every statutory modification and re-enactment thereof for the time being in force.
“address” includes, without limitation, any number or address used for the purposes of communication by way of electronic mail or other electronic communication.
“Adoption Date” means the effective date of adoption of these articles.
“Assistant Secretary” means any person appointed by the Secretary from time to time to assist the Secretary.
“articles” means these articles of association of the Company, as amended from time to time by Special Resolution.
“Clear Days” in relation to the period of notice, means that period excluding the day when the notice is given or deemed to be given and the day for which it is given or on which it is to take effect.
“Chairman” means the Director who is elected by the Directors from time to time to preside as chairman at all meetings of the Board and at general meetings of the Company.
“Company” means the company whose name appears in the heading to these articles.
“Court” means the High Court of Ireland.
“Directors” or “Board” means the directors from time to time and for the time being of the Company or the directors present at a meeting of the board of directors and includes any person occupying the position of director by whatever name called.
7
“Disclosure Notice” means the notice issued in accordance with Section 1062 of the Act or other applicable law;
“electronic communication” has the meaning given to those words in the Electronic Commerce Act 2000.
“electronic signature” has the meaning given to those words in the Electronic Commerce Act 2000.
“Exchange Act” means the U.S. Securities Exchange Act of 1934, as amended and the rules promulgated thereunder.
“Group” means the Company and its subsidiaries from time to time and for the time being.
“Holder” in relation to any share, means the member whose name is entered in the Register as the holder of the share or, where the context permits, the members whose names are entered in the Register as the joint holders of shares.
“Office” means the registered office from time to time and for the time being of the Company.
“Ordinary Resolution” means an ordinary resolution of the Company’s members within the meaning of the Act.
“public announcement” means disclosure in a press release reported by a national news service or in a document publicly filed by the Company with the U.S. Securities and Exchange Commission pursuant to Section 13, 14 or 15(d) of the Exchange Act and the rules and regulations promulgated thereunder.
“Redeemable Shares” means redeemable shares in accordance with the Act.
“Register” means the register of members to be kept as required in accordance with the Act.
“seal” means the common seal of the Company and any duplicate of such common seal of the Company.
“Secretary” means any person appointed to perform the duties of the secretary of the Company and includes any Assistant Secretary or joint secretary.
“Special Resolution” means a special resolution of the Company’s members within the meaning of the Act.
(b)Expressions in these articles referring to writing shall be construed, unless the contrary intention appears, as including references to printing, lithography, photography and any other modes of representing or reproducing words in a visible form except as provided in these articles and/or where it constitutes writing in electronic form sent to the Company, and the Company has agreed to its receipt in such form. Expressions in these articles referring to execution of any document shall include any mode of execution whether under seal or under hand or any mode of electronic signature as shall be approved by the Directors. Expressions in these articles referring to receipt of any electronic communications shall, unless the contrary intention appears, be limited to receipt in such manner as the Company has approved.
8
(c)Unless the contrary intention appears, words or expressions contained in these articles shall bear the same meaning as in the Act or in any statutory modification thereof in force at the date at which these articles become binding on the Company.
(d)A reference to a statute or statutory provision shall be construed as a reference to the laws of Ireland unless otherwise specified and includes:
(i)any subordinate legislation made under it including all regulations, by-laws, orders and codes made thereunder;
(ii)any repealed statute or statutory provision which it re-enacts (with or without modification); and
(iii)any statute or statutory provision which modifies, consolidates, re-enacts or supersedes it.
(e)The masculine gender shall include the feminine and neuter, and vice versa, and the singular number shall include the plural, and vice versa, and words importing persons shall include firms or companies.
(f)Reference to US$, USD, or dollars shall mean the currency of the United States of America and to €, euro, EUR or cent shall mean the currency of Ireland.
SHARE CAPITAL AND VARIATION OF RIGHTS
3.
(a)The share capital of the Company is US$3,005,000,000 and €25,000 divided into 500,000,000 ordinary shares of US$0.01 each, 3,000,000,000,000 preferred shares of US$0.001 each and 25,000 ordinary A shares of €1.00 each.
(b)The rights and restrictions attaching to the ordinary shares shall be as follows:
(i)subject to the right of the Company to set record dates for the purposes of determining the identity of members entitled to notice of and/or to vote at a general meeting, the right to attend and speak at any general meeting of the Company and to exercise one vote per ordinary share held at any general meeting of the Company;
(ii)the right to participate pro rata in all dividends declared by the Company; and
(iii)the right, in the event of the Company’s winding up, to participate pro rata in the total assets of the Company.
The rights attaching to the ordinary shares may be subject to the terms of issue of any series or class of preferred shares allotted by the Directors from time to time in accordance with article 3(d).
(c)The Directors may issue and allot ordinary A shares subject to the rights, privileges, limitations and restrictions set out in this article 3(c):
(i)Income
The holder of an ordinary A share shall not be entitled to receive any dividend or distribution declared, made or paid or any return of capital (save as provided for in this article) and shall not entitle its holder to any further or other right of participation in the assets of the Company.
9
(ii)Capital
On a winding up of, or other return of capital (other than on a redemption of any class of shares in the capital of the Company) by the Company, the holders of ordinary A shares shall be entitled to participate in such return of capital or winding up of the Company, such entitlement to be limited to the repayment of the amount paid up or credited as paid up on such ordinary A shares and shall be paid only after the holders of ordinary shares shall have received payment in respect of such amount as is paid up or credited as paid up on those ordinary shares held by them at that time, plus the payment in cash of $100,000,000 on each such ordinary share.
(iii)Acquisition of Ordinary A Shares
The Company as agent for the holders of ordinary A shares shall have the irrevocable authority to authorise and instruct the Secretary (or any other person appointed for the purpose by the Directors) to acquire, or to accept the surrender of, the ordinary A shares for no consideration and to execute on behalf of such holders such documents as are necessary in connection with such acquisition or surrender, and pending such acquisition or surrender to retain the certificates, to the extent issued, for such ordinary A shares. Any request by the Company to acquire, or for the surrender of, any ordinary A shares may be made by the Directors depositing at the Office a notice addressed to such person as the Directors shall have nominated on behalf of the holders of ordinary A shares. A person whose shares have been acquired or surrendered in accordance with this article shall cease to be a member in respect of such ordinary A shares but shall notwithstanding remain liable to pay the Company all monies which, at the date of acquisition or surrender, were payable by him or her to the Company in respect of such shares, but his or her liability shall cease if and when the Company has received payment in full of all such monies in respect of such shares. A notice issued pursuant to this paragraph shall be deemed to be validly issued notwithstanding the provisions of articles 130 to 135 inclusive.
(iv)Voting
The holders of ordinary A shares shall not be entitled to receive notice of, nor attend, speak or vote at, any general meeting.
The rights attaching to the ordinary A shares may be subject to the terms of issue of any series or class of preferred shares allotted by the Directors from time to time in accordance with article 3(d).
(d)The Directors are authorised to issue all or any of the authorised but unissued preferred shares from time to time in one or more classes or series, and to fix for each such class or series such voting power, full or limited, or no voting power, and such designations, preferences and relative, participating, optional or other special rights and such qualifications, limitations or restrictions thereof, as shall be stated and expressed in the resolution or resolutions adopted by the Board providing for the issuance of such class or series, including, without limitation, the authority to provide that any such class or series may be:
(i)redeemable at the option of the Company, or the Holders, or both, with the manner of the redemption to be set by the Board including, without limitation that any such class or series may be redeemable
10
(A)at such time or times, including upon a fixed date;
(B)at such price or prices; and
(C)for such consideration which may be in cash or in specie (with the ability for each Holder to receive a particular form of consideration being subject to such additional requirements to be set by the Board from time to time);
(ii)entitled to receive dividends (which may be cumulative or non-cumulative) at such rates, on such conditions and at such times, and payable in preference to, or in such relation to, the dividends payable on any other class or classes of shares or any other series;
(iii)entitled to such rights upon the dissolution of, or upon any distribution of the assets of, the Company; or
(iv)convertible into, or exchangeable for, shares of any other class or classes of shares, or of any other series of the same or any other class or classes of shares, of the Company at such price or prices or at such rates of exchange and with such adjustments as the Directors determine,
which rights and restrictions may be as stated in such resolution or resolutions of the Directors as determined by them in accordance with this article 3(d). The Board may at any time before the allotment of any preferred share by further resolution in any way amend the designations, preferences, rights, qualifications, limitations or restrictions, or vary or revoke the designations of such preferred shares.
The rights conferred upon the Holder of any pre-existing shares in the share capital of the Company shall be deemed not to be varied by the creation, issue and allotment of preferred shares in accordance with this article 3(d).
(e)Unless the Board specifically resolves to treat such acquisition as a purchase for the purposes of the Act, an Ordinary Share shall be deemed to be a Redeemable Share on, and from the time of, the existence or creation of an agreement, transaction or trade between the Company and any third party pursuant to which the Company acquires or will acquire ordinary shares, or an interest in ordinary shares, from such third party and the Company is hereby authorised to enter into any such agreement, transaction or trade. In these circumstances, the acquisition of such shares or interest in shares by the Company, save where acquired otherwise than for valuable consideration in accordance with the Act, shall constitute the redemption of a Redeemable Share in accordance with the Act. No resolution, whether special or otherwise, shall be required to be passed to deem any Ordinary Share a Redeemable Share, or to authorise the redemption of such a Redeemable Share and once deemed to be a Redeemable Share such share shall be redeemable at the instance of the Company.
4.Subject to the provisions of the Act and the other provisions of these articles, the Company may:
(a)pursuant to the Act, issue any shares of the Company which are to be redeemed or are liable to be redeemed at the option of the Company or the member on such terms and in such manner as may be determined by the Company in general meeting (by Special Resolution) on the recommendation of the Directors; or
(b)subject to and in accordance with the provisions of the Act and without prejudice to any relevant special rights attached to any class of shares pursuant to the Act, purchase any of its own shares (including any Redeemable Shares and without any obligation to purchase on any pro rata basis as between members or members of the same class) and may cancel any shares so purchased or hold them as treasury shares (as defined in the Act) and may reissue any such shares as shares of any class or classes.
11
5.Without prejudice to any special rights previously conferred on the Holders of any existing shares or class of shares, any share in the Company may be issued with such preferred or deferred or other special rights or such restrictions, whether in regard to dividend, voting, return of capital or otherwise, as the Company may from time to time by Ordinary Resolution determine.
6.
(a)Without prejudice to the authority conferred on the Directors pursuant to article 3 to issue preferred shares in the capital of the Company, if at any time the share capital is divided into different classes of shares, the rights attached to any class may, whether or not the Company is being wound up, be varied or abrogated with the consent in writing of the Holders of three-fourths of the issued shares in that class, or with the sanction of a Special Resolution passed at a separate general meeting of the Holders of the shares of that class, provided that, if the relevant class of Holders has only one Holder, that person present in person or by proxy, shall constitute the necessary quorum. To every such meeting the provisions of article 39 shall apply.
(b)The redemption or purchase of preferred shares or any class of preferred shares shall not constitute a variation of rights of the preferred Holders.
(c)The issue, redemption or purchase of any of the 3,000,000,000,000 preferred shares of US$0.001 shall not constitute a variation of the rights of the Holders of ordinary shares.
(d)The issue of preferred shares or any class of preferred shares which rank pari passu with, or junior to, any existing preferred shares or class of preferred shares shall not constitute a variation of the existing preferred shares or class of preferred shares.
7.The rights conferred upon the Holders of the shares of any class issued with preferred or other rights shall not, unless otherwise expressly provided by the terms of issue of the shares of that class, be deemed to be varied by the creation or issue of further shares ranking pari passu therewith.
8.
(a)Subject to the provisions of these articles relating to new shares, the shares shall be at the disposal of the Directors (and/or by a committee of the Directors or by any other person where such committee or person is so authorised by the Directors), and they may (subject to the provisions of the Act) allot, grant options over or otherwise dispose of them to such persons, on such terms and conditions and at such times as they may consider to be in the best interests of the Company and its members, but so that no share shall be issued at a discount to its nominal value save in accordance with the Act, and so that, save where the Act permits otherwise, the amount payable on application on each share shall not be less than one-quarter of the nominal amount of the share and the whole of any premium thereon.
(b)Subject to any requirement to obtain the approval of members under any laws, regulations or the rules of any stock exchange to which the Company is subject, the Board is authorised, from time to time, in its discretion, to grant such persons, for such periods and upon such terms as the Board deems advisable, options to purchase or subscribe for such number of shares of any class or classes or of any series of any class as the Board may deem advisable, and to cause warrants or other appropriate instruments evidencing such options to be issued.
(c)The Directors are, for the purposes of section 1021 of the Act, generally and unconditionally authorised to exercise all powers of the Company to allot and issue relevant securities (as defined by the said section 1021) up to the amount of Company’s authorised share capital and to allot and issue any shares purchased by the Company pursuant to the provisions of the Act and held as treasury shares and this authority shall expire five years from the Adoption Date. The Company may before the expiry of such authority make an offer or agreement which would or might require relevant securities to be allotted after such expiry and the Directors may allot
12
relevant securities in pursuance of such an offer or agreement notwithstanding that the authority hereby conferred has expired.
(d)The Directors are hereby empowered pursuant to sections 1022 and 1023 of the Act to allot equity securities within the meaning of the said section 1023 of the Act for cash pursuant to the authority conferred by paragraph (c) of this article as if section 1022 of the said Act did not apply to any such allotment. The Company may before the expiry of such authority make an offer or agreement which would or might require equity securities to be allotted after such expiry and the Directors may allot equity securities in pursuance of such an offer or agreement as if the power conferred by this paragraph (d) had not expired.
(e)Nothing in these articles shall preclude the Directors from recognising a renunciation of the allotment of any shares by any allottee in favour of some other person.
9.If by the conditions of allotment of any share the whole or part of the amount or issue price thereof shall be payable by instalments, every such instalment when due shall be paid to the Company by the person who for the time being shall be the Holder of the share.
10.The Company may pay commission to any person in consideration of a person subscribing or agreeing to subscribe, whether absolutely or conditionally, for any shares in the Company or procuring or agreeing to procure subscriptions, whether absolute or conditional, for any shares in the Company on such terms and subject to such conditions as the Directors may determine, including, without limitation, by paying cash or allotting and issuing fully or partly paid shares or any combination of the two. The Company may also, on any issue of shares, pay such brokerage as may be lawful.
11.The Company shall maintain or cause to be maintained the Register in accordance with the Act.
12.If the Board considers it necessary or appropriate, the Company may establish and maintain a duplicate Register at such location or locations within or outside Ireland as the Board thinks fit. The original Register shall be treated as the register of members of the Company for the purposes of these articles and the Act.
13.The Company, or any agent(s) appointed by it to maintain the duplicate Register in accordance with these articles, shall as soon as practicable and on a regular basis record or procure the recording in the original Register of all transfers of shares effected on any duplicate Register and shall at all times maintain the original Register in such manner as to show at all times the member for the time being and the shares respectively held by them, in all respects in accordance with the Act.
14.Except as required by law, no person shall be recognised by the Company as holding any share upon any trust, and the Company shall not be bound by or be compelled in any way to recognise (even when having notice thereof) any equitable, contingent, future or partial interest in any share or any interest in any fractional part of a share or (except only as by these articles or by law otherwise provided) any other rights in respect of any share except an absolute right to the entirety thereof in the Holder. This shall not preclude the Company from requiring the members or the transferee of shares to furnish the Company with information as to the beneficial ownership of any share when such information is reasonably required by the Company.
15.
(a)The Directors may at any time and in their absolute discretion, if they consider it to be in the interests of the Company to do so, give to any shareholder or shareholders a notice (hereinafter referred to as an “Investigation Notice”) requiring such shareholder or shareholders to notify the Company in writing within the prescribed period of full and accurate particulars of all or any of the following matters, namely:
(i)his interest in any shares in the Company;
13
(ii)if his interest in the share does not consist of the entire beneficial interest in it, the interests of all persons having a beneficial interest in the share (provided that one joint shareholder of a share shall not be obliged to give particulars of interests of persons in the share which arise only through another joint shareholder of the Company); and
(iii)any arrangement (whether legally binding or not) entered into by him or any person having any beneficial interest in the share whereby it has been agreed or undertaken or the shareholder of such share can be required to transfer the share or any interest therein to any person (other than a joint shareholder of the share) or to act in relation to any meeting of the Company or of any class of shares of the Company in a particular way or in accordance with the wishes or directions of any other person (other than a person who is a joint shareholder of such share).
(b)Where an intermediary receives a Disclosure Notice and is in possession or control of the information which is required to be provided pursuant to these articles and applicable law, it shall as soon as practicable provide the Company with that information. Any intermediary that receives a Disclosure Notice and is not in possession or control of the information which is required to be provided pursuant to applicable law, shall as soon as practicable:
(i)inform the Company that it is not in possession or control of the information;
(ii)where the intermediary is part of a chain of intermediaries, transmit the request to each other intermediary in the chain known to the first mentioned intermediary as being part of the chain; and
(iii)provide the Company with the details of each intermediary, if any, to which the request has been transmitted under sub-paragraph (ii).
(c)If, pursuant to an Investigation Notice, the person stated to own any beneficial interest in a share or the person in favour of whom any shareholder (or other person having any beneficial interest in the share) has entered into any arrangements referred to in paragraph (a)(iii) is a body corporate, trust, society or any other legal entity or association of individuals and/or entities, the Directors may in their absolute discretion give a further Investigation Notice to the shareholders of, and/or any person whom such shareholder has stated as having any beneficial interest in, such a share requiring them to notify the Company in writing within the prescribed period of full and accurate particulars of the names and Addresses of the individuals who control (whether directly or indirectly and through any number of vehicles, entities or arrangements) the beneficial ownership of all the shares, interests, units or other measure of ownership of such body corporate, trust, society or other entity or association wherever the same shall be incorporated, registered or domiciled or wherever such individuals shall reside provided that if at any stage of such chain of ownership the beneficial interest in any share shall be established to the satisfaction of the Directors to be in the ownership of any body corporate any of whose share capital is listed or dealt in on any bona fide stock exchange, unlisted securities market or over the counter securities market, it shall not be necessary to disclose details of the individuals ultimately controlling the beneficial interests in the shares of such body corporate.
(d)Unless otherwise required by applicable law, where a notice is served pursuant to the terms of this article on the holder of a share and such holder is a central securities depository (or its nominee(s)) acting in its capacity as operator of a securities settlement system, the obligations of the central securities depository (or its nominee(s)) as a holder pursuant to this article shall be limited to disclosing to the Company in accordance with this article such information relating to the ownership of or interests in the share concerned as has been recorded by it pursuant to the rules made and practices instituted by the central securities depository, provided that nothing in this article shall in any other way restrict the powers of the Directors under this article. For the purposes of this article, a person, other than the holder of a share, shall be treated as appearing to be or to have been interested in that share if the holder
14
has informed the Company that the person is, or may be, or has been, or may have been, so interested, or if the Company (after taking account of any information obtained from the registered holder or, pursuant to a Disclosure Notice, from anyone else) knows or has reasonable cause to believe that the person is, or may be, or has been, or may have been, so interested.
(e)Where any member, or any other person with an interest in shares held by such member, is deemed by Section 1048 or 1050 of the Act to have an interest in 3% or more of the issued share capital of the Company, such member or person shall be required to notify the Company both of the existence of such interest and any event which results in the member or person ceasing to be so interested. Such notification shall be made in the same manner and within the same time period as specified in Sections 1052 and 1053 of the Act.
(f)If at any time the Directors are satisfied that:
(i)any member has been served with an Investigation Notice, or
(ii)any member, or any other person appearing to be interested in shares held by such member, has been served with a Disclosure Notice,
and is in default for the prescribed period in supplying to the Company the information thereby required, or, in purported compliance with such a notice has made a statement which is false or inadequate, then the Directors may, in their absolute discretion at any time thereafter by notice (a “Disenfranchisement Notice”) to such member direct that in respect of the shares in relation to which the default occurred (the “Default Shares”) (which expression shall include any further shares which are issued in respect of such shares) the member shall not be entitled to attend or to vote either personally or by proxy at a general meeting of the Company or a meeting of the holders of any class of shares of the Company or to exercise any other rights conferred by membership in relation to general meetings of the Company or meetings of the holders of any class of shares of the Company. Where a Disenfranchisement Notice is served on a central securities depository or its nominee(s) acting in its capacity as operator of a securities settlement system, the provisions of this article shall be treated as applying only to such number of shares as is equal to the number of Default Shares held by the central securities depository or its nominee(s) and not to any other shares held by the central securities depository or its nominee(s).
(g)Where the Default Shares represent at least three per cent. (3%) of the issued shares of that class (or such other percentage as may be determined under the provisions of Section 1052 of the Act), then the Disenfranchisement Notice may additionally direct that:
(i)any dividend (or part thereof) or other money which would otherwise be payable in respect of the Default Shares shall be retained by the Company without any liability to pay interest thereon when such money is finally paid to the member and/or
(ii)no transfer of any shares held by such member shall be registered unless;
(A)the member is not himself in default as regards supplying the information required; and
(B)the transfer is part only of the member's holding and when presented for registration is accompanied by a certificate by the member in a form satisfactory to the Directors to the effect that after due and careful enquiry, the member is satisfied that none of the shares, the subject of the transfer, is a Default Share.
15
(h)The Company shall send to each other person appearing to be interested in the shares, the subject of any Disenfranchisement Notice, a copy of the Disenfranchisement Notice but the failure or omission by the Company to do so shall not invalidate such Disenfranchisement Notice.
(i)Save as herein provided, any Disenfranchisement Notice shall have effect in accordance with its terms for so long as the default in respect of which the Disenfranchisement Notice was issued continues and for a period of one week thereafter provided that the Directors may at the request of the member concerned reduce or waive such one week period if they think fit.
(j)Any Disenfranchisement Notice shall cease to have effect in relation to any shares which are transferred by such member by means of an Approved Transfer (as defined in article 15(k)(iii)).
(k)For the purpose of this article:
(i)a person shall be treated as appearing to be interested in any shares if the member holding such shares has in response to a Disclosure Notice or Investigation Notice either:
(A)named such person as being so interested; or
(B)fails to establish the identities of those interested in the shares;
and (after taking into account the said response and any other relevant Disclosure Notice or Investigation Notice) the Company knows or has reasonable cause to believe that the person in question is or may be interested in the Shares;
(ii)in the case of both an Investigation Notice and a Disclosure Notice, the prescribed period is twenty-eight days from the date of service of the notice except that if the Default Shares represent at least five per cent. (5%) of the issued shares of that class, the prescribed period is fourteen days from such date; and
(iii)a transfer of shares is an “Approved Transfer” if, but only if:
(A)it is a transfer of shares to an offeror by way of or in pursuance of an acceptance of a takeover offer, merger, scheme or arrangement or consolidation of the Company; or
(B)the Directors are satisfied that the transfer is made pursuant to a sale of the whole of the beneficial ownership of the shares to a party unconnected with the member and with other persons appearing to be interested in such shares; or
(C)the transfer results from a sale made through a recognised stock exchange.
16.No person shall be entitled to a share certificate in respect of any Ordinary Share held by them in the share capital of the Company, whether such Ordinary Share was allotted or transferred to them, and the Company shall not be bound to issue a share certificate to any such person entered in the Register.
17.The Company shall not give, whether directly or indirectly and whether by means of a loan, guarantee, the provision of security or otherwise, any financial assistance for the purpose of a purchase or subscription made or to be made by any person of or for any shares in the Company or in its holding company, except as permitted by the Act.
18.
16
(a)The Company shall have a first and paramount lien on every share (not being a fully paid share) for all moneys (whether presently payable or not) payable at a fixed time or called in respect of that share. The Directors, at any time, may declare any share to be wholly or in part exempt from the provisions of this article. The Company’s lien on a share shall extend to all moneys payable in respect of it.
(b)The Company may sell in such manner as the Directors determine any share on which the Company has a lien if a sum in respect of which the lien exists is presently payable and is not paid within fourteen Clear Days after notice demanding payment, and stating that if the notice is not complied with the share may be sold, has been given to the Holder of the share or to the person entitled to it by reason of the death or bankruptcy of the Holder.
(c)To give effect to a sale, the Directors may authorise some person to execute an instrument of transfer of the share sold to, or in accordance with the directions of, the purchaser. The transferee shall be entered in the Register as the Holder of the share comprised in any such transfer and he shall not be bound to see to the application of the purchase moneys nor shall his title to the share be affected by any irregularity in or invalidity of the proceedings in reference to the sale, and after the name of the transferee has been entered in the Register, the remedy of any person aggrieved by the sale shall be in damages only and against the Company exclusively.
(d)The net proceeds of the sale, after payment of the costs, shall be applied in payment of so much of the sum for which the lien exists as is presently payable and any residue (upon surrender to the Company for cancellation of the certificate for the shares sold and subject to a like lien for any moneys not presently payable as existed upon the shares before the sale) shall be paid to the person entitled to the shares at the date of the sale.
19.
(a)Subject to the terms of allotment, the Directors may make calls upon the members in respect of any moneys unpaid on their shares and each member (subject to receiving at least fourteen Clear Days’ notice specifying when and where payment is to be made) shall pay to the Company as required by the notice the amount called on his shares. A call may be required to be paid by instalments. A call may be revoked before receipt by the Company of a sum due thereunder, in whole or in part and payment of a call may be postponed in whole or in part. A person upon whom a call is made shall remain liable for calls made upon him notwithstanding the subsequent transfer of the shares in respect of which the call was made.
(b)A call shall be deemed to have been made at the time when the resolution of the Directors authorising the call was passed.
(c)The joint Holders of a share shall be jointly and severally liable to pay all calls in respect thereof.
(d)If a call remains unpaid after it has become due and payable the person from whom it is due and payable shall pay interest on the amount unpaid from the day it became due until it is paid at the rate fixed by the terms of allotment of the share or in the notice of the call or, if no rate is fixed, at the appropriate rate (as defined by the Act) but the Directors may waive payment of the interest wholly or in part.
(e)An amount payable in respect of a share on allotment or at any fixed date, whether in respect of nominal value or as an instalment of a call, shall be deemed to be a call and if it is not paid the provisions of these articles shall apply as if that amount had become due and payable by virtue of a call.
(f)Subject to the terms of allotment, the Directors may make arrangements on the issue of shares for a difference between the Holders in the amounts and times of payment of calls on their shares.
17
(g)The Directors, if they think fit, may receive from any member willing to advance the same all or any part of the moneys uncalled and unpaid upon any shares held by him, and upon all or any of the moneys so advanced may pay (until the same would, but for such advance, become payable) interest at such rate, not exceeding (unless the Company in general meeting otherwise directs) fifteen percent per annum, as may be agreed upon between the Directors and the member paying such sum in advance.
(h)
(i)If a member fails to pay any call or instalment of a call on the day appointed for payment thereof, the Directors, at any time thereafter and during such times as any part of the call or instalment remains unpaid, may serve a notice on him requiring payment of so much of the call or instalment as is unpaid together with any interest which may have accrued.
(ii)The notice shall name a further day (not earlier than the expiration of fourteen Clear Days from the date of service of the notice) on or before which the payment required by the notice is to be made, and shall state that in the event of non-payment at or before the time appointed the shares in respect of which the call was made will be liable to be forfeited.
(iii)If the requirements of any such notice as aforesaid are not complied with then, at any time thereafter before the payment required by the notice has been made, any shares in respect of which the notice has been given may be forfeited by a resolution of the Directors to that effect. The forfeiture shall include all dividends or other moneys payable in respect of the forfeited shares and not paid before forfeiture. The Directors may accept a surrender of any share liable to be forfeited hereunder.
(iv)On the trial or hearing of any action for the recovery of any money due for any call it shall be sufficient to prove that the name of the member sued is entered in the Register as the Holder, or one of the Holders, of the shares in respect of which such debt accrued, that the resolution making the call is duly recorded in the minute book and that notice of such call was duly given to the member sued, in pursuance of these articles, and it shall not be necessary to prove the appointment of the Directors who made such call nor any other matters whatsoever, but the proof of the matters aforesaid shall be conclusive evidence of the debt.
(i)Α forfeited share may be sold or otherwise disposed of on such terms and in such manner as the Directors think fit and at any time before a sale or disposition the forfeiture may be cancelled on such terms as the Directors think fit. Where for the purposes of its disposal such a share is to be transferred to any person, the Directors may authorise some person to execute an instrument of transfer of the share to that person. The Company may receive the consideration, if any, given for the share on any sale or disposition thereof and may execute a transfer of the share in favour of the person to whom the share is sold or disposed of and thereupon he shall be registered as the Holder of the share and shall not be bound to see to the application of the purchase money, if any, nor shall his title to the share be affected by any irregularity or invalidity in the proceedings in reference to the forfeiture, sale or disposal of the share.
(j)Α person whose shares have been forfeited shall cease to be a member in respect of the forfeited shares, but nevertheless shall remain liable to pay to the Company all moneys which, at the date of forfeiture, were payable by him to the Company in respect of the shares, without any deduction or allowance for the value of the shares at the time of forfeiture but his liability shall cease if and when the Company shall have received payment in full of all such moneys in respect of the shares.
(k)Α statutory declaration that the declarant is a Director or the Secretary of the Company, and that a share in the Company has been duly forfeited on the date stated in the declaration, shall be conclusive evidence of the facts therein stated as against all persons claiming to be entitled to the share.
18
(l)The provisions of these articles as to forfeiture shall apply in the case of non-payment of any sum which, by the terms of issue of a share, becomes payable at a fixed time, whether on account of the nominal value of the share or by way of premium, as if the same had been payable by virtue of a call duly made and notified.
(m)The Directors may accept the surrender of any share which the Directors have resolved to have been forfeited upon such terms and conditions as may be agreed and, subject to any such terms and conditions, a surrendered share shall be treated as if it has been forfeited.
TRANSFER OF SHARES
20.
(a)The instrument of transfer of any share may be executed for and on behalf of the transferor by the Secretary, an Assistant Secretary or any such person that the Secretary or an Assistant Secretary nominates for that purpose (whether in respect of specific transfers or pursuant to a general standing authorisation), and the Secretary, Assistant Secretary or the relevant nominee shall be deemed to have been irrevocably appointed agent for the transferor of such share or shares with full power to execute, complete and deliver in the name of and on behalf of the transferor of such share or shares all such transfers of shares held by the members in the share capital of the Company. Any document which records the name of the transferor, the name of the transferee, the class and number of shares agreed to be transferred, the date of the agreement to transfer shares and the price per share, shall, once executed by the transferor or the Secretary, Assistant Secretary or the relevant nominee as agent for the transferor, and by the transferee where required by the Act, be deemed to be a proper instrument of transfer for the purposes of the Act. The transferor shall be deemed to remain the Holder of the share until the name of the transferee is entered on the Register in respect thereof, and neither the title of the transferee nor the title of the transferor shall be affected by any irregularity or invalidity in the proceedings in reference to the sale should the Directors so determine.
(b)The Company, at its absolute discretion, may, or may procure that a subsidiary of the Company shall, pay Irish stamp duty arising on a transfer of shares on behalf of the transferee of such shares of the Company. If stamp duty resulting from the transfer of shares in the Company which would otherwise be payable by the transferee is paid by the Company or any subsidiary of the Company on behalf of the transferee, then in those circumstances, the Company shall, on its behalf or on behalf of its subsidiary (as the case may be), be entitled to (i) seek reimbursement of the stamp duty from the transferee or transferor, (ii) set-off the stamp duty against any dividends payable to the transferee of those shares and (iii) claim a first and paramount lien on the shares on which stamp duty has been paid by the Company or its subsidiary for the amount of stamp duty paid. The Company’s lien shall extend to all dividends paid on those shares.
(c)Notwithstanding the provisions of these articles and subject to any regulations made under section 1086 of the Act, title to any shares in the Company may also be evidenced and transferred without a written instrument in accordance with section 1086 of the Act or any regulations made thereunder. The Directors shall have power to permit any class of shares to be held in uncertificated form and to implement any arrangements they think fit for such evidencing and transfer which accord with such regulations and in particular shall, where appropriate, be entitled to disapply or modify all or part of the provisions in these articles with respect to the requirement for written instruments of transfer and share certificates (if any), in order to give effect to such regulations.
21.Subject to such of the restrictions of these articles and to such of the conditions of issue of any share warrants as may be applicable, the shares of any member and any share warrant may be transferred by instrument in writing in any usual or common form or any other form which the Directors may approve.
19
22.
(a)The Directors in their absolute discretion and without assigning any reason therefor may decline to register:
(i)any transfer of a share which is not fully paid; or
(ii)any transfer to or by a minor or person of unsound mind;
but this shall not apply to a transfer of such a share resulting from a sale of the share through a stock exchange on which the share is listed.
(b)The Directors may decline to recognise any instrument of transfer unless:
(i)the instrument of transfer is accompanied by any evidence the Directors may reasonably require to show the right of the transferor to make the transfer;
(ii)the instrument of transfer is in respect of one class of share only;
(iii)the instrument of transfer is properly stamped (in circumstances where stamping is required);
(iv)a fee of €10 or such lesser sum as the Directors may from time to time require, is paid to the Company in respect of it;
(v)the instrument of transfer is in favour of not more than four transferees;
(vi)it is lodged at the Office or at such other place as the Directors may appoint;
(vii)the Board is satisfied, acting reasonably, that all applicable consents, authorisations, permissions or approvals of any governmental body or agency in Ireland and any other applicable jurisdiction required to be obtained under relevant law prior to such transfer have been obtained; and
(viii)the Board is satisfied, acting reasonably, that the transfer would not violate the terms of any agreement to which the Company (or any of its subsidiaries) and the transferor are party to or subject.
23.If the Directors refuse to register a transfer, they shall, within two months after the date on which the transfer was lodged with the Company, send to the transferee notice of the refusal.
24.
(a)The Directors may from time to time fix a record date for the purposes of determining the rights of members to notice of and/or to vote at any general meeting of the Company. The record date shall not precede the date upon which the resolution fixing the record date is adopted by the Directors, and the record date shall be not more than eighty nor less than ten days before the date of such meeting. If no record date is fixed by the Directors, the record date for determining members entitled to notice of or to vote at a meeting of the members shall be the close of business on the day next preceding the day on which notice is given. Unless the Directors determine otherwise, a determination of members of record entitled to notice of or to vote at a meeting of members shall apply to any adjournment, recess, rescheduling or postponement of the meeting.
(b)In order that the Directors may determine the members entitled to receive payment of any dividend or other distribution or allotment of any rights or the members entitled to exercise any rights in respect of any change, conversion or exchange of shares, or for the purpose of any other lawful action, the Board may fix a record date, which record date shall not precede the date upon which the resolution fixing the record date is adopted, and which record date shall be not more than thirty nor less than two days prior to such action. If no record date is fixed, the record date for determining
20
members for such purpose shall be at the close of business on the day on which the Directors adopt the resolution relating thereto.
25.Registration of transfers may be suspended at such times and for such period, not exceeding in the whole 30 days in each year, as the Directors may from time to time determine subject to the requirements of the Act.
26.All instruments of transfer shall upon their being lodged with the Company remain the property of the Company and the Company shall be entitled to retain them.
27.Subject to the provisions of these articles, whenever as a result of a consolidation of shares or otherwise any members would become entitled to fractions of a share, the Directors may sell or cause to be sold, on behalf of those members, the shares representing the fractions for the best price reasonably obtainable to any person and distribute the proceeds of sale (subject to any applicable tax and abandoned property laws) in due proportion among those members, and the Directors may authorise some person to execute an instrument of transfer of the shares to, or in accordance with the directions of, the purchaser. The transferee shall not be bound to see to the application of the purchase money nor shall his title to the shares be affected by any irregularity in or invalidity of the proceedings in reference to the sale.
TRANSMISSION OF SHARES
28.In the case of the death of a member, the survivor or survivors where the deceased was a joint Holder, and the personal representatives of the deceased where he was a sole Holder, shall be the only persons recognised by the Company as having any title to his interest in the shares; but nothing herein contained shall release the estate of a deceased joint Holder from any liability in respect of any share which had been jointly held by him with other persons.
29.Any person becoming entitled to a share in consequence of the death or bankruptcy of a member may, upon such evidence being produced as may from time to time properly be required by the Directors and subject as herein provided, elect either to be registered himself as Holder of the share or to have some person nominated by him registered as the transferee thereof, but the Directors shall, in either case, have the same right to decline or suspend registration as they would have had in the case of a transfer of the shares by that member before his death or bankruptcy, as the case may be.
30.If the person so becoming entitled elects to be registered himself, he shall deliver or send to the Company a notice in writing signed by him stating that he so elects. If he elects to have another person registered, he shall testify his election by executing to that person a transfer of the share. All the limitations, restrictions and provisions of these articles relating to the right to transfer and the registration of transfers of shares shall be applicable to any such notice or transfer as aforesaid as if the death or bankruptcy of the member had not occurred and the notice of transfer were a transfer signed by that member.
31.A person becoming entitled to a share by reason of the death or bankruptcy of the Holder shall be entitled to the same dividends and other advantages to which he would be entitled if he were the registered Holder of the share, except that he shall not, before being registered as a member in respect of the share, be entitled in respect of it to exercise any right conferred by membership in relation to the meetings of the Company, so, however, that the Directors may at any time give notice requiring such person to elect either to be registered himself or to transfer the share, and if the notice is not complied with within 90 days, the Directors may thereupon withhold payment of all dividends, bonuses or other moneys payable in respect of the share until the requirements of the notice have been complied with.
ALTERATION OF CAPITAL
32.
21
(a)The Company may from time to time by Ordinary Resolution increase the authorised share capital by such sum, to be divided into shares of such amount, as the resolution shall prescribe.
(b)Subject to the provisions of the Act, the new shares shall be issued upon such terms and conditions and with such rights and privileges annexed thereto as the general meeting resolving upon the creation thereof shall direct, or if no such direction be given, as the Directors shall determine.
(c)Except so far as otherwise provided by the conditions of issue or by these articles, any capital raised by the creation of new shares shall be subject to the provisions herein contained with reference to the payment of calls and instalments, transfer and transmission, forfeiture, lien and otherwise.
33.The Company may by Ordinary Resolution:
(a)divide its share capital into several classes and attach to them respectively any preferential, deferred, qualified or special rights, privileges or conditions;
(b)consolidate and divide all or any of its share capital into shares of larger amount than its existing shares;
(c)subdivide its existing shares, or any of them, into shares of smaller amount than is fixed by the memorandum of association subject, nevertheless, to the Act; or
(d)cancel any shares which, at the date of the passing of the resolution, have not been taken or agreed to be taken by any person and reduce the amount of its authorised share capital by the amount of the shares so cancelled; and
(e)subject to applicable law, change the currency denomination of its share capital.
34.The Company may by Special Resolution (or by Ordinary Resolution where permitted by section 83 of the Act) reduce its company capital (including its share capital, any capital redemption reserve fund or any share premium account or undenominated capital account) in any manner and with and subject to any incident authorised, and consent required, by law.
GENERAL MEETINGS
35.The Company shall in each year hold a general meeting as its annual general meeting in addition to any other meeting in that year, and shall specify the meeting as such in the notices calling it. Not more than fifteen months shall elapse between the date of one annual general meeting of the Company and that of the next.
36.Subject to the Act, all general meetings of the Company may be held outside of Ireland.
37.All general meetings other than annual general meetings shall be called extraordinary general meetings.
38.The Directors may, whenever they think fit, convene an extraordinary general meeting, and extraordinary general meetings shall also be convened on such requisition, or in default may be convened by such requisitionists, as provided in section 178(3) of the Act.
39.All provisions of these articles relating to general meetings of the Company shall, mutatis mutandis, apply to every separate general meeting of the Holders of any class of shares in the capital of the Company, except that:
(a)the necessary quorum shall be two or more persons holding or representing by proxy (whether or not such Holder actually exercises his voting rights in whole, in part or at all at the relevant general meeting) at least one-half in nominal value of the issued shares of the class or, at any adjourned meeting of such Holders, one Holder present
22
in person or by proxy, whatever the amount of his holding, shall be deemed to constitute a meeting; and
(b)each Holder of shares of the class shall have one vote in respect of every share of the class held by him.
40.A Director shall be entitled, notwithstanding that he is not a member, to attend and speak at any general meeting and at any separate meeting of the Holders of any class of shares in the Company.
NOTICE OF GENERAL MEETINGS
41.
(a)Subject to the provisions of the Act allowing a general meeting to be called by shorter notice, an annual general meeting, and an extraordinary general meeting called for the passing of a special resolution, shall be called by not less than twenty-one Clear Days’ notice and all other extraordinary general meetings shall be called by not less than fourteen Clear Days’ notice.
(b)Any notice convening a general meeting shall specify the time and place of the meeting and, in the case of special business, the general nature of that business and, in reasonable prominence, that a member entitled to attend and vote is entitled to appoint a proxy to attend, speak and vote in his place and that a proxy need not be a member of the Company. It shall also give particulars of any Directors who are to retire at the meeting and of any persons who are recommended by the Directors for appointment or re-appointment as Directors at the meeting or in respect of whom notice has been duly given to the Company of the intention to propose them for appointment or re-appointment as Directors at the meeting. Provided that the latter requirement shall only apply where the intention to propose the person has been received by the Company in accordance with, and the proposal is otherwise made in compliance with, the provisions of these articles, including articles 44 to 45 and articles 56 to 65. Subject to any restrictions imposed on any shares, the notice of the meeting shall be given to all the members of the Company as of the record date set by the Directors and to the Directors and the Auditors.
(c)The accidental omission to give notice of a meeting to, or the non-receipt of notice of a meeting by, any person entitled to receive notice shall not invalidate the proceedings at the meeting.
42.Where, by any provision contained in the Act, extended notice is required of a resolution, the resolution shall not be effective (except where the Directors of the Company have resolved to submit it) unless notice of the intention to move it has been given to the Company not less than twenty-eight days (or such shorter period as the Act permit) before the meeting at which it is moved, and the Company shall give to the members notice of any such resolution as required by and in accordance with the provisions of the Act.
PROCEEDINGS AT GENERAL MEETINGS
43.All business shall be deemed special that is transacted at an extraordinary general meeting, and also all that is transacted at an annual general meeting, with the exception of the review by the members of the Company’s affairs declaring a dividend, the consideration of the Company’s statutory financial statements and the reports of the Directors and auditors, the election of Directors, the re-appointment of the retiring auditors and the fixing of the remuneration of the auditors.
44.At any annual general meeting of the members, only such nominations of individuals for election to the Board shall be made, and only such other business shall be conducted or considered, as shall have been properly brought before the meeting. For nominations to be properly made at an annual general meeting, and for other business to be properly brought before an annual general meeting, such nominations and other
23
business must be: (a) specified in the Company’s notice of meeting (or any supplement thereto) given by or at the direction of the Board; (b) otherwise made at the annual general meeting, by or at the direction of the Board; (c) or otherwise properly requested to be brought before the annual general meeting by a member of the Company in accordance with this article 44 and articles 56 to 65. For nominations of individuals for election to the Board or other business to be properly requested by a member to be made at or brought before an annual general meeting pursuant to this article 44(c), a member must (i) be a member at the time of giving of notice of such annual general meeting by or at the direction of the Board, on the record date for determination of members entitled to vote at such meeting, and at the time of the annual general meeting, (ii) be entitled to vote at such annual general meeting and (iii) comply with the procedures set forth in these articles as to such nomination or other business. This article 44 shall be the exclusive means for a member to make nominations or to bring other business (other than matters properly brought under Rule 14a-8 under the Exchange Act and included in the Company’s notice of meeting) before an annual general meeting of members.
45.At any extraordinary general meeting of the members, only such business shall be conducted or considered, as shall have been properly brought before the meeting. To be properly brought before an extraordinary general meeting, such business must be (a) specified in the Company’s notice of meeting (or any supplement thereto) given by or at the direction of the Board, (b) otherwise brought before the extraordinary general meeting, by or at the direction of the Board, or (c) otherwise properly brought before the extraordinary general meeting by any members of the Company pursuant to the valid exercise of power granted to them under the Act to submit a requisition to the Company to convene an extraordinary general meeting (such requisition, an “EGM Request”) and in accordance with this article 45 and articles 56 to 65; provided, however, that nothing herein shall prohibit the Board from submitting additional matters to members at any such extraordinary general meeting. Nominations of persons for election to the Board may be made at an extraordinary general meeting of members at which Directors are to be elected pursuant to the Company’s notice of meeting (a) by or at the direction of the Board, or (b) by any member of the Company who (i) is a member at the time of giving of notice of such extraordinary general meeting, on the record date for determination of members entitled to vote at such meeting and at the time of the extraordinary general meeting, (ii) is entitled to vote at the meeting and (iii) complies with the procedures set forth in these articles as to such nomination. This article 45 shall be the exclusive means for a member to make nominations or bring other business (other than matters properly brought under Rule 14a-8 under the Exchange Act and included in the Company’s notice of meeting) before an extraordinary general meeting of members. For the avoidance of doubt, nothing in these articles shall be construed to confer on any member the right to request an extraordinary general meeting or to make nominations or to bring any other business at any extraordinary general meeting (whether requisitioned by members pursuant to an EGM Request or otherwise), or to expand any rights that members have with respect to any extraordinary general meeting that is held in connection with an EGM Request (including any such extraordinary general meeting that is convened by the Board in response to an EGM Request) beyond those rights provided to members under the Act.
46.No business shall be transacted at any general meeting unless a quorum is present at the time when the meeting proceeds to business. One or more Holders of shares, present in person or by proxy (whether or not such Holder actually exercises his voting rights in whole, in part or at all at the relevant general meeting), entitling them to exercise a majority of the voting power of the Company on the relevant record date shall constitute a quorum.
47.Any general meeting duly called at which a quorum is not present shall be adjourned and the Company shall provide notice pursuant to article 41 in the event that such meeting is to be reconvened.
48.The Chairman, if any, of the Board shall preside as Chairman at every general meeting of the Company, or if there is no such Chairman, or if he is not present at the time appointed for the holding of the meeting or is unwilling to act, any Director of
24
the Company or any other person nominated by the Board (or if the Board has not nominated any such person prior to the meeting or the Chairman or such person is not present within fifteen minutes after the time appointed for the holding of the meeting or is unwilling to act, such an officer or Director or any other person elected by the Directors present at the meeting) shall preside as Chairman of the meeting.
49.If at any general meeting no person nominated in accordance with article 48 is willing to act as Chairman or if no such person is present within fifteen minutes after the time appointed for holding the meeting, the members present shall choose one of their number to be Chairman of the meeting.
50.The Chairman may, with the consent of any meeting at which a quorum is present, and shall if so directed by the meeting, adjourn the meeting from time to time and from place to place without notice other than by announcement of the time and place of the adjourned meeting by the Chairman of the meeting. The Chairman of the meeting may at any time without the consent of the meeting adjourn the meeting to another time and/or place if, in his opinion, it would facilitate the conduct of the business of the meeting to do so or if he is so directed by the Board. Save as aforesaid, it shall not be necessary to give any notice of an adjournment or of the business to be transacted at an adjourned meeting.
51.At any general meeting a resolution put to the vote of the meeting shall be decided by poll.
52.A poll shall be taken in such manner as the Chairman directs, and the result of the poll shall be deemed to be the resolution of the meeting.
53.On a poll taken at any general meeting or any separate meeting of the Holders of any class of shares in the Company, a Holder, whether present in person or by proxy, entitled to more than one vote need not, if he votes, use all his votes or cast all the votes he uses in the same way.
54.Where there is an equality of votes, the Chairman of the meeting shall be entitled to a casting vote in addition to any other vote he may have.
55.Unless the Directors otherwise determine, no member shall be entitled to vote at any general meeting or any separate meeting of the Holders of any class of shares in the Company, either in person or by proxy, or to exercise any privilege as a member in respect of any share held by him unless all monies then payable by him in respect of that share have been paid.
ADVANCE NOTICE OF MEMBER BUSINESS AND NOMINATIONS
56.Subject to article 63, but otherwise without qualification or limitation, for any nominations or any other business to be properly brought before an annual general meeting by a member pursuant to article 44, the member must have given timely notice thereof in writing to the Secretary in proper form, and in accordance with this article 56 and articles 58 to 65, and such other business must otherwise be a proper matter for member action.
(a)To be timely, a member’s notice shall be delivered to the Secretary at the Office not earlier than the close of business on the one hundred and twentieth (120th) day and not later than the close of business on the ninetieth (90th) day prior to the first anniversary of the preceding year’s annual general meeting; provided, however, that in the event that no annual general meeting was held in the previous year or the date of the annual general meeting is more than thirty (30) days before or more than sixty (60) days after such anniversary date, notice by the member must be so delivered not earlier than the close of business on the one hundred and twentieth (120th) day prior to the date of such annual general meeting and not later than the close of business on the later of the ninetieth (90th) day prior to the date of such annual general meeting or, if the first public announcement of the date of such annual general meeting is less than one hundred (100) days prior to the date of such annual general meeting, the tenth (10th) day following the day on which public announcement of the date of such
25
meeting is first made by the Company. In no event shall any adjournment, recess, rescheduling or postponement of an annual general meeting, or the public announcement thereof, commence a new time period for the giving of a member’s notice as described above. For the avoidance of doubt, a member shall not be entitled to make additional or substitute nominations following the expiration of the time periods set forth in these articles.
(b)Notwithstanding anything in article 56(a) to the contrary, in the event that the number of Directors to be elected to the Board is increased by the Board, and there is no public announcement by the Company naming all of the nominees for Director or specifying the size of the increased Board at least ten (10) days prior to the deadline for nominations that would otherwise be applicable under this article 56, a member’s notice required by this article 56 shall also be considered timely, but only with respect to nominees for any new positions created by such increase, if it shall be delivered to the Secretary at the Office not later than the close of business on the tenth (10th) day following the day on which such public announcement is first made by the Company.
57.Subject to article 63, but otherwise without qualification or limitation, for any business to be properly requested to be brought before an extraordinary general meeting by a member, the member must have given timely notice thereof in writing to the Secretary in proper form and in accordance with this article 57 and articles 58 to 65, and such business must otherwise be a proper matter for member action.
(a)Subject to article 63, in the event that an extraordinary general meeting of members is called by a member of the Company pursuant to an EGM Request, the purpose of which is the election of one or more Directors to the Board, the right of any member to nominate an individual or individuals (as the case may be) for election to such position(s) as specified in the Company’s notice of meeting shall be subject to such member having given timely notice thereof. To be timely, a member’s notice shall be delivered to the Secretary at the Office not earlier than the close of business on the one hundred and twentieth (120th) day prior to the date of such extraordinary general meeting and not later than the close of business on the later of the ninetieth (90th) day prior to the date of such extraordinary general meeting or, if the first public announcement of the date of such extraordinary general meeting is less than one hundred (100) days prior to the date of such extraordinary general meeting, the tenth (10th) day following the day on which public announcement is first made of the date of the extraordinary general meeting and, if applicable, of the nominees proposed by the Board to be elected at such meeting. In no event shall any adjournment, recess, rescheduling or postponement of an extraordinary general meeting of members, or the public announcement thereof, commence a new time period for the giving of a member’s notice as described above. For the avoidance of doubt, a member shall not be entitled to make additional or substitute nominations following the expiration of the time periods set forth in these articles.
(b)Notwithstanding anything in article 57(a) to the contrary, in the event that the number of Directors to be elected to the Board is increased by the Board, and there is no public announcement by the Company naming all of the nominees for Director or specifying the size of the increased Board at least ten (10) days prior to the deadline for nominations that would otherwise be applicable under this article 57, a member’s notice required by this article 57 shall also be considered timely, but only with respect to nominees for any new positions created by such increase, if it shall be delivered to the Secretary at the Office not later than the close of business on the 10th day following the day on which such public announcement is first made by the Company.
58.Without prejudice to the generality of articles 56 and 57, to be in proper form, a member’s notice pursuant to articles 44 to 45 and articles 56 to 65 must include the following, as applicable:
(a)As to the member giving the notice and the beneficial owner, if any, on whose behalf the nomination is made or business is brought, as applicable, a member’s notice must set forth: (i) the name and address of such member, as they appear on the Register, of such beneficial owner, if any, and any persons that are acting in concert therewith: (ii) a representation that the member giving the notice is a Holder of shares carrying
26
voting rights entitled to vote at such meeting, will continue to be a Holder of shares carrying voting rights entitled to vote at such meeting through the date of such meeting and intends to appear in person or by proxy at the meeting to make such nomination or to propose such business; (iii) (A) the class or series and number of shares of the Company which are, directly or indirectly, owned of record and owned beneficially by such member, such beneficial owner and their respective affiliates or associates or others acting in concert therewith, (B) any option, warrant, convertible security, share appreciation right, or similar right with an exercise or conversion privilege or a settlement payment or mechanism at a price related to any security of the Company or with a value derived in whole or in part from the value of any security of the Company, or any derivative or synthetic arrangement having the characteristics of a long position in any security of the Company, or any contract, derivative, swap or other transaction or series of transactions designed to produce economic benefits and risks that correspond substantially to the ownership of any security of the Company, including due to the fact that the value of such contract, derivative, swap or other transaction or series of transactions is determined by reference to the price, value or volatility of any security of the Company, whether or not such instrument, contract or right shall be subject to settlement in the underlying securities of the Company, through the delivery of cash or other property, or otherwise, and without regard to whether the Holder, the beneficial owner, if any, or any of their respective affiliates, or associates or others acting in concert therewith, may have entered into transactions that hedge or mitigate the economic effect of such instrument, contract or right, or any other direct or indirect opportunity to profit or share in any profit derived from any increase or decrease in the value of securities of the Company (any of the foregoing, a “Derivative Instrument”) directly or indirectly owned beneficially by such member, the beneficial owner, if any, or any of their respective affiliates or associates, or others acting in concert therewith, (C) any proxy, contract, arrangement, understanding, or relationship pursuant to which such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith has or pursuant to any proxy, contract, understanding or relationship may acquire any right to vote any security of the Company, (D) any agreement, arrangement, understanding, relationship or otherwise, including any repurchase or similar so-called “stock borrowing” agreement or arrangement, involving such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, directly or indirectly, the intent, purpose or effect of which may be to mitigate loss to, transfer to or from any such person, in whole or in part, any of the economic consequences of ownership or reduce the economic risk (of ownership or otherwise) of any security of the Company by, manage the risk of share price changes for, or increase or decrease the voting power of, such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, with respect to any security of the Company, or which provides, directly or indirectly, the opportunity to profit or share in any profit derived from any decrease in the price or value of any securities of the Company (any of the foregoing, a “Short Interest”), (E) any rights to dividends on the shares of the Company owned beneficially by such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, that are separated or separable from the underlying shares of the Company, (F) any proportionate interest in securities of the Company or Derivative Instruments held, directly or indirectly, by a general or limited partnership or similar entity in which such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, is a general partner or, directly or indirectly, beneficially owns an interest in a general partner or is the manager or managing member, or directly or indirectly, beneficially owns any interest in the manager or managing member of such general or limited partnership or similar entity, (G) any performance-related fees (other than an asset-based fee) that such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, is entitled to based on any increase or decrease in the value of securities of the Company or Derivative Instruments or Short Interests, if any; (H) any direct or indirect interest, including any significant equity interests or any Derivative Instruments or Short Interests in any principal competitor of the Company held by such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, and (I) any direct or indirect interest of such member, such beneficial owner or any of their respective affiliates or
27
associates, or others acting in concert therewith, in any contract with, or any litigation involving, the Company, any affiliate of the Company or any principal competitor of the Company (including, in any such case, any employment agreement, collective bargaining agreement or consulting agreement), (iv) if any such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, intends to engage in a solicitation with respect to a nomination or other business pursuant to this article 58, a statement disclosing the name of each participant in such solicitation (as defined in Item 4 of Schedule 14A under the Exchange Act) and if involving a nomination a representation that such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert, therewith intends to deliver a proxy statement and form of proxy to holders of at least sixty-seven percent (67%) of the shares carrying voting rights; (v) a certification that each such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, has complied with all applicable legal requirements in connection with its acquisition of shares or other securities of the Company and such person’s acts or omissions as a member of the Company; (vi) the names and addresses of other members (including beneficial owners) known by any such member, such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, to financially or otherwise materially support (it being understood, for example, that statement of an intent to vote for, or delivery of a revocable proxy to such proponent, does not require disclosure under this section, but solicitation of other members by such supporting member would require disclosure under this section) such nomination(s) or proposal(s), and to the extent known the class and number of all shares of the Company’s share capital owned beneficially or of record by, and any other information contemplated by clause (iii) of this article 58(a) with respect to, such other member(s) or other beneficial owner(s); (vii) all information that would be required to be set forth in a Schedule 13D filed pursuant to Rule 13d-1(a) of the Exchange Act or an amendment pursuant to Rule 13d-2(a) of the Exchange Act if such a statement were required to be filed under the Exchange Act and the rules and regulations promulgated thereunder by such member, such beneficial owner and their respective affiliates or associates, or others acting in concert therewith, if any; and (viii) any other information relating to such member such beneficial owner or any of their respective affiliates or associates, or others acting in concert therewith, if any, that would be required to be disclosed in a proxy statement and form or proxy or other filings required to be made in connection with solicitations of proxies for, as applicable, the business proposal and/or for the election of Directors in a contested election pursuant to Section 14 of the Exchange Act and the rules and regulations promulgated thereunder.
(b)If the notice includes any business other than a nomination of a Director or Directors that the member proposes to bring before the meeting, a member’s notice must, in addition to the matters set forth in article 58(a), also set forth: (i) a brief description of the business desired to be brought before the meeting, the reasons for conducting such business at the meeting and any material interest of such member such beneficial owner and each of their respective affiliates or associates, or others acting in concert therewith, if any, in such business, (ii) the text of the business proposal (including the text of any resolutions proposed for consideration and, in the event that such proposal includes a proposal to amend these articles, the text of the proposed amendment), and (iii) a description of all agreements, arrangements and understandings between such member, such beneficial owner and each of their respective affiliates or associates, or others acting in concert therewith, if any, on the one hand, and any other person or persons (including their names), on the other hand, in connection with the business proposal by such member.
(c)As to each individual, if any, whom the member proposes to nominate for election or re-election to the Board, a member’s notice must, in addition to the matters set forth in article 58(a), also set forth: (i) the name, age, business and residence address of such person; (ii) the principal occupation or employment of such person (present and for the past five (5) years);(iii) the completed and signed questionnaire and representation agreement required by article 64; (iv) all information relating to such individual that would be required to be disclosed in a proxy statement or other filings required to be made in connection with solicitations of proxies for election of
28
Directors in a contested election pursuant to Section 14 of the Exchange Act and the rules and regulations promulgated thereunder (including such individual’s written consent to being named in a proxy statement as a nominee) and a written statement of intent to serve as a Director for the full term if elected; and (v) a description of all direct and indirect compensation and other material monetary agreements, arrangements and understandings during the past three (3) years, and any other material relationships, between or among such member and beneficial owner, if any, and their respective affiliates and associates, or others acting in concert therewith, on the one hand, and each proposed nominee, and his or her respective affiliates and associates, or others acting in concert therewith, on the other hand, including, without limitation all biographical and related party transaction and other information that would be required to be disclosed pursuant to Rule 404 promulgated under Regulation S-Κ under the Exchange Act if the member making the nomination and any beneficial owner on whose behalf the nomination is made, if any, or any affiliate or associate thereof or person acting in concert therewith, were the “registrant” for purposes of such rule and the nominee were a Director or executive officer of such registrant.
(d)In addition, to be considered timely, a member’s notice shall further be updated and supplemented, if necessary, so that the information provided or required to be provided in such notice shall be true and correct as of the record date for determining the Holders entitled to notice of the meeting (or any adjournment, recess, rescheduling or postponement thereof) and as of the date that is ten (10) days prior to the meeting (or any adjournment, recess, rescheduling or postponement thereof), and such update and supplement shall be delivered to the Secretary at the Office not later than (a) the later of (i) ten (10) days after the record date for determining the Holders entitled to notice of the meeting (or any adjournment, recess, rescheduling or postponement thereof) or (ii) the first public announcement of the date of notice of such record date in the case of the update and supplement required to be made as of the record date, and (b) not later than eight (8) days prior to the date for the meeting (or any adjournment, recess, rescheduling or postponement thereof) in the case of the update and supplement required to be made as of ten (10) days prior to the meeting or any adjournment, recess, rescheduling or postponement thereof. The obligation to update and supplement as set forth in this article 58(d) or any other article shall not limit the Company’s rights with respect to any deficiencies in any notice provided by a member, extend any applicable deadlines under these articles or enable or be deemed to permit a member who has previously submitted notice under these articles to amend or update any nomination or business proposal or to submit any new nomination or business proposal, including by changing or adding nominees, matters, business and or resolutions proposed to be brought before a meeting of the members. In addition, if the member giving the notice has delivered to the Company a notice relating to the nomination of Directors, the member giving the notice shall deliver to the Company no later than five (5) business days prior to the date of the meeting or, if practicable, any adjournment, recess, rescheduling or postponement thereof (or, if not practicable, on the first practicable date prior to the date to which the meeting has been adjourned, recessed, rescheduled, or postponed) reasonable evidence that it has complied with the requirements of Rule 14a-19 of the Exchange Act.
(e)The Company may also, as a condition to any such nomination or business being deemed properly brought before an annual or extraordinary general meeting, require any member giving the notice and the beneficial owner, if any, on whose behalf the nomination or business proposal, as applicable, is made, or any proposed nominee to deliver to the Secretary, within five (5) business days of any such request, such other information as may reasonably be required by the Company or the Board, in its sole discretion, to determine (a) the eligibility of such proposed nominee to serve as a Director, (b) whether such nominee qualifies as an “independent director” or “audit committee financial expert” under applicable law, securities exchange rule or regulation, or any publicly disclosed corporate governance guideline or committee charter of the Company or (c) such other information that the Board determines, in its sole discretion, could be material to a reasonable member’s understanding of the independence, or lack thereof, of such nominee. Notwithstanding anything to the contrary, only persons who are nominated in accordance with the procedures set forth
29
in these articles, including, without limitation, articles 44 to 45 and articles 56 to 65 shall be eligible for election as Directors; and
(f)Notwithstanding anything to the contrary in these articles 56 to 65, to the extent the Holder giving the notice is acting solely at the direction of the beneficial owner and not also on its own behalf or in concert with a beneficial owner, and is not an affiliate or associate or such beneficial owner, information otherwise required by clauses (iii), (iv), (v) and (vi) of article 58(a) shall not be required of or with respect to such Holder.
59.Notwithstanding the provisions of these articles, a member giving the notice shall also comply with all applicable requirements of the Exchange Act and the rules and regulations thereunder with respect to the matters set forth in these articles; provided, however, that any references in these articles to the Exchange Act or the rules promulgated thereunder are not intended to and shall not limit the separate and additional requirements set forth in these articles with respect to nominations or proposals as to any other business to be considered.
60.Only persons who are nominated by members in accordance with the procedures set forth in article 44 to 45 and articles 56 to 65 shall be eligible to be elected at an annual or extraordinary general meeting of members of the Company to serve as Directors and only such business shall be conducted at a meeting of members as shall have been brought before the meeting in accordance with the procedures set forth in articles 44 to 45 and articles 56 to 65. The procedures set forth in articles 44 to 45 and articles 56 to 65 for nomination for the election of Directors by members are in addition to, and not in limitation of, any procedures now in effect or hereafter adopted by or at the direction of the Board or any committee thereof.
61.Notwithstanding the foregoing provisions of article 44 and articles 56 to 65, if the member giving the notice (or a qualified representative thereof) does not appear at the annual or extraordinary general meeting of members of the Company to present a nomination or proposed business, such nomination shall be disregarded and such proposed business shall not be transacted, notwithstanding that proxies in respect of such vote may have been received by the Company.
62.Except as otherwise provided by law, the Board or the Chairman of the meeting shall have the power (a) to determine whether a nomination or any business proposed to be brought before the meeting was made or proposed, as the case may be, in accordance with the procedures set forth in articles 44 and 45 and articles 56 to 65 (including whether the member or beneficial owner, if any, on whose behalf the nomination or business proposal is made solicited (or is part of a group which solicited) or did not so solicit, as the case may be, proxies in support of such member’s nominee or business proposal in compliance with such member’s representation as required by article 59(a)(v)) and (b) if any nomination or proposal was not made or proposed in compliance with articles 44 and 45 and articles 56 to 65, or if any of the information provided to the Company pursuant to articles 44 and 45 and articles 56 to 65 was inaccurate, to declare that such nomination shall be disregarded or that such proposed business shall not be transacted.
63.Nothing in these articles shall be deemed to affect any rights: (a) of members to request inclusion of business proposals in the Company’s proxy statement pursuant to Rule 14a-8 under the Exchange Act; (b) of the holders of any series of preferred shares if and to the extent provided for under law, the memorandum of association or these articles or (c) of members of the Company to bring business before an extraordinary general meeting pursuant to the valid exercise of power granted to them under Section 178 of the Act. Subject to Rule 14a-8 under the Exchange Act, nothing in these articles shall be construed to permit any member, or give any member the right, to include or have disseminated or described in the Company’s proxy statement any nomination of Director or Directors or any other business proposal, unless such Member has complied with these articles, including the provision to the Company of notices required thereunder in a timely manner.
30
64.To be eligible to be a nominee of any member for election or re-election as a Director of the Company, a person must deliver (in accordance with the time periods prescribed for delivery of notice under these articles 56 to 65) to the Secretary at the Office a written questionnaire with respect to the background and qualification of such individual and the background of any other person or entity on whose behalf, directly or indirectly, the nomination is being made, and a written representation and agreement (in the form of such questionnaire and representation and agreement provided by the Secretary, which form shall be provided by the Secretary upon written request of any member giving the notice and the beneficial owner, if any, on whose behalf the nomination or business proposal, as applicable, is made; provided such written request identifies both the member making such request and the beneficial owner(s), if any, on whose behalf such request is being made) that such individual:
(a)(1) is not and will not become a party to: (a) any agreement, arrangement or understanding with, and has not given any commitment or assurance to, any person or entity as to how such person, if elected as a Director, will act or vote on any issue or question (a “Voting Commitment”) that has not been disclosed to the Company; and (b) any Voting Commitment that could limit or interfere with such individual’s ability to comply, if elected as a Director, with such individual’s fiduciary duties under applicable law; and (2) is not and will not become a party to any agreement, arrangement or understanding with any person or entity other than the Company with respect to any direct or indirect compensation, reimbursement or indemnification in connection with service or action as a Director that has not been disclosed therein;
(b)agrees to promptly provide to the Company such other information as the Company may reasonably request and;
(c)in such individual’s personal capacity and on behalf of any person or entity on whose behalf, directly or indirectly, the nomination is being made, would be in compliance, if elected as a Director, and will comply with all applicable corporate governance, conflict of interest, confidentiality and stock ownership and trading policies and guidelines of the Company publicly disclosed from time to time.
65.Any individual who is nominated for election to the Board, including pursuant to these articles 44 and 45 and articles 56 to 65 shall tender an irrevocable resignation in advance of the general meeting. Unless otherwise resolved by the Board, such resignation shall become effective if the Board resolves that (a) the information provided to the Company by an individual who is nominated for election to the Board, including pursuant to any of articles 44 to 45 and articles 56 to 65, as applicable, or, if applicable, by the member who nominated such person under any of articles 44 to 45 and articles 56 to 65, applicable, and the beneficial owner, if any, on whose behalf the nomination was made, was untrue in any material respect or omitted to state a material fact necessary in order to make the statements made, in light of the circumstances under which they were made, not misleading; or (b) such individual or, if applicable, the member who nominated such individual under any of articles 44 to 45 and articles 56 to 65, as applicable, and the beneficial owner, if any, on whose behalf the nomination was made, shall have breached any representations or obligations owed to the Company under these articles.
VOTES OF MEMBERS
66.Subject to any special rights or restrictions as to voting for the time being attached by or in accordance with these articles to any class of shares, every member who is present in person or by proxy shall have one vote for each share of which he is the Holder.
67.When there are joint Holders, the vote of the senior who tenders a vote, whether in person or by proxy, shall be accepted to the exclusion of the votes of the other joint Holders; and for this purpose, seniority shall be determined by the order in which the names stand in the Register.
31
68.A member of unsound mind, or in respect of whom an order has been made by any court having jurisdiction (whether in Ireland or elsewhere) in matters concerning mental disorder, may vote, on a poll, by his committee, receiver, guardian or other person appointed by that court and any such committee, receiver, guardian or other person may vote by proxy on a poll. Evidence to the satisfaction of the Directors of the authority of the person claiming to exercise the right to vote shall be received at the Office or at such other address as is specified in accordance with these articles for the receipt of appointments of proxy and in default the right to vote shall not be exercisable.
69.No objection shall be raised to the qualification of any voter except at the meeting or adjourned meeting at which the vote objected to is given or tendered, and every vote not disallowed at such meeting shall be valid for all purposes. Any such objection made in due time shall be referred to the Chairman of the meeting, whose decision shall be final and conclusive.
70.Votes may be given either personally or by proxy.
71.
(a)Every member entitled to attend and vote at a general meeting may appoint a proxy to attend, speak and vote on his behalf and may appoint more than one proxy to attend, speak and vote at the same meeting. The appointment of a proxy shall be in any form which the Directors may approve (subject to compliance with any requirements as to form prescribed by the Act and the Exchange Act) and, if required by the Company, shall be signed by or on behalf of the appointor. In relation to written proxies, a body corporate must sign a form of proxy under its common seal (if applicable) or under the hand of a duly authorised officer or attorney thereof. A proxy need not be a member of the Company. The appointment of a proxy in electronic or other form shall only be effective in such manner as the Directors may approve and subject to any requirements of the Act. An instrument or other form of communication appointing or evidencing the appointment of a proxy or a corporate representative (other than a standing proxy or representative) together with such evidence as to its due execution as the Board may from time to time require, may be returned to the address or addresses stated in the notice of meeting or adjourned meeting or any other information or communication by such time or times as may be specified by the Board in the notice of meeting or adjourned meeting or in any other such information or communication (which times may differ when more than one place is so specified) or, if no such time is specified, at any time prior to the holding of the relevant meeting or adjourned meeting at which the appointee proposes to vote, and, subject to the Act, if not so delivered the appointment shall not be treated as valid.
(b)Without limiting the foregoing, the Directors may from time to time permit appointments of a proxy to be made by means of an electronic or internet communication or facility and may in a similar manner permit supplements to, or amendments or revocations of, any such electronic or internet communication or facility to be made. For the avoidance of doubt, such appointments of proxy as made by electronic or internet communication or facility as permitted by the Directors will be deemed to be deposited at the place specified for such purpose once received by the Company. The Directors may in addition prescribe the method of determining the time at which any such electronic or internet communication or facility is to be treated as received by the Company. The Directors may treat any such electronic or internet communication or facility which purports to be or is expressed to be sent on behalf of a Holder of a share as sufficient evidence of the authority of the person sending that instruction to send it on behalf of that Holder.
72.Any body corporate which is a member of the Company may authorise such person as it thinks fit to act as its representative at any meeting of the Company or of any class of members of the Company and the person so authorised shall be entitled to exercise the same powers on behalf of the body corporate which he represents as that body corporate could exercise if it were an individual member of the Company. The Company may require evidence from the body corporate of the due authorisation of such person to act as the representative of the relevant body corporate.
32
73.An appointment of proxy relating to more than one meeting (including any adjournment thereof) having once been received by the Company for the purposes of any meeting shall not require to be delivered, deposited or received again by the Company for the purposes of any subsequent meeting to which it relates.
74.Receipt by the Company of an appointment of proxy in respect of a meeting shall not preclude a member from attending and voting at the meeting or at any adjournment thereof. An appointment proxy shall be valid, unless the contrary is stated therein, as well for any adjournment of the meeting as for the meeting to which it relates.
75.
(a)A vote given in accordance with the terms of an appointment of proxy or a resolution authorising a representative to act on behalf of a body corporate shall be valid notwithstanding the death or insanity of the principal, or the revocation of the appointment of proxy or of the authority under which the proxy was appointed or of the resolution authorising the representative to act or transfer of the share in respect of which the proxy was appointed or the authorisation of the representative to act was given, provided that no intimation in writing (whether in electronic form or otherwise) of such death, insanity, revocation or transfer shall have been received by the Company at the Office before the commencement of the meeting or adjourned meeting at which the appointment of proxy is used or at which the representative acts.
(b)The Directors may send, at the expense of the Company, by post, electronic mail or otherwise, to the members forms for the appointment of a proxy (with or without stamped envelopes for their return) for use at any general meeting or at any class meeting, either in blank or nominating any one or more of the Directors or any other persons in the alternative.
76.Subject to the Act and the Exchange Act, a resolution in writing signed by all of the members for the time being entitled to attend and vote on such resolution at a general meeting (or being bodies corporate by their duly authorised representatives) shall be as valid and effective for all purposes as if the resolution had been passed at a general meeting of the Company duly convened and held, and may consist of several documents in like form each signed by one or more persons, and if described as a special resolution shall be deemed to be a special resolution within the meaning of the Act. Any such resolution shall be served on the Company.
DIRECTORS
77.The number of Directors shall be as the Board may determine from time to time, but shall not be less than two nor more than twenty unless otherwise determined by the Company in a general meeting. The continuing Directors may act notwithstanding any vacancy in their body, provided that if the number of the Directors is reduced below the prescribed minimum the remaining Director or Directors shall appoint forthwith an additional Director or additional Directors to make up such minimum or shall convene a general meeting of the Company for the purpose of making such appointment. If, at any annual general meeting of the Company, the number of Directors is reduced below the prescribed minimum due to the failure of any Directors to be re-elected, then in those circumstances, the two Directors which receive the highest number of votes in favour of re-election shall be re-elected and shall remain Directors until such time as additional Directors have been appointed to replace them as Directors. If, at any annual general meeting of the Company, the number of Directors is reduced below the prescribed minimum in any circumstances where one Director is re-elected, then that Director shall hold office until the next annual general meeting and the Director which (excluding the re-elected Director) receives the highest number of votes in favour of re-election shall be re-elected and shall remain a Director until such time as one or more additional Directors have been appointed to replace him or her. If there are no Director or Directors able or willing to act then any two members may summon a general meeting for the purpose of appointing Directors. Any additional Director so appointed shall hold office (subject to the provisions of the Act and these articles) only until the conclusion of the annual
33
general meeting of the Company next following such appointment unless he is re-elected during such meeting.
78.Each Director (whether or not an employee) shall be paid a fee or other remuneration for their services at such rate and/or on such basis as may from time to time be determined by the Board. The Directors may also be paid all travelling, hotel and other expenses properly incurred by them in attending and returning from meetings of the Directors or any committee of the Directors or general meetings of the Company or in connection with the business of the Company. The amount, rate or basis of the fees, remuneration or expenses paid to the Directors shall not require approval or ratification by the Company in a general meeting.
79.If any Director shall be called upon to perform extra services which in the opinion of the Directors are outside the scope of the ordinary duties of a Director, the Company may remunerate such Director either by a fixed sum or by a percentage of profits or otherwise as may be determined by a resolution passed at a meeting of the Directors and such remuneration may be either in addition to or in substitution for any other remuneration to which he may be entitled as a Director.
80.A Director (whether or not a member of the Company) shall be entitled to attend and speak at general meetings.
81.Unless the Company otherwise directs, a Director of the Company may be or become a Director or other officer of, or otherwise interested in, any company promoted by the Company or in which the Company may be interested as Holder or otherwise, and no such Director shall be accountable to the Company for any remuneration or other benefits received by him as a Director or officer of, or from his interest in, such other company.
BORROWING POWERS
82.Subject to the Act, the Directors may exercise all the powers of the Company to borrow or raise money, and to mortgage or charge its undertaking, property, assets and uncalled capital or any part thereof and to issue debentures, debenture stock and other securities whether outright or as collateral security for any debt, liability or obligation of the Company or of any third party, without any limitation as to amount.
POWERS AND DUTIES OF THE DIRECTORS
83.The business of the Company shall be managed by the Directors, who may pay all expenses incurred in promoting and registering the Company and may exercise all such powers of the Company as are not, by the Act or by these articles, required to be exercised by the Company in general meeting, subject, nevertheless, to any of these articles and to the provisions of the Act.
84.The Directors may from time to time and at any time by power of attorney appoint any company, firm or person or body of persons, whether nominated directly or indirectly by the Directors, to be the attorney or attorneys of the Company for such purposes and with such powers, authorities and discretions (not exceeding those vested in or exercisable by the Directors under these articles) and for such period and subject to such conditions as they may think fit, and any such power of attorney may contain such provisions for the protection of persons dealing with any such attorney as the Directors may think fit, and may also authorise any such attorney to delegate all or any of the powers, authorities and discretions vested in him.
85.The Company may exercise the powers conferred by the Act with regard to having an official seal for use abroad and such powers shall be vested in the Directors.
86.
(a)Each Director is expressly permitted (for the purposes of Section 228(1)(d) of the Act) to use vehicles, telephones, computers, accommodation and any other Company
34
property as may be specified by the directors where such use is approved by the Board or by any person so authorised by the Board or as permitted by their terms of employment or appointment.
(b)A Director who is in any way, whether directly or indirectly, interested in a contract or proposed contract with the Company shall declare the nature of his interest at a meeting of the Directors in accordance with the Act.
(c)As recognised by section 228(1)(e) of the Act, the Directors may agree to restrict their power to exercise an independent judgement but only where this has been approved by a resolution of the Board of the Company.
(d)Nothing in section 228(1)(d) or section 228(1)(f) of the Act shall restrict a Director from engaging directly or indirectly in the same or similar business activities or lines of business as the Company or any of its subsidiaries. To the fullest extent permitted by applicable law, the Company renounces any interest or expectancy of the Company and its subsidiaries in, or in being offered an opportunity to participate in, business opportunities that may from time to time be presented to Directors other than in their role as directors of the Company, even if the opportunity is one that the Company or its subsidiaries might reasonably be expected to have pursued or had the ability or desire to pursue if granted the opportunity to do so. The Directors shall have no duty to communicate or offer such business opportunity to the Company and, to the fullest extent permitted by applicable law, shall not be deemed to have breached any fiduciary or other duty solely by reason of the fact that such Director pursues or acquires such business opportunity, directs such business opportunity to another person or fails to present such business opportunity, or information regarding such business opportunity, to the Company or any of its subsidiaries. Without prejudice to the generality of the foregoing, a business opportunity shall not be deemed to be an opportunity of the Company if it is an opportunity that the Company is not financially able or contractually permitted or legally able to undertake, or that is, by its nature, not in line with the Company’s business or is of no advantage to it or is one in which the Company has no interest or reasonable prospect.
87.Save as otherwise provided by these articles, a Director shall not vote at a meeting of the Directors or a committee of Directors on any resolution concerning a matter in which he has, directly or indirectly, an interest which is material or a duty which conflicts or may conflict with the interests of the Company. A Director shall not be counted in the quorum present at a meeting in relation to any such resolution on which he is not entitled to vote.
(a)A Director shall be entitled (in the absence of some other material interest than is indicated below) to vote (and be counted in the quorum) in respect of any resolutions concerning any of the following matters, namely:
(i)the giving of any security, guarantee or indemnity to him in respect of money lent by him to the Company or any of its subsidiary or associated companies or obligations incurred by him or by any other person at the request of or for the benefit of the Company or any of its subsidiary or associated companies;
(ii)the giving of any security, guarantee or indemnity to a third party in respect of a debt or obligation of the Company or any of its subsidiary or associated companies for which he himself has assumed responsibility in whole or in part and whether alone or jointly with others under a guarantee or indemnity or by the giving of security;
(iii)any proposal concerning any offer of shares or debentures or other securities of or by the Company or any of its subsidiary or associated companies for subscription, purchase or exchange in which offer he is or is to be interested as a participant in the underwriting or sub-underwriting thereof;
(iv)any proposal concerning any other company in which he is interested, directly or indirectly and whether as an officer or member or otherwise howsoever, provided that he is not the Holder of or beneficially interested in
35
1% or more of the issued shares of any class of such company or of the voting rights available to members of such company (or of a third company through which his interest is derived) (any such interest being deemed for the purposes of this article to be a material interest in all circumstances);
(v)any proposal concerning the adoption, modification or operation of a superannuation fund or retirement benefits scheme under which he may benefit;
(vi)any proposal concerning the adoption, modification or operation of any scheme or plan for the remuneration or reward of any employee, officer or Director (or any of them) of the Company and/or any subsidiary thereof, including (without limitation) in relation to the award or acquisition of shares (or any interest in shares), under which the Director benefits or may benefit; or
(vii)any proposal concerning the giving of any indemnity pursuant to article 139(a) or the discharge of the cost of any insurance coverage purchased or maintained pursuant to article 94 and article 139(b).
(b)Where proposals are under consideration concerning the appointment (including fixing or varying the terms of appointment) of two or more Directors to offices or employments with the Company or any company in which the Company is interested, such proposals may be divided and considered in relation to each Director separately and in such case each of the Directors concerned (if not debarred from voting under sub-paragraph (a)(iv) of this article) shall be entitled to vote (and be counted in the quorum) in respect of each resolution except that concerning his own appointment,
(c)If a question arises at a meeting of Directors or of a committee of Directors as to the materiality of a Director’s interest or as to the right of any Director to vote and such question is not resolved by his voluntarily agreeing to abstain from voting, such question may be referred, before the conclusion of the meeting, to the Chairman of the meeting and his ruling in relation to any Director other than himself shall be final and conclusive. In relation to the Chairman, such question may be resolved by a resolution of a majority of the Directors (other than the Chairman) present at the meeting at which the question first arises.
(d)For the purposes of this article, an interest of a person who is the spouse or a minor child of a Director shall be treated as an interest of the Director.
(e)The Company by Ordinary Resolution may suspend or relax the provisions of this article to any extent or ratify any transaction not duly authorised by reason of a contravention of this article.
88.A Director may hold and be remunerated in respect of any other office or place of profit under the Company or any other company in which the Company may be interested (other than the office of auditor of the Company or any subsidiary thereof) in conjunction with his office of Director for such period and on such terms as to remuneration and otherwise as the Directors may determine, and no Director or intending Director shall be disqualified by his office from contracting or being interested, directly or indirectly, in any contract or arrangement with the Company or any such other company either with regard to his tenure of any such other office or place of profit or as vendor, purchaser or otherwise nor shall any Director so contracting or being so interested be liable to account to the Company for any profits and advantages accruing to him from any such contract or arrangement by reason of such Director holding that office or of the fiduciary relationship thereby established.
89.The Directors may exercise the voting powers conferred by shares of any other company held or owned by the Company in such manner in all respects as they think fit and in particular they may exercise their voting powers in favour of any resolution appointing the Directors or any of them as Directors or officers of such other company or providing for the payment of remuneration or pensions to the Directors or officers of such other company.
36
90.Any Director may act by himself or his firm in a professional capacity for the Company, and he or his firm shall be entitled to remuneration for professional services as if he were not a Director, but nothing herein contained shall authorise a Director or his firm to act as auditor to the Company.
91.All cheques, promissory notes, drafts, bills of exchange and other negotiable instruments and all receipts for money paid to the Company shall be signed, drawn, accepted, endorsed or otherwise executed, as the case may be, by such person or persons and in such manner as the Directors shall from time to time by resolution determine.
92.The Directors shall cause minutes to be made in books provided for the purpose:
(a)of all appointments of officers made by the Directors;
(b)of the names of the Directors present at each meeting of the Directors and of any committee of the Directors; and
(c)of all resolutions and proceedings at all meetings of the Company and of the Directors and of committees of Directors.
93.The Directors may procure the establishment and maintenance of or participate in, or contribute to any non-contributory or contributory pension or superannuation fund, scheme or arrangement or life assurance scheme or arrangement for the benefit of, and pay, provide for or procure the grant of donations, gratuities, pensions, allowances, benefits or emoluments to any persons (including Directors or other officers) who are or shall have been at any time in the employment or service of the Company or of any company which is or was a subsidiary of the Company or of the predecessor in business of the Company or any such subsidiary or holding Company and the wives, widows, families, relatives or dependants of any such persons. The Directors may also procure the establishment and subsidy of or subscription to and support of any institutions, associations, clubs, funds or trusts calculated to be for the benefit of any such persons as aforesaid or otherwise to advance the interests and well being of the Company or of any such other Company as aforesaid, or its members, and payments for or towards the insurance of any such persons as aforesaid and subscriptions or guarantees of money for charitable or benevolent objects or for any exhibition or for any public, general or useful object. Provided that any Director shall be entitled to retain any benefit received by him under this article, subject only, where the Act require, to disclosure to the members and the approval of the Company in general meeting.
DISQUALIFICATION OF DIRECTORS
94.The office of a Director shall be vacated ipso facto if the Director:
(a)is restricted or disqualified to act as a Director under the Act; or
(b)resigns his office by notice in writing to the Company or in writing offers to resign and the Directors resolve to accept such offer; or
(c)is requested to resign in writing by not less than three quarters of the other Directors; or
(d)is removed from office under article 99.
APPOINTMENT, ROTATION AND REMOVAL OF DIRECTORS
95.At every annual general meeting of the Company, all of the Directors shall retire from office unless re-elected by Ordinary Resolution at the annual general meeting. A Director retiring at a meeting shall retain office until the close or adjournment of the meeting.
37
96.Every Director shall be eligible to stand for re-election at an annual general meeting in accordance with articles 44 to 65.
97.If a Director offers himself for re-election, he shall be deemed to have been re-elected, unless at such meeting the Ordinary Resolution for the re-election of such Director has been defeated.
98.The Company may from time to time by Special Resolution increase or reduce the maximum number of Directors.
99.The Company may, by Ordinary Resolution, of which notice has been given in accordance with the Act, remove any Director before the expiration of his period of office notwithstanding anything in these articles or in any agreement between the Company and such Director. Such removal shall be without prejudice to any claim such Director may have for damages for breach of any contract of service between him and the Company.
100.The Company may, by Ordinary Resolution, appoint another person in place of a Director removed from office under article 99 and without prejudice to the powers of the Directors under article 77, the Company in general meeting by Ordinary Resolution may appoint any person to be a Director either to fill a casual vacancy or as an additional Director; provided that the appointment does not cause the number of Directors to exceed the maximum number of Directors as determined by the Board in accordance with article 77, and further provided that if, at the time the Company files its definitive proxy statement for any general meeting of the Company, the number of persons who are at such time validly nominated in accordance with these articles for election or re-election as Directors (such person collectively, the “Director Nominees”) exceeds the maximum number of Directors as determined by the Board in accordance with article 77 (the “Available Director Positions”, and such an election, a “contested election”) (whether to fill a vacancy pursuant to this article 100 arising from a removal or if members have the right to nominate directors for election at an annual general meeting or otherwise), then those Director Nominees in number equal to the Available Director Positions who receive the highest number of votes in favour of their election (whether or not such votes in favour represent, with respect to each such Director Nominee, a majority of the votes cast in respect of the election of such Director Nominee) by the members present in person or represented by proxy at such meeting and entitled to vote on the election of Directors shall be appointed Directors. For clarity, notwithstanding the withdrawal of any nominations for Directors in a contested election subsequent to the time the Company files its definitive proxy statement, the plurality voting provisions of the proviso in this article 100 will continue to apply to the election of Directors at any such meeting. If such members do not appoint a Director to fill such vacancy within 45 days after the occurrence of such vacancy, the Board may appoint a Director to fill such vacancy until the next meeting of the members held for the purpose of electing Directors. If not re-appointed at such general meeting, such Director shall vacate office at the conclusion thereof. The Directors are not entitled to appoint alternate directors.
101.The Directors may appoint a person who is willing to act to be a Director, either to fill a vacancy or as an additional Director, provided that the appointment does not cause the number of Directors to exceed the maximum number of Directors as determined by the Board in accordance with article 77. A Director so appointed shall hold office only until the next following annual general meeting. If not re-appointed at such annual general meeting, such Director shall vacate office at the conclusion thereof. The Directors are not entitled to appoint alternate directors.
102.
(a)The Directors may appoint (and remove) any person to fill the position of Secretary (including more than one Secretary to act as joint secretary).
(b)It shall be the duty of the Secretary to make and keep records of the votes, doings and proceedings of all meetings of the members and Board of the Company, and of its committees, and to authenticate records of the Company. The Secretary shall be
38
appointed by the Directors for such term, at such remuneration and upon such conditions as they may think fit; and any Secretary so appointed may be removed by them.
(c)A provision of the Act or these articles requiring or authorising a thing to be done by or to a Director and the Secretary shall not be satisfied by its being done by or to the same person acting both as Director and as, or in place of, the Secretary.
(d)The Secretary may appoint (and remove) any one or more persons as Assistant Secretary from time to time, who may be delegated such functions of the Secretary as the Secretary may determine from time to time.
(e)Without limitation to the preceding paragraph, the Secretary may delegate any of his functions to such one or more persons (including individuals, bodies corporate or firms) as may be nominated by the Secretary from time to time.
(f)In addition to the Board’s power to delegate to committees pursuant to article 107, the Board may delegate any of its powers to any individual Director or member of the management of the Company or any of its subsidiaries as it sees fit; any such individual shall, in the exercise of the powers so delegated, conform to any regulations that may be imposed on them by the Board. The Board shall also have the power to appoint and remove officers of the Company including, but not limited to, chief executive officer, president, vice president, treasurer, controller and assistant treasurer.
PROCEEDINGS OF DIRECTORS
103.
(a)The Directors may meet together for the dispatch of business, adjourn and otherwise regulate their meetings as they may think fit. The quorum necessary for the transaction of the business of the Directors shall be a majority of the Directors in office at the time when the meeting is convened. Questions arising at any meeting shall be decided by a majority of votes cast by Directors present or represented at such meeting. Each Director present and voting shall have one vote.
(b)Any Director may participate in a meeting of the Directors by means of telephonic or other such communication whereby all persons participating in the meeting can hear each other speak, and participation in a meeting in this manner shall be deemed to constitute presence in person at such meeting and any director may be situated in any part of the world for any such meeting.
104.The Chairman or any four Directors may, and the Secretary on the requisition of the Chairman or any four Directors shall, at any time summon a meeting of the Directors.
105.The continuing Directors may act notwithstanding any vacancy in their number but, if and so long as their number is reduced below the number fixed by or pursuant to these articles as the minimum number of Directors, the continuing Directors or Director may act for the purpose of increasing the number of Directors to that number or of summoning a general meeting of the Company but for no other purpose.
106.The Directors may elect a Chairman of their meetings and determine the period for which he is to hold office. Any Director may be elected no matter by whom he was appointed but if no such Chairman is elected, or if at any meeting the Chairman is not present within five minutes after the time appointed for holding the same, the Directors present may choose one of their number to be Chairman of the meeting.
107.The Board may from time to time designate committees of the Board, with such powers and duties as the Board may decide to confer on such committees, and shall, for those committees and any others provided for herein, elect a director or directors to serve as the member or members, designating, if it desires, other directors as alternate members who may replace any absent or disqualified member at any meeting of the committee. Adequate provision shall be made for notice to members of
39
all meetings; a majority of the members shall constitute a quorum unless the committee shall consist of one or two members, in which event one member shall constitute a quorum; and all matters shall be determined by a majority vote of the members present. Action may be taken by any committee without a meeting if all members thereof consent thereto in writing, and the writing or writings are filed with the minutes of the proceedings of such committees.
108.A committee may elect a chairman of its meeting. If no such chairman is elected, or if at any meeting the chairman is not present within five minutes after the time appointed for holding the same, the members present may choose one of their number to be chairman of the meeting.
109.All acts done by any meeting of the Directors or of a committee of Directors or by any person acting as a Director shall, notwithstanding that it be afterwards discovered that there was some defect in the appointment of any such Director or person acting as aforesaid, or that they or any of them were disqualified, be as valid as if every such person had been duly appointed and was qualified to be a Director.
110.Notwithstanding anything in these articles or in the Act which might be construed as providing to the contrary, notice of every meeting of the Directors shall be given to all Directors either by mail, telephone, email, or any other electronic means on not less than 48 hours’ notice, or on such shorter notice as person or persons calling such meeting may deem necessary or appropriate and which is reasonable in the circumstances and shall specify the purpose of such meeting and provide other customary information regarding the topics to be considered. Any Director may waive any notice required to be given under these articles, and the attendance of a Director at a meeting shall be deemed to be a waiver by such Director.
111.A resolution or other document in writing (in electronic form or otherwise) signed (whether by electronic signature, advanced electronic signature or otherwise as approved by the Directors) by all the Directors entitled to receive notice of a meeting of Directors or of a committee of Directors shall be as valid as if it had been passed at a meeting of Directors or (as the case may be) a committee of Directors duly convened and held and may consist of several documents in the like form each signed by one or more Directors, and such resolution or other document or documents when duly signed may be delivered or transmitted (unless the Directors shall otherwise determine either generally or in any specific case) by facsimile transmission, electronic mail or some other similar means of transmitting the contents of documents.
THE SEAL
112.
(a)The Directors shall ensure that the Seal (including any official securities seal kept pursuant to the Act) shall be used only by the authority of the Directors or of a committee authorised by the Directors and that every instrument to which the seal shall be affixed shall be signed by a Director or some other person appointed by the Directors for that purpose.
(b)The Company may exercise the powers conferred by the Act with regard to having an official seal for use abroad and such powers shall be vested in the Directors.
DIVIDENDS AND RESERVES
113.The Company in general meeting may declare dividends, but no dividends shall exceed the amount recommended by the Directors.
114.The Directors may from time to time pay to the members such interim dividends as appear to the Directors to be justified by the profits of the Company.
40
115.No dividend or interim dividend shall be paid otherwise than in accordance with the provisions of the Act.
116.The Directors may, before recommending any dividend, set aside out of the profits of the Company such sums as they think proper as a reserve or reserves which shall, at the discretion of the Directors, be applicable for any purpose to which the profits of the Company may be properly applied and pending such application may at the like discretion either be employed in the business of the Company or be invested in such investments as the Directors may lawfully determine. The Directors may also, without placing the same to reserve, carry forward any profits which they may think it prudent not to divide.
117.Subject to the rights of persons, if any, entitled to shares with special rights as to dividend, all dividends shall be declared and paid according to the amounts paid or credited as paid on the shares in respect whereof the dividend is paid. All dividends shall be apportioned and paid proportionately to the amounts paid or credited as paid on the shares during any portion or portions of the period in respect of which the dividend is paid; but if any share is issued on terms providing that it shall rank for dividend as from a particular date, such share shall rank for dividend accordingly.
118.The Directors may deduct from any dividend payable to any member all sums of money (if any) immediately payable by him to the Company in relation to the shares of the Company.
119.Any general meeting declaring a dividend or bonus and any resolution of the Directors declaring an interim dividend may direct payment of such dividend or bonus or interim dividend wholly or partly by the distribution of specific assets and in particular of paid up shares, debentures or debenture stocks of any other company or in any one or more of such ways, and the Directors shall give effect to such resolution, and where any difficulty arises in regard to such distribution, the Directors may settle the same as they think expedient, and in particular may fix the value for distribution of such specific assets or any part thereof and may determine that cash payments shall be made to any members upon the footing of the value so fixed, in order to adjust the rights of all the parties, and may vest any such specific assets in trustees as may seem expedient to the Directors.
120.Any dividend or other moneys payable in respect of any share may be paid by cheque or warrant sent by post, at the risk of the person or persons entitled thereto, to the registered address of the Holder or, where there are joint Holders, to the registered address of that one of the joint Holders who is first named on the members Register or to such person and to such address as the Holder or joint Holders may in writing direct. Every such cheque or warrant shall be made payable to the order of the person to whom it is sent and payment of the cheque or warrant shall be a good discharge to the Company. Any joint Holder or other person jointly entitled to a share as aforesaid may give receipts for any dividend or other moneys payable in respect of the share. Any such dividend or other distribution may also be paid by any other method (including payment in a currency other than US$, electronic funds transfer, direct debit, bank transfer or by means of a relevant system) which the Directors consider appropriate and any member who elects for such method of payment shall be deemed to have accepted all of the risks inherent therein. The debiting of the Company’s account in respect of the relevant amount shall be evidence of good discharge of the Company’s obligations in respect of any payment made by any such methods.
121.No dividend shall bear interest against the Company.
122.If the Directors so resolve, any dividend which has remained unclaimed for twelve years from the date of its declaration shall be forfeited and cease to remain owing by the Company. The payment by the Directors of any unclaimed dividend or other moneys payable in respect of a share into a separate account shall not constitute the Company a trustee in respect thereof.
41
ACCOUNTS
123.
(a)The Company shall cause to be kept adequate accounting records, whether in the form of documents, electronic form or otherwise, that:
(i)correctly record and explain the transactions of the Company;
(ii)will enable, at any time, the assets, liabilities, financial position and profit or loss of the Company to be determined with reasonable accuracy;
(iii)will enable the Directors to ensure that any financial statements of the Company comply with the requirements of the Act; and
(iv)will enable those financial statements of the Company to be readily and properly audited.
Accounting records shall be kept on a continuous and consistent basis and entries therein shall be made in a timely manner and be consistent from year to year. Adequate accounting records shall be deemed to have been maintained if they comply with the provisions of the Act and explain the Company’s transactions and facilitate the preparation of financial statements that give a true and fair view of the assets, liabilities, financial position and profit and loss of the Company and, if relevant, the Group and include any information and returns referred to in section 283(2) of the Act.
The Company may send by post, electronic mail or any other means of electronic communication a summary financial statement to its members or persons nominated by any member. The Company may meet, but shall be under no obligation to meet, any request from any of its members to be sent additional copies of its full report and accounts or summary financial statement or other communications with its members provided that, where the Directors elect to send summary financial statements to the members, any member may require that he be sent a copy of the statutory financial statements of the Company.
(b)The accounting records shall be kept at the Office or, subject to the provisions of the Act, at such other place as the Directors think fit and shall be open at all reasonable times to the inspection of the Directors.
(c)In accordance with the provisions of the Act, the Directors shall cause to be prepared and to be laid before the annual general meeting of the Company from time to time such statutory financial statements and reports as are required by the Act to be prepared and laid before such meeting.
(d)A copy of every statutory financial statement of the Company (including every document required by law to be annexed thereto) which is to be laid before the annual general meeting of the Company together with a copy of the Directors’ report, or summary financial statements prepared in accordance with section 1119 of the Act, and Auditors’ report shall be sent by post, electronic mail or any other means of communication (electronic or otherwise), not less than twenty-one Clear Days before the date of the annual general meeting, to every person entitled under the provisions of the Act to receive them; provided that in the case of those documents sent by electronic mail or any other means of electronic communication, such documents shall be sent with the consent of the recipient, to the address of the recipient notified to the Company by the recipient for such purposes.
124.The Directors shall determine from time to time whether and to what extent and at what times and places and under what conditions or regulations the accounts and books of the Company or any of them shall be open to the inspection of members, not
42
being Directors, and no member (not being a Director) shall have any right of inspecting any account or book or document of the Company except as conferred by the Act or authorised by the Directors or by the Company in general meeting. No member shall be entitled to require discovery of or any information respecting any detail of the Company’s trading, or any matter which is or may be in the nature of a trade secret, mystery of trade, or secret process which may relate to the conduct of the business of the Company and which in the opinion of the Directors it would be inexpedient in the interests of the members of the Company to communicate to the public.
CAPITALISATION OF PROFITS
125.Without prejudice to any powers conferred on the Directors as aforesaid and subject to the Directors’ authority to issue and allot shares under articles 8(c) and 8(d), the Directors may resolve to capitalise any part of the amount for the time being standing to the credit of any of the Company’s reserve accounts (including any capital redemption reserve fund, share premium account, any undenominated capital, any sum representing unrealised revaluation reserves, merger reserves or other reserve account not available for distribution) or to the credit of the profit and loss account which is not available for distribution by applying such sum in paying up in full unissued shares to be allotted as fully paid bonus shares to those members of the Company who would have been entitled to that sum if it were distributable and had been distributed by way of dividend (and in the same proportions). Whenever such a resolution is passed in pursuance of-this article, the Directors shall make all appropriations and applications of the amounts resolved to be capitalised thereby and all allotments and issues of fully paid shares or debentures, if any. Any such capitalisation will not require approval or ratification by the members of the Company.
126.Without prejudice to any powers conferred on the Directors by these articles, and subject to the Directors’ authority to issue and allot shares under articles 8(c) and 8(d), the Directors may resolve that any sum for the time being standing to the credit of any of the Company’s reserve accounts (including any reserve account available for distribution) or to the credit of the profit and loss account be capitalised and applied on behalf of the members who would have been entitled to receive that sum if it had been distributed by way of dividend (and in the same proportions) either in or towards paying up amounts for the time being unpaid on any shares held by them respectively, or in paying up in full unissued shares or debentures of the Company of a nominal amount equal to the sum capitalised (such shares or debentures to be allotted and distributed and credited as fully paid up to and amongst such Holders in the proportions aforesaid) or partly in one way and partly in another, so, however, that the only purposes for which sums standing to the credit of the capital redemption reserve fund or the share premium account or any undenominated capital shall be applied shall be those permitted by the Act.
127.The Directors may from time to time at their discretion, subject to the provisions of the Act and, in particular, to their being duly authorised pursuant to Section 1021 of the Act, to allot the relevant shares, offer to the Holders of ordinary shares the right to elect to receive in lieu of any dividend or proposed dividend or part thereof an allotment of additional ordinary shares credited as fully paid. In any such case the following provisions shall apply.
(i)The basis of allotment shall be determined by the Directors so that, as nearly as may be considered convenient in the Directors’ absolute discretion, the value (calculated by reference to the average quotation) of the additional ordinary shares (excluding any fractional entitlement) to be allotted in lieu of any amount of dividend shall equal such amount. For such purpose the “average quotation” of an ordinary share shall be the average of the five amounts resulting from determining whichever of the following ((A), (B) or (C) specified below) in respect of ordinary shares shall be appropriate for each of the first five business days on which ordinary shares are quoted “ex” the relevant dividend and as determined from the information published by
43
the New York Stock Exchange reporting the business done on each of these five business days:
(A)if there shall be more than one dealing reported for the day, the average of the prices at which such dealings took place; or
(B)if there shall be only one dealing reported for the day, the price at which such dealing took place; or
(C)if there shall not be any dealing reported for the day, the average of the closing bid and offer prices for the day;
and if there shall be only a bid (but not an offer) or an offer (but not a bid) price reported, or if there shall not be any bid or offer price reported, for any particular day then that day shall not count as one of the said five business days for the purposes of determining the average quotation. If the means of providing the foregoing information as to dealings and prices by reference to which the average quotation is to be determined is altered or is replaced by some other means, then the average quotation shall be determined on the basis of the equivalent information published by the relevant authority in relation to dealings on the New York Stock Exchange or its equivalent.
(ii)The Directors shall give notice in writing (whether in electronic form or otherwise) to the Holders of ordinary shares of the right of election offered to them and shall send with or following such notice forms of election and specify the procedure to be followed and the place at which, and the latest date and time by which, duly completed forms of election must be lodged in order to be effective. The Directors may also issue forms under which Holders may elect in advance to receive new ordinary shares instead of dividends in respect of future dividends not yet declared (and, therefore, in respect of which the basis of allotment shall not yet have been determined).
(iii)The dividend (or that part of the dividend in respect of which a right of election has been offered) shall not be payable on ordinary shares in respect of which the right of election as aforesaid has been duly exercised (the “Subject Ordinary Shares”) and in lieu thereof additional ordinary shares (but not any fraction of a share) shall be allotted to the Holders of the Subject Ordinary Shares on the basis of allotment determined aforesaid and for such purpose the Directors shall capitalise, out of such of the sums standing to the credit of any of the Company’s reserves (including any capital redemption reserve fund or share premium account) or to the credit of the profit and loss account as the Directors may determine, a sum equal to the aggregate nominal amount of additional ordinary shares to be allotted on such basis and apply the same in paying up in full the appropriate number of unissued ordinary shares for allotment and distribution to and amongst the holders of the Subject Ordinary Shares on such basis.
128.
(a)Any additional ordinary shares allotted pursuant to articles 125, 126 or 127 shall rank pari passu in all respects with the fully paid ordinary shares then in issue save only as regards participation in the relevant dividend or share election in lieu.
(b)The Directors may do all acts and things considered necessary or expedient to give effect to any capitalisation pursuant to articles 125, 126 or 127 with full power to the Directors to make such provisions as they think fit where shares would otherwise have been distributable in fractions (including provisions whereby, in whole or in part, fractional entitlements are disregarded and the benefit of fractional entitlements accrues to the Company rather than to the holders concerned). The Directors may authorise any person to enter on behalf of all the Holders interested into an agreement with the Company providing for such capitalisation and matters incidental thereto and
44
any agreement made under such authority shall be effective and binding on all concerned.
(c)The Directors may on any occasion determine that rights of election shall not be offered to any Holders of ordinary shares who are citizens or residents of any territory where the making or publication of an offer of rights of election or any exercise of rights of election or any purported acceptance of the same would or might be unlawful, and in such event the provisions aforesaid shall be read and construed subject to such determination.
ΑUDΙΤ
129.Auditors shall be appointed and their duties regulated in accordance with the Act or any statutory amendment thereof.
NOTICES
130.Any notice to be given, served, sent or delivered pursuant to these articles shall be in writing (whether in electronic form or otherwise).
131.
(a)A notice or document to be given, served, sent or delivered in pursuance of these articles may be given to, served on or delivered to any member by the Company;
(i)by handing same to him or his authorised agent;
(ii)by leaving the same at his registered address;
(iii)by sending the same by the post in a pre-paid cover addressed to him at his registered address; or
(iv)by sending, with the consent of the member, the same by means of electronic mail or other means of electronic communication approved by the Directors, with the consent of the member, to the address of the member notified to the Company by the member for such purpose (or if not so notified, then to the address of the member last known to the Company) and this article 131(a)(iv) constitutes permission of the use of electronic means within the meaning of 218(3)(d) of the Act.
(b)For the purposes of these articles and the Act, a document shall be deemed to have been sent to a member if a notice is given, served, sent or delivered to the member and the notice specifies the website or hotlink or other electronic link at or through which the member may obtain a copy of the relevant document.
(c)Where a notice or document is given, served or delivered pursuant to sub-paragraph (a)(i) or (ii) of this article, the giving, service or delivery thereof shall be deemed to have been effected at the time the same was handed to the member or his authorised agent, or left at his registered address (as the case may be).
(d)Where a notice or document is given, served or delivered pursuant to sub-paragraph (a)(iii) of this article, the giving, service or delivery thereof shall be deemed to have been effected at the expiration of twenty-four hours after the cover containing it was posted. In proving service or delivery it shall be sufficient to prove that such cover was properly addressed, stamped and posted.
(e)Where a notice or document is given, served or delivered pursuant to sub-paragraph (a)(iv) of this article, the giving, service or delivery thereof shall be deemed to have been effected at the expiration of twelve hours after despatch.
(f)Every legal personal representative, committee, receiver, curator bonis or other legal curator, assignee in bankruptcy, examiner or liquidator of a member shall be bound
45
by a notice given as aforesaid if sent to the last registered address of such member, or, in the event of notice given or delivered pursuant to sub-paragraph (a)(iv), if sent to the address notified by the Company by the member for such purpose notwithstanding that the Company may have notice of the death, lunacy, bankruptcy, liquidation or disability of such member.
(g)Notwithstanding anything contained in this article the Company shall not be obliged to take account of or make any investigations as to the existence of any suspension or curtailment of postal services within or in relation to all or any part of any jurisdiction or other area other than Ireland.
(h)Any requirement in these articles for the consent of a member in regard to the receipt by such member of electronic mail or other means of electronic communications approved by the Directors, including the receipt of the Company’s audited accounts and the directors’ and auditor’s reports thereon, shall be deemed to have been satisfied where the Company has written to the member informing him/her of its intention to use electronic communications for such purposes and the member has not, within four weeks of the issue of such notice, served an objection in writing on the Company to such proposal. Where a member has given, or is deemed to have given, his/her consent to the receipt by such member of electronic mail or other means of electronic communications approved by the Directors, he/she may revoke such consent at any time by requesting the Company to communicate with him/her in documented form; provided, however, that such revocation shall not take effect until five days after written notice of the revocation is received by the Company.
(i)Without prejudice to the provisions of sub-paragraphs (a)(i) and (ii) of this article, if at any time by reason of the suspension or curtailment of postal services in any territory, the Company is unable effectively to convene a general meeting by notices sent through the post, a general meeting may be convened by a public announcement and such notice shall be deemed to have been duly served on all members entitled thereto at noon on the day on which the said public announcement is made. In any such case the Company shall put a full copy of the notice of the general meeting on its website.
132.A notice may be given by the Company to the joint Holders of a share by giving the notice to the joint Holder whose name stands first in the Register in respect of the share and notice so given shall be sufficient notice to all the joint Holders.
133.
(a)Every person who becomes entitled to a share shall before his name is entered in the Register in respect of the share, be bound by any notice in respect of that share which has been duly given to a person from whom he derives his title.
(b)A notice may be given by the Company to the persons entitled to a share in consequence of the death or bankruptcy of a member by sending or delivering it, in any manner authorised by these articles for the giving of notice to a member, addressed to them at the address, if any, supplied by them for that purpose. Until such an address has been supplied, a notice may be given in any manner in which it might have been given if the death or bankruptcy had not occurred.
134.The signature (whether electronic signature, an advanced electronic signature or otherwise) to any notice to be given by the Company may be written (in electronic form or otherwise) or printed.
135.A member present, either in person or by proxy, at any meeting of the Company or the Holders of any class of shares in the Company shall be deemed to have received notice of the meeting and, where requisite, of the purposes for which it was called.
WINDING UP
136.If the Company shall be wound up and the assets available for distribution among the members as such shall be insufficient to repay the whole of the paid up or credited as
46
paid up share capital, such assets shall be distributed so that, as nearly as may be, the losses shall be borne by the members in proportion to the capital paid up or credited as paid up at the commencement of the winding up on the shares held by them respectively. And if in a winding up the assets available for distribution among the members shall be more than sufficient to repay the whole of the share capital paid up or credited as paid up at the commencement of the winding up, the excess shall be distributed among the members in proportion to the capital at the commencement of the winding up paid up or credited as paid up on the said shares held by them respectively. Provided that this article shall not affect the rights of the Holders of shares issued upon special terms and conditions.
137.
(a)In case of a sale by the liquidator under the Act, the liquidator may by the contract of sale agree so as to bind all the members for the allotment to the members directly of the proceeds of sale in proportion to their respective interests in the Company and may further by the contract limit a time at the expiration of which obligations or shares not accepted or required to be sold shall be deemed to have been irrevocably refused and be at the disposal of the Company, but so that nothing herein contained shall be taken to diminish, prejudice or affect the rights of dissenting members conferred by the said section.
(b)The power of sale of the liquidator shall include a power to sell wholly or partially for debentures, debenture stock, or other obligations of another company, either then already constituted or about to be constituted for the purpose of carrying out the sale.
138.If the Company is wound up, the liquidator, with the sanction of a Special Resolution and any other sanction required by the Act, may divide among the members in specie or kind the whole or any part of the assets of the Company (whether they shall consist of property of the same kind or not), and, for such purpose, may value any assets and determine how the division shall be carried out as between the members or different classes of members. The liquidator, with the like sanction, may vest the whole or any part of such assets in trustees upon such trusts for the benefit of the contributories as, with the like sanction, he determines, but so that no member shall be compelled to accept any assets upon which there is a liability.
INDEMNITY
139.
(a)Subject to the provisions of and so far as may be admitted by the Act, every Director and the Secretary of the Company shall be entitled to be indemnified by the Company against all costs, charges, losses, expenses and liabilities incurred by him in the execution and discharge of his duties or in relation thereto including any liability incurred by him in defending any proceedings, civil or criminal, which relate to anything done or omitted or alleged to have been done or omitted by him as an officer or employee of the Company and in which judgement is given in his favour (or the proceedings are otherwise disposed of without any finding or admission of any material breach of duty on his part) or in which he is acquitted or in connection with any application under any statute for relief from liability in respect of any such act or omission in which relief is granted to him by the Court.
(b)The Directors shall have power to purchase and maintain for any Director, the Secretary or other employees of the Company insurance against any such liability as referred to in the Act.
(c)As far as is permissible under the Act, the Company shall indemnify any current or former executive officer of the Company (excluding any present or former Directors of the Company or Secretary of the Company), or any person who is serving or has served at the request of the Company as a director or executive officer of another company, joint venture, trust or other enterprise, including any Company subsidiary (each individually, a “Covered Person”), against any expenses, including attorney’s fees, judgements, fines, and amounts paid in settlement actually and reasonably
47
incurred by him or her in connection with any threatened, pending, or completed action, suit or proceeding, whether civil, criminal, administrative or investigative, to which he or she was or is threatened to be made a party, or is otherwise involved (a “proceeding”), by reason of the fact that he or she is or was a Covered Person; provided, however, that this provision shall not indemnify any Covered Person against any liability arising out of (a) any fraud or dishonesty in the performance of such Covered Person’s duty to the Company, or (b) such Covered Party’s conscious, intentional or wilful breach of the obligation to act honestly and in good faith with a view to the best interests of the Company. Notwithstanding the preceding sentence, this section shall not extend to any matter which would render it void pursuant to the Act or to any person holding the office of auditor in relation to the Company.
(d)In the case of any threatened, pending or completed action, suit or proceeding by or in the name of the Company, the Company shall indemnify each Covered Person against expenses, including attorneys’ fees, actually and reasonably incurred in connection with the defence or the settlement thereof, except no indemnification shall be made in respect of any claim, issue or matter as to which such person shall have been adjudged to be liable for fraud or dishonesty in the performance of his or her duty to the Company, or for conscious, intentional or wilful breach of his or her obligation to act honestly and in good faith with a view to the best interests of the Company, unless and only to the extent that the Court or the court in which such action or suit was brought shall determine upon application that despite the adjudication of liability, but in view of all the circumstances of the case, such Covered Person is fairly and reasonably entitled to indemnity for such expenses as the court shall deem proper. Notwithstanding the preceding sentence, this section shall not extend to any matter which would render it void pursuant to the Act or to any person holding the office of auditor in relation to the Company.
(e)Any indemnification under this article (unless ordered by a court) shall be made by the Company only as authorised in the specific case upon a determination that indemnification of the Covered Person is proper in the circumstances because such person has met the applicable standard of conduct set forth in this article. Such determination shall be made by any person or persons having the authority to act on the matter on behalf of the Company. To the extent, however, that any Covered Person has been successful on the merits or otherwise in defence of any proceeding, or in defence of any claim, issue or matter therein, such Covered Person shall be indemnified against expenses (including attorneys’ fees) actually and reasonably incurred by such person in connection therewith, without necessity of authorisation in the specific case.
(f)As far as permissible under the Act, expenses, including attorneys’ fees, incurred in defending any proceeding for which indemnification is permitted pursuant to this article shall be paid by the Company in advance of the final disposition of such proceeding upon receipt by the Board of an undertaking by the particular indemnitee to repay such amount if it shall ultimately be determined that he or she is not entitled to be indemnified by the Company pursuant to these articles.
(g)It being the policy of the Company that indemnification of the persons specified in this article shall be made to the fullest extent permitted by law, the indemnification provided by this article shall not be deemed exclusive (a) of any other rights to which those seeking indemnification or advancement of expenses may be entitled under these articles, any agreement, any insurance purchased by the Company, vote of members or disinterested directors, or pursuant to the direction (however embodied) of any court of competent jurisdiction, or otherwise, both as to action in his or her official capacity and as to action in another capacity while holding such office, or (b) of the power of the Company to indemnify any person who is or was an employee or agent of the Company or of another company, joint venture, trust or other enterprise which he or she is serving or has served at the request of the Company, to the same extent and in the same situations and subject to the same determinations as are hereinabove set forth. As used in this article, references to the “Company” include all constituent companies in a scheme of arrangement, consolidation or merger in which the Company or a predecessor to the Company by scheme of arrangement, consolidation or merger was involved. The indemnification provided by this article
48
shall continue as to a person who has ceased to be a Covered Person and shall inure to the benefit of their heirs, executors, and administrators.
UNTRACED HOLDERS
140.
(a)The Company shall be entitled to sell at the best price reasonably obtainable any share or stock of a member or any share or stock to which a person is entitled by transmission if and provided that:
(i)for a period of twelve years (not less than three dividends having been declared and paid) no cheque or warrant sent by the Company through the post in a prepaid letter addressed to the member or to the person entitled by transmission to the share or stock at his address on the Register or other last known address given by the member or the person entitled by transmission to which cheques and warrants are to be sent has been cashed and no communication has been received by the Company from the member or the person entitled by transmission; and
(ii)at the expiration of the said period of twelve years the Company has given notice by advertisement in a leading Dublin newspaper and a newspaper circulating in the area in which the address referred to in paragraph (a) of this article is located of its intention to sell such share or stock; and
(iii)the Company has not during the further period of three months after the date of the advertisement and prior to the exercise of the power of sale received any communication from the member or person entitled by transmission.
(b)To give effect to any such sale the Company may appoint any person to execute as transferor an instrument of transfer of such share or stock and such instrument of transfer shall be as effective as if it had been executed by the registered Holder of or person entitled by transmission to such share or stock. The Company shall account to the member or other person entitled to such share or stock for the net proceeds of such sale by carrying all monies in respect thereof to a separate account which shall be a permanent debt of the Company and the Company shall be deemed to be a debtor and not a trustee in respect thereof for such member or other person. Monies carried to such separate account may either be employed in the business of the Company or invested in such investments (other than shares of the Company or its holding company if any) as the Directors may from time to time think fit.
(c)To the extent necessary in order to comply with any laws or regulations to which the Company is subject in relation to escheatment, abandonment of property or other similar or analogous laws or regulations (“Applicable Escheatment Laws”), the Company may deal with any share of any member and any unclaimed cash payments relating to such share in any manner which it sees fit, including (but not limited to) transferring or selling such share and transferring to third parties any unclaimed cash payments relating to such share.
(d)The Company may only exercise the powers granted to it in sub-paragraph (a) above in circumstances where it has complied with, or procured compliance with, the required procedures (as set out in the Applicable Escheatment Laws) with respect to attempting to identify and locate the relevant member of the Company.
(e)Any stock transfer form to be executed by the Company in order to sell or transfer a share pursuant to sub-paragraph (a) may be executed in accordance with article 20(a).
DESTRUCTION OF DOCUMENTS
141.The Company may implement such document destruction policies as it so chooses in relation to any type of documents (whether in paper, electronic or other formats), and in particular (without limitation to the foregoing) may destroy:
49
(a)any dividend mandate or any variation or cancellation thereof or any notification of change of name or address, at any time after the expiry of two years from the date such mandate variation, cancellation or notification was recorded by the Company;
(b)any instrument of transfer of shares which has been registered, at any time after the expiry of six years from the date of registration; and
(c)any other document on the basis of which any entry in the Register was made, at any time after the expiry of six years from the date an entry in the Register was first made in respect of it,
and it shall be presumed conclusively in favour of the Company that every share certificate (if any) so destroyed was a valid certificate duly and properly sealed and that every instrument of transfer so destroyed was a valid and effective instrument duly and properly registered and that every other document destroyed hereunder was a valid and effective document in accordance with the recorded particulars thereof in the books or records of the Company provided always that:
(i)the foregoing provisions of this article shall apply only to the destruction of a document in good faith and without express notice to the Company that the preservation of such document was relevant to a claim;
(ii)nothing contained in this article shall be construed as imposing upon the Company any liability in respect of the destruction of any such document earlier than as aforesaid or in any case where the conditions of proviso (a) above are not fulfilled; and
(iii)references in this article to the destruction of any document include references to its disposal in any manner.
SHAREHOLDER RIGHTS PLAN
142.Subject to applicable law, the Directors are hereby expressly authorised to adopt any shareholder rights plan (a “Rights Plan”), upon such terms and conditions as the Directors deem expedient and in the best interests of the Company, including, without limitation, where the Directors are of the opinion that a Rights Plan could grant them additional time to gather relevant information or pursue strategies in response to or anticipation of, or could prevent, a potential change of control of the Company or accumulation of shares in the Company or interests therein.
143.The Directors may exercise any power of the Company to grant rights (including approving the execution of any documents relating to the grant of such rights) to subscribe for ordinary shares or preferred shares in the share capital of the Company (“Rights”) in accordance with the terms of a Rights Plan.
144.For the purposes of effecting an exchange of Rights for ordinary shares or preferred shares in the share capital of the Company (an “Exchange”), the Directors may:
(a)resolve to capitalise an amount standing to the credit of the reserves of the Company (including, but not limited to, the share premium account, capital redemption reserve and profit and loss account), whether or not available for distribution, being an amount equal to the nominal value of the ordinary shares or preferred shares which are to be exchanged for the Rights; and
(b)apply that sum in paying up in full ordinary shares or preferred shares and allot such shares, credited as fully paid, to those holders of Rights who are entitled to them under an Exchange effected pursuant to the terms of a Rights Plan.
145.The common law duties of the Directors to the Company are hereby deemed amended and modified such that the adoption of a Rights Plan and any actions taken thereunder by the Directors (if so approved by the Directors) shall be deemed to constitute an
50
action in the best interests of the Company in all circumstances, and any such action shall be deemed to be immediately confirmed, approved and ratified.
BUSINESS COMBINATION
146.
(a)Notwithstanding anything to the contrary contained in these articles, the Company shall not engage in any business combination with any Interested Member for a period of three years following the time that such member became an Interested Member, unless:
(i)prior to such time the Directors approved either the business combination or the transaction which resulted in the member becoming an Interested Member;
(ii)upon consummation of the transaction which resulted in the member becoming an Interested Member, the Interested Member owned at least 85% of the voting shares of the Company outstanding at the time the transaction commenced, excluding for purposes of determining the voting shares outstanding (but not the outstanding voting shares owned by the Interested Member) those shares owned (A) by persons who are directors and also officers and (B) employee shares plans in which employee participants do not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or
(iii)at or subsequent to such time the business combination is approved by the Directors and authorised by way of Special Resolution without the Interested Member.
(b)The Directors shall have the power and duty to determine, on the basis of information known to them after reasonable inquiry, all facts necessary to determine compliance with this article, including, without limitation, (i) whether a Person is an Interested Member, (ii) the number of shares or other securities beneficially owned by any Person, (iii) whether a Person is an Affiliate or Associate of another, and (iv) the fair market value of the Company’s securities or securities of any subsidiary of the Company, and the good faith determination of the Directors on such matters shall be conclusive and binding for all the purposes of this article.
(c)As used in this article only, the term:
(i)“Affiliate” means a person that directly, or indirectly through one or more intermediaries, controls or is controlled by, or is under common control with, another person.
(ii)“Associate”, when used to indicate a relationship with any person, means: (A) any company, partnership, unincorporated association or other entity of which such person is a director, officer or partner or is, directly or indirectly, the owner of 20% or more of any class of voting shares; (B) any trust or other estate in which such person has at least a 20% beneficial interest or as to which such person serves as trustee or in a similar fiduciary capacity; and (C) any relative or spouse of such person, or any relative of such spouse, who has the same residence as such person.
(iii)“Business combination”, when used in reference to any company and any Interested Member of such company, means:
(A)any scheme of arrangement, merger or consolidation of the Company or any direct or indirect majority-owned subsidiary of the Company with (1) the Interested Member, or (2) any other company, partnership, unincorporated association or other entity if the scheme of arrangement, merger or consolidation is caused by the Interested Member;
51
(B)any sale, lease, exchange, mortgage, pledge, transfer or other disposition (in one transaction or a series of transactions), except proportionately as a member of such company, to or with the Interested Member, whether as part of a dissolution or otherwise, of assets of the Company or of any direct or indirect majority-owned subsidiary of the Company which assets have an aggregate market value equal to 10% or more of either the aggregate market value of all the assets of the Company determined on a consolidated basis or the aggregate market value of all the outstanding shares of the Company;
(C)any transaction which results in the issuance or transfer by the Company or by any direct or indirect majority-owned subsidiary of the Company of any shares of the Company or of such subsidiary to the Interested Member, except: (1) pursuant to the exercise, exchange or conversion of securities exercisable for, exchangeable for or convertible into shares of such company or any such subsidiary which securities were outstanding prior to the time that the Interested Member became such; (2) pursuant to a dividend or distribution paid or made, or the exercise, exchange or conversion of securities exercisable for, exchangeable for or convertible into shares of such company or any such subsidiary which security is distributed, pro rata to all holders of a class or series of shares of such company subsequent to the time the Interested Member became such; (3) pursuant to an exchange offer by the Company to purchase shares made on the same terms to all holders of said shares; or (4) any issuance or transfer of shares by the Company; provided however, that in no case under items (3) and (4) of this subparagraph shall there be an increase in the Interested Member’s proportionate share of the shares of any class or series of the Company or of the voting shares of the Company;
(D)any transaction involving the Company or any direct or indirect majority-owned subsidiary of the Company which has the effect, directly or indirectly, of increasing the proportionate share of the shares of any class or series, or securities convertible into the shares of any class or series, of the Company or of any such subsidiary which is owned by the Interested Member, except as a result of immaterial changes due to fractional share adjustments or as a result of any purchase or redemption of any shares of shares not caused, directly or indirectly, by the Interested Member; or
(E)any receipt by the Interested Member of the benefit, directly or indirectly (except proportionately as a member of such company), of any loans, advances, guarantees, pledges or other financial benefits (other than those expressly permitted in subparagraphs (A)-(D) of this paragraph) provided by or through the Company or any direct or indirect majority-owned subsidiary.
(iv)“Control”, including the terms “controlling”, “controlled by” and “under common control with”, means the possession, directly or indirectly, of the power to direct or cause the direction of the management and policies of a person, whether through the ownership of voting shares, by contract or otherwise. A person who is the owner of 20% or more of the outstanding voting shares of any company, partnership, unincorporated association or other entity shall be presumed to have control of such entity, in the absence of proof by a preponderance of the evidence to the contrary. Notwithstanding the foregoing, a presumption of control shall not apply where such person holds voting shares, in good faith and not for the purpose of circumventing this article, as an agent, bank, broker, nominee, custodian or trustee for one or more owners who do not individually or as a group have control of such entity.
52
(v)“Interested Member” means any Person, including its Affiliates and Associates (other than the Company and any direct or indirect majority-owned subsidiary of the Company), that is, or was at any time within the three-year period immediately prior to the date in question, the Owner of 15% or more of the outstanding voting shares of the Company; provided, however, that the term “Interested Member” shall not include any person whose ownership of shares in excess of the 15% limitation set forth herein is the result of action taken solely by the Company; provided that such person shall be an Interested Member if thereafter such person acquires additional voting shares of the Company, except as a result of further corporate action not caused, directly or indirectly, by such person. For the purpose of determining whether a person is an Interested Member, the voting shares of the Company deemed to be outstanding shall include shares deemed to be owned by the person through application of (viii) of this subsection but shall not include any other unissued shares of such company which may be issuable pursuant to any agreement, arrangement or understanding, or upon exercise of conversion rights, warrants or options, or otherwise.
(vi)“Person” means any individual, company, partnership, unincorporated association or other entity.
(vii)“Shares” means, with respect to any company, capital shares and, with respect to any other entity, any equity interest.
(viii)“Voting shares” means, with respect to any company, shares of any class or series entitled to vote generally in the election of directors and, with respect to any entity that is not a company, any equity interest entitled to vote generally in the election of the governing body of such entity. Every reference to a percentage of voting shares shall refer to such percentage of the votes of such voting shares.
(ix)“Owner”, including the terms “own” and “owned”, when used with respect to any Shares, means a person that individually or with or through any of its Affiliates or Associates:
(A)beneficially owns such Shares, directly or indirectly; or
(B)has (1) the right to acquire such Shares (whether such right is exercisable immediately or only after the passage of time) pursuant to any agreement, arrangement or understanding, or upon the exercise of conversion rights, exchange rights, warrants or options, or otherwise; provided, however, that a person shall not be deemed the Owner of Shares tendered pursuant to a tender or exchange offer made by such person or any of such person’s affiliates or associates until such tendered Shares are accepted for purchase or exchange; or (2) the right to vote such shares pursuant to any agreement, arrangement or understanding; provided, however, that a person shall not be deemed the Owner of any Shares because of such person’s right to vote such Shares if the agreement, arrangement or understanding to vote such shares arises solely from a revocable proxy or consent given in response to a proxy or consent solicitation made to 10 or more persons; or
(C)has any agreement, arrangement or understanding for the purpose of acquiring, holding, voting (except voting pursuant to a revocable proxy or consent as described in item (2) of subparagraph (B) of this paragraph), or disposing of such Shares with any other person that beneficially owns, or whose Affiliates or Associates beneficially own, directly or indirectly, such Shares.
53
We, the several persons whose names and addressed are subscribed, wish to be formed into a company in pursuance of this memorandum of association and we agree to take the number of shares in the capital of the company set opposite our respective names.
Names, addresses and descriptions of subscribers | Number of shares taken by each subscriber | ||||
/s/ Fiona McGowan-Smyth For and on behalf of Fand Limited Arthur Cox Building Earlsfort Terrace Dublin 2 Corporate Body | One Ordinary Share | ||||
/s/ Fiona McGowan-Smyth For and on behalf of DIJR Nominees Limited Arthur Cox Building Earlsfort Terrace Dublin 2 Corporate Body | One Ordinary Share | ||||
/s/ Fiona McGowan-Smyth For and on behalf of AC Administration Services Limited Arthur Cox Building Earlsfort Terrace Dublin 2 Corporate Body | One Ordinary Share | ||||
/s/ Fiona McGowan-Smyth For and on behalf of Arthur Cox Nominees Limited Arthur Cox Building Earlsfort Terrace Dublin 2 Corporate Body | One Ordinary Share | ||||
54
/s/ Fiona McGowan-Smyth For and on behalf of Aurther Cox Registrars Limited Arthur Cox Building Earlsfort Terrace Dublin 2 Corporate Body | One Ordinary Share | ||||
/s/ Fiona McGowan-Smyth For and on behalf of Arthur Cox Trust Services Limited Arthur Cox Building Earlsfort Terrace Dublin 2 Corporate Body | One Ordinary Share | ||||
/s/ Fiona McGowan-Smyth For and on behalf of Arthur Cox Trustees Limited Arthur Cox Building Earlsfort Terrace Dublin 2 Corporate Body | One Ordinary Share | ||||
Dated the 21st day of December 2012 /s/ Mairéad Foley Witnesses to the above signatures: Name: Mairéad Foley Address: Arthur Cox Building, Earlsfort Terrace, Dublin 2 Occupation: Company Secretary | |||||
55
Companies Act 2014
A PUBLIC COMPANY LIMITED BY SHARES
MEMORANDUM AND ARTICLES OF ASSOCIATION
OF
KEENOVA THERAPEUTICS PUBLIC LIMITED COMPANY
(as amended by Special Resolution passed on 8 October 2025)
Arthur Cox
Arthur Cox Building
Earlsfort Terrace
Dublin
56
Exhibit 4.1
DESCRIPTION OF THE REGISTRANT’S SECURITIES
REGISTERED PURSUANT TO SECTION 12 OF THE
SECURITIES EXCHANGE ACT OF 1934
The following description of the share capital of Keenova Therapeutics plc (“Keenova” or the “Company”) is a summary. This summary does not purport to be complete and is qualified in its entirety by reference to the Irish Companies Act 2014 (the “Companies Act”) and the complete text of Keenova’s memorandum and articles of association (the “Memorandum and Articles of Association”), each of which are incorporated by reference as an exhibit to the Annual Report on Form 10-K of which this exhibit is a part. You should read those laws and documents carefully. As used in this exhibit, “we,” and “our” refer to Keenova.
Description of Ordinary Shares
Legal Name; Formation; Fiscal Year; Registered Office
The legal name of the company is Keenova Therapeutics public limited company. Keenova was incorporated in Ireland as a public limited company on January 9, 2013 with company registration number 522227. Keenova’s fiscal year ends on 31 December and Keenova’s registered address is College Business & Technology Park, Cruiserath, Blanchardstown, Dublin 15, Ireland.
Share Capital
The authorized share capital of Keenova is $3,005,000,000 and €25,000 , divided into 500,000,000 ordinary shares with a par value of $0.01 per share, 3,000,000,000,000 preferred shares with a par value of $0.001 per share and 25,000 ordinary A shares with a par value of €1.00 per share.
Keenova may issue shares subject to the maximum prescribed by its authorized share capital contained in its Memorandum and Articles of Association.
As a matter of Irish company law, the directors of a company may cause the company to issue new ordinary or preferred shares without shareholder approval once authorized to do so by the articles of association of the company or by an ordinary resolution adopted by the shareholders at a general meeting. Under the Memorandum and Articles of Association, the board of directors of Keenova was granted authority to issue shares up to the amount of its authorized share capital. This authority will expire five years from the date on which the Memorandum and Articles of Association were adopted, at which point it may be renewed by the shareholders of the company by an ordinary resolution. An ordinary resolution requires over 50% of the votes of a company’s shareholders cast at a general meeting (in person or by proxy).
The authorized share capital may be increased or reduced (but not below the number of issued ordinary shares, preferred shares or ordinary A shares, as applicable) by way of an ordinary resolution of Keenova’s shareholders, but not below the number of shares then outstanding. The shares comprising the authorized share capital of Keenova may be divided into shares of such par value as the resolution prescribes.
The rights and restrictions to which the ordinary shares are subject are prescribed in Keenova’s Memorandum and Articles of Association. Keenova’s Memorandum and Articles of Association entitle the board of directors, without shareholder approval, to determine the terms of the preferred shares issued by Keenova. Preferred shares may be preferred as to dividends, rights on a winding up, voting or in such manner as the directors of Keenova may resolve. The preferred shares may also be redeemable at the option of the holder of the preferred shares or at the option of Keenova, and may be convertible into or exchangeable for shares of any other class or classes of Keenova, depending on the terms of such preferred shares. The issuance of preferred shares is subject to applicable law.
Irish law does not recognize fractional shares held of record; accordingly, Keenova’s Memorandum and Articles of Association do not provide for the issuance of fractional ordinary shares of Keenova, and the official Irish register of Keenova will not reflect any fractional ordinary shares.
1
Whenever an alteration or reorganization of the share capital of Keenova would result in any Keenova shareholder becoming entitled to fractions of a share, the Keenova board of directors may, on behalf of those shareholders that would become entitled to fractions of a share, arrange for the sale of the shares representing fractions and the distribution of the net proceeds of sale in due proportion among the shareholders who would have been entitled to the fractions. For the purpose of any such sale the board may authorize some person to transfer the shares representing fractions to the purchaser, who shall not be bound to see to the application of the purchase money, nor shall his title to the shares be affected by any irregularity or invalidity in the proceedings relating to the sale.
Preemption Rights, Share Warrants and Share Options
Under Irish law, certain statutory preemption rights apply automatically in favor of Keenova’s shareholders where securities are to be issued for cash, unless Keenova is authorized to opt out of these preemption rights. Under the Memorandum and Articles of Association, Keenova has opted out of these pre-emption rights for a period of five years. This opt-out will expire five years from the date on which the Memorandum and Articles of Association were adopted, at which point it may be renewed by the shareholders of the company by a special resolution of the shareholders. A special resolution requires not less than 75% of the votes of Keenova’s shareholders cast at a general meeting (in person or by proxy). If the opt-out is not renewed, shares issued for cash must be offered to pre-existing shareholders of Keenova pro-rata to their existing shareholding before the shares can be issued to any new shareholders. The statutory preemption rights do not apply where shares are issued for non-cash consideration (such as in a stock-for-stock acquisition) and do not apply to the issue of non-equity shares (that is, shares that have the right to participate only up to a specified amount in any income or capital distribution) or when shares are issued pursuant to an employee option or similar equity plan.
The Memorandum and Articles of Association of Keenova provide that, subject to any shareholder approval requirement under any laws, regulations or the rules of any stock exchange to which Keenova is subject, the board is authorized, from time to time, in its discretion, to grant such persons, for such periods and upon such terms as the board deems advisable, options to purchase or subscribe for such number of shares of any class or classes or of any series of any class as the board may deem advisable, and to cause warrants or other appropriate instruments evidencing such options to be issued. The Companies Act provides that directors may issue share warrants or options without shareholder approval once authorized to do so by the memorandum and articles of association or an ordinary resolution of shareholders. Under Irish law, the board may issue shares upon exercise of validly issued warrants or options without shareholder approval or authorization.
Dividends
Under Irish law, dividends and distributions may only be made from “distributable reserves.” Distributable reserves, broadly, means the accumulated realized profits of Keenova less accumulated realized losses of Keenova and includes reserves created by way of capital reduction. In addition, no distribution or dividend may be made unless the net assets of Keenova are equal to, or in excess of, the aggregate of Keenova’s called up share capital plus its undistributable reserves and the distribution does not reduce Keenova’s net assets below such aggregate. Undistributable reserves include the share premium account, the capital redemption reserve fund and the amount by which Keenova’s accumulated unrealized profits, so far as not previously utilized by any capitalization, exceed Keenova’s accumulated unrealized losses, so far as not previously written off in a reduction or reorganization of capital.
The determination as to whether or not Keenova has sufficient distributable reserves to fund a dividend must be made by reference to the “relevant financial statements” of Keenova. The “relevant financial statements” are either the last set of unconsolidated annual audited financial statements or unaudited financial statements prepared in accordance with the Companies Act, which give a “true and fair view” of Keenova’s unconsolidated financial position and accord with accepted accounting practice.
The mechanism as to who declares a dividend and when a dividend becomes payable is governed by the Memorandum and Articles of Association of Keenova. Keenova’s Memorandum and Articles of Association authorize the directors to declare such dividends as appear justified from the profits of Keenova without the approval of the shareholders at a general meeting. The board of directors may also recommend a dividend to be approved and declared by the shareholders at a general meeting. Any general meeting declaring a dividend and any resolution of the directors declaring a dividend may direct that the payment be made by distribution of
2
assets, shares or cash, no dividend issued may exceed the amount recommended by the directors. No dividend issued may exceed the amount recommended by the directors. The dividends can be declared and paid in the form of assets, shares or cash.
The directors of Keenova may deduct from any dividend payable to any shareholder all sums of money (if any) immediately payable by such shareholder to Keenova in relation to the shares of Keenova.
The directors of Keenova are also entitled to issue shares with preferred rights to participate in dividends declared by Keenova. The holders of such preferred shares may, depending on their terms, be entitled to claim arrears of a declared dividend out of subsequently declared dividends in priority to ordinary shareholders. The holders of ordinary A shares are not entitled to receive any dividend.
Share Repurchases and Redemptions
Overview
Keenova’s Memorandum and Articles of Association provide that unless the board of directors specifically resolves to treat such acquisition as a purchase for the purposes of the Companies Act, any ordinary share or an interest in any ordinary share which Keenova has acquired or agreed to acquire from a third party is deemed to be a redeemable share. Accordingly, for Irish company law purposes, the repurchase of ordinary shares by Keenova may technically be effected as a redemption of those shares as described below under “—Share Repurchases and Redemptions—Repurchases and Redemptions by Keenova.” If such shares were not to be deemed to be redeemable shares, their repurchase by Keenova would be subject to additional requirements imposed by Irish law. Neither Irish law nor any constituent document of Keenova places limitations on the right of non-resident or foreign owners to vote or hold Keenova ordinary shares. Except where otherwise noted, when we refer elsewhere in this exhibit to repurchasing or buying back ordinary shares of Keenova, we are referring to the redemption of ordinary shares by Keenova or the purchase of Keenova ordinary shares by a subsidiary of Keenova, in each case in accordance with the Keenova Memorandum and Articles of Association and Irish company law as described below.
Repurchases and Redemptions by Keenova
Under Irish law, a company can issue redeemable shares and redeem them out of distributable reserves (which are described above under “—Dividends”) or the proceeds of a new issue of shares for that purpose. The issue of redeemable shares may only be made by Keenova where the nominal value of the issued share capital that is not redeemable is not less than 10% of the nominal value of the total issued share capital of Keenova. All redeemable shares must also be fully paid and the terms of redemption of the shares must provide for payment on redemption. Based on the provision of Keenova’s Memorandum and Articles of Association described above, shareholder approval is not required to redeem Keenova ordinary shares.
The board of directors of Keenova is also entitled to issue preferred shares which may be redeemed at the option of either Keenova or the shareholder, depending on the terms of such preferred shares. For additional information on redeemable shares, see “—Share Capital.”
Keenova may also be given an additional general authority by its shareholders to purchase its own shares as overseas market purchases on a recognized stock exchange such as the New York Stock Exchange or the Nasdaq stock market, which would take effect on the same terms and be subject to the same conditions as applicable to purchases by Keenova’s subsidiaries as described below.
Repurchased and redeemed shares may be cancelled or held as treasury shares. The nominal value of treasury shares held by Keenova at any time must not exceed 10% of the nominal value of the issued share capital of Keenova. While Keenova holds shares as treasury shares, it cannot exercise any voting rights in respect of those shares. Treasury shares may be cancelled by Keenova or re-issued subject to certain conditions.
Purchases by Subsidiaries of Keenova
Under Irish law, it may be permissible for an Irish or non-Irish subsidiary to purchase ordinary shares of Keenova either as overseas market purchases on a recognized stock exchange or off-market. A general authority
3
of the shareholders of Keenova is required to allow a subsidiary of Keenova to make on-market purchases of Keenova ordinary shares; however, as long as this general authority has been granted, no specific shareholder authority for a particular on-market purchase by a subsidiary of Keenova ordinary shares is required.
In order for a subsidiary of Keenova to make an on-market purchase of Keenova’s ordinary shares, such shares must be purchased on a “recognized stock exchange.” Each of the New York Stock Exchange and the Nasdaq stock market are specified as a recognized stock exchange for this purpose by Irish company law.
For an off-market purchase by a subsidiary of Keenova, the proposed purchase contract must be authorized by special resolution of the shareholders of Keenova before the contract is entered into. The person whose shares are to be bought back cannot vote in favor of the special resolution and, for at least 21 days prior to the special resolution, the purchase contract must be on display or must be available for inspection by shareholders at the registered office of Keenova.
The number of shares held by the subsidiaries of Keenova at any time will count as treasury shares and will be included in any calculation of the permitted treasury share threshold of 10% of the nominal value of the issued share capital of Keenova. While a subsidiary holds Keenova ordinary shares, it cannot exercise any voting rights in respect of those shares. The acquisition of the ordinary shares of Keenova by a subsidiary must be funded out of distributable reserves of the subsidiary.
Lien on Shares, Calls on Shares and Forfeiture of Shares
Keenova’s Memorandum and Articles of Association provide that Keenova will have a first and paramount lien on every share for all moneys, whether presently due or not, payable in respect of such Keenova ordinary share. Subject to the terms of their allotment, directors may call for any unpaid amounts in respect of any shares to be paid, and if payment is not made, the shares may be forfeited. These provisions are standard inclusions in the memorandum and articles of association of an Irish company limited by shares such as Keenova and will only be applicable to Keenova shares that have not been fully paid up.
Bonus Shares
Under Keenova’s Memorandum and Articles of Association, the board may resolve to capitalize any amount for the time being standing to the credit of Keenova’s reserves accounts or to the credit of the profit and loss account which is not available for distribution by applying such sum in paying up in full unissued shares to be allotted as fully paid-up bonus shares to shareholders of Keenova who would have been entitled to that sum if it were distributable and had been distributed by way of dividend (and in the same proportions).
Consolidation and Division; Subdivision
Under the Memorandum and Articles of Association, Keenova may, by ordinary resolution, consolidate and divide all or any of its share capital into shares of larger par value than its existing shares or subdivide its shares into smaller amounts than are fixed by its Memorandum and Articles of Association.
Reduction of Share Capital
Keenova may, by ordinary resolution, reduce its authorized but unissued share capital in any way. Keenova also may, by special resolution and subject to confirmation by the Irish High Court, reduce or cancel its issued share capital (which includes share premium) in any way permitted by the Companies Act.
Annual General Meetings of Shareholders
Keenova held its first annual general meeting on March 20, 2014, and is required to hold subsequent annual general meetings at intervals of no more than 15 months thereafter, provided that an annual general meeting is held in each calendar year following the first annual general meeting, no more than nine months after Keenova’s fiscal year end. Any annual general meeting may be held outside Ireland pursuant to the Memorandum and Articles of Association and subject to the requirements of Irish law. Because of the 15-month requirement described in this paragraph, Keenova’s Memorandum and Articles of Association include a provision reflecting this requirement of Irish law.
4
Notice of an annual general meeting must be given to all Keenova shareholders and to the auditors of Keenova. The Memorandum and Articles of Association of Keenova provide for a minimum notice period of 21 days, which is the minimum permitted under Irish law.
The only matters which must, as a matter of Irish company law, be transacted at an annual general meeting are the review by the members of the company’s affairs, presentation of the statutory financial statements and reports of the directors and auditors, the appointment of new auditors and the fixing of the auditor’s remuneration (or delegation of same). If no resolution is made in respect of the reappointment of an existing auditor at an annual general meeting, the existing auditor will be deemed to have continued in office.
At any annual general meeting, only such business may be conducted as has been brought before the meeting (i) by or at the direction of the board of directors, (ii) in certain circumstances, at the direction of the Irish High Court, (iii) as required by law or (iv) such business that the chairman of the meeting or the board of directors determines is properly brought before the meeting. Shareholders entitled to vote at an annual general meeting may make nominations of candidates for election to the board of directors pursuant to the Memorandum and Articles of Association.
Extraordinary General Meetings of Shareholders
Extraordinary general meetings of Keenova may be convened by (i) the board of directors, (ii) on requisition of the shareholders holding not less than 10% of the paid-up share capital of Keenova carrying voting rights, (iii) on requisition of Keenova’s auditors upon their resignation or (iv) in exceptional cases, by court order. Extraordinary general meetings are generally held for the purposes of approving shareholder resolutions of Keenova as may be required from time to time. At any extraordinary general meeting only such business shall be conducted as is either (i) set forth in the notice of the meeting, (ii) brought before the meeting by or at the direction of the board of directors, or (iii) properly brought before the meeting by any members of Keenova pursuant to the valid exercise of power granted to them under the Companies Act and in accordance with the Memorandum and Articles of Association.
Notice of an extraordinary general meeting must be given to all Keenova shareholders and to the auditors of Keenova. Under Irish law and the Memorandum and Articles of Association, the minimum notice periods are 21 days’ notice in writing for an extraordinary general meeting to approve a special resolution and 14 days’ notice in writing for any other extraordinary general meeting. General meetings may be called by shorter notice in accordance with the terms of the Companies Act.
In the case of an extraordinary general meeting convened by shareholders of Keenova, the proposed purpose of the meeting must be set out in the requisition notice. The requisition notice can contain any resolution. Upon receipt of this requisition notice, the board of directors has 21 days to convene a meeting of Keenova’s shareholders to vote on the matters set out in the requisition notice. This meeting must be held within two months of the receipt of the requisition notice. If the board of directors does not convene the meeting within such 21-day period, the requisitioning shareholders, or any of them representing more than one half of the total voting rights of all of them, may themselves convene a meeting, which meeting must be held within three months of the receipt of the requisition notice.
If the directors become aware that the net assets of Keenova are half or less of the amount of Keenova’s called-up share capital, the directors of Keenova must convene an extraordinary general meeting of Keenova’s shareholders not later than 28 days from the date that they learn of this fact. This meeting must be convened for the purposes of considering whether any, and if so what, measures should be taken to address the situation.
Voting
Where a vote is to be taken at a general meeting, every shareholder has one vote for each ordinary share that he or she holds as of the record date for the meeting. Voting rights may be exercised by shareholders registered in Keenova’s share register as of the record date for the meeting or by a duly appointed proxy of such a registered shareholder, which proxy need not be a shareholder. Where interests in shares are held by a nominee trust company, this company may exercise the rights of the beneficial holders on their behalf as their proxy. All proxies must be appointed in the manner prescribed by Keenova’s Memorandum and Articles of Association.
5
The Memorandum and Articles of Association of Keenova permit the appointment of proxies by the shareholders to be notified to Keenova electronically.
Except where a greater majority is required by the Companies Act, any question, business or resolution proposed at any general meeting shall be decided by a simple majority of the votes cast.
Keenova’s Memorandum and Articles of Association provide that all resolutions are decided on a poll. In the case of an equality of votes on a poll, the chair of the meeting has a casting vote.
In accordance with Keenova’s Memorandum and Articles of Association, the board of directors may from time to time cause Keenova to issue preferred or any other class or series of shares. These shares may have such voting rights, if any, as may be specified in the terms of such shares (i.e. they may carry more votes per share than ordinary shares or may entitle their holders to a class vote on such matters as may be specified in the terms of the shares).
The holders of ordinary A shares shall not be entitled to receive notice of, nor attend, speak or vote at, general meetings of shareholders. Treasury shares and shares held by subsidiaries will not be entitled to vote at general meetings of shareholders.
Irish company law requires “special resolutions” of the shareholders at a general meeting to approve certain matters. A special resolution requires not less than 75% of the votes cast of Keenova’s shareholders present in person or by proxy at a general meeting. This may be contrasted with “ordinary resolutions,” which require a simple majority of the votes of Keenova’s shareholders cast in person or by proxy at a general meeting. Examples of matters requiring special resolutions include:
•amending the objects (i.e., main purposes) of Keenova;
•amending the Memorandum and Articles of Association of Keenova;
•approving a change of name of Keenova;
•authorizing the entering into of a guarantee or provision of security in connection with a loan, quasi-loan or credit transaction to a director or a person who is deemed to be “connected” to a director for the purposes of the Companies Act;
•opting-out of preemption rights on the issuance of new shares;
•re-registration of Keenova from a public limited company to a private company;
•variation of class rights attaching to classes of shares;
•purchasing Keenova’s ordinary shares off-market;
•any reduction of Keenova’s issued share capital;
•resolving that Keenova be wound up by the Irish courts;
•sanctioning a compromise/scheme of arrangement;
•resolving in favor of a shareholders’ voluntary winding-up;
•re-designation of shares into different share classes; and
•setting the re-issue price of treasury shares.
Unanimous Shareholder Consent to Action Without Meeting
The Companies Act provides that shareholders may approve an ordinary or special resolution of shareholders without a meeting only if (a) all shareholders entitled to attend and vote on such resolution sign the written resolution and (b) the company’s memorandum and articles of association permit written resolutions of shareholders. Keenova’s Memorandum and Articles of Association permit unanimous written resolutions of shareholders entitled to attend and vote on such resolution, subject to the Companies Act and the U.S. Securities Exchange Act of 1934, as amended (the “Exchange Act”).
Variation of Class Rights Attaching to Shares
Variation of all or any special rights attached to any class of shares of Keenova is addressed in the Memorandum and Articles of Association of Keenova as well as the Companies Act. Any variation of class rights attaching to the issued shares of Keenova must be approved by a special resolution of the shareholders of the class affected. Keenova’s Memorandum and Articles of Association expressly provide that any issue of preferred shares
6
(whatever the rights attaching to them) will be deemed not to be a variation of the rights of ordinary shareholders.
The provisions of the Memorandum and Articles of Association of Keenova relating to general meetings shall apply to every such general meeting of the holders of any class of shares with certain exceptions in relation to quorum and voting.
Quorum for General Meetings
The presence, in person or by proxy, of the holders of shares in Keenova entitling them to exercise a majority of the voting power of Keenova constitutes a quorum for the conduct of business. No business may take place at a general meeting of Keenova if a quorum is not present in person or by proxy. The board of directors has no authority to waive quorum requirements stipulated in the Memorandum and Articles of Association of Keenova. Abstentions and broker non-votes will be counted as present for purposes of determining whether there is a quorum in respect of the proposals.
Requirements for Advance Notification of Director Nominations and Proposals of Shareholders
Keenova’s Memorandum and Articles of Association provide that with respect to an annual or extraordinary general meeting of shareholders, nominations of persons for election to its board of directors and the proposal of business to be considered by shareholders may be made only (i) pursuant to Keenova’s notice of meeting, (ii) by the board of directors, (iii) by any shareholders pursuant to the valid exercise of power granted to them under the Companies Act, or (iv) such nomination or business that the chairman of the meeting or the board of directors determines is properly brought before the meeting by a shareholder who is entitled to vote at the meeting and who has complied with the advance notice procedures provided for in the Memorandum and Articles of Association.
In order to comply with the advance notice procedures of Keenova’s Memorandum and Articles of Association, a shareholder must give written notice to Keenova’s secretary on a timely basis. To be timely for an annual general meeting, notice must be delivered not earlier than the close of business on the 120th day and not later than the close of business on the 90th day prior to the first anniversary of the preceding year’s annual general meeting, provided, however, that in the event that no annual general meeting was held in the previous year or the date of the annual general meeting is more than 30 days before or more than 60 days after such anniversary date, notice by the member must be so delivered not earlier than the close of business on the 120th day prior to the date of such annual general meeting and not later than the close of business on the later of the 90th day prior to the date of such annual general meeting or, if the first public announcement of the date of such annual general meeting is less than 100 days prior to the date of such annual general meeting, the 10th day following the day on which public announcement is first made of the date of the annual general meeting. In no event shall the public announcement of an adjournment, recess, rescheduling or postponement of an annual general meeting commence a new time period (or extend any time period) for the giving of a shareholder’s notice.
To be timely for an extraordinary general meeting, notice must be delivered not earlier than the close of business on the 120th day prior to the date of such extraordinary general meeting and not later than the close of business on the 90th day prior to the date of such extraordinary general meeting or, if the first public announcement of the date of such extraordinary general meeting is less than 100 days prior to the date of such extraordinary general meeting, the 10th day following the day on which public announcement is first made of the date of the extraordinary general meeting and of the nominees proposed by the board of directors to be elected at such meeting. In no event shall the public announcement of an adjournment, recess, rescheduling or postponement of an extraordinary general meeting commence a new time period (or extend any time period) for the giving of a shareholder’s notice.
In addition, whether relating to an annual or extraordinary general meeting, to be timely, a shareholder’s notice must be updated and supplemented, if necessary, so the information provided or required to be provided is true and correct as of the record date for the meeting and as of the date that is 10 days prior to the meeting or any adjournment, recess, rescheduling or postponement thereof. Such update and supplement shall be delivered to Keenova’s secretary not later than (i) the later of (a) 10 days after the record date for the meeting or (b) the first public announcement of the date of notice of such record date in the case of the update and supplement required to be made as of the record date, and (ii) 8 days prior to the meeting or any adjournment, recess, rescheduling or
7
postponement thereof in the case of the update and supplement required to be made as of 10 days prior to the meeting or any adjournment, recess, rescheduling or postponement thereof. In addition, with respect to nominations, the shareholder must provide to Keenova reasonable evidence of compliance with Rule 14a‑19 under the Exchange Act no later than 5 business days prior to the meeting or, if practicable, any adjournment, recess, rescheduling or postponement thereof (or, if not practicable, on the first practicable date prior to the date to which the meeting has been adjourned, recessed, rescheduled, or postponed).
For nominations to the board, the notice must include (i) the name, age, business and residence address of such director nominee, (ii) principal occupation or employment of such director nominee (present and for the past 5 years), (iii) completed and signed questionnaire and representation and agreement as prescribed in the Memorandum and Articles of Association, (iv) all information about the director nominee that is required to be disclosed by SEC rules regarding the solicitation of proxies for the election of directors pursuant to Section 14 under the Exchange Act (including such person’s written consent to being named in the proxy statement as a nominee and to serving as a director for the full term if elected), and(v) a description of all direct and indirect compensation and other material monetary agreements or arrangements during the past three years, any other material relationships between the nominating shareholder, and their affiliates and associates or others acting in concert, and the proposed nominee and his or her affiliates and associates and other concert parties (including, but not limited to, all biographical and related party transaction and other information that would be required to be disclosed pursuant to Rule 404 promulgated under Regulation S-K under the Exchange Act). In addition to the notice, Keenova can require the nominating shareholder, before the applicable annual or extraordinary general meeting, to provide within 5 business days of any such request such other information as Keenova may reasonably require to determine the (a) eligibility of the proposed nominee, (b) whether the proposed nominee qualifies as an “independent director” or “audit committee financial expert”, or (c) such information that the board of directors determines could be material to reasonably understand the independence, or lack thereof, of such nominee.
For other business that a shareholder proposes to bring before the meeting, the notice must include a brief description of the business, the reasons for proposing the business at the meeting, the text of the proposal or wording (including the text of any proposed resolutions for consideration and if such business includes a proposal to amend the Memorandum and Articles of Association of Keenova, the text of the proposed amendment), a discussion of any material interest of the shareholder or beneficial owner and each of their respective affiliates or associates, or others acting in concert therewith in the business and a description of all arrangements between the shareholder(s) or beneficial owner(s) and each of their respective affiliates or associates, or others acting in concert therewith and any other person or persons in connection with the proposal.
Whether the notice relates to a nomination to the board of directors or to other business to be proposed at the meeting, the notice also must include information about (i) the shareholder (including the name and address as they appear on the register) and of such beneficial owner if any, and any persons acting in concert, (ii) a representation that the shareholder holds shares carrying voting rights entitled to vote at such meeting, will continue to be a holder of such shares carrying voting rights entitled to vote at such meeting through the date of such meeting, and intends to appear in person or by proxy at the meeting to make such nomination or propose such business, (iii) the shareholder’s holdings of Keenova shares (as well as “Derivative Instruments” or “Short Interests” with respect to Keenova shares, as defined in the Memorandum and Articles of Association), (iv) any arrangements giving the shareholder the right to vote shares of Keenova, (v) any rights to dividends on the Keenova shares that are separated or separable from the underlying Keenova shares, (vi) any proportionate interest in Keenova’s shares or “Derivative Instruments,” held by a general or limited partnership in which the shareholder has an interest, (vii) any performance-related fees (other than an asset-based fee) that the shareholder is entitled to base on any increase or decrease in the value of the Keenova shares, “Derivative Instruments,” or “Short Interests” (viii) any significant equity interests or any “Derivative Instruments” or “Short Interests” in any of Keenova’s principal competitors held by the shareholder, (ix) any interest of the shareholder in any contract with, or any litigation involving, Keenova or any of its affiliates or principal competitors, (x) where the notice involves a nomination or other soliciting activity, the identity of each participant in the solicitation and a representation of the member’s intent to solicit proxies in support thereof, including an undertaking to deliver a proxy statement and form of proxy to holders representing at least 67% of the voting power, (xi) certification that the shareholder, any beneficial owner and their affiliates and associates have complied with all applicable legal requirements in connection with their acquisition and holding of Keenova securities and their conduct as shareholders, (xii) the names and addresses of other shareholders known to the notifying member to be financially or otherwise materially supporting the nomination or proposal and, to
8
the extent known, details of their holdings and relevant Derivative Instruments or Short Positions, (xiii) any information that would be required to be set forth in a Schedule 13-D filed pursuant to the Exchange Act, and (xiv) any other information that would be required to be disclosed by SEC rules regarding solicitation of proxies for the director nomination and/or other business to be proposed at the meeting.
The chairman of the meeting shall have the power and duty to determine whether a nomination or any business proposed to be brought before the meeting was made or proposed in accordance with these procedures (as set out in Keenova’s Memorandum and Articles of Association), and if any proposed business is not in compliance with these provisions, to declare that no action shall be taken in respect of such defective proposal and that it shall be disregarded.
In addition, the Companies Act provides that shareholders holding not less than 10% of the total voting rights may call an extraordinary general meeting for the purpose of considering director nominations or other proposals, as described above under “—Extraordinary General Meetings of Shareholders.”
Inspection of Books and Records
Under Irish law, shareholders have the right to: (i) receive a copy of the Memorandum and Articles of Association of Keenova and any act of the Irish legislature which alters the Memorandum and Articles of Association of Keenova, (ii) inspect and obtain copies of the minutes and resolutions of general meetings of Keenova, (iii) inspect and receive a copy of the register of shareholders, register of directors and secretaries, register of directors’ interests and other statutory registers maintained by Keenova, (iv) receive copies of statutory financial statements and directors’ and auditors’ reports which have previously been sent to shareholders prior to an annual general meeting, and (v) receive any statutory financial statement of a subsidiary company of Keenova which have previously been sent to shareholders prior to an annual general meeting for the preceding 10 years. The auditors of Keenova also have the right to inspect all books, records and vouchers of Keenova. The auditors’ report must be circulated to the shareholders 21 days before the annual general meeting with Keenova’s financial statements prepared in accordance with the Companies Act and must be available to the shareholders at Keenova’s annual general meeting.
Acquisitions
There are a number of mechanisms for acquiring an Irish public limited company, including:
(i)a court-approved scheme of arrangement under the Companies Act. A scheme of arrangement with shareholders requires a court order from the High Court of Ireland and the approval of a majority in number representing 75% in value of the shareholders present and voting in person or by proxy at a meeting called to approve the scheme;
(ii)through a tender offer or takeover offer by a third party for all of the shares of Keenova. Where the holders of 80% or more of Keenova’s shares have accepted an offer by a bidder for their shares in Keenova, the remaining shareholders may be statutorily required to also transfer their shares to such bidder. If the bidder does not exercise its “squeeze out” right, then the non-accepting shareholders also have a statutory right to require the bidder to acquire their shares on the same terms. If shares of Keenova were listed on the main market of the Irish Stock Exchange or another regulated stock exchange in the European Economic Area (the European Economic Area includes all member states of the E.U. and Norway, Iceland and Liechtenstein), this threshold would be increased to 90%; and
(iii)it is also possible for Keenova to be acquired by way of a merger with an E.U.-incorporated public company under the E.U. Cross Border Merger Directive 2017/1132 (as amended). Such a merger must be approved by a special resolution. If Keenova is being merged with another E.U. public company under the E.U. Cross Border Merger Directive 2017/1132 (as amended) and the consideration payable to Keenova’s shareholders is not all in the form of cash, Keenova’s shareholders may be entitled to require their shares to be acquired at fair value.
Irish law does not generally require shareholder approval for a sale, lease or exchange of all or substantially all of a company’s property and assets. However, Keenova’s Memorandum and Articles of Association provide that certain transactions require shareholder approval, as described below under “Anti-Takeover Provisions – Business Combinations with Interested Shareholders”.
Appraisal Rights
9
Generally, under Irish law, shareholders of an Irish company do not have dissenters’ or appraisal rights. Under the European Communities (Cross-Border Mergers) Regulations 2008 (as amended) governing the merger of an Irish company limited by shares such as Keenova and a company incorporated in the European Economic Area, a shareholder (i) who voted against the special resolution approving the transaction or (ii) of a company in which 90% of the shares are held by the other party to the transaction has the right to request that the company acquire its shares for cash at a price determined in accordance with the share exchange ratio set out in the transaction.
In an acquisition effected by a scheme of arrangement under Irish law, 100% of the ordinary shares of a company may be acquired following a shareholder resolution approved by at least a majority in number of the registered shareholders representing 75% of votes cast and approved by the Irish High Court.
In the event of a takeover of Keenova by a third party in accordance with the Companies Act where the holders of 80% or more in value of a class of Keenova’s shares (excluding any shares already beneficially owned by the bidder) have accepted an offer for their shares, the remaining shareholders in that class may be statutorily required to transfer their shares, unless, within one month, the non-tendering shareholders can obtain an Irish court order otherwise providing. If the bidder does not exercise this “squeeze out” right, the non-accepting shareholders also have a statutory right to require the bidder to acquire their shares on the same terms as the original offer, or such other terms as the bidder and the non-tendering shareholders may agree or on such terms as an Irish court, on application of the bidder or non-tendering shareholder, may order.
Disclosure of Interests in Shares
Under the Companies Act, there is a notification requirement for shareholders who acquire or cease to be interested in 3% of the shares of an Irish public company. A shareholder of Keenova must notify Keenova (but not the public at large) if as a result of a transaction the shareholder will be interested in 3% or more of any class of shares of Keenova carrying voting rights; or if as a result of a transaction a shareholder who was interested in more than 3% of any class of shares of Keenova carrying voting rights ceases to be so interested. Where a shareholder is interested in more than 3% of any class of shares of Keenova carrying voting rights, any alteration of his or her interest that brings his or her total holding through the nearest whole percentage number, whether an increase or a reduction, must be notified to Keenova (but not the public at large). The relevant percentage figure is calculated by reference to the aggregate par value of the class of shares in which the shareholder is interested as a proportion of the entire par value of the issued shares of that class. Where the percentage level of the shareholder’s interest does not amount to a whole percentage, this figure may be rounded down to the next whole number. All such disclosures must be notified to Keenova within five business days of the transaction or alteration of the shareholder’s interests that gave rise to the requirement to notify. Where a person fails to comply with the notification requirements described above, no right or interest of any kind whatsoever in respect of any shares in Keenova concerned, held by such person, will be enforceable by such person, whether directly or indirectly, by action or legal proceeding. However, such person may apply to the Irish High Court to have the rights attaching to the shares concerned reinstated.
In addition to the above disclosure requirement, Keenova, under the Companies Act, may by notice in writing require a person whom Keenova knows or has reasonable cause to believe to be or, at any time during the three years immediately preceding the date on which such notice is issued, to have been interested in shares comprised in Keenova’s relevant share capital: (i) to indicate whether or not it is the case, and (ii) where such person holds or has during that time held an interest in any class of shares of Keenova carrying voting rights to give such further information as may be required by Keenova, including particulars of such person’s own past or present interests in such class of shares of Keenova. Any information given in response to the notice is required to be given in writing within such reasonable time as may be specified in the notice.
Where such a notice is served by Keenova on a person who is or was interested in shares of Keenova carrying voting rights and that person fails to give Keenova any information required within the reasonable time specified, Keenova may apply to the court for an order directing that the affected shares be subject to certain restrictions.
Under the Companies Act, the restrictions that may be placed on the shares by the court are:
10
(i)any transfer of those shares, or in the case of unissued shares any transfer of the right to be issued with shares and any issue of shares, is void;
(ii)no voting rights are exercisable in respect of those shares;
(iii)no further shares may be issued in right of those shares or in pursuance of any offer made to the holder of those shares; and
(iv)no payment may be made of any sums due from Keenova on those shares, whether in respect of capital or otherwise.
Where the shares in Keenova are subject to these restrictions, the court may order the shares to be sold and may also direct that the shares will cease to be subject to these restrictions.
In addition to the above statutory obligations, Keenova’s Memorandum and Articles of Association also empower the board to issue investigation notices and disclosure notices requiring information about interests in Keenova’s shares (including the identity of ultimate beneficial owners and interests held through a chain of intermediaries); failure to comply may result in the board serving a disenfranchisement notice, designating the relevant shares as “Default Shares” (as defined in the Memorandum and Articles of Association).
Anti-Takeover Provisions
Business Combinations with Interested Shareholders
Keenova’s Memorandum and Articles of Association include a provision which generally prohibits Keenova from engaging in a business combination with an interested shareholder (defined as “Interested Member”) for a period of three years following the date the person became an interested shareholder, unless, in general:
•the Keenova board of directors approved the transaction which resulted in the shareholder becoming an “Interested Member”;
•upon consummation of the transaction which resulted in the shareholder becoming an “Interested Member”, the shareholder owned at least 85% of the voting shares outstanding at the time of commencement of such transaction, excluding for purposes of determining the number of voting shares outstanding (but not the outstanding voting shares owned by the “Interested Member”), voting shares owned by persons who are directors and also officers and by certain employee share plans; or
•at or subsequent to such time the business combination is approved by the Keenova board of directors and authorized by a special resolution of Keenova’s shareholders (excluding the “Interested Member”).
A “Business Combination” is generally defined in the Memorandum and Articles of Association as a merger, scheme of arrangement, asset or share sale or other transaction resulting in a financial benefit to the interested shareholder. An “Interested Member” is generally defined as a person who, together with affiliates and associates, owns or, within three years prior to the date in question, owned 15% or more of the outstanding voting shares of Keenova.
Shareholder Rights Plans and Share Issuances
Irish law does not expressly prohibit companies from issuing share purchase rights or adopting a shareholder rights plan (commonly known as a “poison pill”) as an anti-takeover measure. However, there is no directly relevant case law on the validity of such plans under Irish law.
Keenova’s Memorandum and Articles of Association allow the board to adopt a shareholder rights plan upon such terms and conditions as the board deems expedient and in the best interests of Keenova, subject to applicable law.
The board also has power to cause Keenova to issue any of its authorized and unissued shares on such terms and conditions as the board may determine (as described under “—Share Capital”) and any such action must be taken in the best interests of Keenova. It is possible, however, that the terms and conditions of any issue of preferred shares could discourage a takeover or other transaction that holders of some or a majority of the ordinary shares believe to be in their best interests or in which holders might receive a premium for their shares over the then market price of the shares.
11
Irish Takeover Rules
The Irish Takeover Panel (the “Panel”) is a statutory body responsible for monitoring and supervising public company takeovers and over relevant transactions in Ireland and was established by the Irish Takeover Panel Act, 1997 (the “Act”). For purposes of the Act, a relevant company includes public limited companies or other bodies corporate incorporated in Ireland whose securities are currently being traded, or (if the subject of a takeover or other relevant proposal) were traded within the previous five years, on the Irish Stock Exchange, the London Stock Exchange, the New York Stock Exchange and/or Nasdaq. In October 2024, based solely on a submission to the Panel on behalf of Keenova, the Panel granted a waiver from the general application of the Irish Takeover Rules (the “Rules”) until 12 October 2025, being the end of the five-year period that the Rules would have remained applicable to Mallinckrodt following its delisting on 12 October 2020.
Corporate Governance
The Memorandum and Articles of Association of Keenova delegate the day-to-day management of Keenova to its board of directors. The board of directors may then delegate management of Keenova to committees, executives or to a management team, but regardless, the directors remain responsible, as a matter of Irish law, for the proper management of the affairs of Keenova. Committees may meet and adjourn as they determine proper. Unless otherwise determined by the board of directors, the quorum necessary for the transaction of business at any committee meeting shall be a majority of the members of such committee then in office unless the committee shall consist of one or two members, in which case one member shall constitute a quorum.
Election of Directors
The Companies Act provides for a minimum of two directors. Keenova’s Memorandum and Articles of Association provide for a minimum of two directors and a maximum of 20 directors. The shareholders of Keenova may from time to time increase or reduce the maximum number, or increase the minimum number, of directors by a special resolution amending the Memorandum and Articles of Association.
At each annual general meeting of Keenova, all the directors shall retire from office and be eligible for re-election. Upon the resignation or termination of office of any director, if a new director shall be appointed to the board he will be designated to fill the vacancy arising. In the event that an election results in either only one or no directors receiving the required majority vote, either the nominee or each of the two nominees receiving the greatest number of votes in favor of his or her election, in accordance with Keenova’s Memorandum and Articles of Association, hold office until his or her successor shall be elected.
Each nominee shall be appointed by ordinary resolution. Any nominee for director who does not receive a majority of the votes cast is not elected to the board of directors of Keenova and the position that would have been filled by such nominee will become vacant. Incumbent directors who are subject to re-election who do not receive a majority of the votes cast at the annual general meeting of Keenova are not re-elected to the board of directors, and immediately following the annual general meeting, will no longer be members of the board of directors unless re-appointed in accordance with Keenova’s Memorandum and Articles of Association. In the event that the number of persons who are validly nominated for election or re-election as directors at any general meeting exceeds the maximum number of directors, then those nominees in number equal to the available director positions who receive the highest number of votes in favour of their election by the shareholders present in person or represented by proxy at such meeting and entitled to vote on the election of directors shall be appointed.
No person shall be appointed director unless nominated in accordance with the Memorandum and Articles of Association of Keenova. Keenova’s Memorandum and Articles of Association provide that with respect to an annual or extraordinary general meeting of shareholders, nominations of persons for election to the board of directors and the proposal of business to be considered by shareholders may be made only (i) pursuant to Keenova’s notice of meeting, (ii) by the board of directors, (iii) by any shareholders pursuant to the valid exercise of power granted to them under the Companies Act, (iv) a shareholder who is entitled to vote at the meeting and who has complied with the advance notice procedures provided for in Keenova’s Memorandum and Articles of Association or (v) by holders of any class of shares in Keenova then in issue having special rights to nominate or appoint directors in accordance with the terms of issue of such class or series, but only to the extent provided in such terms of issue. In addition, the Companies Act provides that shareholders holding
12
not less than 10% of the total voting rights may call an extraordinary general meeting for the purpose of considering director nominations or other proposals.
Directors shall be appointed as follows:
(i)by shareholders by ordinary resolution at the annual general meeting in each year or at any extraordinary general meeting called for the purpose;
(ii)by the board in accordance with the Memorandum and Articles of Association of Keenova; or
(iii)so long as there is in office a sufficient number of directors to constitute a quorum of the board in accordance with the Memorandum and Articles of Association of Keenova, the directors shall have the power at any time and from time to time to appoint any person to be director, either to fill a vacancy in the board or as an addition to the existing directors but so that the total number of directors shall not any time exceed the maximum number provided for in the Memorandum and Articles of Association. A director so appointed shall hold office only until the next following annual general meeting.
Vacancies on the Board of Directors
Keenova’s Memorandum and Articles of Association provide that the directors have the authority to appoint one or more directors to the Keenova board of directors, subject to the maximum number of directors allowed for in the Memorandum and Articles of Association. A vacancy caused by the removal of a director may be filled at the meeting at which the director is removed by ordinary resolution of Keenova’s shareholders, subject to compliance with the applicable advance notice requirements for the election of directors, see above at “- Requirements for Advance Notification of Director Nominations and Proposals of Shareholders”. If shareholders do not appoint a director to fill such vacancy within 45 days after the occurrence of such vacancy, it may be filled by the board of directors.
Any director appointed by the other directors will hold office until the next annual general meeting of Keenova. During any vacancy on the board, the remaining directors will have full power to act as the board but, if and so long as, their number is reduced below the minimum number, the continuing directors may act for increasing the number of directors to that minimum number or for summoning a general meeting of Keenova but for no other purpose.
Removal of Directors
The Companies Act provides that, notwithstanding anything contained in the memorandum and articles of association of a company or in any agreement between that company and a director, the shareholders may, by an ordinary resolution, remove a director from office before the expiration of his or her term at a meeting held on no less than 28 days’ notice and at which the director is entitled to be heard. Accordingly, the shareholders of Keenova may by an ordinary resolution remove a director from office before the expiration of his or her term (notwithstanding any agreement between Keenova and the director). The power of removal is without prejudice to any claim for damages for breach of contract (e.g., employment contract) which the director may have against Keenova in respect of his or her removal.
Amendment of Governing Documents
Irish companies, including Keenova, may only alter their memorandum and articles of association with the approval of a special resolution of a general meeting of the company.
Duration; Dissolution; Rights upon Liquidation
Keenova’s corporate existence has unlimited duration. Keenova may be dissolved at any time by way of either a shareholders’ voluntary winding up or a creditors’ voluntary winding up. In the case of a shareholders’ voluntary winding up, a special resolution of the shareholders of Keenova is required. Keenova may also be dissolved by way of court order on the application of a creditor, or by the Irish Companies Registration Office as an enforcement measure where Keenova has failed to file certain returns. Keenova may also be dissolved by the Irish Corporate Enforcement Authority in Ireland where the affairs of Keenova have been investigated by an inspector and it appears from the report or any information obtained by the Irish Corporate Enforcement Authority that Keenova should be wound up.
13
The rights of the shareholders to a return of Keenova’s assets on dissolution or winding up, following the settlement of all claims of creditors, may be prescribed in Keenova’s Memorandum and Articles of Association or the terms of any preferred shares issued by the directors of Keenova from time to time. The holders of preferred shares in particular may have the right to priority in a dissolution or winding up of Keenova. If the Memorandum and Articles of Association contain no specific provisions in respect of a dissolution or winding up, then, subject to the priorities of any creditors, the assets will be distributed to shareholders in proportion to the paid-up par value of the shares held. Keenova’s Memorandum and Articles of Association provide that the ordinary shareholders of Keenova are entitled to participate pro rata in a winding up, but their right to do so may be subject to the rights of any preferred shareholder to participate under the terms of any series or class of preferred shares. Keenova’s Memorandum and Articles of Association provide that holders of Keenova’s ordinary A shares are entitled to participate on winding up of Keenova, such entitlement to be limited to the repayment of the amount paid up or credited as paid up on those ordinary A shares, and such amount is payable solely after the ordinary shareholders have first received an amount equal to the paid‑up or credited as paid‑up amount on those ordinary shares, together with US$100,000,000 in cash per ordinary share.
Uncertificated Shares
Holders of ordinary shares of Keenova do not have the right to require Keenova to issue certificates for their shares. Keenova only issues uncertificated ordinary shares.
Stock Exchange Listing
Keenova’s ordinary shares are not currently listed on any Irish Stock Exchange (including Euronext Dublin) or any other exchange. Prior to our emergence from its 2023 Chapter 11 cases and Irish examinership proceedings (together, the “2023 Bankruptcy Proceedings”), our ordinary shares were traded on the NYSE American LLC (“NYSE American”) under the ticker symbol “MNK.” On September 6, 2023, the NYSE filed a Form 25 with the SEC to delist our ordinary shares from the NYSE American. The delisting became effective September 16, 2023. Our ordinary shares began trading on the Pink Open Market (formerly known as the OTC Pink Marketplace) on August 29, 2023 under the symbol “MNKTQ.” On the Effective Date, upon emergence from the 2023 Bankruptcy Proceedings, all of our outstanding ordinary shares were cancelled and we ceased trading on the Pink Open Market. Following our emergence from the 2023 Bankruptcy Proceedings, our ordinary shares are not listed on any Irish Stock Exchange (including Euronext Dublin) or any other exchange.
No Sinking Fund
The Keenova ordinary shares have no sinking fund provisions.
Transfer and Registration of Shares
Keenova’s official share register is maintained by its transfer agent and the transfer agent’s affiliates. Registration in this share register is determinative of membership in Keenova. A shareholder of Keenova who holds shares beneficially is not the holder of record of such shares. Instead, the depository or other nominee is the holder of record of such shares. Accordingly, a transfer of shares from a person who holds such shares beneficially to a person who also holds such shares beneficially through the same depository or other nominee is not registered in Keenova’s official share register, as the depository or other nominee remains the record holder of such shares. Under Irish law, rights attaching to Keenova’s shares, including those outlined in this Exhibit 4.1 are generally only exercisable by the legal owner of the relevant shares on Keenova’s official Irish share register. A shareholder holding through a depository may only exercise such rights by either procuring the transfer of the shares from the depository into their direct legal ownership or by procuring the exercise by the depository nominee of those rights on their behalf in accordance with the applicable terms, procedures and rules of the depository.
A written instrument of transfer is required under Irish law in order to register on Keenova’s official share register any transfer of shares (i) from a person who holds such shares directly to any other person, (ii) from a person who holds such shares beneficially to a person who holds such shares directly, or (iii) from a person who holds such shares beneficially to another person who holds such shares beneficially where the transfer involves a change in the depository or other nominee that is the record owner of the transferred shares. An instrument of transfer also is required for a shareholder who directly holds shares to transfer those shares into his or her own
14
broker account (or vice versa). Such instruments of transfer may give rise to Irish stamp duty which must be paid prior to registration of the transfer on Keenova’s official Irish share register. However, a shareholder who directly holds shares may transfer those shares into his or her own broker account (or vice versa) without giving rise to Irish stamp duty, provided there is no change in the ultimate beneficial ownership of the shares as a result of the transfer and the transfer is not made in contemplation of a sale of the shares by a beneficial owner to a third party.
Keenova may, in its absolute discretion, pay (or cause one of its affiliates to pay) any stamp duty. Keenova’s Memorandum and Articles of Association provide that, in the event of any such payment, Keenova (i) may seek reimbursement from the buyer, (ii) will have a lien against the Keenova ordinary shares acquired by such buyer and any dividends paid on such shares and (iii) may set-off the amount of the stamp duty against future dividends on such shares.
Keenova’s Memorandum and Articles of Association delegate to Keenova’s secretary and certain other persons and delegates the authority to execute an instrument of transfer on behalf of a transferring party.
The directors of Keenova, in their absolute discretion, may decline to recognize any instrument of transfer unless (i) it is accompanied by such evidence as the directors may reasonably require to show the right of the transferor to make the transfer, (ii) it is in respect of one class of share only, (iii) the instrument of transfer is properly stamped (if required to be stamped), (iv) a fee of EUR10 or such lesser amount as the directors may from time to time require, is paid to Keenova in respect of it, (v) it is in favor of not more than four transferees, (vi) it is lodged at the registered office of Keenova or at such other place as the directors may appoint, (vii) the board of directors is satisfied, acting reasonably, that all applicable consents, authorisations, permission or approvals of any governmental body or agency in Ireland and any other applicable jurisdiction required under law for the transfer are obtained, and (viii) the board of directors is satisfied, acting reasonably, the tranfer would not violate the terms of any agreement that Keenova (or any of its subsidiaries) and the transferor are a party or subject to. In the case of a transfer of shares by means other than a sale through a stock exchange on which the shares are listed, the directors have absolute discretion and without assigning any reason therefor to decline to register such transfer of a share that is not fully paid or that is transferred to or by a minor or person of unsound mind.
The transferor shall be deemed to remain the legal holder of the share until the name of the transferee is entered on the register in respect thereof.
The registration of transfers may be suspended by the directors at such times and for such period, not exceeding 30 days in each year, as the directors may from time to time determine.
Transfer Agent and Registrar
The transfer agent and registrar for Keenova ordinary shares is Computershare Trust Company, N.A.
15
| Exhibit 4.7 | ||
Execution Version | ||
FIFTH SUPPLEMENTAL INDENTURE
Supplemental Indenture (this “Supplemental Indenture”), dated as of December 17, 2025, among Keenova International Holdings LLC, a Delaware limited liability company (“Keenova International”), and Sonorant Therapeutics Limited, a private limited company under the laws of Ireland (company registration number 648696) (“Sonorant Therapeutics”) (Keenova International and Sonorant Therapeutics, each, a “Guaranteeing Subsidiary” and together, the “Guaranteeing Subsidiaries”), which Guaranteeing Subsidiaries are subsidiaries of Endo LP (or its permitted successor), Endo Finance Holdings LP (f/k/a Endo Finance Holdings, Inc.), a Delaware limited partnership (the “Issuer”), KT Finance Inc., a Delaware corporation (the “Co-Issuer”), Endo, LP, a Delaware limited partnership (f/k/a Endo, Inc.) (the “Parent”), the Subsidiary Guarantors (as defined in the Indenture referred to herein) and Computershare Trust Company, National Association, as trustee (in such capacity, the “Trustee”) and as notes collateral agent (in such capacity, the “Notes Collateral Agent”) under the Indenture referred to below.
W I T N E S S E T H
WHEREAS, the Issuer and the Guarantors have heretofore executed and delivered to the Trustee an indenture, dated as of April 23, 2024, by and among the parties thereto (as amended, supplemented or otherwise modified from time to time, the “Indenture”), providing for the issuance of 8.500% Senior Secured Notes due 2031 (the “Notes”);
WHEREAS, the Indenture provides that under certain circumstances the Guaranteeing Subsidiaries shall execute and deliver to the Trustee and the Notes Collateral Agent a supplemental indenture pursuant to which the Guaranteeing Subsidiaries shall unconditionally guarantee all of the Issuer’s Obligations under the Notes and the Indenture on the terms and conditions set forth herein and in the Indenture (the “Note Guarantee”); and
WHEREAS, pursuant to Section 9.01 of the Indenture, the Trustee and the Notes Collateral Agent are authorized to execute and deliver this Supplemental Indenture.
NOW, THEREFORE, in consideration of the foregoing and for other good and valuable consideration, the receipt of which is hereby acknowledged, the Guaranteeing Subsidiaries and the Trustee mutually covenant and agree for the equal and ratable benefit of the Holders as follows:
1. Capitalized Terms. Capitalized terms used herein without definition shall have the meanings assigned to them in the Indenture.
2. Agreement To Guarantee. Each Guaranteeing Subsidiary hereby provides an unconditional Guarantee on the terms and subject to the conditions set forth in the Note Guarantee and in the Indenture including but not limited to Article 10 thereof.
3. No Recourse Against Others. No director, officer, employee, incorporator or stockholder of the Issuer or any Guarantor, as such, will have any liability for any obligations of the Issuer or any Guarantor under the Notes, the Indenture, the Note Guarantees or for any claim based on, in respect of, or by reason of, such obligations or their creation. Each Holder of Notes by accepting a Note waives and releases all such liability. The waiver and release are part of the consideration for issuance of the Notes. The waiver may not be effective to waive liabilities under the federal securities laws and the laws of certain foreign jurisdictions.
4. NEW YORK LAW TO GOVERN; WAIVER OF JURY TRIAL. THIS SUPPLEMENTAL INDENTURE SHALL BE GOVERNED BY, AND CONSTRUED IN ACCORDANCE WITH THE LAWS OF THE STATE OF NEW YORK. THE ISSUER AND EACH OF THE GUARANTORS CONSENT AND IRREVOCABLY SUBMIT TO THE JURISDICTION OF ANY NEW YORK STATE OR U.S. FEDERAL COURT LOCATED IN THE BOROUGH OF MANHATTAN, CITY OF NEW YORK, COUNTY OF NEW YORK, STATE OF NEW YORK IN RELATION TO ANY LEGAL ACTION OR PROCEEDING (I) ARISING OUT OF, RELATING TO OR IN CONNECTION WITH THE INDENTURE, AS SUPPLEMENTED, THE NOTES, THE GUARANTEES AND ANY RELATED DOCUMENTS (OTHER THAN ANY SECURITY DOCUMENTS WHICH SPECIFY A DIFFERENT JURISDICTION) AND/OR (II) ARISING UNDER ANY U.S. FEDERAL OR U.S. STATE SECURITIES LAWS IN RESPECT OF THE NOTES, THE GUARANTEES AND ANY SECURITIES ISSUED PURSUANT TO THE TERMS OF THE INDENTURE, AS SUPPLEMENTED. THE ISSUER AND EACH OF THE GUARANTORS WAIVE ANY OBJECTION TO PROCEEDINGS IN ANY SUCH COURTS, WHETHER ON THE GROUND OF VENUE OR ON THE GROUND THAT THE PROCEEDINGS HAVE BEEN BROUGHT IN AN INCONVENIENT FORUM. EACH GUARANTEEING SUBSIDIARY, TO THE EXTENT ORGANIZED OUTSIDE OF THE UNITED STATES, HEREBY APPOINTS THE ISSUER, AS ITS AGENT FOR SERVICE OF PROCESS IN ANY SUCH SUIT, ACTION OR PROCEEDING AND AGREES THAT SERVICE OF PROCESS UPON SAID AUTHORIZED AGENT SHALL BE DEEMED IN EVERY RESPECT EFFECTIVE SERVICE OF PROCESS UPON IT IN ANY SUCH SUIT, ACTION OR PROCEEDING. EACH GUARANTEEING SUBSIDIARY AGREES TO DELIVER, UPON THE EXECUTION AND DELIVERY OF THIS SUPPLEMENTAL INDENTURE, A WRITTEN ACCEPTANCE BY SUCH AGENT OF ITS APPOINTMENT AS SUCH AGENT. EACH GUARANTEEING SUBSIDIARY, TO THE EXTENT ORGANIZED OUTSIDE OF THE UNITED STATES, FURTHER AGREES TO TAKE ANY AND ALL ACTION, INCLUDING THE FILING OF ANY AND ALL SUCH DOCUMENTS AND INSTRUMENTS, AS MAY BE REASONABLY NECESSARY TO CONTINUE SUCH DESIGNATION AND APPOINTMENT OF THE ISSUER IN FULL FORCE AND EFFECT FOR SO LONG AS THE INDENTURE, AS SUPPLEMENTED, REMAINS IN FORCE. THE ISSUER, EACH OF THE GUARANTORS AND THE TRUSTEE HEREBY IRREVOCABLY WAIVE, TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY AND ALL RIGHT TO TRIAL BY JURY IN ANY LEGAL
PROCEEDING ARISING OUT OF OR RELATING TO THIS SUPPLEMENTAL INDENTURE, THE NOTES OR THE TRANSACTIONS CONTEMPLATED HEREBY.
5. Counterparts. The parties may sign any number of copies of this Supplemental Indenture. Each signed counterpart shall be deemed an original, but all of them together represent the same agreement. The exchange of copies of this Supplemental Indenture and of signature pages by facsimile or PDF transmission shall constitute effective execution and delivery of this Supplemental Indenture as to the parties hereto and may be used in lieu of the original Supplemental Indenture for all purposes. Signatures of the parties hereto transmitted by facsimile or PDF or other electronic signatures shall be deemed to be their original signatures for all purposes.
6. Effect of Headings. The Section headings herein are for convenience only and shall not affect the construction hereof .
7. The Trustee And The Notes Collateral Agent. The Trustee and the Notes Collateral Agent shall not be responsible in any manner whatsoever for or in respect of the validity or sufficiency of this Supplemental Indenture or for or in respect of the recitals contained herein, all of which recitals are made solely by the Guaranteeing Subsidiaries and the Issuer.
[Signature pages follow]
IN WITNESS WHEREOF, the parties hereto have caused this Supplemental Indenture to be duly executed and attested, all as of the date first above written.
| Keenova International Holdings LLC, as Guaranteeing Subsidiary | |||||
| By: | /s/ Matthew Peters | ||||
| Name: Matthew Peters | |||||
| Title: Vice President of Tax and Treasurer | |||||
| Sonorant Therapeutics Limited, as Guaranteeing Subsidiary | |||||
| By: | /s/ Alasdair Fenlon | ||||
| Name: Alasdair Fenlon | |||||
| Title: Director | |||||
[Signature Page to the Endo Notes Joinder – Fifth Supplemental Indenture]
Endo Finance Holdings LP, as Issuer | |||||
| By: | /s/ Matthew Peters | ||||
| Name: Matthew Peters | |||||
| Title: Vice President of Tax and Treasurer | |||||
| Endo LP, as Parent | |||||
| By: | /s/ Matthew Peters | ||||
| Name: Matthew Peters | |||||
| Title: Vice President of Tax and Treasurer | |||||
KT Finance Inc, as Co-Issuer | |||||
| By: | /s/ Matthew Peters | ||||
| Name: Matthew Peters | |||||
| Title: Vice President of Tax and Treasurer | |||||
[Signature Page to the Endo Notes Joinder – Fifth Supplemental Indenture]
Computershare Trust Company, National Association, as Trustee and Notes Collateral Agent | |||||
| By: | /s/ Katherine M. O’Brien Mathis | ||||
| Name: Katherine M. O’Brien Mathis | |||||
| Title: Vice President | |||||
[Signature Page to the Endo Notes Joinder – Fifth Supplemental Indenture]

Exhibit 10.47 Execution Version THIRD AMENDMENT THIRD AMENDMENT, dated as of November 3, 2025 (this “Amendment”), to the Credit Agreement, dated as of April 23, 2024 (as amended by that certain First Amendment, dated as of October 29, 2024, and that certain Second Amendment (Technical Amendment), dated as of July 17, 2025 and as further amended, restated, amended and restated, supplemented or otherwise modified from time to time prior to the date hereof, the “Credit Agreement”, and as amended by this Amendment, the “Amended Credit Agreement”), among Endo LP (f/k/a Endo, Inc.), as parent (“Parent”), Endo Finance Holdings LP (f/k/a Endo Finance Holdings, Inc.), as the borrower (the “Borrower”), Goldman Sachs Bank USA, as Administrative Agent, Collateral Agent, Issuing Bank and Swingline Lender, and each other lender from time to time party thereto (collectively, the “Lenders”). Capitalized terms not otherwise defined in this Amendment have the same meanings as specified in the Credit Agreement. W I T N E S S E T H: WHEREAS, the Borrower, the Lenders party hereto (constituting the Required Lenders under the Credit Agreement) and the Administrative Agent desire to amend the Credit Agreement, in accordance with Section 9.02(b) thereof, on the terms set forth in Annex A hereto, as further set forth below; and WHEREAS, the Borrower, the Lenders party hereto (constituting the Required Lenders under the Credit Agreement) and the Administrative Agent are willing to agree to this Amendment on the terms set forth herein. NOW, THEREFORE, in consideration of these premises and for other good and valuable consideration, the sufficiency and receipt of all of which is hereby acknowledged, the parties hereto hereby agree as follows: SECTION 1. Amendments. Effective as of the Third Amendment Effective Date, the Credit Agreement is hereby amended to delete the stricken text (indicated textually in the same manner as the following example: stricken text) and to add the double-underlined text (indicated textually in the same manner as the following example: double-underlined text) as set forth on Annex A hereto. SECTION 2. Effectiveness. This Amendment and the transactions contemplated hereby shall become effective as of the date (the “Third Amendment Effective Date”) on which the following conditions have been satisfied (or waived): (i) the Administrative Agent (or its counsel) shall have received a duly executed and completed counterpart hereof that bears the signature of (A) the Borrower, (B) each Lender party hereto (constituting the Required Lenders under the Credit Agreement) and (C) the Administrative Agent; (ii) the Administrative Agent shall have received a certificate dated the Third Amendment Effective Date executed by a Responsible Officer of the Borrower certifying to the representations and warranties set forth in Section 3(b) and (c) of this Amendment; (iii) the Administrative Agent shall have received all fees due and payable thereto on or prior to the Third Amendment Effective Date and, to the extent invoiced at least two (2) Business Days prior to the Third Amendment Effective Date, reimbursement of all reasonable and out-of-pocket expenses (including reasonable fees, charges and

2 disbursements of Simpson Thacher & Bartlett LLP) in connection with this Amendment required to be reimbursed and paid by the Loan Parties under the Credit Agreement; (iv) the Borrower shall have paid, or caused to be paid, to the Administrative Agent, for the account of each Lender party hereto, a consent fee equal to 0.10% of the aggregate principal amount of Loans and Commitments of such Lender outstanding on the Third Amendment Effective Date; and (v) the Administrative Agent shall have received all documentation and other information required by regulatory authorities under applicable “know your customer” and anti-money laundering rules and regulations, including without limitation, the USA PATRIOT Act, requested not less than ten (10) Business Days prior to the date hereof. SECTION 3. Representations and Warranties. The Borrower represents and warrants to each of the Lenders and the Administrative Agent that on and as of the Third Amendment Effective Date: (a) this Amendment has been duly authorized, executed and delivered by it, this Amendment constitutes the valid and binding obligation of the Borrower and the Amended Credit Agreement constitutes the valid and binding obligation of each of the Loan Parties, in each case enforceable against them in accordance with their respective terms, subject to applicable bankruptcy, insolvency, reorganization, moratorium or other laws affecting creditors’ rights generally and subject to general principles of equity, regardless of whether considered in a proceeding in equity or at law; (b) the representations and warranties of Parent and the Borrower set forth in the Credit Agreement are true and correct in all material respects (other than to the extent qualified by materiality or “Material Adverse Effect”, in which case, such representations and warranties are true and correct), except in the case of any such representation and warranty that expressly relates to an earlier date, in which case such representation and warranty was true and correct in all material respects (other than to the extent qualified by materiality or “Material Adverse Effect”, in which case such representation and warranty was true and correct) on and as of such earlier date; and (c) no Default or Event of Default has occurred and is continuing. SECTION 4. Effect of Amendment. (a) Except as expressly set forth herein, this Amendment shall not by implication or otherwise limit, impair, constitute a waiver of or otherwise affect the rights and remedies of the Lenders or the Administrative Agent under the Credit Agreement or any other Loan Document, and shall not alter, modify, amend or in any way affect any of the terms, conditions, obligations, covenants or agreements contained in the Credit Agreement or any other provision of the Credit Agreement or of any other Loan Document, all of which are ratified and affirmed in all respects and shall continue in full force and effect. Nothing herein shall be deemed to entitle Parent, the Borrower or any other Loan Party to a consent to, or a waiver, amendment, modification or other change of, any of the terms, conditions, obligations, covenants or agreements contained in the Credit Agreement, the Amended Credit Agreement or any other Loan Document in similar or different circumstances. The Borrower agrees that, notwithstanding the execution and delivery of this Amendment, the Liens previously granted to the Collateral Agent pursuant to the Loan Documents shall be and remain in full force and effect and that any rights and remedies of the Collateral Agent and the Lenders thereunder and obligations of the Loan Parties thereunder shall be and remain in full force and effect, shall not be affected, impaired or discharged thereby and shall secure all of the Loan Parties’ indebtedness, obligations and liabilities owed to the Secured Parties under the Amended Credit

3 Agreement and the other Loan Documents. Nothing herein can or may be construed as a novation of the Amended Credit Agreement or any other Loan Document. (b) On and after the Third Amendment Effective Date, each reference in the Amended Credit Agreement to “this Agreement”, “hereunder”, “hereof”, “herein”, or words of like import, and each reference to the Credit Agreement in any other Loan Document shall be deemed a reference to the Amended Credit Agreement. This Amendment shall constitute a “Loan Document” for all purposes of the Credit Agreement and the other Loan Documents. SECTION 5. General. (a) GOVERNING LAW; WAIVER OF JURY TRIAL, ETC. THIS AMENDMENT AND THE RIGHTS AND OBLIGATIONS OF THE PARTIES UNDER THIS AMENDMENT SHALL BE CONSTRUED IN ACCORDANCE WITH AND GOVERNED BY THE LAWS OF THE STATE OF NEW YORK. EACH PARTY HERETO HEREBY WAIVES, TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY RIGHT IT MAY HAVE TO A TRIAL BY JURY IN ANY LEGAL PROCEEDING DIRECTLY OR INDIRECTLY ARISING OUT OF OR RELATING TO THIS AMENDMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY (WHETHER BASED ON CONTRACT, TORT OR ANY OTHER THEORY). EACH PARTY HERETO (A) CERTIFIES THAT NO REPRESENTATIVE, AGENT OR ATTORNEY OF ANY OTHER PARTY HAS REPRESENTED, EXPRESSLY OR OTHERWISE, THAT SUCH OTHER PARTY WOULD NOT, IN THE EVENT OF LITIGATION, SEEK TO ENFORCE THE FOREGOING WAIVER AND (B) ACKNOWLEDGES THAT IT AND THE OTHER PARTIES HERETO HAVE BEEN INDUCED TO ENTER INTO THIS AMENDMENT BY, AMONG OTHER THINGS, THE MUTUAL WAIVERS AND CERTIFICATIONS IN THIS SECTION 5(a). Sections 9.09(b), (c) and (d) of the Amended Credit Agreement are hereby incorporated by reference, mutatis mutandis. (b) Counterparts. This Amendment may be executed by one or more of the parties to this Amendment on any number of separate counterparts, and all of said counterparts taken together shall be deemed to constitute one and the same instrument. Delivery of an executed signature page of this Amendment by email or facsimile transmission (or other electronic transmission) shall be effective as delivery of a manually executed counterpart hereof. Delivery of an executed counterpart of a signature page of this Amendment by facsimile or by email as a “.pdf” or “.tif” attachment shall be effective as delivery of a manually executed counterpart of this Amendment. The words “execution,” “signed,” “signature,” “delivery,” and words of like import in or relating to this Amendment shall be deemed to include Electronic Signatures, deliveries or the keeping of records in any electronic form (including deliveries by telecopy, emailed pdf or any other electronic means that reproduces an image of an actual executed signature page), each of which shall be of the same legal effect, validity or enforceability as a manually executed signature, physical delivery thereof or the use of a paper-based recordkeeping system, as the case may be. Nevertheless, upon the request of the Administrative Agent, any Electronic Signature shall be promptly followed by a manually executed counterpart. (c) Headings. The headings of this Amendment are used for convenience of reference only, are not part of this Amendment and shall not affect the construction of, or be taken into consideration in interpreting, this Amendment. [remainder of page intentionally left blank]

[Signature Page to Third Amendment] IN WITNESS WHEREOF, the parties hereto have caused this Amendment to be duly executed and delivered by their respective duly authorized officers as of the date first above written. ENDO FINANCE HOLDINGS LP, as Borrower By EFHI GP Limited, its General Partner By: /s/ Matthew Peters Name: Matthew T. Peters Title: Vice President of Tax and Treasurer

[Signature Page to Third Amendment] GOLDMAN SACHS BANK USA, as Administrative Agent and Lender By: /s/ Luke Qiu Name: Luke Qiu Title: Authorized Signatory

Execution Version Annex A to Third Amendment Second Amendment (Technical Amendment) CREDIT AGREEMENT dated as of April 23, 2024, as amended by the First Amendment, dated as of October 29, 2024, as amended by the Second Amendment (Technical Amendment), dated as of July 17, 2025, as amended by the Third Amendment, dated as of November 3, 2025, among ENDO LP (f/k/a ENDO, INC.), as Parent, ENDO FINANCE HOLDINGS LP (f/k/a ENDO FINANCE HOLDINGS, INC.), as the Borrower Representative, The Lenders Party Hereto, GOLDMAN SACHS BANK USA, as Administrative Agent, Collateral Agent, Issuing Bank and Swingline Lender, GOLDMAN SACHS BANK USA, JPMORGAN CHASE BANK, N.A., BARCLAYS BANK PLC, DEUTSCHE BANK SECURITIES INC., MORGAN STANLEY SENIOR FUNDING, INC., BANCO SANTANDER, S.A., NEW YORK BRANCH, TCBI SECURITIES, INC. (D/B/A TEXAS CAPITAL SECURITIES) and BANK OF AMERICA, N.A., as Joint Lead Arrangers and Joint Bookrunners

i Table of Contents Page Article I Definitions ........................................................................................................................ 1 Section 1.01 Defined Terms ................................................................................................... 1 Section 1.02 Classification of Loans and Borrowings ......................................................... 78 Section 1.03 Terms Generally .............................................................................................. 78 Section 1.04 Accounting Terms; GAAP; Pro Forma Calculations ...................................... 80 Section 1.05 Status of Obligations and Secured Obligations ........................................... 8384 Section 1.06 Special Luxembourg Provisions ...................................................................... 84 Section 1.07 Cashless Rollovers ........................................................................................... 85 Section 1.08 Divisions .......................................................................................................... 85 Article II The Credits .................................................................................................................... 85 Section 2.01 Commitments and Loans ................................................................................. 85 Section 2.02 Loans and Borrowings ..................................................................................... 86 Section 2.03 Requests for Borrowings ................................................................................. 87 Section 2.04 Determination of Dollar Amounts ................................................................... 88 Section 2.05 Swingline Loans .............................................................................................. 89 Section 2.06 Letters of Credit ............................................................................................... 90 Section 2.07 Funding of Borrowings .................................................................................... 97 Section 2.08 Interest Elections ............................................................................................. 97 Section 2.09 Termination and Reduction of Commitments ................................................. 99 Section 2.10 Repayment and Amortization of Loans; Evidence of Debt ........................... 100 Section 2.11 Prepayment of Loans ............................................................................... 101102 Section 2.12 Fees ................................................................................................................ 105 Section 2.13 Interest ........................................................................................................... 107 Section 2.14 Alternate Rate of Interest ............................................................................... 108 Section 2.15 Increased Costs .............................................................................................. 111 Section 2.16 Break Funding Payments ............................................................................... 112 Section 2.17 Taxes .............................................................................................................. 113 Section 2.18 Payments Generally; Allocations of Proceeds; Pro Rata Treatment; Sharing of Set-offs ..................................................................................... 117 Section 2.19 Mitigation Obligations; Replacement of Lenders ......................................... 122 Section 2.20 Incremental Credit Extensions....................................................................... 122 Section 2.21 Judgment Currency ........................................................................................ 125 Section 2.22 Defaulting Lenders .................................................................................. 125126 Section 2.23 Extensions of Loans and Commitments ........................................................ 128 Section 2.24 Loan Repurchases .................................................................................... 131132 Section 2.25 Refinancing Amendment ............................................................................... 133 Section 2.26 Illegality ......................................................................................................... 134 Article III Representations and Warranties ................................................................................. 135 Section 3.01 Organization; Powers; Subsidiaries ............................................................... 135 Section 3.02 Authorization; Enforceability ........................................................................ 136

ii Section 3.03 Governmental Approvals; No Conflicts ........................................................ 136 Section 3.04 Financial Condition; No Material Adverse Change ...................................... 136 Section 3.05 Properties ................................................................................................. 136137 Section 3.06 Litigation, Environmental and Labor Matters ............................................... 137 Section 3.07 Compliance with Laws and Agreements ....................................................... 137 Section 3.08 Investment Company Status .......................................................................... 138 Section 3.09 Taxes .............................................................................................................. 138 Section 3.10 Benefit Plans .................................................................................................. 138 Section 3.11 Disclosure ...................................................................................................... 138 Section 3.12 Federal Reserve Regulations ......................................................................... 139 Section 3.13 Security Interest in Collateral ........................................................................ 139 Section 3.14 Solvency ........................................................................................................ 139 Section 3.15 Sanctions; Anti-Corruption............................................................................ 139 Section 3.16 Beneficial Ownership Certificate .................................................................. 140 Section 3.17 Affected Financial Institutions ...................................................................... 140 Section 3.18 Luxembourg Regulatory Matters ................................................................... 140 Article IV Conditions ............................................................................................................ 140141 Section 4.01 Effective Date .......................................................................................... 140141 Section 4.02 Each Credit Event .......................................................................................... 143 Article V Affirmative Covenants ................................................................................................ 143 Section 5.01 Financial Statements and Other Information ........................................... 143144 Section 5.02 Notices of Material Events ............................................................................ 146 Section 5.03 Existence; Conduct of Business .................................................................... 147 Section 5.04 Payment of Obligations ................................................................................. 147 Section 5.05 Maintenance of Properties; Insurance ........................................................... 147 Section 5.06 Books and Records; Inspection Rights .................................................... 147148 Section 5.07 Compliance with Laws and Material Contractual Obligations ..................... 148 Section 5.08 Use of Proceeds ............................................................................................. 148 Section 5.09 Subsidiary Guarantors; Pledges; Additional Collateral; Further Assurances ................................................................................................. 149 Section 5.10 Designation of Subsidiaries ........................................................................... 151 Section 5.11 Ratings ........................................................................................................... 152 Section 5.12 Post-Closing Obligations. .............................................................................. 152 Article VI Negative Covenants ................................................................................................... 152 Section 6.01 Indebtedness ............................................................................................ 152153 Section 6.02 Liens ........................................................................................................ 158159 Section 6.03 Fundamental Changes and Asset Sales ......................................................... 162 Section 6.04 Investments, Loans, Advances, Guarantees and Acquisitions ...................... 165 Section 6.05 Swap Agreements .................................................................................... 168169 Section 6.06 Transactions with Affiliates ..................................................................... 168169 Section 6.07 Restricted Payments ................................................................................ 169170 Section 6.08 Restrictive Agreements ............................................................................ 171172 Section 6.09 Amendments to Subordinated Indebtedness............................................ 172173

iii Section 6.10 Sale and Leaseback Transactions ............................................................ 172173 Section 6.11 Financial Covenant ........................................................................................ 173 Article VII Events of Default ................................................................................................ 173174 Section 7.01 Events of Default ..................................................................................... 173174 Section 7.02 Right to Cure ........................................................................................... 176177 Article VIII The Administrative Agent and the Collateral Agent ........................................ 178179 Article IX Miscellaneous ...................................................................................................... 183184 Section 9.01 Notices ..................................................................................................... 183184 Section 9.02 Waivers; Amendments ............................................................................ 185186 Section 9.03 Expenses; Indemnity; Damage Waiver ................................................... 189190 Section 9.04 Successors and Assigns ........................................................................... 191192 Section 9.05 Survival .................................................................................................... 200201 Section 9.06 Integration; Counterparts; Electronic Signature ...................................... 200201 Section 9.07 Severability .............................................................................................. 200201 Section 9.08 Right of Setoff ......................................................................................... 201202 Section 9.09 Governing Law; Jurisdiction; Consent to Service of Process; Foreign Process Agent ....................................................................................... 201202 Section 9.10 WAIVER OF JURY TRIAL ................................................................ 202203 Section 9.11 Headings .................................................................................................. 202203 Section 9.12 Confidentiality ......................................................................................... 202203 Section 9.13 Release of Liens and Guarantees ............................................................. 203204 Section 9.14 USA Patriot Act ....................................................................................... 204205 Section 9.15 Appointment for Perfection ..................................................................... 204205 Section 9.16 No Fiduciary Relationship ....................................................................... 204205 Section 9.17 Interest Rate Limitation ........................................................................... 205206 Section 9.18 Additional Borrowers .............................................................................. 205206 Section 9.19 Acknowledgement and Consent to Bail-In of Affected Financial Institution ............................................................................................. 206207 Section 9.20 Intercreditor Agreement .......................................................................... 207208 Section 9.21 Certain ERISA Matters. ........................................................................... 207208 Section 9.22 Acknowledgment Regarding Any Supported QFCs ............................... 208209 Article X Parent Guaranty .................................................................................................... 210211 Section 10.01 Guaranty .................................................................................................. 210211 Section 10.02 Obligations Unconditional ....................................................................... 210211 Section 10.03 Reinstatement .......................................................................................... 211212 Section 10.04 Certain Additional Waivers ..................................................................... 211212 Section 10.05 Remedies ................................................................................................. 211212 Section 10.06 Rights of Contribution ............................................................................. 212213 Section 10.07 Guarantee of Payment; Continuing Guarantee ........................................ 212213

iv SCHEDULES: Schedule 1.01A – Agreed Security Principles Schedule 2.01 – Commitments Schedule 2.06 – Existing Letters of Credit Schedule 3.01 – Subsidiaries Schedule 3.06 – Material Litigation Schedule 3.07 – Compliance with Laws Schedule 5.12 – Post-Closing Obligations Schedule 6.01 – Existing Indebtedness Schedule 6.02 – Existing Liens Schedule 6.04 – Existing Investments Schedule 6.08 – Existing Restrictions EXHIBITS: Exhibit A – Additional Borrower Joinder Exhibit B-1 – Form of Assignment and Assumption Exhibit B-2 – Form of Affiliated Lender Assignment and Assumption Exhibit C – Auction Procedures Exhibit D – Form of Letter of Credit Request Exhibit E – Form of Solvency Certificate Exhibit F-1 – Form of U.S. Tax Compliance Certificate Exhibit F-2 – Form of U.S. Tax Compliance Certificate Exhibit F-3 – Form of U.S. Tax Compliance Certificate Exhibit F-4 – Form of U.S. Tax Compliance Certificate

CREDIT AGREEMENT, dated as of April 23, 2024 (this “Agreement”), among Endo LP (f/k/a Endo, Inc.), a Delaware corporationlimited partnership (“Parent”), Endo Finance Holdings LP (f/k/a Endo Finance Holdings, Inc.), a Delaware corporationlimited partnership (the “Borrower Representative”), the Additional Borrowers from time to time party hereto, the LENDERS from time to time party hereto and Goldman Sachs Bank USA, as Administrative Agent, Collateral Agent, Issuing Bank and Swingline Lender. The parties hereto agree as follows: ARTICLE I DEFINITIONS Section 1.01 Defined Terms. As used in this Agreement, the following terms have the meanings specified below: “2024 Refinancing Cashless Settlement Term Lender” has the meaning set forth in the First Amendment. “2024 Refinancing Term Lender” means, as of any date of determination, each Lender that holds 2024 Refinancing Term Loan Commitments or 2024 Refinancing Term Loans. “2024 Refinancing Term Loan Commitments” means, collectively, (i) the 2024 Refinancing Term Loan Exchange Commitment and (ii) the Additional 2024 Refinancing Term Loan Commitment. The aggregate principal amount of the 2024 Refinancing Term Loan Commitments on the First Amendment Effective Date is $1,500,000,000. “2024 Refinancing Term Loan Exchange Commitment” means the agreement of each 2024 Refinancing Cashless Settlement Term Lender to exchange its Existing Initial Term Loans for an equal aggregate principal amount of 2024 Refinancing Term Loans to the Borrower on the First Amendment Effective Date (or such lesser amount as allocated to such 2024 Refinancing Cashless Settlement Term Lender by the Lead Arrangers on or prior to the First Amendment Effective Date). “2024 Refinancing Term Loans” means (i) the term loans made or continued, as the case may be, by the 2024 Refinancing Term Lenders to the Borrower Representative on the First Amendment Effective Date pursuant to Section 1 of the First Amendment and (ii) any Incremental Term Loans (which do not constitute Other Term Loans) made from time to time pursuant to Section 2.20 from and after the First Amendment Effective Date. “ABR”, when used in reference to any Loan or Borrowing, refers to a Loan, or the Loans comprising such Borrowing, bearing interest at a rate determined by reference to the Alternate Base Rate. “Additional 2024 Refinancing Term Lender” means a Person with an Additional 2024 Refinancing Term Loan Commitment on the First Amendment Effective Date. For the avoidance of doubt, an Existing Initial Term Lender immediately prior to the First Amendment Effective Date may also be an Additional 2024 Refinancing Term Lender.

2 “Additional 2024 Refinancing Term Loan” means a Loan that is made pursuant to Section 1(a) of the First Amendment. “Additional 2024 Refinancing Term Loan Commitment” means, with respect to each Additional 2024 Refinancing Term Lender, the commitment of such Additional 2024 Refinancing Term Lender to make Additional 2024 Refinancing Term Loans to the Borrower on the First Amendment Effective Date. The amount of each Lender’s Additional 2024 Refinancing Term Loan Commitment as of the First Amendment Effective Date is set forth under the heading “Additional 2024 Refinancing Term Loan Commitments” on Schedule I to the First Amendment. “Additional Borrower Joinder” means a joinder agreement substantially in the form of Exhibit A. “Additional Borrowers” means, collectively, the Restricted Subsidiaries which are designated as a Borrower by Parent pursuant to Section 9.18(a). “Adjusted Daily Simple CORRA” means an interest rate per annum equal to (a) the Daily Simple CORRA, plus (b) 0.29547%; provided that if the Adjusted Daily Simple CORRA as so determined would be less than the Floor, such rate shall be deemed to be equal to the Floor for the purposes of this Agreement. “Adjusted Term CORRA Rate” means, for the purposes of any calculation, the rate per annum equal to (a) Term CORRA for such calculation plus (b) 0.29547% for a one month interest period or 0.32138% for a three month interest period; provided that if the Adjusted Term CORRA Rate as so determined would be less than the Floor, such rate shall be deemed to be equal to the Floor for the purposes of this Agreement. “Administrative Agent” means Goldman Sachs Bank USA, in its capacity as administrative agent for the Lenders hereunder. “Administrative Agent Fee Letter” shall mean that certain Administrative Agent Fee Letter, dated as of the Effective Date, by and among the Borrower Representative and the Administrative Agent. “Administrative Questionnaire” means an Administrative Questionnaire in a form supplied by the Administrative Agent. “Affected Financial Institution” means (a) any EEA Financial Institution or (b) any UK Financial Institution. “Affiliate” means, with respect to a specified Person, another Person that directly, or indirectly through one or more intermediaries, Controls or is Controlled by or is under common Control with the Person specified. “Affiliated Lender” means, at any time, any Lender that is an Affiliate of Parent (other than (a) Parent or any Subsidiary, (b) any Debt Fund Affiliate or (c) any natural person) at such time.

3 “Affiliated Lender Assignment and Assumption” has the meaning specified in Section 9.04(g)(vi). “Affiliated Lender Cap” has the meaning specified in Section 9.04(g). “Agent Parties” has the meaning assigned to such term in Section 9.01(c). “Agreed Currencies” means (i) Dollars, (ii) euros, (iii) Japanese Yen, (iv) Pounds Sterling, (v) Canadian Dollars and (vi) any other Foreign Currency agreed to by the Administrative Agent and each of the Multicurrency Tranche Lenders. “Agreed Security Principles” means the Agreed Security Principles set forth on Schedule 1.01A. For the avoidance of doubt, the Agreed Security Principles shall only apply to Guarantees proposed to be granted by, assets of, and Equity Interests in, Foreign Subsidiaries. “Agreement” has the meaning assigned to such term in the preamble hereto. “Alternate Base Rate” means, for any day, a rate per annum equal to the greatest of (a) the Prime Rate in effect on such day, (b) the NYFRB Rate in effect on such day plus 1⁄2 of 1% and (c) Term SOFR for a one month Interest Period as published two U.S. Government Securities Business Days prior to such day (or if such day is not a U.S. Government Securities Business Day, the immediately preceding U.S. Government Securities Business Day) plus 1%; provided that for the purpose of this definition, Term SOFR for any day shall be based on the Term SOFR Reference Rate at approximately 5:00 a.m. Chicago time on such day (or any amended publication time for the Term SOFR Reference Rate, as specified by the CME Term SOFR Administrator in the Term SOFR Reference Rate methodology). Any change in the Alternate Base Rate due to a change in the Prime Rate, the NYFRB Rate or Term SOFR shall be effective from and including the effective date of such change in the Prime Rate, the NYFRB Rate, the Federal Funds Effective Rate or Term SOFR, respectively. If the Alternate Base Rate is being used as an alternate rate of interest pursuant to Section 2.14 (for the avoidance of doubt, only until the Benchmark Replacement has been determined pursuant to Section 2.14(b)), then the Alternate Base Rate shall be the greater of clauses (a) and (b) above and shall be determined without reference to clause (c) above. “Ancillary Document” has the meaning assigned to such term in Section 9.06. “Anti-Corruption Laws” means all laws, rules, and regulations of any jurisdiction applicable to the Borrower or any of its Affiliates from time to time concerning or relating to bribery or corruption. “Applicable Excess Cash Flow Percentage” means 50%, with a step down to 25% upon the achievement and maintenance of a First Lien Net Leverage Ratio of less than or equal to 2.50 to 1.00 and a further step down to 0% upon the achievement and maintenance of a First Lien Net Leverage Ratio of less than or equal to 2.00 to 1.00. “Applicable Lender” has the meaning assigned to such term in Section 2.06(d). “Applicable Percentage” means, (a) with respect to any Multicurrency Tranche Lender in respect of a Multicurrency Tranche Credit Event, its Multicurrency Tranche Percentage,
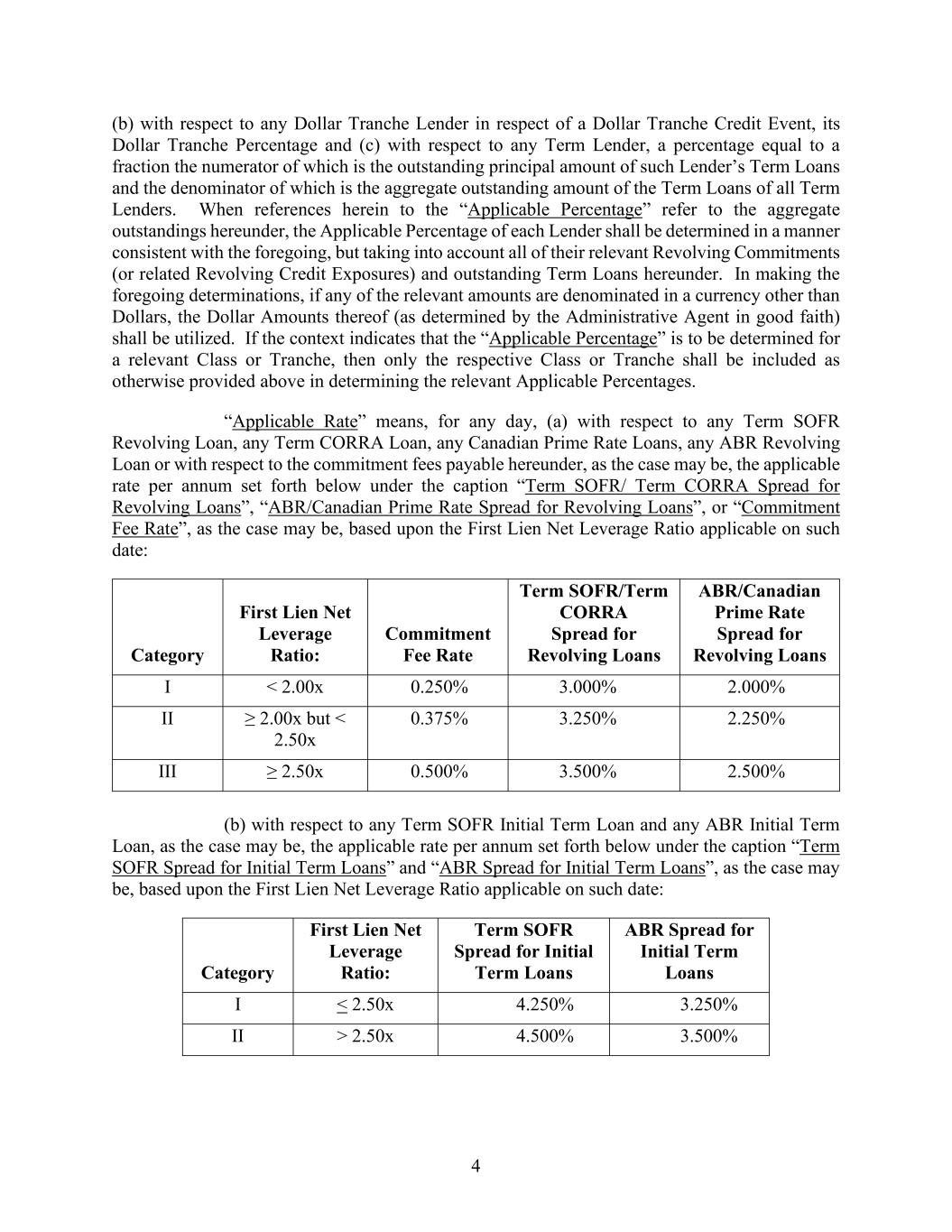
4 (b) with respect to any Dollar Tranche Lender in respect of a Dollar Tranche Credit Event, its Dollar Tranche Percentage and (c) with respect to any Term Lender, a percentage equal to a fraction the numerator of which is the outstanding principal amount of such Lender’s Term Loans and the denominator of which is the aggregate outstanding amount of the Term Loans of all Term Lenders. When references herein to the “Applicable Percentage” refer to the aggregate outstandings hereunder, the Applicable Percentage of each Lender shall be determined in a manner consistent with the foregoing, but taking into account all of their relevant Revolving Commitments (or related Revolving Credit Exposures) and outstanding Term Loans hereunder. In making the foregoing determinations, if any of the relevant amounts are denominated in a currency other than Dollars, the Dollar Amounts thereof (as determined by the Administrative Agent in good faith) shall be utilized. If the context indicates that the “Applicable Percentage” is to be determined for a relevant Class or Tranche, then only the respective Class or Tranche shall be included as otherwise provided above in determining the relevant Applicable Percentages. “Applicable Rate” means, for any day, (a) with respect to any Term SOFR Revolving Loan, any Term CORRA Loan, any Canadian Prime Rate Loans, any ABR Revolving Loan or with respect to the commitment fees payable hereunder, as the case may be, the applicable rate per annum set forth below under the caption “Term SOFR/ Term CORRA Spread for Revolving Loans”, “ABR/Canadian Prime Rate Spread for Revolving Loans”, or “Commitment Fee Rate”, as the case may be, based upon the First Lien Net Leverage Ratio applicable on such date: Category First Lien Net Leverage Ratio: Commitment Fee Rate Term SOFR/Term CORRA Spread for Revolving Loans ABR/Canadian Prime Rate Spread for Revolving Loans I < 2.00x 0.250% 3.000% 2.000% II ≥ 2.00x but < 2.50x 0.375% 3.250% 2.250% III ≥ 2.50x 0.500% 3.500% 2.500% (b) with respect to any Term SOFR Initial Term Loan and any ABR Initial Term Loan, as the case may be, the applicable rate per annum set forth below under the caption “Term SOFR Spread for Initial Term Loans” and “ABR Spread for Initial Term Loans”, as the case may be, based upon the First Lien Net Leverage Ratio applicable on such date: Category First Lien Net Leverage Ratio: Term SOFR Spread for Initial Term Loans ABR Spread for Initial Term Loans I < 2.50x 4.250% 3.250% II > 2.50x 4.500% 3.500%
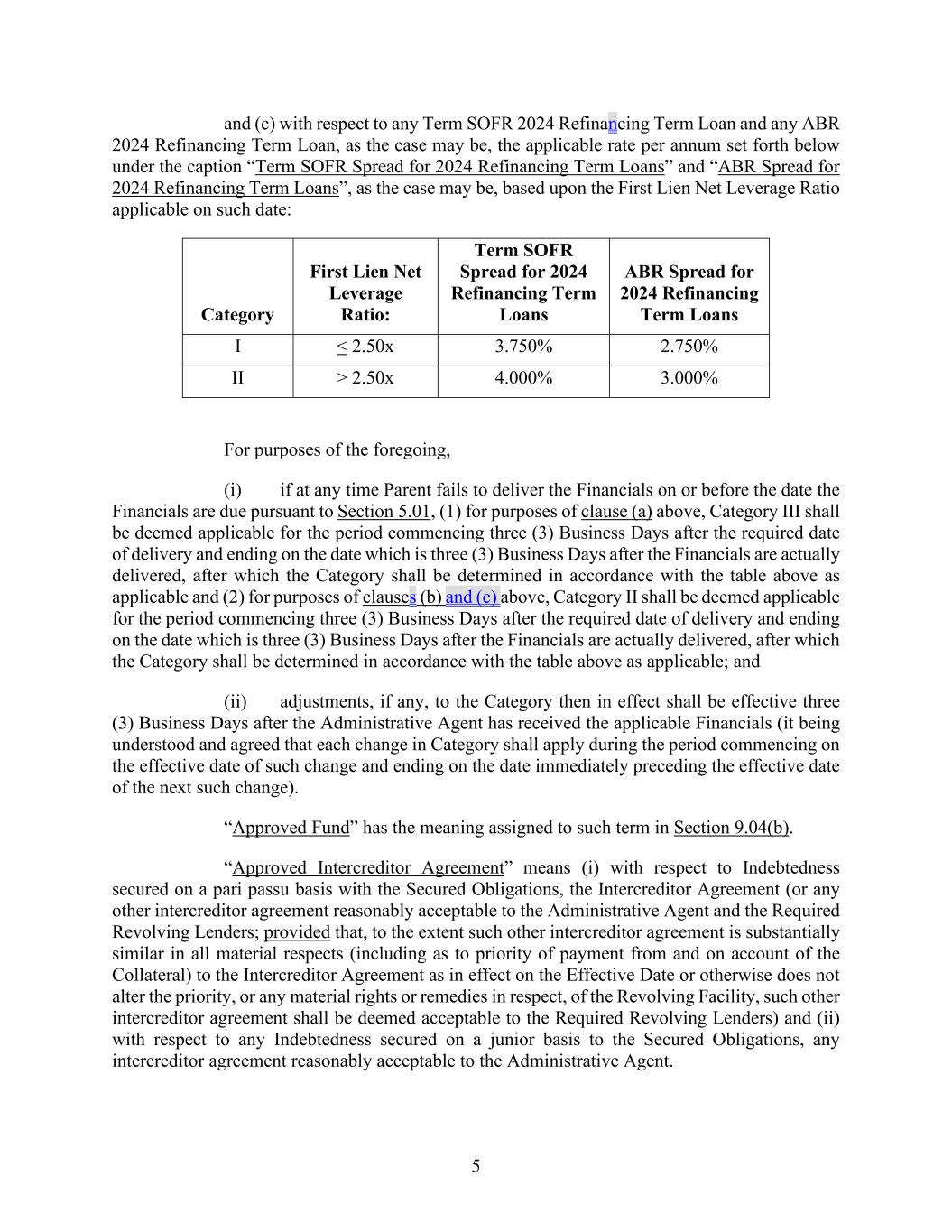
5 and (c) with respect to any Term SOFR 2024 Refinancing Term Loan and any ABR 2024 Refinancing Term Loan, as the case may be, the applicable rate per annum set forth below under the caption “Term SOFR Spread for 2024 Refinancing Term Loans” and “ABR Spread for 2024 Refinancing Term Loans”, as the case may be, based upon the First Lien Net Leverage Ratio applicable on such date: Category First Lien Net Leverage Ratio: Term SOFR Spread for 2024 Refinancing Term Loans ABR Spread for 2024 Refinancing Term Loans I < 2.50x 3.750% 2.750% II > 2.50x 4.000% 3.000% For purposes of the foregoing, (i) if at any time Parent fails to deliver the Financials on or before the date the Financials are due pursuant to Section 5.01, (1) for purposes of clause (a) above, Category III shall be deemed applicable for the period commencing three (3) Business Days after the required date of delivery and ending on the date which is three (3) Business Days after the Financials are actually delivered, after which the Category shall be determined in accordance with the table above as applicable and (2) for purposes of clauses (b) and (c) above, Category II shall be deemed applicable for the period commencing three (3) Business Days after the required date of delivery and ending on the date which is three (3) Business Days after the Financials are actually delivered, after which the Category shall be determined in accordance with the table above as applicable; and (ii) adjustments, if any, to the Category then in effect shall be effective three (3) Business Days after the Administrative Agent has received the applicable Financials (it being understood and agreed that each change in Category shall apply during the period commencing on the effective date of such change and ending on the date immediately preceding the effective date of the next such change). “Approved Fund” has the meaning assigned to such term in Section 9.04(b). “Approved Intercreditor Agreement” means (i) with respect to Indebtedness secured on a pari passu basis with the Secured Obligations, the Intercreditor Agreement (or any other intercreditor agreement reasonably acceptable to the Administrative Agent and the Required Revolving Lenders; provided that, to the extent such other intercreditor agreement is substantially similar in all material respects (including as to priority of payment from and on account of the Collateral) to the Intercreditor Agreement as in effect on the Effective Date or otherwise does not alter the priority, or any material rights or remedies in respect, of the Revolving Facility, such other intercreditor agreement shall be deemed acceptable to the Required Revolving Lenders) and (ii) with respect to any Indebtedness secured on a junior basis to the Secured Obligations, any intercreditor agreement reasonably acceptable to the Administrative Agent.

6 “Asset Sale” means any Disposition of property or series of related Dispositions of property in respect of which either the fair market value of such property or the Disposition Consideration payable to Parent or any of its Restricted Subsidiaries exceeds $5,000,000. “Asset Sale Step Down” has the meaning set forth in Section 2.11(c). “Assignment and Assumption” means an assignment and assumption agreement entered into by a Lender and an assignee (with the consent of any party whose consent is required by Section 9.04), and accepted by the Administrative Agent, in the form of Exhibit B-1 or any other form approved by the Administrative Agent. “Attributable Receivables Indebtedness” means the principal amount of Indebtedness (other than any subordinated Indebtedness owing by a Receivables Entity to a Receivables Seller or a Receivables Seller to another Receivables Seller in connection with the transfer, sale and/or pledge of Permitted Receivables Facility Assets) which (i) if a Permitted Receivables Facility is structured as a secured lending agreement or other similar agreement, constitutes the principal amount of such Indebtedness or (ii) if a Permitted Receivables Facility is structured as a purchase agreement or other similar agreement, would be outstanding at such time under such Permitted Receivables Facility if the same were structured as a secured lending agreement rather than a purchase agreement or such other similar agreement. “Auction Manager” has the meaning assigned to such term in Section 2.24(a). “Auction Procedures” means the auction procedures with respect to Purchase Offers set forth in Exhibit C hereto. “Auto-Extension Letter of Credit” has the meaning assigned to such term in Section 2.06(c). “Availability Period” means the period from and including the Effective Date to but excluding the earlier of the Maturity Date with respect to the Revolving Commitments (or with respect to any Extended Revolving Commitments, the Maturity Date with respect thereto) and the date of termination of all of the Revolving Commitments. “Available Amount” means, at any time, an amount equal to, without duplication: (a) the sum of: (i) the greater of (x) $230,000,000 and (y) 30.0% of Consolidated EBITDA as of the end of the Reference Period; plus (ii) the greater of: (1) 50.0% of the Consolidated Net Income of Parent and the Restricted Subsidiaries for the period (taken as one accounting period) commencing on April 1, 2024 to the end of the most recently ended fiscal quarter for which Financials of Parent have been delivered, or in the case such Consolidated Net Income for such period is a deficit, minus 100.0% of such deficit; and

7 (2) 100.0% of Retained Excess Cash Flow of Parent and the Restricted Subsidiaries for the period (taken as one accounting period) commencing on April 1, 2024 to the end of the most recently ended fiscal quarter for which Financials of Parent have been delivered; plus (iii) 100.0% of the aggregate Net Proceeds and the fair market value (as determined in good faith by Parent) of marketable securities or other property received by Parent and its Restricted Subsidiaries since the Effective Date from any capital contributions to, or the sale or issuance of Equity Interests of Parent (other than (i) Disqualified Equity Interests, (ii) Equity Interests issued or sold to a Restricted Subsidiary or an employee stock ownership plan or similar trust to the extent such sale to an employee stock ownership plan or similar trust is financed by loans from or Guaranteed by Parent or any Restricted Subsidiary unless such loans have been repaid with cash on or prior to the date of determination, (iii) Equity Interests the Net Proceeds of which are used to repay long-term Indebtedness for borrowed money (other than (x) revolving loans or (y) Indebtedness of a Person, or Indebtedness secured by a Lien on the assets, being acquired in connection with acquisitions permitted hereunder for which Parent issues Equity Interests as consideration) and (iv) any exercise of the cure rights set forth in Section 7.02); plus (iv) 100% of the Net Proceeds of Indebtedness and Disqualified Equity Interests of Parent and its Restricted Subsidiaries, in each case, issued after the Effective Date, which have been exchanged or converted into Equity Interests (other than of Disqualified Equity Interests) of Parent, together with any cash and Permitted Investments and the fair market value (as determined in good faith by Parent) of any assets that are received by Parent or any Restricted Subsidiary upon such exchange or conversion; plus (v) 100% of the aggregate Net Proceeds and the fair market value of marketable securities or other property received by Parent and its Restricted Subsidiaries since the Effective Date from Dispositions of Investments made using the Available Amount; plus (vi) 100% of the returns, profits, distributions and similar amounts received in cash or Permitted Investments by Parent and its Restricted Subsidiaries on Investments made using the Available Amount (including Investments in Unrestricted Subsidiaries); plus (vii) 100% of (x) the Investments of Parent and its Restricted Subsidiaries made using the Available Amount in any Unrestricted Subsidiary that has been re- designated as a Restricted Subsidiary or that has been merged or consolidated with or into Parent or any of its Restricted Subsidiaries (up to the fair market value (as determined in good faith by Parent) of the Investments of Parent and its Restricted Subsidiaries in such Unrestricted Subsidiary at the time of such re-designation or merger or consolidation) and (y) the fair market value (as determined in good faith by Parent) of the assets of any Unrestricted Subsidiary acquired by such

8 Unrestricted Subsidiary with the proceeds of Investments of Parent and its Restricted Subsidiaries made using the Available Amount in such Unrestricted Subsidiary that have been transferred, conveyed or otherwise distributed to Parent and its Restricted Subsidiaries (up to the fair market value (as determined in good faith by Parent) of the Investments of Parent and its respective Restricted Subsidiaries in such Unrestricted Subsidiary at the time of such transfer, conveyance or other distribution); plus (viii) 100% of the aggregate amount of any Declined Prepayment Amounts; minus, without duplication, (b) an amount equal to the sum of (i) Restricted Payments made pursuant to Section 6.07(j), plus (ii) Investments made pursuant to Section 6.04(cc), in each case, after the Effective Date and prior to such time or contemporaneously therewith. “Available Revolving Commitment” means, at any time with respect to any Lender, the Revolving Commitments of such Lender then in effect minus the Revolving Credit Exposure of such Lender at such time; it being understood and agreed that any Lender’s Swingline Exposure shall not be deemed to be a component of the Revolving Credit Exposure for purposes of calculating the commitment fee under Section 2.12(a). “Available Tenor” means, as of any date of determination and with respect to the then-current Benchmark, as applicable, any tenor for such Benchmark (or component thereof) or payment period for interest calculated with reference to such Benchmark, as applicable, that is or may be used for determining the length of an Interest Period for any term rate or otherwise or for determining any frequency of making payments of interest calculated pursuant to this Agreement as of such date and not including, for the avoidance of doubt, any tenor for such Benchmark that is then-removed from the definition of “Interest Period” pursuant to clause (e) of Section 2.14. “Bail-In Action” means the exercise of any Write-Down and Conversion Powers by the applicable Resolution Authority in respect of any liability of an Affected Financial Institution. “Bail-In Legislation” means, (a) with respect to any EEA Member Country implementing Article 55 of Directive 2014/59/EU of the European Parliament and of the Council of the European Union, the implementing law, regulation rule or requirement for such EEA Member Country from time to time which is described in the EU Bail-In Legislation Schedule and (b) with respect to the United Kingdom, Part I of the United Kingdom Banking Act 2009 (as amended from time to time) and any other law, regulation or rule applicable in the United Kingdom relating to the resolution of unsound or failing banks, investment firms or other financial institutions or their affiliates (other than through liquidation, administration or other insolvency proceedings). “Banking Services” means each and any of the following bank services provided to Parent or any Subsidiary by any Lender or any of its Affiliates: (a) credit cards for commercial customers (including, without limitation, “commercial credit cards” and purchasing cards), (b) stored value cards and (c) treasury management services (including, without limitation,

9 controlled disbursement, automated clearinghouse transactions, return items, overdrafts and interstate depository network services). “Banking Services Agreement” means any agreement entered into by the Borrower or any Subsidiary in connection with Banking Services. “Banking Services Obligations” means any and all obligations of Parent or any other Loan Party, whether absolute or contingent and howsoever and whensoever created, arising, evidenced or acquired (including all renewals, extensions and modifications thereof and substitutions therefor) in connection with Banking Services. “Bankruptcy Code” means title 11 of the United States Code entitled “Bankruptcy”, as now or hereafter in effect, and any successor thereto. “Bankruptcy Event” means, with respect to any Person, such Person becomes the subject of a bankruptcy or insolvency proceeding, or has had a receiver, conservator, trustee, administrator, custodian, examiner, assignee for the benefit of creditors or similar Person charged with the reorganization or liquidation of its business appointed for it, or, in the good faith determination of the Administrative Agent, has taken any action in furtherance of, or indicating its consent to, approval of, or acquiescence in, any such proceeding or appointment; provided that a Bankruptcy Event shall not result solely by virtue of any ownership interest, or the acquisition of any ownership interest, in such Person by a Governmental Authority or instrumentality thereof; provided, further, that such ownership interest does not result in or provide such Person with immunity from the jurisdiction of courts within the United States of America or from the enforcement of judgments or writs of attachment on its assets or permit such Person (or such Governmental Authority or instrumentality) to reject, repudiate, disavow or disaffirm any contracts or agreements made by such Person. “Benchmark” means, initially, with respect to any Term Benchmark Loan, the applicable Relevant Rate; provided that if a Benchmark Transition Event and its related Benchmark Replacement Date have occurred with respect to such Relevant Rate or the then- current Benchmark, then “Benchmark” means the applicable Benchmark Replacement to the extent that such Benchmark Replacement has replaced such prior benchmark rate pursuant to clause (b) or clause (c) of Section 2.14. “Benchmark Replacement” means, for any Available Tenor, the first alternative set forth in the order below that can be determined by the Administrative Agent for the applicable Benchmark Replacement Date: (1) in the case of any Loan denominated in Dollars, Daily Simple SOFR; (2) in the case of any Loan denominated in Canadian Dollars, Adjusted Daily Simple CORRA; (3) the sum of: (a) the alternate benchmark rate that has been selected by the Administrative Agent and the Parent as the replacement for the then-current Benchmark for the applicable Corresponding Tenor giving due consideration to (i)

10 any selection or recommendation of a replacement benchmark rate or the mechanism for determining such a rate by the relevant Governmental Authority or (ii) any evolving or then-prevailing market convention for determining a benchmark rate as a replacement for the then-current Benchmark for syndicated credit facilities denominated in the applicable Agreed Currency at such time and (b) if applicable, the related Benchmark Replacement Adjustment. If the Benchmark Replacement as determined pursuant to clause (1), (2) or (3) above would be less than the Floor, the Benchmark Replacement will be deemed to be the Floor for the purposes of this Agreement and the other Loan Documents. “Benchmark Replacement Adjustment” means, with respect to any replacement of the then-current Benchmark with an Unadjusted Benchmark Replacement for any applicable Interest Period and Available Tenor for any setting of such Unadjusted Benchmark Replacement, the spread adjustment, or method for calculating or determining such spread adjustment, (which may be a positive or negative value or zero) that has been selected by the Administrative Agent and the Parent for the applicable Corresponding Tenor giving due consideration to (i) any selection or recommendation of a spread adjustment, or method for calculating or determining such spread adjustment, for the replacement of such Benchmark with the applicable Unadjusted Benchmark Replacement by the relevant Governmental Authority on the applicable Benchmark Replacement Date and/or (ii) any evolving or then-prevailing market convention for determining a spread adjustment, or method for calculating or determining such spread adjustment, for the replacement of such Benchmark with the applicable Unadjusted Benchmark Replacement for syndicated credit facilities denominated in the applicable Agreed Currency at such time. “Benchmark Replacement Conforming Changes” means, with respect to any Benchmark Replacement and/or any Term Benchmark Loan, any technical, administrative or operational changes (including changes to the definition of “Alternate Base Rate,” the definition of “Canadian Prime Rate,” the definition of “Benchmark,” the definition of “Business Day,” the definition of “CORRA Business Day,” the definition of “U.S. Government Securities Business Day,” the definition of “Interest Period,” timing and frequency of determining rates and making payments of interest, timing of borrowing requests or prepayment, conversion or continuation notices, length of lookback periods, the applicability of breakage provisions, and other technical, administrative or operational matters) that the Administrative Agent decides in its reasonable discretion (in consultation with Parent) may be appropriate to reflect the adoption and implementation of such Benchmark Replacement and/or Term Benchmark Loan and to permit the administration thereof by the Administrative Agent in a manner substantially consistent with market practice (or, if the Administrative Agent decides that adoption of any portion of such market practice is not administratively feasible or if the Administrative Agent determines that no market practice for the administration of such Benchmark Replacement and/or Term Benchmark Loan exists, in such other manner of administration as the Administrative Agent decides (in consultation with Parent) is reasonably necessary in connection with the administration of this Agreement and the other Loan Documents). “Benchmark Replacement Date” means, with respect to any Benchmark, the earliest to occur of the following events with respect to such then-current Benchmark:

11 (1) in the case of clause (1) or (2) of the definition of “Benchmark Transition Event,” the later of (a) the date of the public statement or publication of information referenced therein and (b) the date on which the administrator of such Benchmark (or the published component used in the calculation thereof) permanently or indefinitely ceases to provide all Available Tenors of such Benchmark (or such component thereof); (2) in the case of clause (3) of the definition of “Benchmark Transition Event,” the date of the public statement or publication of information referenced therein; or (3) in the case of clause (4) of the definition of “Benchmark Transition Event”, the date of determination by the Administrative Agent referenced therein. For the avoidance of doubt, (i) if the event giving rise to the Benchmark Replacement Date occurs on the same day as, but earlier than, the Reference Time in respect of any determination, the Benchmark Replacement Date will be deemed to have occurred prior to the Reference Time for such determination and (ii) the “Benchmark Replacement Date” will be deemed to have occurred in the case of clause (1) or (2) with respect to any Benchmark upon the occurrence of the applicable event or events set forth therein with respect to all then-current Available Tenors of such Benchmark (or the published component used in the calculation thereof). “Benchmark Transition Event” means, with respect to any Benchmark, the occurrence of one or more of the following events with respect to such then-current Benchmark: (4) a public statement or publication of information by or on behalf of the administrator of such Benchmark (or the published component used in the calculation thereof) announcing that such administrator has ceased or will cease to provide all Available Tenors of such Benchmark (or such component thereof), permanently or indefinitely, provided that, at the time of such statement or publication, there is no successor administrator that will continue to provide any Available Tenor of such Benchmark (or such component thereof); (5) a public statement or publication of information by the regulatory supervisor for the administrator of such Benchmark (or the published component used in the calculation thereof), the Federal Reserve Board, the NYFRB, the CME Term SOFR Administrator, the Term CORRA Administrator, the Bank of Canada, an insolvency official with jurisdiction over the administrator for such Benchmark (or such component), a resolution authority with jurisdiction over the administrator for such Benchmark (or such component) or a court or an entity with similar insolvency or resolution authority over the administrator for such Benchmark (or such component), in each case which states that the administrator of such Benchmark (or such component) has ceased or will cease to provide all Available Tenors of such Benchmark (or such component thereof) permanently or indefinitely; provided that, at the time of such statement or publication, there is no successor administrator that will continue to provide any Available Tenor of such Benchmark (or such component thereof);

12 (6) a public statement or publication of information by the regulatory supervisor for the administrator of such Benchmark (or the published component used in the calculation thereof) announcing that all Available Tenors of such Benchmark (or such component thereof) are no longer representative, or, as of a specified future date will no longer be representative; or (7) the Administrative Agent determines, in consultation with the Borrower, that reporting of the Benchmark for all Available Tenors has ceased or will cease. For the avoidance of doubt, a “Benchmark Transition Event” will be deemed to have occurred with respect to any Benchmark if a public statement or publication of information set forth above has occurred with respect to each then-current Available Tenor of such Benchmark (or the published component used in the calculation thereof). “Benchmark Unavailability Period” means, with respect to any Benchmark, the period (if any) (x) beginning at the time that a Benchmark Replacement Date pursuant to clauses (1) or (2) of that definition has occurred if, at such time, no Benchmark Replacement has replaced such then-current Benchmark for all purposes hereunder and under any Loan Document in accordance with Section 2.14 and (y) ending at the time that a Benchmark Replacement has replaced such then-current Benchmark for all purposes hereunder and under any Loan Document in accordance with Section 2.14. “Benefit Plans” means any of (a) an “employee benefit plan” (as defined in ERISA) that is subject to Title I of ERISA, (b) a “plan” as defined in and subject to Section 4975 of the Code or (c) any person whose assets include (for purposes of ERISA Section 3(42) or otherwise for purposes of Title I of ERISA or Section 4975 of the Code) the assets of any such “employee benefit plan” or “plan.” “Beneficial Ownership Certificate” means a certification regarding beneficial ownership required by the Beneficial Ownership Regulation. “Beneficial Ownership Regulation” means 31 C.F.R. § 1010.230. “Big Boy Letter” means a letter from a Lender acknowledging that (1) an assignee may have information regarding Parent, any Borrower and any Subsidiary of Parent, their ability to perform the Obligations or any other material information that has not previously been disclosed to the Administrative Agent and the Lenders (“Excluded Information”), (2) the Excluded Information may not be available to such Lender, (3) such Lender has independently and without reliance on any other party made its own analysis and determined to assign Term Loans to such assignee pursuant to Section 9.04(g) and Section 9.04(k) notwithstanding its lack of knowledge of the Excluded Information and (4) such Lender waives and releases any claims it may have against the Administrative Agent, such assignee, Parent, any Borrower and the Subsidiaries of Parent with respect to the nondisclosure of the Excluded Information; or otherwise in form and substance reasonably satisfactory to such assignee, the Administrative Agent and assigning Lender. “Blocking Law” has the meaning assigned to such term in Section 5.08(c).

13 “Board” means the Board of Governors of the Federal Reserve System of the United States of America. “Borrower” means the Borrower Representative and/or any Additional Borrower (subject to Section 9.18), as applicable. “Borrower Materials” has the meaning assigned to such term in the final paragraph of Section 5.01. “Borrowing” means (a) Revolving Loans of the same Class, Type and currency made, converted or continued on the same date and, in the case of Term Benchmark Loans, as to which a single Interest Period is in effect, (b) Term Loans of the same Class and Type made on the same date and, in the case of Term Benchmark Loans meeting the foregoing requirements, as to which a single Interest Period is in effect or (c) a Swingline Loan. “Borrowing Request” means a request by the applicable Borrower for a Borrowing in accordance with Section 2.03. “Business Day” means any day that is not a Saturday, Sunday or other day on which commercial banks in New York City are authorized or required by law to remain closed; provided that, when used in connection with (i) a Borrowing denominated in Canadian Dollars and in relation to the calculation or computation of Term CORRA or the Canadian Prime Rate, the term “Business Day” shall also exclude any day that is not a CORRA Business Day and (ii) an ABR Loan or a Term SOFR Loan, the term “Business Day” shall also exclude any day on which banks are not open for dealings in the relevant Agreed Currency in the applicable market or the principal financial center of the country of such Agreed Currency (and, if the Borrowings or LC Disbursements which are the subject of a borrowing, drawing, payment, reimbursement or rate selection are denominated in euro, the term “Business Day” shall also exclude any day on which the TARGET payment system is not open for the settlement of payments in euro). “Canadian Dollar Revolving Loans” means Multicurrency Tranche Revolving Loans denominated in Canadian Dollars. Each Canadian Dollar Revolving Loan shall be a Term CORRA Loan or Canadian Prime Rate Loan. “Canadian Dollars” or “CAD” refers to the lawful currency of Canada (expressed in Canadian dollars). “Canadian Domiciled Subsidiary” means each Restricted Subsidiary incorporated or otherwise organized under the laws of Canada or any province or territory thereof. “Canadian Prime Rate” means, on any day, the rate determined by the Administrative Agent to be the higher of (i) the rate of interest quoted in the print edition of The Wall Street Journal, Money Rates Section as the “Canadian Prime Rate”, as in effect from time to time, or if The Wall Street Journal ceases to quote such rate, the highest per annum interest rate published by the Bank of Canada as its policy rate and (ii) the Adjusted Term CORRA Rate for an Interest Period of one (1) month plus 1.00%; provided that if any the above rates shall be less than the Floor, such rate shall be deemed to be equal to the Floor for the purposes of this Agreement. Any change in the Canadian Prime Rate due to a change in the quoted or published prime rate

14 pursuant to the foregoing clause (i) or the Adjusted Term CORRA Rate shall be effective from and including the effective date of such change in such prime rate or Adjusted Term CORRA Rate. “Canadian Prime Rate Loans” means any Canadian Dollar Revolving Loan during the period which it bears interest at a rate determined by reference to the Canadian Prime Rate. “Canadian Statutory Liens” means any Lien in respect of any property or assets of a Canadian Domiciled Subsidiary created by or arising pursuant to any applicable legislation in favour of any Person (such as but not limited to a Governmental Authority), including, without limitation, a Lien for the purpose of securing such Canadian Domiciled Subsidiary’s obligation to deduct and remit employee source deductions and goods and services tax pursuant to the Income Tax Act (Canada), the Excise Tax Act (Canada), the Canada Pension Plan (Canada), the Employment Insurance Act (Canada) and any legislation in any jurisdiction similar to or enacted in replacement of the foregoing from time to time. “Capital Expenditures” means, without duplication, any expenditures for any purchase or other acquisition of any asset which would be classified as a fixed or capital asset on a consolidated balance sheet of Parent and its Restricted Subsidiaries prepared in accordance with GAAP. “Capital Lease Obligations” of any Person means the obligations of such Person to pay rent or other amounts under any lease of (or other arrangement conveying the right to use) real or personal property, or a combination thereof, which obligations are required to be classified and accounted for as capital leases on a balance sheet of such Person under GAAP, and the amount of such obligations shall be the capitalized amount thereof determined in accordance with GAAP; provided, however, all obligations of any Person that are or would have been treated as operating leases (including for avoidance of doubt, any network lease or any Operating IRU) for purposes of GAAP prior to the issuance by the Financial Accounting Standards Board on February 25, 2016 of an Accounting Standards Update (the “ASU”) shall continue to be accounted for as operating leases for purposes of all financial definitions and calculations for purpose of this Agreement (whether or not such operating lease obligations were in effect on such date) notwithstanding the fact that such obligations are required in accordance with the ASU (on a prospective or retroactive basis or otherwise) to be treated as Capital Lease Obligations in the financial statements to be delivered pursuant to Section 5.01. “Capitalized Software Expenditures” means, for any period, the aggregate of all expenditures (whether paid in cash or accrued as liabilities) by a Person and its Restricted Subsidiaries during such period in respect of licensed or purchased software or internally developed software and software enhancements that, in conformity with GAAP, are or are required to be reflected as capitalized costs on the consolidated balance sheet of a Person and its Restricted Subsidiaries. “Captive Insurance Subsidiary” means any Subsidiary of Parent that is subject to regulation as an insurance company (or any Subsidiary thereof). “Cash Collateralized” means, with respect to any Letter of Credit, as of any date, that the applicable Borrower shall have deposited in the LC Collateral Account, in the name of the

15 Administrative Agent and for the benefit of the Lenders, an amount in cash equal to 103% of the LC Exposure as of such date plus any accrued and unpaid interest thereon pursuant to such documentation and arrangements as are reasonably satisfactory to the Administrative Agent. “Cash Collateralize” shall have the correlative meaning. “Change in Control” means (a) the acquisition of ownership, directly or indirectly, beneficially or of record, by any Person or group (within the meaning of the Securities Exchange Act of 1934 and the rules of the SEC thereunder as in effect on the Effective Date) other than one or more Permitted Holders, of Equity Interests representing more than 50% of the aggregate ordinary voting power represented by the issued and outstanding Equity Interests of Parent; (b) the occurrence of a change of control, or other similar provision, as defined in any agreement or instrument evidencing any Material Indebtedness (triggering a default or mandatory prepayment, which default or mandatory prepayment has not been waived in writing); or (c) any Borrower ceasing to be a direct or indirect wholly-owned subsidiary of Parent. Notwithstanding the foregoing, a transaction will not be deemed to constitute a Change in Control if (1) Parent becomes a direct or indirect wholly-owned Subsidiary of a holding company and (2)(A) the direct or indirect holders of the voting stock of such holding company immediately following that transaction are substantially the same as the holders of such voting stock immediately prior to that transaction or (B) immediately following that transaction no Person (other than a holding company satisfying the requirements of this sentence) is the beneficial owner, directly or indirectly, of more than 50% of the voting stock of such holding company. “Change in Law” means the occurrence, after the Effective Date, of any of the following: (a) the adoption or taking effect of any law, rule, regulation or treaty, (b) any change in any law, rule, regulation or treaty or in the administration, interpretation or application thereof by any Governmental Authority or (c) the making or issuance of any request, rules, guideline, requirement or directive (whether or not having the force of law) by any Governmental Authority; provided, however, that notwithstanding anything herein to the contrary, (i) the Dodd-Frank Wall Street Reform and Consumer Protection Act and all requests, rules, guidelines, requirements and directives thereunder, issued in connection therewith or in implementation thereof shall be deemed to be a “Change in Law” regardless of the date enacted, adopted, issued or implemented and (ii) all reports, notes, guidelines, rules, requests and directives promulgated by the Bank for International Settlements, the Basel Committee on Banking Supervision (or any successor or similar authority) or the United States or foreign regulatory authorities, in each case pursuant to Basel III, shall, in each case, be deemed to be a “Change in Law” regardless of the date enacted, adopted, issued or implemented. “Chapter 11 Cases” means the jointly administered chapter 11 bankruptcy cases of PLC and certain of its affiliates as debtors and debtors in possession. “Class”, when used in reference to (a) any Loan or Borrowing, refers to whether such Loan, or the Loans comprising such Borrowing, are Loans of a particular Tranche; provided that any Loans within a Tranche having different Maturity Dates, currency of denomination (except pursuant to a Class of revolving commitments allowing extensions of credit thereunder in multiple currencies), interest rates, repayments or other terms shall be regarded as separate Classes of Loans and Borrowings for purposes of this Agreement, (b) any Commitment, refers to whether such Commitment is a Commitment of a particular Tranche; provided that any Commitments

16 within a Tranche having different Maturity Dates, currency of denomination (except pursuant to a Class of revolving commitments allowing extensions of credit thereunder in multiple currencies), interest rates, repayments or other terms shall be regarded as separate Classes of Commitments for purposes of this Agreement and (c) any Lender, refers to whether such Lender is a Lender of a particular Tranche; provided that any Lender holding Loans or Commitments within a Tranche having different Maturity Dates, currency of denomination (except pursuant to a Class of revolving commitments allowing extensions of credit thereunder in multiple currencies), interest rates, repayments or other terms shall be regarded as a Lender with respect to separate Classes of Loans and/or Commitments (as applicable) for purposes of this Agreement. “CME Term SOFR Administrator” means CME Group Benchmark Administration Limited as administrator of the forward-looking term Secured Overnight Financing Rate (SOFR) (or a successor administrator). “Code” means the Internal Revenue Code of 1986, as amended from time to time. “Collateral” means any and all assets of a Loan Party covered by the Collateral Documents, but only so long as the Collateral Documents are then in effect, and any and all other assets of any Loan Party, now existing or hereafter acquired and wherever located, that may at any time be or become subject to a security interest or Lien in favor of the Collateral Agent, on behalf of itself and the Secured Parties, to secure the Secured Obligations; provided that Collateral shall exclude Excluded Assets. “Collateral Agent” means Goldman Sachs Bank USA, in its capacity as collateral agent for the Secured Parties. “Collateral Documents” means, collectively, the US Security Agreement, the Irish Security Documents, the Luxembourg Pledge Agreement, the Mortgages and all other agreements, instruments and documents executed in connection with this Agreement that are intended to create, perfect or evidence Liens to secure the Secured Obligations, and shall also include, without limitation, all other security agreements, pledge agreements, mortgages, short-form intellectual property security agreements, deeds of trust, loan agreements, notes, guarantees, subordination agreements, intercreditor agreements, pledges and each of the other agreements, instruments or documents that creates, perfects or evidences, or purports to create, perfect or evidence a Lien in favor of the Collateral Agent for the benefit of the Secured Parties. “Commitment” means, with respect to each Lender, the sum of such Lender’s Multicurrency Tranche Commitment, Dollar Tranche Commitment, Term Loan Commitment, Incremental Revolving Commitment, Other Refinancing Revolving Commitment and Incremental Term Loan Commitment. “Commodity Exchange Act” means the Commodity Exchange Act (7 U.S.C. § 1 et seq.), as amended from time to time, and any successor statute. “Competitor” shall mean any person that is a bona fide operating company engaged in the same or a line of similar business as the Borrower or its Subsidiaries.

17 “Compliance Certificate” means a certificate of a Financial Officer of Parent required to be delivered with the Financials pursuant to Section 5.01(c). “Compliance Date” means the last day of any Reference Period that the aggregate outstanding Revolving Credit Exposure (other than (a) undrawn Letters of Credit in an amount not to exceed $20 million and (b) Letters of Credit to the extent Cash Collateralized or backstopped (whether drawn or undrawn) on terms reasonably acceptable to the applicable Issuing Bank) exceeds an amount equal to 40% of the Revolving Commitments then in effect. “Computation Date” has the meaning assigned to such term in Section 2.04(c). “Connection Income Taxes” means Other Connection Taxes that are imposed on or measured by net income (however denominated) or that are franchise Taxes or branch profits Taxes. “Consolidated Current Assets” means, with respect to Parent and its Restricted Subsidiaries on a consolidated basis at any date of determination, all assets (other than cash and Permitted Investments) that would, in accordance with GAAP, be classified on a consolidated balance sheet of Parent and its Restricted Subsidiaries as current assets at such date of determination, other than amounts related to current or deferred Taxes based on income or profits (but excluding (i) assets held for sale, (ii) permitted loans to third parties, (iii) Plan assets, (iv) deferred bank fees, and (v) derivative financial instruments). “Consolidated Current Liabilities” means, with respect to Parent and its Restricted Subsidiaries on a consolidated basis at any date of determination, all liabilities that would, in accordance with GAAP, be classified on a consolidated balance sheet of Parent and its Restricted Subsidiaries as current liabilities at such date of determination, other than (i) the current portion of any Indebtedness, (ii) the current portion of interest, (iii) accruals for current or deferred Taxes based on income or profits, (iv) accruals of any costs or expenses related to restructuring reserves, (v) the aggregate amount of outstanding Revolving Loans and Swingline Loans and LC Exposure and (vi) the current portion of pension liabilities. “Consolidated Depreciation and Amortization Expense” means, with respect to any Person for any period, the total amount of depreciation and amortization expense of such Person and its Restricted Subsidiaries, including the amortization of intangible assets, deferred financing fees, debt issuance costs, commissions, fees and expenses and the amortization of Capitalized Software Expenditures of such Person and its Restricted Subsidiaries for such period on a consolidated basis and otherwise determined in accordance with GAAP. “Consolidated EBITDA” means, with respect to any Person, the Consolidated Net Income of such Person and its Restricted Subsidiaries for such period: (c) increased (without duplication) by the following, in each case (other than clauses (x) and (xiv)) to the extent deducted (and not added back) in determining Consolidated Net Income for such period: (i) total interest expense and, to the extent not reflected in such total interest expense, any losses on Swap Obligations or other derivative instruments

18 entered into for the purpose of hedging interest rate risk, net of interest income and gains on such Swap Obligations or such derivative instruments, and bank and letter of credit fees, letter of guarantee and bankers’ acceptance fees and costs of surety bonds in connection with financing activities, together with items excluded from the definition of “Consolidated Interest Expense”; plus (ii) provision for taxes based on income, profits, revenue or capital, including federal, foreign and state income, franchise, excise, value added and similar taxes, property taxes and similar taxes, and foreign withholding taxes paid or accrued during such period (including any future taxes or other levies that replace or are intended to be in lieu of taxes, and any penalties and interest related to taxes or arising from tax examinations) and the net tax expense associated with any adjustments made pursuant to the definition of “Consolidated Net Income,” plus (iii) Consolidated Depreciation and Amortization Expense for such period; plus (iv) any non-recurring charges, costs, fees and expenses directly incurred or paid directly as a result of discontinued operations; plus (v) any cost, expense or other charge (including any legal fees and expenses) associated with the bankruptcy proceedings and related restructuring, adequate protection payments and the application of fresh-start accounting, or the payment of any legal settlement, fine, judgment or order, including all settlement payments paid to Governmental Authorities, as described in Parent and/or PLC’s public filings with the SEC or, in connection with the Chapter 11 Cases, the Reorganization Plan or the Reorganization Plan Documents; plus (vi) Milestone Payments and Upfront Payments; plus (vii) minority interest expense, the amount of any non-controlling interest consisting of income attributable to non-controlling interests of third parties in any non-wholly-owned Restricted Subsidiary, excluding cash distributions in respect thereof, and the amount of any reductions in arriving at Consolidated Net Income resulting from the application of Accounting Standards Codification Topic No. 810, Consolidation; plus (viii) (i) the amount of board of director or similar fees and (ii) the amount of payments made to optionholders of such Person in connection with, or as a result of, any distribution being made to equityholders of such Person, which payments are being made to compensate such optionholders as though they were equityholders at the time of, and entitled to share in, such distribution, in each case to the extent permitted hereunder; plus (ix) the amount of loss or discount on sale of any Receivables Assets to any Restricted Subsidiary or Receivables Entity in connection with a Permitted Receivables Facility; plus

19 (x) cash receipts (or any netting arrangements resulting in reduced cash expenditures) not representing Consolidated EBITDA or Consolidated Net Income in any prior period to the extent non-cash gains relating to such income were deducted in the calculation of Consolidated EBITDA pursuant to clause (b) below for any previous period and not added back; plus (xi) any costs or expenses incurred pursuant to any management equity plan, stock option plan or any other management or employee benefit plan, agreement or any stock subscription or shareholder agreement, to the extent that such costs or expenses are funded with cash proceeds contributed to the capital of such Person or net cash proceeds of an issuance of Equity Interests of such Person (other than Disqualified Equity Interest); plus (xii) any net pension or other post-employment benefit costs representing amortization of unrecognized prior service costs, actuarial losses, including amortization of such amounts arising in prior periods, amortization of the unrecognized net obligation (and loss or cost) existing at the date of initial application of FASB Accounting Standards Codification Topic 715— Compensation—Retirement Benefits, and any other items of a similar nature, plus (xiii) [reserved]; plus (xiv) the amount of “run-rate” cost savings, synergies and operating expense reductions related to restructurings, cost savings initiatives or other initiatives that are projected by Parent in good faith to result from actions either taken or with respect to which substantial steps have been taken or are expected to be taken within 18 months after the end of such period, calculated as though such cost savings, synergies and operating expense reductions had been realized on the first day of such period and net of the amount of actual benefits received during such period from such actions; provided that (A) any such pro forma adjustments in respect of such cost savings, synergies and operating expense reductions shall not exceed 20.0% of Consolidated EBITDA (prior to giving effect to such pro forma adjustment) as of the end of the Reference Period at such time, (B) such cost savings and synergies are reasonably expected and factually supportable in the good faith judgment of Parent and (C) no cost savings or synergies shall be added pursuant to this clause (xiv) to the extent duplicative of any expenses or charges otherwise added to Consolidated EBITDA, whether through a pro forma adjustment or otherwise, for such period (it is understood and agreed that “run rate” means the full recurring benefit that is associated with any action taken or with respect to which substantial steps have been taken or are expected to be taken, whether prior to or following the Effective Date) (which adjustments may be incremental to (but not duplicative of) pro forma cost savings, synergies or operating expense reduction adjustments made pursuant to Section 1.04); provided that such cost savings, synergies and operating expenses are reasonably identifiable and factually supportable; plus

20 (xv) the aggregate amount of all other non-cash charges, expenses or losses reducing Consolidated Net Income during such period (including all reserves taken during such period on account of contingent cash payments that may be required in a future period) and (d) decreased (without duplication) by the following, in each case to the extent included in determining Consolidated Net Income for such period: (1) any cash payments made during such period in respect of items described in clause (xv) above subsequent to the period in which the relevant non- cash expenses or losses were incurred; (2) any non-recurring income or gains directly as a result of discontinued operations; (3) any unrealized income or gains in respect of Swap Agreements; and (4) the amount of any loss attributable to non-controlling interests of third parties in any non-wholly owned Restricted Subsidiary added to (and not deducted from) Consolidated Net Income in such period. For the avoidance of doubt, Consolidated EBITDA shall be calculated, including pro forma adjustments, in accordance with Section 1.04. “Consolidated First Lien Secured Debt” means, as of any date of determination, the aggregate principal amount of Indebtedness of Parent and its Restricted Subsidiaries outstanding on such date, determined on a consolidated basis in accordance with GAAP, consisting only of Indebtedness for borrowed money, Capital Lease Obligations and purchase money Indebtedness, in each case secured by a first priority lien on any asset or property of Parent or any other Loan Party; provided, Consolidated First Lien Secured Debt will not include Non-Recourse Indebtedness, undrawn amounts under revolving credit facilities and Indebtedness in respect of any (1) letter of credit, bank guarantees and performance or similar bonds, except to the extent of obligations in respect of drawn standby letters of credit which have not been reimbursed within two (2) Business Days and (2) Swap Obligations. “Consolidated Interest Coverage Ratio” means, as of any date of determination, the ratio of (a) Consolidated EBITDA for the most recently completed four fiscal quarters to (b) Consolidated Interest Expense for such period. “Consolidated Interest Expense” means, with reference to any period, the interest expense (including without limitation interest expense under Capital Lease Obligations that is treated as interest in accordance with GAAP) of Parent and its Restricted Subsidiaries calculated on a consolidated basis for such period with respect to (a) all outstanding Indebtedness of Parent and its Restricted Subsidiaries allocable to such period in accordance with GAAP (including, without limitation, all commissions, discounts and other fees and charges owed with respect to letters of credit and bankers acceptance financing and net costs and benefits under interest rate Swap Agreements to the extent such net costs and benefits are allocable to such period in

21 accordance with GAAP) and (b) the interest component of all Attributable Receivables Indebtedness of Parent and its Restricted Subsidiaries for such period. “Consolidated Net Income” means, with respect to any Person for any period, the net income (loss) of such Person and its Restricted Subsidiaries for such period determined on a consolidated basis in accordance with GAAP, excluding (and excluding the effect of), without duplication, (1) extraordinary, non-recurring or unusual gains, losses, fees, costs, charges or expenses (including relating to any strategic initiatives and accruals and reserves in connection with such gains, losses, charges or expenses); restructuring costs, charges, accruals or reserves; severance and relocation costs and expenses, one-time compensation costs and expenses, consulting fees, signing, retention or completion bonuses, and executive recruiting costs; costs and expenses incurred in connection with strategic initiatives; transition costs and duplicative running costs; costs incurred in connection with acquisitions (or purchases of assets) prior to or after the Effective Date (including integration costs); business optimization expenses; operating expenses attributable to the implementation of cost-savings initiatives; (2) the cumulative effect of a change in accounting principles and changes as a result of the adoption or modification of accounting policies during such period whether effected through a cumulative effect adjustment or a retroactive application, in each case in accordance with GAAP; (3) Transaction Expenses; (4) any gain (loss) on asset sales, disposals or abandonments (other than asset sales, disposals or abandonments in the ordinary course of business); (5) the net income for such period of any Person that is an Unrestricted Subsidiary and, solely for the purpose of determining the amount available for Restricted Payments under clause (a)(vii) of the definition of “Available Amount”, the net income for such period of any Person that is not a Subsidiary or that is accounted for by the equity method of accounting, in each case except to the extent of any dividends or distributions or other payments that are actually paid in cash or Permitted Investments (or to the extent converted into cash or Permitted Investments) to such Person or a Restricted Subsidiary thereof in respect of such period; (6) solely for the purpose of determining the amount available for Restricted Payments under clause (a)(vii) of the definition of “Available Amount”, the net income for such period of any Restricted Subsidiary (other than any Guarantor) to the extent that the declaration or payment of dividends or similar distributions by that Restricted Subsidiary of its net income is not at the date of determination permitted without any prior governmental approval (which has not been obtained) or, directly or indirectly, by the operation of the terms of its charter or any agreement, instrument, judgment, decree, order, statute, rule or governmental regulation applicable to that Restricted Subsidiary or its stockholders, unless such restriction with respect to the payment of dividends or similar

22 distributions has been legally waived; provided that Consolidated Net Income of a Person will be increased by the amount of dividends or other distributions or other payments actually paid in cash or Permitted Investments (or to the extent converted into cash or Permitted Investments), or the amount that could have been paid in cash or Permitted Investments without violating any such restriction or requiring any such approval, to such Person or a Restricted Subsidiary thereof in respect of such period, to the extent not already included therein; (7) effects of adjustments (including the effects of such adjustments pushed down to such Person and its Restricted Subsidiaries) related to the application of recapitalization accounting or purchase accounting (including in the inventory, property and equipment, software, goodwill, intangible assets, in process research and development, deferred revenue and debt line items); (8) income (loss) from the early extinguishment or conversion of (a) Indebtedness, (b) Swap Obligations or (c) other derivative instruments; (9) any impairment charge or asset write-off or write-down in each case, pursuant to GAAP, and the amortization of intangibles arising pursuant to GAAP; (10) (a) any equity based or non-cash compensation charge or expense, including any such charge or expense arising from grants of stock appreciation, equity incentive programs or similar rights, stock options, restricted stock or other rights to, and any cash charges associated with the rollover, acceleration or payout of, Equity Interests by management of such Person or of a Restricted Subsidiary, (b) noncash compensation expense resulting from the application of Accounting Standards Codification Topic No. 718, Compensation—Stock Compensation or Accounting Standards Codification Topic 505-50, Equity-Based Payments to Non-Employees, and (c) any income (loss) attributable to deferred compensation plans or trusts; (11) any fees, expenses or charges incurred during such period, or any amortization thereof for such period, in connection with any acquisition, Investment, Asset Sale, disposition, incurrence or repayment of Indebtedness (including such fees, expenses or charges related to the offering and issuance of the Senior Secured Notes), issuance of Equity Interests, recapitalization, refinancing transaction or amendment or modification of any debt instrument (including any amendment or other modification of the Senior Secured Notes and other securities and this Agreement) and including, in each case, any such transaction whether consummated on, after or prior to the Effective Date and any such transaction undertaken but not completed, and any charges or nonrecurring merger costs incurred during such period as a result of any such transaction, in each case whether or not successful or consummated (including, for the avoidance of doubt, the effects of expensing all transaction related expenses in accordance with Accounting Standards Codification Topic No. 805, Business Combinations); (12) accruals and reserves that are established or adjusted in connection with the Transactions, an Investment or an acquisition that are required to be established or adjusted

23 as a result of the Transactions, such Investment or such acquisition, in each case in accordance with GAAP; (13) any expenses, charges or losses to the extent covered by insurance that are, directly or indirectly, reimbursed or reimbursable by a third party, and any expenses, charges or losses that are covered by indemnification or other reimbursement provisions only to the extent that such amount is in fact reimbursed within 365 days of the date of such determination (with a deduction in the applicable future period for any amount so excluded to the extent not so reimbursed within such 365 days); (14) any non-cash gain (loss) attributable to the mark to market movement in the valuation of Swap Obligations or other derivative instruments pursuant to FASB Accounting Standards Codification Topic 815—Derivatives and Hedging or mark to market movement of other financial instruments pursuant to FASB Accounting Standards Codification Topic 825—Financial Instruments; (15) any net unrealized gain or loss (after any offset) resulting in such period from currency transaction or translation gains or losses including those related to currency remeasurements of Indebtedness (including any net loss or gain resulting from (a) Swap Obligations for currency exchange risk and (b) resulting from intercompany indebtedness) and any other foreign currency transaction or translation gains and losses, to the extent such gain or losses are non-cash items; (16) any adjustments resulting from the application of Accounting Standards Codification Topic No. 460, Guarantees, or any comparable regulation; (17) any non-cash rent expense; (18) any non-cash expenses, accruals or reserves related to adjustments to historical tax exposures; and (19) earn-out and contingent consideration obligations (including to the extent accounted for as bonuses or otherwise) and adjustments thereof and purchase price adjustments. In addition, to the extent not already included in the Consolidated Net Income of such Person and its Restricted Subsidiaries, Consolidated Net Income will include the amount of proceeds received or receivable from business interruption insurance, the amount of any expenses or charges incurred by such Person or its Restricted Subsidiaries during such period that are, directly or indirectly, reimbursed or reimbursable by a third party, and amounts that are covered by indemnification or other reimbursement provisions in connection with any acquisition, Investment or any sale, conveyance, transfer or other disposition of assets permitted hereunder only to the extent that such amount is in fact reimbursed within 365 days of the date of such determination (with a deduction in the applicable future period for any amount so excluded to the extent not so reimbursed within such 365 days). For the avoidance of doubt, Consolidated Net Income shall be calculated, including pro forma adjustments, in accordance with Section 1.04.

24 “Consolidated Secured Debt” means, the aggregate principal amount of Indebtedness of Parent and its Restricted Subsidiaries outstanding on such date, determined on a consolidated basis in accordance with GAAP, consisting only of Indebtedness for borrowed money, Capital Lease Obligations and purchase money Indebtedness, in each case secured by a lien on any asset or property of Parent or any other Loan Party; provided, Consolidated Secured Debt will not include Non-Recourse Indebtedness, undrawn amounts under revolving credit facilities and Indebtedness in respect of any (1) letter of credit, bank guarantees and performance or similar bonds, except to the extent of obligations in respect of drawn standby letters of credit which have not been reimbursed within two (2) Business Days and (2) Swap Obligations. “Consolidated Total Assets” means, as of the date of any determination thereof, total assets of Parent and its Restricted Subsidiaries calculated in accordance with GAAP on a consolidated basis as of such date (and, in the case of any determination relating to any Specified Transaction, on a pro forma basis including any property or assets being acquired in connection therewith). “Consolidated Total Indebtedness” means, as of any date of determination, the aggregate principal amount of Indebtedness of Parent and its Restricted Subsidiaries outstanding on such date, determined on a consolidated basis in accordance with GAAP, consisting only of Indebtedness for borrowed money, Capital Lease Obligations and purchase money Indebtedness; provided, Consolidated Total Indebtedness will not include Non-Recourse Indebtedness, undrawn amounts under revolving credit facilities and Indebtedness in respect of any (1) letter of credit, bank guarantees and performance or similar bonds, except to the extent of obligations in respect of drawn standby letters of credit which have not been reimbursed within two (2) Business Days and (2) Swap Obligations. “Consolidated Working Capital” means, at any time, Consolidated Current Assets (but excluding therefrom all cash and Permitted Investments) less Consolidated Current Liabilities at such time; provided that increases or decreases in Consolidated Working Capital shall be calculated without regard to any changes in Consolidated Current Assets or Consolidated Current Liabilities as a result of (x) any reclassification in accordance with GAAP of assets or liabilities, as applicable, between current and noncurrent, (y) the effects of purchase accounting or (z) the effect of fluctuations in the amount of accrued or contingent obligations, assets or liabilities under Swap Agreements. “Control” means the possession, directly or indirectly, of the power to direct or cause the direction of the management or policies of a Person, whether through the ability to exercise voting power, by contract or otherwise. “Controlling” and “Controlled” have meanings correlative thereto. “Controlled Affiliate” has the meaning assigned to such term in Section 3.15(a). “Controlled Foreign Corporation” means any Subsidiary of Parent (i) which is a “controlled foreign corporation” within the meaning of Section 957 of the Code or (ii) that has no material assets other than Equity Interests (including any debt instrument treated as equity for U.S. federal income tax purposes) or other securities of Persons described in clause (i).

25 “Convertible Debt Security” means debt securities, the terms of which provide for conversion into, or exchange for, Equity Interests (other than Disqualified Equity Interests) of Parent, cash in lieu thereof and/or a combination of such Equity Interests and cash in lieu thereof. “CORRA” means the Canadian Overnight Repo Rate Average administered and published by the Bank of Canada (or any successor administrator). “CORRA Business Day” means, for any Loan denominated in Canadian Dollars, any day except for (i) a Saturday, (ii) a Sunday or (iii) a day on which commercial banks in Toronto are authorized or required by law to remain closed. “CORRA Determination Date” has the meaning specified in the definition of “Daily Simple CORRA”. “CORRA Rate Day” has the meaning specified in the definition of “Daily Simple CORRA”. “Corresponding Tenor” with respect to any Available Tenor means, as applicable, either a tenor (including overnight) or an interest payment period having approximately the same length (disregarding business day adjustment) as such Available Tenor. “Credit Agreement Refinancing Indebtedness” means any (a) Permitted Pari Passu Secured Refinancing Debt, (b) Permitted Junior Secured Refinancing Debt, (c) Permitted Unsecured Refinancing Debt or (d) Indebtedness incurred pursuant to a Refinancing Amendment, in each case, issued, incurred or otherwise obtained (including by means of the extension or renewal of existing Indebtedness) in exchange for, or to extend, renew, replace or refinance, in whole or part, any existing Class of Term Loans or Revolving Commitments (including any successive Credit Agreement Refinancing Indebtedness) (“Refinanced Debt”); provided that (i) such exchanging, extending, renewing, replacing or refinancing Indebtedness (including, if such Indebtedness includes any Other Refinancing Revolving Commitments, the unused portion of such Other Refinancing Revolving Commitments) is in an original aggregate principal amount not greater than the aggregate principal amount of the Refinanced Debt (and, in the case of Refinanced Debt consisting, in whole or in part, of unused Revolving Commitments, Incremental Revolving Commitments, Extended Revolving Commitments or Other Refinancing Revolving Commitments, the amount thereof) except by an amount equal to unpaid accrued interest and premium (including tender premium) thereon plus reasonable upfront fees and original issue discount on such exchanging, extending, renewing, replacing or refinancing Indebtedness, plus other reasonable and customary fees and expenses in connection with such exchange, modification, refinancing, refunding, renewal, replacement or extension, (ii) such Indebtedness has a later maturity date no earlier than, and, except in the case of Other Refinancing Revolving Commitments, a Weighted Average Life to Maturity equal to or greater than, the Refinanced Debt (other than such Indebtedness incurred under the Inside Maturity Basket), (iii) the terms and conditions of such Indebtedness (except as otherwise provided in clause (ii) above and with respect to pricing, premiums and optional prepayment or redemption terms) are substantially identical to, or (taken as a whole) are no more favorable to the lenders or holders providing such Indebtedness in any material respect, than those applicable to the Loans or Commitments being refinanced (as determined in good faith by Parent), or, except with respect Indebtedness incurred pursuant to a

26 Refinancing Amendment pursuant to clause (d) above, are otherwise current market terms (in each case except for covenants or other provisions applicable only to periods after the Latest Maturity Date at the time of incurrence of such Indebtedness); (iv) such Refinanced Debt shall be repaid, defeased or satisfied and discharged, and all accrued interest, fees and premiums (if any) in connection therewith shall be paid, on the date such Credit Agreement Refinancing Indebtedness is issued, incurred or obtained in accordance with Section 2.11(c)(2) and (v) such Indebtedness may be incurred in the form of a bridge or other interim credit facility intended to be refinanced with long-term indebtedness (and such bridge or other interim credit facility shall be deemed to satisfy clause (ii) of this definition so long as (x) such credit facility includes customary “rollover” provisions and (y) assuming such credit facility were to be extended pursuant to such “rollover” provisions, such extended credit facility would comply with clause (ii) above) and in which case, on or prior to the first anniversary of the incurrence of such “bridge” or other interim credit facility, clause (iii) in this definition shall not prohibit the inclusion of customary terms for “bridge” facilities, including customary mandatory prepayment, repurchase or redemption provisions. “Credit Event” means a Borrowing, the issuance of a Letter of Credit, an LC Disbursement or any of the foregoing. “Credit Exposure” means, as to any Lender at any time, the sum of (a) such Lender’s Revolving Credit Exposure at such time, plus (b) an amount equal to the aggregate principal amount of its Term Loans outstanding at such time. “Credit Party” means the Administrative Agent, the Issuing Bank, the Swingline Lender or any other Lender. “Daily Simple CORRA” means, for any day (a “CORRA Rate Day”), a rate per annum equal to CORRA for the day (such day “CORRA Determination Date”) that is five (5) CORRA Business Days prior to (i) if such CORRA Rate Day is a CORRA Business Day, such CORRA Rate Day or (ii) if such CORRA Rate Day is not a CORRA Business Day, the CORRA Business Day immediately preceding such CORRA Rate Day, in each case, as such CORRA is published by the Term CORRA Administrator on the Term CORRA Administrator’s website. Any change in Daily Simple CORRA due to a change in CORRA shall be effective from and including the effective date of such change in CORRA without notice to the Borrower Representative. If by 5:00 p.m. (Toronto time) on any given CORRA Determination Date, CORRA in respect of such CORRA Determination Date has not been published on the Term CORRA Administrator’s website and a Benchmark Replacement Date with respect to the Daily Simple CORRA has not occurred, then CORRA for such CORRA Determination Date will be CORRA as published in respect of the first preceding CORRA Business Day for which such CORRA was published on the Term CORRA Administrator’s website, so long as such first preceding CORRA Business Day is not more than five (5) Business Days prior to such CORRA Determination Day. “Daily Simple SOFR” means, for any day (a “SOFR Rate Day”), a rate per annum equal to SOFR for the day (such day “SOFR Determination Date”) that is five (5) U.S. Government Securities Business Days prior to (i) if such SOFR Rate Day is a U.S. Government Securities Business Day, such SOFR Rate Day or (ii) if such SOFR Rate Day is not a U.S. Government Securities Business Day, the U.S. Government Securities Business Day immediately preceding such SOFR Rate Day, in each case, as such SOFR is published by the SOFR Administrator on the

27 SOFR Administrator’s Website. Any change in Daily Simple SOFR due to a change in SOFR shall be effective from and including the effective date of such change in SOFR without notice to the Borrower Representative. If by 5:00 p.m. (New York City time) on the second (2nd) U.S. Government Securities Business Day immediately following any SOFR Determination Date, SOFR in respect of such SOFR Determination Date has not been published on the SOFR Administrator’s Website and a Benchmark Replacement Date with respect to the Daily Simple SOFR has not occurred, then SOFR for such SOFR Determination Date will be SOFR as published in respect of the first preceding U.S. Government Securities Business Day for which such SOFR was published on the SOFR Administrator’s Website. “Daily Simple SOFR Loan”, when used in reference to any a Loan or Borrowing, means a Loan, or the Loans comprising such Borrowing, bearing interest at rate determined by reference to Daily Simple SOFR. “Debt Fund Affiliate” means any Lender that is an Affiliate of Parent and is a bona fide debt fund or an investment vehicle that is primarily engaged in making, purchasing, holding or otherwise investing in commercial loans, bonds and similar extensions of credit in the ordinary course of its business and whose managers have fiduciary duties to the investors therein independent of or in addition to their duties to any Affiliate of Parent that beneficially owns Equity Interests of Parent. “Debtor Relief Law” means the Bankruptcy Code, and all other liquidation, conservatorship, bankruptcy, assignment for the benefit of creditors, moratorium, rearrangement, receivership, insolvency, reorganization, examinership or similar debtor relief laws of the United States or other applicable jurisdictions from time to time in effect and affecting the rights of creditors generally. “Debtors” shall have the meaning given to such term in the definition of “Reorganization Plan Transactions”. “Declined Prepayment Amount” has the meaning assigned to such term in Section 2.11(f). “Default” means any event or condition which constitutes an Event of Default or which upon notice, lapse of time or both would, unless cured or waived, become an Event of Default. “Defaulting Lender” means any Lender that (a) has failed, within three (3) Business Days of the date required to be funded or paid, to (i) fund any portion of its Loans, (ii) fund any portion of its participations in Letters of Credit or Swingline Loans or (iii) pay over to any Credit Party any other amount required to be paid by it hereunder, unless, in the case of clause (i) above, such Lender notifies the Administrative Agent in writing that such failure is the result of such Lender’s good faith determination that a condition precedent to funding (specifically identified and including the particular default, if any) has not been satisfied, (b) has notified Parent or any Credit Party in writing, or has made a public statement to the effect, that it does not intend or expect to comply with any of its funding obligations under this Agreement (unless such writing or public statement indicates that such position is based on such Lender’s good faith determination

28 that a condition precedent (specifically identified and including the particular default, if any) to funding a loan under this Agreement cannot be satisfied), (c) has failed, within three (3) Business Days after request by a Credit Party, acting in good faith, to provide a certification in writing from an authorized signatory of such Lender that it will comply with its obligations (and is financially able to meet such obligations) to fund prospective Loans and participations in then outstanding Letters of Credit and Swingline Loans under this Agreement; provided that such Lender shall cease to be a Defaulting Lender pursuant to this clause (c) upon such Credit Party’s receipt of such certification in form and substance satisfactory to it and the Administrative Agent (d) has become the subject of a Bankruptcy Event or (e) become the subject of a Bail-In Action. “Deposit Accounts” shall have the meaning set forth in Article 9 of the UCC. “Designated Representative” means, with respect to any series of Permitted Pari Passu Secured Refinancing Debt, Permitted First Lien Indebtedness or Permitted Junior Secured Refinancing Debt, the trustee, administrative agent, collateral agent, security agent or similar agent under the indenture or agreement pursuant to which such Indebtedness is issued, incurred or otherwise obtained, as the case may be, and each of their successors in such capacities. “Discharge of Priority Revolving Credit Obligations” shall have the meaning given to such term in the Intercreditor Agreement. “Disposition” means a sale, transfer, lease, disposition or Exclusive License. “Disposition Consideration” means (a) for any Disposition (other than an Exclusive License), the aggregate fair market value of any assets sold, transferred, leased or otherwise disposed of and (b) for any Exclusive License, the aggregate cash payment paid to Parent or any Restricted Subsidiary on or prior to the consummation of the Exclusive License (and which, for the avoidance of doubt, shall not include any royalty, earnout, contingent payment or any other deferred payment that may be payable thereafter). “Disqualified Equity Interest” means, with respect to any Person, any Equity Interest in such Person that by its terms (or by the terms of any security into which it is convertible or for which it is exchangeable, either mandatorily or at the option of the holder thereof), or upon the happening of any event or condition: (e) matures or is mandatorily redeemable (other than solely for Equity Interests in such Person or of Parent that do not constitute Disqualified Equity Interests and cash in lieu of fractional shares of such Equity Interests), whether pursuant to a sinking fund obligation or otherwise; (f) is convertible or exchangeable at the option of the holder thereof for Indebtedness or Equity Interests (other than solely for Equity Interests in such Person or of Parent that do not constitute Disqualified Equity Interests and cash in lieu of fractional shares of such Equity Interests); or (g) is or may be redeemable (other than solely for Equity Interests in such Person or of Parent that do not constitute Disqualified Equity Interests and cash in lieu of fractional shares of such Equity Interests) or is or may be required to be repurchased by

29 such Person or any of its Affiliates (other than, at the option of such Person, solely for Equity Interests in such Person or of Parent that do not constitute Disqualified Equity Interests and cash in lieu of fractional shares of such Equity Interests), in whole or in part, at the option of the holder thereof; in each case, on or prior to the date that occurs 91 days after the Latest Maturity Date; provided that (i) any Equity Interests that would constitute Disqualified Equity Interests solely because the holders thereof have the right to require Parent to repurchase such Disqualified Equity Interests upon the occurrence of a change of control or asset sale shall not constitute Disqualified Equity Interests if the terms of such Equity Interests (and all securities into which it is convertible or for which it is ratable or exchangeable) provide that Parent may not repurchase or redeem any such Equity Interests (and all securities into which it is convertible or for which it is ratable or exchangeable) pursuant to such provision unless the Obligations are fully satisfied simultaneously therewith and (ii) only the portion of the Equity Interests meeting one of the foregoing clauses (a) through (c) prior to the date that is ninety-one (91) days after the Latest Maturity Date will be deemed to be Disqualified Equity Interests. Notwithstanding the preceding sentence, (A) if such Equity Interest is issued pursuant to any plan for the benefit of directors, officers, employees, members of management, managers or consultants or by any such plan to such directors, officers, employees, members of management, managers or consultants, in each case in the ordinary course of business of Parent or any Restricted Subsidiary, such Equity Interest shall not constitute Disqualified Equity Interests solely because it may be required to be repurchased by the issuer thereof in order to satisfy applicable statutory or regulatory obligations, and (B) no Equity Interest held by any future, present or former employee, director, officer, manager, member of management or consultant (or their respective Affiliates or immediate family members) of Parent (or any Subsidiary) shall be considered Disqualified Equity Interests because such stock is redeemable or subject to repurchase pursuant to any management equity subscription agreement, stock option, stock appreciation right or other stock award agreement, stock ownership plan, put agreement, stockholder agreement or similar agreement that may be in effect from time to time. “Disqualified Lenders” means (a)(i) Persons identified in writing by the Borrower Representative to the Administrative Agent from time to time that are Competitors of Parent, the Borrower Representative or their respective Subsidiaries and (ii) any Affiliates of any such Competitors that are either (x) separately identified in writing by the Borrower Representative to the Administrative Agent from time to time or (y) known or clearly identifiable solely on the basis of such Affiliate’s name and (b) other Persons separately identified by the Borrower Representative to the Administrative Agent in writing prior to the Effective Date or any of their Affiliates that are either (x) separately identified in writing by the Borrower Representative to the Administrative Agent prior to the Effective Date or (y) known or clearly identifiable solely on the basis of such Affiliate’s name. With respect to any Disqualified Lender that is designated after the Effective Date, such designation shall not have retroactive effect. “Dollar Amount” of any currency at any date means (i) the amount of such currency if such currency is Dollars or (ii) the equivalent in such currency of Dollars if such currency is a Foreign Currency, calculated on the basis of the Exchange Rate for such currency, on or as of the most recent Revaluation Date provided for in Section 2.04.

30 “Dollar Tranche Commitment” means, with respect to each Dollar Tranche Lender, the commitment, if any, of such Dollar Tranche Lender to make Dollar Tranche Revolving Loans and to acquire participations in Dollar Tranche Letters of Credit and Swingline Loans hereunder, as such commitment may be (a) reduced or terminated from time to time pursuant to Section 2.09, (b) increased from time to time pursuant to Section 2.20 and (c) reduced or increased from time to time pursuant to assignments by or to such Lender pursuant to Section 9.04. The amount of each Dollar Tranche Lender’s Dollar Tranche Commitment as of the Effective Date is set forth on Schedule 2.01, or in the Assignment and Assumption (or other documentation contemplated by this Agreement) pursuant to which such Dollar Tranche Lender shall have assumed its Dollar Tranche Commitment, as applicable. The aggregate principal amount of the Dollar Tranche Commitments on the Effective Date is $0. “Dollar Tranche Credit Event” means a Dollar Tranche Revolving Borrowing of any Class, the issuance of a Dollar Tranche Letter of Credit, an LC Disbursement with respect to a Dollar Tranche Letter of Credit or any of the foregoing. “Dollar Tranche LC Exposure” means, at any time, the sum of (a) the aggregate undrawn Dollar Amount of all outstanding Dollar Tranche Letters of Credit at such time plus (b) the aggregate Dollar Amount of all LC Disbursements in respect of Dollar Tranche Letters of Credit that have not yet been reimbursed by or on behalf of the Borrower at such time. The Dollar Tranche LC Exposure of any Dollar Tranche Lender at any time shall be its Dollar Tranche Percentage of the total Dollar Tranche LC Exposure at such time. “Dollar Tranche Lender” means a Lender with a Dollar Tranche Commitment or holding Dollar Tranche Revolving Loans. “Dollar Tranche Letter of Credit” means any standby or commercial letter of credit issued under the Dollar Tranche Commitments pursuant to this Agreement. “Dollar Tranche Percentage” the percentage equal to a fraction the numerator of which is such Lender’s Dollar Tranche Commitment and the denominator of which is the aggregate Dollar Tranche Commitments of all Dollar Tranche Lenders (if the Dollar Tranche Commitments of any Class have terminated or expired, the Dollar Tranche Percentages shall be determined based upon the Dollar Tranche Commitments of such Class most recently in effect, giving effect to any assignments). “Dollar Tranche Revolving Borrowing” means a Borrowing comprised of Dollar Tranche Revolving Loans of any Class. “Dollar Tranche Revolving Credit Exposure” means, with respect to any Dollar Tranche Lender at any time, and without duplication, the sum of the outstanding principal amount of such Dollar Tranche Lender’s Dollar Tranche Revolving Loans and its Dollar Tranche LC Exposure and its Swingline Exposure at such time. “Dollar Tranche Revolving Loan” means a Loan made by a Dollar Tranche Lender pursuant to Section 2.01(b). Each Dollar Tranche Revolving Loan shall be a Term SOFR Revolving Loan denominated in Dollars or an ABR Revolving Loan denominated in Dollars.

31 “Dollars” or “$” refers to lawful money of the United States of America. “Domestic Subsidiary” means a Subsidiary organized under the laws of a jurisdiction located in the United States of America. “Drug Acquisition” means any acquisition (including any license or any acquisition of any license) solely or primarily of all or any portion of the rights in respect of one or more drugs or pharmaceutical products, whether in development or on market (including related Intellectual Property), but not of Equity Interests in any Person or any operating business unit. “ECP” means an “eligible contract participant” as defined in the Commodity Exchange Act and the regulations thereunder. “EEA Financial Institution” means (a) any credit institution or investment firm established in any EEA Member Country which is subject to the supervision of an EEA Resolution Authority, (b) any entity established in an EEA Member Country which is a parent of an institution described in clause (a) of this definition, or (c) any financial institution established in an EEA Member Country which is a Subsidiary of an institution described in clauses (a) or (b) of this definition and is subject to consolidated supervision with its parent. “EEA Member Country” means any of the member states of the European Union, Iceland, Liechtenstein, and Norway. “EEA Resolution Authority” means any public administrative authority or any person entrusted with public administrative authority of any EEA Member Country (including any delegee) having responsibility for the resolution of any EEA Financial Institution. “Effective Date” means April 23, 2024. “Effective Yield” means, as to any Loans of any Class, the effective yield on such Loans as reasonably determined by the Administrative Agent, taking into account (a) the applicable interest rate margins, (b) any interest rate floors or similar devices, (c) all recurring fees and other fees, including upfront or similar fees or original issue discount (in the case of clause (c), amortized over the shorter of (i) the life of such Loans and (ii) the four years following the date of incurrence thereof) payable generally to Lenders making such Loans, but excluding (x) any arrangement, commitment, structuring or other fees payable in connection therewith that are not generally shared with the Lenders thereunder and (y) any customary consent fees paid generally to consenting Lenders; provided that differences in the Effective Yield of Loans denominated in Dollars from loans denominated in other currencies shall be calculated by the Administrative Agent in good faith but ignoring differences due to the currency differences or underlying base rates employed (so long as reasonably equivalent in nature) (but giving effect to any differences in interest rate margins, spreads or upfront fees or floors as otherwise required above). “Electronic Signature” has the meaning assigned to such term in Section 9.06. “Eligible Transferee” has the meaning assigned to such term in Section 9.04(b)(i).

32 “Engagement Letter” means that certain Engagement Letter, dated as of March 31, 2024, by and among the Borrower, PLC and the Lead Arrangers. “Enterprise Transformative Event” means any merger, acquisition, Investment, dissolution, liquidation, consolidation or Disposition, in any such case by Parent, any Borrower or any Restricted Subsidiary that is either (a) not permitted by the terms of any Loan Document immediately prior to the consummation of such transaction or (b) if permitted by the terms of the Loan Documents immediately prior to the consummation of such transaction, would not provide Parent, the Borrower and the Restricted Subsidiaries with adequate flexibility under the Loan Documents for the continuation or expansion of their combined operations following such consummation, as reasonably determined by Parent acting in good faith. “Environmental Laws” means all laws, rules, regulations, codes, ordinances, or binding orders, decrees, judgments, injunctions, notices or agreements issued, promulgated or entered into by any Governmental Authority, relating to pollution or protection of the environment, including management or reclamation of natural resources, and the management, Release or threatened Release of any Hazardous Material or to occupational health and safety matters, as such occupational health and safety matters relate to exposure or handling of Hazardous Materials. “Environmental Liability” means any liability, contingent or otherwise (including any liability for damages, costs of environmental remediation, fines, penalties or indemnities), of Parent or any Restricted Subsidiary directly or indirectly resulting from or based upon (a) violation of any Environmental Law, (b) the generation, use, handling, transportation, storage, treatment or disposal of any Hazardous Materials, (c) exposure to any Hazardous Materials, (d) the Release or threatened Release of any Hazardous Materials into the environment or (e) any contract, agreement or other consensual arrangement pursuant to which liability is assumed or imposed with respect to any of the foregoing. “Equity Interests” means shares of capital stock, partnership interests, membership interests in a limited liability company, beneficial interests in a trust or other equity ownership interests in a Person, and any warrants, options or other rights entitling the holder thereof to purchase or acquire any of the foregoing; provided that “Equity Interests” shall not include Convertible Debt Securities or Permitted Convertible Debt Hedge Transactions. “Equivalent Amount” of any currency with respect to any amount of Dollars at any date shall mean the equivalent in such currency of such amount of Dollars, calculated on the basis of the Exchange Rate for such other currency at 11:00 a.m., London time, on the date on or as of which such amount is to be determined. “ERISA” means the Employee Retirement Income Security Act of 1974, as amended from time to time. “ERISA Affiliate” means (i) any trade or business (whether or not incorporated) that, together with Parent, is treated as a single employer under Section 414(b) or (c) of the Code or, solely for purposes of Section 302 of ERISA and Section 412 of the Code, is treated as a single employer under Section 414 of the Code and (ii) any entity (whether or not incorporated) that is under common control with Parent within the meaning of Section 4001(a)(14) of ERISA.

33 “ERISA Event” means (a) any “reportable event”, as defined in Section 4043 of ERISA or the regulations issued thereunder with respect to a Plan (other than an event for which the 30-day notice period is waived); (b) failure to satisfy the minimum funding standard under Section 412 of the Code or Section 302 of ERISA, whether or not waived with respect to a Plan; (c) the filing pursuant to Section 412(d) of the Code or Section 303(d) of ERISA of an application for a waiver of the minimum funding standard with respect to any Plan; (d) the incurrence by Parent or any of its ERISA Affiliates of any liability under Title IV of ERISA with respect to the termination of any Plan other than the PBGC premiums due but not delinquent under Section 4007 of ERISA; (e) a determination that any Plan is, or is expected to be considered an at-risk plan within the meaning of Section 430 of the Code or Section 303 of ERISA; (f) the receipt by Parent or any ERISA Affiliate from the PBGC or a plan administrator of any notice relating to an intention to terminate any Plan or Plans or to appoint a trustee to administer any Plan; (g) the incurrence by Parent or any of its ERISA Affiliates of any liability with respect to the withdrawal or partial withdrawal of Parent or any of its ERISA Affiliates from any Multiemployer Plan; (h) the receipt by Parent or any ERISA Affiliate of any notice, or the receipt by any Multiemployer Plan from Parent or any ERISA Affiliate of any notice, concerning the imposition upon Parent or any of its ERISA Affiliates of Withdrawal Liability or a determination that a Multiemployer Plan is, or is expected to be, insolvent, within the meaning of Title IV of ERISA; (i) the receipt by Parent or any ERISA Affiliate of any notice, or the receipt by any Multiemployer Plan from Parent or any ERISA Affiliate of any notice, that a Multiemployer Plan is in endangered or critical status under Section 432 of the Code or Section 305 of ERISA; (j) the occurrence of any event which would trigger the full or partial wind up of any occupational pension scheme (within the meaning of section 2 of the Irish Pension Act 1990 (as amended) (the “Pensions Act”)) sponsored by Parent or its Subsidiaries or in which Parent or its Subsidiaries participates (an “Irish Pension Scheme”); (k) the failure by an Irish Pension Scheme to meet the minimum funding standard prescribed by Part IV of the Pensions Act; (l) where any funding proposal (within the meaning of section 49 of the Pensions Act) which has been put in place to address a deficit within an Irish Pension Scheme goes off-track (within the meaning of the Irish Pensions Authority’s prescribed guidance under section 49 of the Pensions Act); (m) where a prosecution for an offence is brought under section 3 of the Pensions Act against the sponsoring employer, a participating employer, trustees, administrator or other agent concerning an Irish Pension Scheme or where the Irish Pensions Authority brings proceedings before the Irish High Court concerning an Irish Pension Scheme under Part IX of the Pensions Act; (n) where the Irish Pensions Authority commences an investigation of or appoints an authorised person in respect of an Irish Pension Scheme in accordance with its powers under Part II of the Pensions Act; (o) where the Irish Pensions Authority serves an advisory notice on the trustees of an Irish Pension Scheme or requires the trustees of an Irish Pension Scheme to provide it with an external report in accordance with its powers under Part IIA of the Pensions Act; (p) where the Irish Financial Services and Pensions Ombudsman either makes a determination against or brings enforcement proceedings against the sponsoring employer, a participating employer, trustees, administrator or other agent concerning an Irish Pension Scheme; or (q) where any arbitration proceedings or proceedings before the Irish High Court are initiated relating to a dispute between the sponsoring employer and/or any participating employer and the trustees and/or members of an Irish Pension Scheme. “Escrow Debt” means Indebtedness incurred in connection with any transaction permitted hereunder for so long as proceeds thereof have been deposited into an escrow account

34 on customary terms to secure such Indebtedness pending the application of such proceeds to finance such transaction. “EU” means the European Union. “EU Bail-In Legislation Schedule” means the EU Bail-In Legislation Schedule published by the Loan Market Association (or any successor person), as in effect from time to time. “euro” means the single currency of the participating member states of the EU. “Event of Default” has the meaning assigned to such term in Section 7.01. “Excess Cash Flow” means, for any period, the remainder (if positive) of (a) the sum of, without duplication, (i) Consolidated Net Income for such period, (ii) the decrease, if any, in Consolidated Working Capital from the first day to the last day of such period (other than any such decreases arising from Permitted Acquisitions or Dispositions of any person by Parent or any of its Restricted Subsidiaries), (iii) the amount of expenses for Taxes paid or accrued to the extent same reduced Consolidated Net Income for such period, (iv) any expense related to Swap Agreements which decreased Consolidated Net Income for such period, (v) non-cash charges, losses or expenses deducted in calculating Consolidated Net Income such period, (vi) cash charges or expenses reducing Consolidated Net Income during such period in respect of expenditures for which a deduction from Excess Cash Flow was made in a prior period and (vii) items not included in Excess Cash Flow in a previous period as items that were committed to be spent in a future period which are not actually spent during the subsequent period, minus (b) the sum of, without duplication, (i) the aggregate amount of all Capital Expenditures, Capitalized Software Expenditures or acquisitions of Intellectual Property in cash made by Parent and its Restricted Subsidiaries not expensed during such period (except to the extent made with proceeds of long- term Indebtedness (other than any Indebtedness under any revolving credit facilities)), (ii) the aggregate amount of permanent principal repayments of Indebtedness of Parent and its Restricted Subsidiaries (including (x) the principal component of payments made on Capital Lease Obligations of Parent and its Restricted Subsidiaries during such period and (y) the aggregate principal amount of any mandatory prepayment of Term Loans pursuant to Section 2.11(c)(1), to the extent required due to the circumstances described in clauses (a) or (b) of the definition of “Prepayment Event” that resulted in an increase to Consolidated Net Income and not in excess of such increase), but excluding (A) all repayments and prepayments of Term Loans (other than payments required pursuant to Section 2.10 and mandatory prepayments described in clause (y) of the foregoing parenthetical), (B) all repayments and prepayments of Revolving Loans, Swingline Loans or loans under any Incremental Revolving Commitment or other revolving credit or similar facility unless such prepayments are accompanied by a corresponding permanent reduction of the related revolving or similar commitments and (C) any such repayments and prepayments to the extent made with proceeds of long-term Indebtedness (other than any Indebtedness under any revolving credit facilities), (iii) the increase, if any, in Consolidated Working Capital from the first day to the last day of such period (other than any such increase in Consolidated Working Capital arising from a Permitted Acquisition or Disposition of any person by Parent and/or any of its Restricted Subsidiaries), (iv) to the extent included or not deducted in calculating Consolidated Net Income, the aggregate amount of all cash payments made in respect of all Permitted

35 Acquisitions and other Investments (including earn-out obligations, Milestone Payments, working capital or similar adjustments paid in connection therewith and in connection with acquisitions or Investments consummated prior to the Effective Date) permitted by Section 6.04 consummated (or committed or budgeted to be consummated in the next succeeding period) by Parent and its Restricted Subsidiaries (other than intercompany Investments among Parent and its Restricted Subsidiaries or Investments in cash or Permitted Investments) during such period or prior to the applicable Excess Cash Payment Date, except to the extent financed with long-term Indebtedness (other than any Indebtedness under any revolving credit facilities), (v) to the extent not expensed or not deducted in calculating Consolidated Net Income, the aggregate amount of any premium, make-whole or penalty payments actually paid, except to the extent financed with long-term Indebtedness (other than any Indebtedness under any revolving credit facilities) during such period that are required to be made in connection with any prepayment of Indebtedness, (vi) cash payments by Parent and its Restricted Subsidiaries during such period in respect of long-term liabilities of Parent and its Restricted Subsidiaries other than Indebtedness, except to the extent financed with long-term Indebtedness (other than any Indebtedness under any revolving credit facilities), (vii) cash expenditures for costs and expenses in connection with acquisitions or Dispositions and the issuance of Equity Interests or Indebtedness or amendments or modifications to any Indebtedness to the extent not deducted in arriving at such Consolidated Net Income (in each case, including any such transactions consummated prior to the Effective Date or transactions undertaken but not completed), except to the extent financed with long-term Indebtedness (other than any Indebtedness under any revolving credit facilities), (viii) the aggregate amount of expenditures actually made by Parent and its Restricted Subsidiaries in cash during such period (including expenditures for the payment of financing fees) to the extent that such expenditures are not expensed during such period, (ix) any payment of cash to be amortized or expensed over a future period and recorded as a long-term asset (so long as any related amortization or expense in a future period shall be added back in the calculation of Excess Cash Flow in such future period), (x) reimbursable or insured expenses incurred during such fiscal year to the extent that reimbursement has not yet been received (in which case the respective reimbursement shall increase Excess Cash Flow in the period in which it is received), (xi) the aggregate amount of Taxes actually paid or payable by Parent and its Restricted Subsidiaries in cash during such period, (xii) to the extent not expensed or not deducted in calculating Consolidated Net Income, the aggregate amount of any permitted Restricted Payments actually made in cash during such period by Parent and by any Restricted Subsidiary to any Person other than Parent or the Restricted Subsidiaries, in each case, pursuant to Section 6.07, except to the extent financed with long term Indebtedness (other than any Indebtedness under any revolving credit facilities), (xiii) cash expenditures made in respect of Swap Agreements during such period, (xiv) the aggregate net amount of non-cash gains, non-cash income and non-cash credits included in calculating Consolidated Net Income during such period and cash losses, charges, expenses, costs and fees excluded by virtue of the definition of “Consolidated Net Income”, (xv) without duplication of amounts deducted from Excess Cash Flow in other periods, and at the option of Parent, (1) the aggregate consideration required to be paid in cash by Parent or any of its Restricted Subsidiaries pursuant to binding contracts (the “Contract Consideration”) entered into prior to or during such period and (2) any planned cash expenditures by Parent or any of its Restricted Subsidiaries (the “Planned Expenditures”), in the case of each of the preceding clauses (1) and (2), relating to Permitted Acquisitions or other Investments, Capital Expenditures, Restricted Payments, acquisitions of Intellectual Property and any scheduled payment of Indebtedness that was

36 permitted by the terms of this Agreement to be incurred and paid, in each case, to be consummated or made, as applicable, during the period of four consecutive fiscal quarters of Parent following the end of such period (except to the extent financed with the proceeds of long-term Indebtedness (other than revolving credit facilities)); provided that to the extent that the aggregate amount of internally generated cash flow actually utilized to finance such Permitted Acquisitions or other Investments, Capital Expenditures, Restricted Payments, acquisitions of Intellectual Property or permitted scheduled payments of Indebtedness that were permitted by the terms of this Agreement to be incurred and paid during such following period of four consecutive fiscal quarters is less than the Contract Consideration and Planned Expenditures, the amount of such shortfall shall be added to the calculation of Excess Cash Flow, at the end of such period of four consecutive fiscal quarters, (xvi) any fees, expenses or charges incurred during such period, in connection with any acquisition, Investment, Disposition, incurrence or repayment of Indebtedness, issuance of Equity Interests, refinancing transaction or amendment or modification of any debt instrument and including, in each case, any such transaction consummated prior to the Effective Date and any such transaction undertaken but not completed, and any charges or non-recurring merger costs incurred during such period as a result of any such transaction, in each case whether or not successful, and (xvii) at the option of Parent, any amounts in respect of Capital Expenditures, Investments, Permitted Acquisitions, Indebtedness and Restricted Payments which could have been deducted if made in such period, but which are made after the end of such period and prior to the Excess Cash Payment Date (which amounts, if so deducted in accordance with this clause (xvii), shall not affect the calculation of Excess Cash Flow in any future period). Notwithstanding anything in the definition of any term used in the definition of Excess Cash Flow to the contrary, all components of Excess Cash Flow shall be computed for Parent and its Restricted Subsidiaries on a consolidated basis. “Excess Cash Payment Date” means the date occurring five (5) Business Days after the date on which Parent’s annual audited financial statements are required to be delivered pursuant to Section 5.01(a) (commencing with the fiscal year of Parent ended December 31, 2025). “Exchange Rate” means, on any day, with respect to any Foreign Currency, the rate at which such Foreign Currency may be exchanged into Dollars, displayed by ICE Data Services as the “ask price” at approximately 11:00 a.m., Local Time, on such date for such Foreign Currency. In the event that such rate does not appear on any Reuters World Currency Page, the Exchange Rate with respect to such Foreign Currency shall be determined by reference to such other publicly available service for displaying exchange rates as may be reasonably selected by the Administrative Agent or, in the event no such service is selected, such Exchange Rate shall instead be calculated on the basis of the arithmetical mean of the buy and sell spot rates of exchange of the Administrative Agent for such Foreign Currency on the applicable market at 11:00 a.m., Local Time, on such date for the purchase of Dollars with such Foreign Currency, for delivery two Business Days later; provided that if at the time of any such determination, for any reason, no such spot rate is being quoted, the Administrative Agent, after consultation with Parent, may use any reasonable method it deems appropriate to determine such rate, and such determination shall be conclusive absent manifest error. “Excluded Accounts” (i) any Deposit Account of a Loan Party that is used by such Loan Party solely as a payroll account for the employees of such Loan Party, (ii) Deposit Accounts consisting of withheld income taxes and federal, state or local employment taxes in such amounts as are required in the reasonable judgment of the Loan Party in the ordinary course of business to

37 be paid to the Internal Revenue Service or state or local government agencies with respect to current or former employees of any of the Loan Parties, (iii) Deposit Accounts consisting of amounts required to be paid over to an employee benefit plan pursuant to DOL Reg. Sec. 2510.3- 102 on behalf of or for the benefit of employees of one or more Loan Parties, (iv) any Deposit Account the maximum daily balance of which does not exceed $1,000,000 individually, or in the aggregate, together with the maximum daily balance of all such other Deposit Accounts excluded pursuant to this clause (iv) at any time, $3,000,000 and (v) zero balance accounts so long as the balance in such account is zero at the end of each Business Day. “Excluded Assets” means (a) motor vehicles, aircraft and other assets subject to certificates of title, (b) leasehold interests in real property (except leasehold interests of the kind described in Section (E)1(y) of the Agreed Security Principles), (c) any fee-owned real property with an appraised value of less than $20,000,000 (it being understood there shall be no requirement to obtain any landlord or other third party waivers, estoppels or collateral access letters) or any fixtures affixed to any real property to the extent (A) such real property does not constitute Collateral and (B) a security interest in such fixtures may not be perfected by a UCC or similar financing statement in the jurisdiction of organization of the applicable Loan Party, (d) any assets to the extent a security interest in such assets would result in material adverse Tax consequences as reasonably determined by Parent in consultation with the Administrative Agent; (e) any lease, license, contract, property right or agreement, or any property subject to a purchase money security interest, capital lease obligation or similar arrangement, in each case to the extent that a grant of a security interest therein would violate or invalidate such lease, license or agreement or purchase money or similar arrangement or create a right of termination in favor of any other party thereto (other than Parent or any of its Subsidiaries) or otherwise require consent thereunder (other than from Parent or a Restricted Subsidiary) after giving effect to the applicable anti-assignment provisions of the UCC or other applicable law, other than proceeds and receivables thereof, the assignment of which is expressly deemed effective under the UCC or other applicable law, notwithstanding such prohibition; (f) any Excluded Equity Interests, (g) any assets to the extent expressly excluded pursuant to the Agreed Security Principles, (h) any Margin Stock, (i) any applications for trademarks or service marks filed in the United States Patent and Trademark Office or any successor thereto (the “PTO”) on the basis of the applicant’s intent-to-use such trademark or service mark, prior to the filing of an amendment with the PTO under 15 U.S.C. §1051(c) that brings the application into conformity with 15 U.S.C. §1051(a) or the filing of a verified statement of use with the PTO under 15 U.S.C. §1051(d) that has been examined and accepted by the PTO, to the extent, if any, that and solely during the period, if any, in which, the grant of a security interest therein would impair the validity or enforceability of any trademark or service mark registration that issues from such intent-to-use application under applicable federal law, (j) any Excluded Accounts, (k) commercial tort claims that, in the reasonable determination of Parent, are not expected to result in a judgment in excess of $1,000,000, (l) letter of credit rights (other than to the extent consisting of supporting obligations that can be perfected solely by the filing of a UCC or similar financing statement (it being understood that no actions shall be required to perfect a security interest in letter of credit rights other than filing of a UCC or similar financing statement)), (m) any governmental licenses or state or local franchises, charters and authorizations, to the extent a security interest in any such license, franchise, charter or authorization is prohibited or restricted thereby (excluding any prohibition or restriction that is ineffective under the UCC or other applicable law), (n) assets to the extent the pledge thereof or grant of security interests therein (x) is prohibited or restricted by applicable law, rule or regulation, (y) would cause the destruction,

38 invalidation or abandonment of such asset under applicable law, rule or regulation, or (z) requires any consent, approval, license or other authorization of any third party or Governmental Authority (excluding any prohibition or restriction that is ineffective under the UCC or other applicable law), (o) assets where the cost of obtaining a security interest therein is excessive in relation to the practical benefit to the Lenders afforded thereby as reasonably determined between Parent and the Administrative Agent, (p) acquired property (including property acquired through acquisition or merger of another entity) if at the time of such acquisition the granting of a security interest therein or the pledge thereof is prohibited by any contract or other agreement (in each case, not created in contemplation thereof) to the extent and for so long as such contract or other agreement prohibits such security interest or pledge (excluding any prohibition or restriction that is ineffective under the UCC or other applicable law) and (q) Permitted Receivables Facility Assets. “Excluded Equity Interests” means (a) any portion of the issued and outstanding Equity Interests of a Pledge Subsidiary not required to be subject to a perfected lien in favor of the Administrative Agent in accordance with Section 5.09(b), (b) Equity Interests in non-wholly- owned Subsidiaries or in entities where a Loan Party holds 50% or less of the outstanding Equity Interests of such entity, to the extent a pledge of such Equity Interests is prohibited by the organizational documents, or agreements with the other equity holders, of such entity, (c) Equity Interests in any Excluded Subsidiary (other than an Excluded Subsidiary that is a Guarantor and except to the extent a security interest therein can be perfected by filing a Uniform Commercial Code financing statement (or similar filing statements)), (d) other than any Subsidiary Guarantor that is an Irish Subsidiary or Luxembourg Subsidiary, Equity Interests of a Controlled Foreign Corporation in excess of 65% of the total combined voting power of all classes of Equity Interests of such Controlled Foreign Corporation entitled to vote, (e) any other Equity Interests (or any portion thereof) to the extent expressly excluded pursuant to the Agreed Security Principles, and (f) to the extent reasonably agreed to by the Administrative Agent, any Equity Interests or membership interests in an unlimited liability company. “Excluded Subsidiary” means: (a) any Subsidiary that is not a wholly-owned Subsidiary of Parent, (b) any Subsidiary, including any regulated entity that is subject to net worth or net capital or similar capital and surplus restrictions, that is prohibited or restricted by applicable law, accounting policies or by contractual obligation existing on the Effective Date (or, with respect to any Subsidiary acquired by Parent or a Restricted Subsidiary after the Effective Date (and so long as such contractual obligation was not incurred in contemplation of such acquisition), on the date such Subsidiary is so acquired) from providing a Guaranty, or if such Guaranty would require governmental (including regulatory) or third party consent, approval, license or authorization (except to the extent that such consent, approval, license or authorization has been obtained), (c) any Receivables Entity, (d) any special purpose vehicle (or similar entity), (e) any Captive Insurance Subsidiary,

39 (f) any not for profit Subsidiary, (g) any Immaterial Subsidiary, (h) any Unrestricted Subsidiary, (i) any Foreign Subsidiary or Controlled Foreign Corporation (other than any Irish Subsidiary or Luxembourg Subsidiary that is a Subsidiary Guarantor), (j) any Restricted Subsidiary acquired with Indebtedness assumed pursuant to Section 6.01(f) to the extent such Restricted Subsidiary would be prohibited from providing a Guaranty or consent would be required (that has not been obtained), pursuant to the terms of such Indebtedness, (k) any Subsidiary with respect to which the Guaranty would result in material adverse Tax consequences to the Borrower or one of its Subsidiaries as reasonably determined by the Parent, and (l) any other Subsidiary with respect to which the Parent and the Administrative Agent reasonably determine that the burden or cost of providing the Guaranty shall outweigh the benefits to be obtained by the Lenders therefrom. Notwithstanding anything to the contrary herein, for the avoidance of doubt, in no event shall (i) any Subsidiary that owns any Material IP on the Effective Date be an Excluded Subsidiary and (ii) any Foreign Subsidiary that is party to the Subsidiary Guaranty on the Effective Date be an Excluded Subsidiary. “Excluded Swap Obligation” means, with respect to any Guarantor, any Swap Obligation if, and to the extent that, all or a portion of the Guarantee of such Guarantor of, or the grant by such Guarantor of a security interest to secure, such Swap Obligation (or any Guarantee thereof) is or becomes illegal under the Commodity Exchange Act or any rule, regulation or order of the Commodity Futures Trading Commission (or the application or official interpretation of any thereof) by virtue of such Guarantor’s failure for any reason to constitute an ECP at the time the Guarantee of such Guarantor or the grant of such security interest becomes effective with respect to such Swap Obligation. If a Swap Obligation arises under a master agreement governing more than one swap, such exclusion shall apply only to the portion of such Swap Obligation that is attributable to swaps for which such Guarantee or security interest is or becomes illegal. For purposes of this definition, “Swap Obligation” means, with respect to any Guarantor, any obligation of Parent or any Restricted Subsidiary to pay or perform under any agreement, contract or transaction that constitutes a “swap” within the meaning of section 1a(47) of the Commodity Exchange Act. “Excluded Taxes” means any of the following Taxes imposed on or with respect to a Recipient or required to be withheld or deducted from a payment to a Recipient, (a) Taxes imposed on or measured by net income (however denominated), franchise Taxes, and branch profits Taxes, in each case, (i) imposed as a result of such Recipient being organized under the laws of, or having its principal office or, in the case of any Lender, its applicable lending office located in, the jurisdiction imposing such Tax (or any political subdivision thereof) or (ii) that are

40 Other Connection Taxes, (b) in the case of a Lender, any U.S. federal withholding tax that is imposed on amounts payable to or for the account of such Lender with respect to an applicable interest in a Loan or Commitment pursuant to a law in effect on the date on which (i) such Lender acquires such interest in the Loan or Commitment (other than pursuant to an assignment request by the Borrower under Section 2.19(b)) or (ii) such Lender changes its lending office, except in each case to the extent that, pursuant to Section 2.17, amounts with respect to such Taxes were payable either to such Lender’s assignor immediately before such Lender became a party hereto or to such Lender immediately before it changed its lending office, (c) any Taxes attributable to such Recipient’s failure to comply with Section 2.17(e), Section 2.17(f), or Section 2.17(g), (d) any withholding Taxes imposed under FATCA and (e) any Taxes imposed in relation to the Luxembourg law dated 23 December 2005 introducing a final withholding tax on interest payable to Luxembourg resident individual beneficial owners of the payment, as amended from time to time. “Exclusive License” means any license of material intellectual property owned by the Parent or any Restricted Subsidiary with a term greater than five (5) years and made on an exclusive basis (other than exclusivity of immaterial scope or in respect of an immaterial jurisdiction). “Exclusively License” shall have the correlative meaning. “Existing Letters of Credit” has the meaning assigned to such term in Section 2.06(a). “Existing Initial Term Lender” has the meaning set forth in the First Amendment. “Existing Initial Term Loan” has the meaning set forth in the First Amendment. “Exit Transactions” means (a) the United States Bankruptcy Court for the Southern District of New York’s issuance of an order confirming the Reorganization Plan (the “Confirmation Order”) at Docket No. 3960 which Confirmation Order shall not be stayed or vacated, and which shall be otherwise in full force and effect without modification in any way materially adverse to the Lenders without the consent of the Administrative Agent, (b) the completion of the Exit Financing Approval Process (as defined in the Confirmation Order) in accordance with the terms of the Confirmation Order pursuant to the Exit Financing Term Sheet Documents (as defined in the Confirmation Order), which Exit Financing Term Sheet Documents shall be in form and substance reasonably acceptable to the Administrative Agent, and (c) the satisfaction or waiver of all conditions precedent to the effectiveness of the Reorganization Plan (to the extent such waiver is not materially adverse to the Lenders) and occurrence of the Effective Date (as defined in the Reorganization Plan) under the Reorganization Plan (which may occur substantially simultaneously with the Effective Date (as defined in this Agreement)) which Reorganization Plan is in full force and effect, without modification in any way materially adverse to the Lenders without the consent of the Administrative Agent. “Extended Commitments” means the Extended Term Loan Commitment and the Extended Revolving Commitment. “Extended Loans” means the Extended Term Loans and the Extended Revolving Loans.

41 “Extended Revolving Commitment” shall have the meaning given to such term in Section 2.23(a)(ii). “Extended Revolving Loans” means Revolving Loans made by one or more Lenders to the Borrower pursuant to Section 2.23. “Extended Term Loan Commitment” means the commitment of any Lender, established pursuant to Section 2.23, to make Extended Term Loans to a Borrower. “Extended Term Loans” shall have the meaning given to such term in Section 2.23(a)(iv). “Extending Revolving Lender” shall have the meaning given to such term in Section 2.23(a)(ii). “Extending Term Lender” shall have the meaning given to such term in Section 2.23(a)(iv). “Extension” shall have the meaning given to such term in Section 2.23(a). “Extension Offer” shall have the meaning given to such term in Section 2.23(a). “FATCA” means Sections 1471 through 1474 of the Code, as of the Effective Date (or any amended or successor version that is substantively comparable and not materially more onerous to comply with), any current or future regulations or official interpretations thereof and any agreements entered into pursuant to Section 1471(b)(1) of the Code and any fiscal or regulatory legislation, rules or practices adopted pursuant to any intergovernmental agreement, treaty or convention among Governmental Authorities and implementing such Sections of the Code. “Federal Funds Effective Rate” means, for any day, the greater of (i) 0.00% or (ii) the weighted average (rounded upwards, if necessary, to the next 1/100 of 1%) of the rates on overnight Federal funds transactions with members of the Federal Reserve System arranged by Federal funds brokers, as published on the next succeeding Business Day by the Federal Reserve Bank of New York, or, if such rate is not so published for any day that is a Business Day, the average (rounded upwards, if necessary, to the next 1/100 of 1%) of the quotations for such day for such transactions received by the Administrative Agent from three Federal funds brokers of recognized standing selected by it. “Financial Covenant” means the covenant in Section 6.11. “Financial Officer” means the chief financial officer, principal accounting officer, treasurer or controller of Parent. “Financials” means the annual or quarterly financial statements, and accompanying certificates and other documents, of Parent and its Subsidiaries required to be delivered pursuant to Section 5.01(a) or 5.01(b).

42 “First Amendment” means that certain First Amendment to this Agreement, dated as of October 29, 2024. “First Amendment Effective Date” has the meaning assigned to such term in the First Amendment. “First Lien Net Leverage Ratio” means the ratio of (a) Consolidated First Lien Secured Debt minus the aggregate amount of cash and Permitted Investments of Parent and its Restricted Subsidiaries on such date that (x) would not appear as “restricted” on a consolidated balance sheet of Parent and its Restricted Subsidiaries or (y) are restricted or secured in favor of the Indebtedness incurred under this Agreement or other Indebtedness secured by a pari passu or junior Lien on the Collateral as permitted under this Agreement to (b) Consolidated EBITDA of Parent and its Restricted Subsidiaries for such Reference Period, in each case on a pro forma basis with such pro forma adjustments as are appropriate and consistent with Section 1.04. “Flood Insurance Laws” means, collectively, (i) the National Flood Insurance Act of 1968 as now or hereafter in effect or any successor statute thereto, (ii) the Flood Disaster Protection Act of 1973 as now or hereafter in effect or any successor statute thereto, (iii) the National Flood Insurance Reform Act of 1994 as now or hereafter in effect or any successor statute thereto and (iv) the Flood Insurance Reform Act of 2004 and the Biggert –Waters Flood Insurance Reform Act of 2012, as now or hereafter in effect or any successor statute thereto, in each case, together with all statutory and regulatory provisions consolidating, amending, replacing, supplementing, implementing or interpreting any of the foregoing, as amended or modified from time to time. “Floor” means 0.00% (in the case of Revolving Loans) and 0.50% (in the case of Initial Term Loans and 2024 Refinancing Term Loans). “Foreign Currencies” means Agreed Currencies other than Dollars. “Foreign Currency LC Exposure” means, at any time, the sum of (a) the Dollar Amount of the aggregate undrawn and unexpired amount of all outstanding Foreign Currency Letters of Credit at such time plus (b) the aggregate principal Dollar Amount of all LC Disbursements in respect of Foreign Currency Letters of Credit that have not yet been reimbursed at such time. “Foreign Currency Letter of Credit” means a Multicurrency Tranche Letter of Credit denominated in a Foreign Currency. “Foreign Currency Payment Office” of the Administrative Agent means, for each Foreign Currency, the office, branch, affiliate or correspondent bank of the Administrative Agent for such currency as specified from time to time by the Administrative Agent to Parent and each Lender. “Foreign Lender” means any Lender that is not a “United States person” as defined in Section 7701(a)(30) of the Code.

43 “Foreign Loan Party” means each Foreign Subsidiary that is a Borrower or a Subsidiary Guarantor. “Foreign Subsidiary” means any Subsidiary which is not a Domestic Subsidiary. “GAAP” means generally accepted accounting principles in the United States of America. “Governmental Authority” means the federal and state governments of the United States of America, any other nation or any political subdivision thereof, whether state or local, and any agency, authority, instrumentality, regulatory body, agency, tribunal, court, central bank or other entity exercising executive, legislative, judicial, taxing, regulatory or administrative powers or functions of or pertaining to government (including any supra-national bodies such as the European Union or the European Central Bank). “Granting Lender” has the meaning assigned to such term in Section 9.04(f). “Guarantee” of or by any Person (the “guarantor”) means any obligation, contingent or otherwise, of the guarantor guaranteeing or having the economic effect of guaranteeing any Indebtedness or other obligation of any other Person (the “primary obligor”) in any manner, whether directly or indirectly, and including any obligation of the guarantor, direct or indirect, (a) to purchase or pay (or advance or supply funds for the purchase or payment of) such Indebtedness or other obligation or to purchase (or to advance or supply funds for the purchase of) any security for the payment thereof, (b) to purchase or lease property, securities or services for the purpose of assuring the owner of such Indebtedness or other obligation of the payment thereof, (c) to maintain working capital, equity capital or any other financial statement condition or liquidity of the primary obligor so as to enable the primary obligor to pay such Indebtedness or other obligation or (d) as an account party in respect of any letter of credit or letter of guaranty issued to support such Indebtedness or obligation; provided that the term Guarantee shall not include endorsements for collection or deposit in the ordinary course of business. The amount of any Guarantee shall be deemed to be an amount equal to the lesser of (a) the stated or determinable amount of the primary payment obligation in respect of which such Guarantee is made and (b) the maximum amount for which the guaranteeing Person may be liable pursuant to the terms of the instrument embodying such Guarantee, unless such primary payment obligation and the maximum amount for which such guaranteeing Person may be liable are not stated or determinable, in which case the amount of the Guarantee shall be such guaranteeing Person’s maximum reasonably possible liability in respect thereof as reasonably determined by Parent in good faith. “Guarantor” means Parent and the Subsidiary Guarantors. “Guaranty” means the Subsidiary Guaranty and the Guarantee set forth in Article X. “Hazardous Materials” means all explosive or radioactive substances or wastes and all hazardous or toxic substances, wastes or other pollutants, including petroleum or petroleum distillates, asbestos or asbestos containing materials, polychlorinated biphenyls, radon gas, infectious or medical wastes and all other substances or wastes of similar nature regulated pursuant to any Environmental Law.

44 “Holding Company” means, with respect to any Lender, any Person as to which such Lender is, directly or indirectly, a subsidiary. “Immaterial Asset Sale” means any Disposition of property or series of related Dispositions of in respect of which the fair market value of such property and the Disposition Consideration payable to Parent or any of its Restricted Subsidiaries is equal to or less than $20,000,000 for any single Disposition or $40,000,000 in the aggregate in any fiscal year of Parent. “Immaterial Subsidiary” means any Restricted Subsidiary that is not a Material Subsidiary. “Incremental Amendment” means an Incremental Amendment among the applicable Borrower, the Administrative Agent and one or more Incremental Term Lenders and/or Incremental Revolving Lenders entered into pursuant to Section 2.20. “Incremental Amount” means, at any time after the Effective Date, an amount not to exceed (a) in respect of (i) Incremental Revolving Facilities, $100,000,000 and (ii) Incremental Term Facilities and Incremental Equivalent Debt, the greater of (x) $760,000,000 and (y) 100.0% of Consolidated EBITDA as of the end of the Reference Period plus (b)(i) in the case of Indebtedness secured on a pari passu basis with the Obligations, the First Lien Net Leverage Ratio, at the time of incurrence of such Incremental Amount (subject to Section 1.04) and after giving effect thereto on a pro forma basis in accordance with Section 1.04, is less than or equal to 3.50 to 1.00 (assuming for purposes of such calculation that any Incremental Revolving Commitments being incurred at the time of such calculation are fully drawn and without netting cash proceeds of any Incremental Loans or Incremental Equivalent Debt), (ii) in the case of Incremental Term Facilities and Incremental Equivalent Debt secured on a junior basis to the Obligations, the Secured Net Leverage Ratio, at the time of incurrence of such Incremental Amount (subject to Section 1.04) and after giving effect thereto on a pro forma basis in accordance with Section 1.04, is less than or equal to 5.00 to 1.00, or (iii) in the case of unsecured Incremental Term Facilities and Incremental Equivalent Debt, either (x) the Total Net Leverage Ratio, at the time of incurrence of such Incremental Amount (subject to Section 1.04) and after giving effect thereto on a pro forma basis in accordance with Section 1.04, is less than or equal to 6.00 to 1.00 or (y) the Consolidated Interest Coverage Ratio, at the time of incurrence of such Incremental Amount (subject to Section 1.04) and after giving effect thereto on a pro forma basis in accordance with Section 1.04, is more than or equal to 2.00 to 1.00, in each case, an unlimited amount (this clause (b), the “Incremental Ratio Basket”); provided that, if the ratios set forth in clause (b) is satisfied on such date on a pro forma basis, any such Indebtedness may, at the sole discretion of the applicable Borrower, be incurred under clause (b) regardless of whether there is capacity to incur such Indebtedness under clause (a). “Incremental Commitments” means the Incremental Term Loan Commitment and the Incremental Revolving Commitment. “Incremental Equivalent Debt” is defined in Section 6.01(w). “Incremental Loans” means the Incremental Term Loans and the Incremental Revolving Loans.

45 “Incremental Revolving Commitment” means any increase to an existing Class of Revolving Commitments provided pursuant to Section 2.20. “Incremental Revolving Lender” means a Lender with a Revolving Commitment or an outstanding Revolving Loan as a result of an Incremental Revolving Commitment. “Incremental Revolving Loans” means additional Revolving Loans made by one or more Lenders to the Borrower pursuant to Section 2.20. “Incremental Term Lender” means a Lender with an Incremental Term Loan Commitment or an outstanding Incremental Term Loan. “Incremental Term Loan Commitment” means the commitment of any Lender, established pursuant to Section 2.20, to make Incremental Term Loans to the applicable Borrower. “Incremental Term Loans” means Term Loans made by one or more Lenders to the applicable Borrower, pursuant to Section 2.20. Incremental Term Loans may be made in the form of additional Term Loans or, to the extent permitted by Section 2.20 and provided for in the relevant Incremental Amendment, Other Term Loans. “Indebtedness” of any Person means, without duplication, (a) all obligations of such Person for borrowed money, (b) all obligations of such Person evidenced by bonds, debentures, notes or similar instruments, (c) all obligations of such Person under conditional sale or other title retention agreements relating to property acquired by such Person (excluding trade accounts payable incurred in the ordinary course of business), (d) all obligations of such Person in respect of the deferred purchase price of property or services (including payments or other arrangements representing acquisition consideration, in each case entered into in connection with an acquisition, but excluding (i) accounts payable not more than 60 days overdue incurred in the ordinary course of business, (ii) deferred compensation and (iii) any purchase price adjustment, royalty, earnout, contingent payment or deferred payment of a similar nature incurred in connection with an acquisition), (e) all Capital Lease Obligations and synthetic lease obligations of such Person, (f) all obligations, contingent or otherwise, of such Person as an account party in respect of letters of credit and letters of guaranty, (g) all obligations, contingent or otherwise, of such Person in respect of bankers’ acceptances, (h) all Indebtedness of others secured by (or for which the holder of such Indebtedness has an existing right, contingent or otherwise, to be secured by) any Lien on property owned or acquired by such Person, whether or not the Indebtedness secured thereby has been assumed; provided that, if such Person has not assumed or otherwise become liable in respect of such Indebtedness, such obligations shall be deemed to be in an amount equal to the lesser of (i) the amount of such Indebtedness and (ii) fair market value of such property at the time of determination (in Parent’s good faith estimate), (i) all Guarantees by such Person of Indebtedness of others, (j) all Attributable Receivables Indebtedness of such Person and (k) all obligations of such Person to purchase, redeem, retire, defease or otherwise make any payment in respect of any Disqualified Equity Interests; provided that, Indebtedness shall not include Escrow Debt. The Indebtedness of any Person shall include the Indebtedness of any other entity (including any partnership in which such Person is a general partner) to the extent such Person is liable therefor as a result of such Person’s ownership interest in or other relationship with such entity, except to the extent the terms of such Indebtedness provide that such Person is not liable therefor.

46 “Indemnified Taxes” means Taxes imposed on or with respect to any payments made by or on account of any obligation of the Borrower hereunder or under any other Loan Document other than (i) Excluded Taxes and (ii) Other Taxes. “Independent Assets or Operations” means, with respect to any parent entity of Parent, that such parent entity’s total assets, revenues or income from continuing operations before income taxes and cash flows from operating activities (excluding in each case amounts related to its investment in Parent and its Restricted Subsidiaries), determined in accordance with GAAP and as shown on the most recent balance sheet of such Person, is more than 5.0% of such Person’s corresponding consolidated amount. “Information Memorandum” means any confidential information memorandum or lender presentation relating to Parent and its Subsidiaries and the loans and commitments hereunder. “Initial Term Lender” means, as of any date of determination, each Lender that holds Initial Term Loan Commitments or Initial Term Loans. “Initial Term Loan Commitments” means, with respect to each Initial Term Lender, the commitment, if any, of such Initial Term Lender to make or continue, as the case may be, Initial Term Loans hereunder, as such commitment may be (a) reduced or terminated from time to time pursuant to Section 2.09 and (b) reduced or increased from time to time pursuant to assignments by or to such Lender pursuant to Section 9.04. The amount of each Initial Term Lender’s Initial Term Loan Commitment as of the Effective Date is set forth on Schedule 2.01, or in the Assignment and Assumption (or other documentation contemplated by this Agreement) pursuant to which such Initial Term Lender shall have assumed its Initial Term Loan Commitment, as applicable. The aggregate principal amount of the Initial Term Loan Commitments on the Effective Date is $1,500,000,000. “Initial Term Loans” means (i) the term loans made or continued, as the case may be, by the Initial Term Lenders to the Borrower Representative on the Effective Date pursuant to Section 2.01(a) and (ii) any Incremental Term Loans (which do not constitute Other Term Loans) made from time to time pursuant to Section 2.20 prior to the First Amendment Effective Date. Each Initial Term Loan shall be a Term SOFR Loan denominated in Dollars or an ABR Loan denominated in Dollars. “Inside Maturity Basket” means Indebtedness that has a maturity date that is earlier than the Latest Maturity Date with respect to the then-existing Term Loans; provided, however, (i) the maturity date of such Indebtedness may not be earlier than the date that is 91 days following the Latest Maturity Date applicable to the Revolving Commitments and (ii) the aggregate principal amount of such Indebtedness shall not exceed the greater of (x) $380,000,000 and (y) 50.0% of Consolidated EBITDA as of the end of the Reference Period at any time outstanding. “Insolvency or Liquidation Proceeding” means, with respect to any Person, (a) any voluntary or involuntary case or proceeding under any Debtor Relief Law with respect to any such Person, (b) any other voluntary or involuntary insolvency, reorganization or bankruptcy case or proceeding, or any receivership, liquidation, reorganization, examinership or other similar case or

47 proceeding or private or judicial foreclosure with respect to any such Person or with respect to all or any material portion of its assets, (c) any liquidation, dissolution, reorganization or winding up of any such Person whether voluntary or involuntary and whether or not involving insolvency or bankruptcy or (d) any assignment for the benefit of creditors or any other marshalling of all or any material part of the assets and liabilities of any such Person. In addition, in respect of any Luxembourg Guarantor, “Insolvency or Liquidation Proceeding” shall also mean a Luxembourg Insolvency Event. “Intellectual Property” has the meaning assigned to such term in the US Security Agreement. “Intercreditor Agreement” means the Intercreditor Agreement, dated as of the Effective Date, among the Collateral Agent, as representative for the Secured Parties, Computershare Trust Company, as collateral agent for the holders of the Senior Secured Notes, the Parent, and each of the other Loan Parties party thereto and agents from time to time parties thereto. “Interest Election Request” means a request by a Borrower to convert or continue a Borrowing in accordance with Section 2.08. “Interest Payment Date” means (a) with respect to any ABR Loan and any Canadian Prime Rate Loan (other than a Swingline Loan), the last Business Day of each March, June, September and December and the applicable Maturity Date, (b) with respect to any Term Benchmark Loan, the last day of the Interest Period applicable to the Borrowing of which such Loan is a part and, in the case of a Term Benchmark Borrowing with an Interest Period of more than three months’ duration, each day prior to the last day of such Interest Period that occurs at intervals of three months’ duration after the first day of such Interest Period and the applicable Maturity Date and (c) with respect to any Swingline Loan, the day that such Loan is required to be repaid and the Latest Maturity Date with respect to any Revolving Commitments. “Interest Period” means with respect to (x) any Term Benchmark Borrowing denominated in Dollars, the period commencing on the date of such Borrowing and ending on the numerically corresponding day in the calendar month that is one, three or six months thereafter (or, if acceptable to all Lenders, twelve months thereafter), as the applicable Borrower may elect and (y) any Term Benchmark Borrowing denominated in Canadian Dollars, the period commencing on the date of such Borrowing and ending on the numerically corresponding day in the calendar month that is one or three months thereafter (or some other period subject to the availability for the Benchmark applicable to the relevant Loan or Commitment for Canadian Dollars), as the applicable Borrower may elect; provided that (i) if any Interest Period would end on a day other than a Business Day, such Interest Period shall be extended to the next succeeding Business Day unless such next succeeding Business Day would fall in the next calendar month, in which case such Interest Period shall end on the next preceding Business Day and (ii) any Interest Period that commences on the last Business Day of a calendar month (or on a day for which there is no numerically corresponding day in the last calendar month of such Interest Period) shall end on the last Business Day of the last calendar month of such Interest Period. For purposes hereof, the date of a Borrowing initially shall be the date on which such Borrowing is made and thereafter shall be the effective date of the most recent conversion or continuation of such Borrowing.

48 “Intermediate Parent Entity” means any direct or indirect parent company of the Borrower Representative that is a Restricted Subsidiary of Parent. “Investment” has the meaning assigned to such term in Section 6.04. “Irish Debenture” means that certain Irish law debenture (including any and all supplements thereto), dated as of the Effective Date, among Parent and each Irish Loan Party party thereto and the Collateral Agent, for the benefit of the Secured Parties. “Irish Loan Party” means each Irish Subsidiary that is a Borrower or a Subsidiary Guarantor. “Irish Pension Scheme” has the meaning assigned to such term in the definition of “ERISA Event”. “Irish Security Documents” means the Irish Debenture, the Irish Share Charge and any other pledge or security agreement governed by the laws of Ireland entered into by any Loan Party (as required by this Agreement or any other Loan Document). “Irish Share Charge” means that certain Irish law share charge (including any and all supplements thereto) with respect to the shares in Operand Pharmaceuticals HoldCo I Limited, dated as of the Effective Date, between Endo US Holdings Luxembourg I S.a.r.l. and the Collateral Agent, for the benefit of the Secured Parties. “Irish Subsidiary” means any Restricted Subsidiary that is incorporated under the laws of Ireland. “ISDA Definitions” means the 2006 ISDA Definitions published by the International Swaps and Derivatives Association, Inc. or any successor thereto, as amended or supplemented from time to time, or any successor definitional booklet for interest rate derivatives published from time to time by the International Swaps and Derivatives Association, Inc. or such successor thereto. “Issuing Bank” means solely with respect to standby Letters of Credit, Goldman Sachs Bank USA and its successors in such capacity as provided in Section 2.06; provided that, solely with respect to the Existing Letters of Credit, each issuer thereof shall be deemed to be an Issuing Bank (and each reference in this Agreement to the “Issuing Bank” solely when made in respect of the Existing Letters of Credit, shall be deemed to refer to each issuer thereof). The Issuing Bank may, in its discretion, arrange for one or more Letters of Credit to be issued by Affiliates of the Issuing Bank, in which case the term “Issuing Bank” shall include any such Affiliate with respect to Letters of Credit issued by such Affiliate. “Japanese Yen” or “¥” means the lawful currency of Japan. “Latest Maturity Date” means, at any date of determination, the latest Maturity Date applicable to any Loan or Commitment hereunder at such time, including the latest maturity date of any Other Refinancing Term Loan, any Other Refinancing Term Commitment, any Other Refinancing Revolving Commitment, any Other Term Loan, any Extended Term Loan, any

49 Extended Commitment, any Incremental Term Loan or any Incremental Revolving Commitments, in each case as extended in accordance with this Agreement from time to time. “LC Collateral Account” has the meaning assigned to such term in Section 2.06(j). “LC Disbursement” means a payment made by the Issuing Bank pursuant to a Letter of Credit. “LC Exposure” means, at any time, the sum of (a) the aggregate undrawn Dollar Amount of all outstanding Letters of Credit at such time plus (b) the aggregate Dollar Amount of all LC Disbursements that have not yet been reimbursed by or on behalf of the Borrower at such time. The LC Exposure of any Multicurrency Tranche Lender at any time shall be its Multicurrency Tranche Percentage of the total Multicurrency Tranche LC Exposure at such time and the LC Exposure of any Dollar Tranche Lender at any time shall be its Dollar Tranche Percentage of the total Dollar Tranche LC Exposure at such time. “Lead Arrangers” means each of Goldman Sachs Bank USA, JPMorgan Chase Bank, N.A., Barclays Bank PLC, Deutsche Bank Securities Inc., Morgan Stanley Senior Funding, Inc., Banco Santander, S.A., New York Branch, TCBI Securities, Inc. (doing business as Texas Capital Securities) and Bank of America, N.A. “Lenders” means the Persons listed on Schedule 2.01 and any other Person that shall have become a Lender hereunder pursuant to Section 2.20, Section 2.25 or pursuant to an Assignment and Assumption, other than any such Person that ceases to be a party hereto pursuant to an Assignment and Assumption. Unless the context otherwise requires, the term “Lenders” includes the Swingline Lender. “Letter of Credit” means any Multicurrency Tranche Letter of Credit or Dollar Tranche Letter of Credit. “Lien” means, with respect to any asset, (a) any mortgage, deed of trust, lien, statutory lien, pledge, hypothecation, encumbrance, charge or security interest in, on or of such asset and (b) the interest of a vendor or a lessor under any conditional sale agreement, capital lease or title retention agreement (or any financing lease having substantially the same economic effect as any of the foregoing) relating to such asset. “Limited Condition Transactions” means (i) any Permitted Acquisition or other investment permitted hereunder by Parent or one or more of its Restricted Subsidiaries whose consummation is not conditioned on the availability of, or on obtaining, third-party financing and (2) any redemption, repurchase, defeasance, satisfaction and discharge or repayment of Indebtedness requiring irrevocable notice in advance of such redemption, repurchase, defeasance, satisfaction and discharge or repayment. “Loan Documents” means this Agreement, any promissory notes issued pursuant to Section 2.10(f) of this Agreement, any Letter of Credit applications, the Collateral Documents, the Subsidiary Guaranty, any Incremental Amendment, Extension Amendment or Refinancing Amendment, the Intercreditor Agreement and any other intercreditor agreements and subordination agreements, and all written notices and certificates executed and/or delivered to the

50 Administrative Agent pursuant to this Agreement. Any reference in the Agreement or any other Loan Document to a Loan Document shall include all appendices, exhibits or schedules thereto, and all amendments, restatements, supplements or other modifications thereto, and shall refer to the Agreement or such Loan Document as the same may be in effect at any and all times such reference becomes operative. “Loan Parties” means, collectively, Parent, the Borrower and the Subsidiary Guarantors. “Loans” means the loans made by the Lenders to the Borrower pursuant to this Agreement. “Local Time” means (i) New York City time in the case of a Loan, Borrowing or LC Disbursement denominated in Dollars and (ii) Toronto time in the case of a Loan, Borrowing or LC Disbursement denominated in Canadian Dollars. “Luxembourg” means the Grand Duchy of Luxembourg. “Luxembourg Companies Register” means the Luxembourg Register of Commerce and Companies (Registre de Commerce et des Sociétés, Luxembourg). “Luxembourg Guarantor” means any Subsidiary Guarantor incorporated and existing under the Duchy of Luxembourg, or whose registered office or place of effective management is located in Luxembourg. “Luxembourg Insolvency Event” means, in relation to each Luxembourg Guarantor or any of its assets, any corporate action, legal proceedings or other procedure or step in relation to administrative dissolution without liquidation (dissolution administrative sans liquidation), reprieve from payment (sursis de paiement), out-of-court mutual agreement (réorganisation extra- judiciaire par accord amiable), reorganization by amicable agreement (reorganisation par accord amiable), judicial reorganisation in the form of a stay to enter into a mutual agreement (sursis en vue de la conclusion d'un accord amiable), judicial reorganisation by collective agreement (réorganisation judiciaire par accord collectif), judicial reorganisation by transfer of assets or activities (réorganisation judiciaire par transfert sous autorité de justice), conciliation (conciliation), bankruptcy (faillite) and any other insolvency proceedings (including without limitation administrative dissolution without liquidation proceedings (procédure de dissolution administrative sans liquidation)) under any applicable law. “Luxembourg Loan Party” means each Luxembourg Subsidiary that is a Borrower or a Subsidiary Guarantor. “Luxembourg Pledge Agreement” means that certain Luxembourg law Share Pledge Agreement (including any and all supplements thereto), dated as of the Effective Date, between Endo Enterprise, Inc., as pledgor, and the Collateral Agent, for the benefit of the Secured Parties.

51 “Luxembourg Subsidiary” means any Restricted Subsidiary incorporated and existing under the Grand Duchy of Luxembourg, or whose registered office or place of effective management is located in Luxembourg. “Majority in Interest” means, at any time (i) in the case of any Class of Revolving Lenders, Lenders having Revolving Credit Exposures with respect to such Class of Revolving Loans and unused Revolving Commitments with respect to such Class of Revolving Loans representing more than 50% of the sum of the aggregate Revolving Credit Exposures with respect to such Class of Revolving Loans and the unused aggregate Revolving Commitments with respect to such Class of Revolving Loans at such time and (ii) in the case of the Term Lenders of any Class, Lenders holding outstanding Term Loans of such Class representing more than 50% of all Term Loans of such Class outstanding at such time; provided that the unused Commitments of, and the portion of the Revolving Credit Exposure or Term Loans, as applicable, held or deemed held by, any Defaulting Lender or Net Short Lenders shall be excluded for purposes of making a determination of the Majority in Interest; provided, further, that, to the same extent specified in Section 9.04(h) with respect to determination of Required Lenders, the Loans of any Affiliated Lender shall in each case be excluded for purposes of making a determination of Majority in Interest unless the action in question affects such Affiliated Lender in a disproportionately adverse manner than its effect on the other Lenders. In making the above calculations, the Dollar Amounts (as determined in good faith by the Administrative Agent) of all amounts denominated in currencies other than Dollars shall be utilized. If the context indicates that the “Majority in Interest” is to be determined for a relevant Class or Tranche, then only the respective Class or Tranche shall be included as otherwise provided above in determining the applicable Majority in Interest. The determination of Majority in Interest with respect to any Class of Revolving Lenders shall also exclude the exposure related to any Persons excluded in the determination of the Required Revolving Lenders. “Margin Stock” has the meaning assigned to such term in Regulation U of the Board. “Material Adverse Effect” means a material adverse effect on (a) the business, assets, property or condition (financial or otherwise) of Parent and its Subsidiaries taken as a whole or (b) the validity or enforceability of this Agreement or any and all other Loan Documents or the rights or remedies of the Administrative Agent and the Lenders thereunder. “Material Indebtedness” means Indebtedness (other than the Loans and Letters of Credit), or obligations in respect of one or more Swap Agreements, of any one or more of Parent and its Restricted Subsidiaries in an aggregate principal amount exceeding the greater of (x) $150,000,000 and (y) 20.0% of Consolidated EBITDA as of the end of the Reference Period. For purposes of determining Material Indebtedness, the “principal amount” of the obligations of Parent or any Restricted Subsidiary in respect of any Swap Agreement at any time shall be the aggregate amount (giving effect to any netting agreements) that Parent or such Restricted Subsidiary would be required to pay if such Swap Agreement were terminated at such time. “Material IP” means all trademarks, trade names, copyrights, patents and other Intellectual Property that constitutes Collateral and is material to the business of the Borrower and its Restricted Subsidiaries, taken as a whole.

52 “Material Subsidiary” means each Restricted Subsidiary (i) which, as of the most recent fiscal quarter of Parent, for the period of four consecutive fiscal quarters then ended, for which financial statements have been delivered pursuant to Section 5.01, contributed greater than five percent (5%) of Parent’s Consolidated EBITDA for such period or (ii) which contributed greater than five percent (5%) of Parent’s Consolidated Total Assets as of such date; provided that, if at any time the aggregate amount of Consolidated EBITDA or Consolidated Total Assets attributable to all Restricted Subsidiaries that are not Material Subsidiaries exceeds ten percent (10%) of Consolidated EBITDA of Parent and its Restricted Subsidiaries for any such period or ten percent (10%) of Consolidated Total Assets of Parent and its Restricted Subsidiaries as of the end of any such fiscal quarter, Parent (or, in the event Parent has failed to do so within forty-five (45) days, the Administrative Agent) shall designate sufficient Restricted Subsidiaries as “Material Subsidiaries” to eliminate such excess, and such designated Restricted Subsidiaries shall for all purposes of this Agreement constitute Material Subsidiaries. For purposes of determining whether any entity is a “Material Subsidiary,” (i) all intercompany balances and activity between the entity being tested and its subsidiaries, on the one hand, and Parent and its subsidiaries, on the other hand, shall be excluded and (ii) any assets held by the entity being tested that would be classified as “restricted” on a consolidated balance sheet of such entity with its subsidiaries and which are intended to fund payments related to mesh device related claims shall be excluded. Notwithstanding anything to the contrary contained herein, any Borrower hereunder shall be deemed at all times to be a Material Subsidiary. “Maturity Date” means (i) with respect to the Initial Term Loans and 2024 Refinancing Term Loans that, in each case, have not been extended pursuant to Section 2.23, the date occurring seven years after the Effective Date, (ii) with respect to the Revolving Commitments of the Revolving Lenders that have not been extended pursuant to Section 2.23, the date occurring five years after the Effective Date and (iii) with respect to any other tranche of Term Loans or Revolving Commitments (including any Extended Term Loans, Other Term Loans, Other Refinancing Term Commitments, Incremental Revolving Commitments and Other Refinancing Revolving Commitments), the maturity dates specified therefor in the applicable Incremental Amendment, Extension Amendment or Refinancing Amendment; provided that, in each case, if any such day is not a Business Day, the Maturity Date shall be the Business Day immediately succeeding such day. “Milestone Payments” means payments made under contractual arrangements existing during the period of twelve months ending on the Effective Date or contractual arrangements arising thereafter, in each case in connection with any Permitted Acquisition to sellers (or licensors) of the assets or Equity Interests acquired (or licensed) therein based on the achievement of specified revenue, profit or other performance targets (financial or otherwise). “Minimum Extension Condition” shall have the meaning given to such term in Section 2.23(b). “Minimum Tranche Amount” shall have the meaning given to such term in Section 2.23(b). “Moody’s” means Moody’s Investors Service, Inc.

53 “Mortgage” means each mortgage, deed of trust or other agreement which conveys or evidences a Lien in favor of the Collateral Agent, for the benefit of the Secured Parties, on real property of a Loan Party, including any amendment, restatement, modification or supplement thereto. “Mortgage Instruments” means such title reports, title insurance, flood certifications and flood insurance, opinions of counsel, surveys, appraisals and environmental reports and other similar information and related certifications as are reasonably requested by, and in form and substance reasonably acceptable to, the Administrative Agent from time to time. “Multicurrency Tranche Commitment” means, with respect to each Multicurrency Tranche Lender, the commitment, if any, of such Multicurrency Tranche Lender to make Multicurrency Tranche Revolving Loans and to acquire participations in Multicurrency Tranche Letters of Credit hereunder, as such commitment may be (a) reduced or terminated from time to time pursuant to Section 2.09, (b) increased from time to time pursuant to Section 2.20 and (c) reduced or increased from time to time pursuant to assignments by or to such Lender pursuant to Section 9.04. The amount of each Multicurrency Tranche Lender’s Multicurrency Tranche Commitment as of the Effective Date is set forth on Schedule 2.01, or in the Assignment and Assumption (or other documentation contemplated by this Agreement) pursuant to which such Multicurrency Tranche Lender shall have assumed its Multicurrency Tranche Commitment, as applicable. The aggregate principal Dollar Amount of the Multicurrency Tranche Commitments on the Effective Date is $400,000,000. “Multicurrency Tranche Credit Event” means a Multicurrency Tranche Revolving Borrowing of any Class, the issuance of a Multicurrency Tranche Letter of Credit, an LC Disbursement with respect to a Multicurrency Tranche Letter of Credit or any of the foregoing. “Multicurrency Tranche LC Exposure” means, at any time, the sum of (a) the aggregate undrawn Dollar Amount of all outstanding Multicurrency Tranche Letters of Credit at such time plus (b) the aggregate Dollar Amount of all LC Disbursements in respect of Multicurrency Tranche Letters of Credit that have not yet been reimbursed by or on behalf of the Borrower at such time. The Multicurrency Tranche LC Exposure of any Multicurrency Tranche Lender at any time shall be its Multicurrency Tranche Percentage of the total Multicurrency Tranche LC Exposure at such time. “Multicurrency Tranche Lender” means a Lender with a Multicurrency Tranche Commitment or holding Multicurrency Tranche Revolving Loans. “Multicurrency Tranche Letter of Credit” means any standby or commercial letter of credit issued under the Multicurrency Tranche Commitments pursuant to this Agreement. “Multicurrency Tranche Percentage” the percentage equal to a fraction the numerator of which is such Lender’s Multicurrency Tranche Commitment and the denominator of which is the aggregate Multicurrency Tranche Commitments of all Multicurrency Tranche Lenders (if the Multicurrency Tranche Commitments of any Class have terminated or expired, the Multicurrency Tranche Percentages shall be determined based upon the Multicurrency Tranche Commitments of such Class most recently in effect, giving effect to any assignments).

54 “Multicurrency Tranche Revolving Borrowing” means a Borrowing comprised of Multicurrency Tranche Revolving Loans of any Class. “Multicurrency Tranche Revolving Credit Exposure” means, with respect to any Multicurrency Tranche Lender at any time, and without duplication, the sum of the outstanding principal amount of such Multicurrency Tranche Lender’s Multicurrency Tranche Revolving Loans and its Multicurrency Tranche LC Exposure at such time. “Multicurrency Tranche Revolving Loan” means a Loan made by a Multicurrency Tranche Lender pursuant to Section 2.01(c). Each Multicurrency Tranche Revolving Loan shall be a Term Benchmark Loan denominated in an Agreed Currency (subject to the limitation set forth in Section 2.01(c)(iv)) or a Canadian Prime Rate Loan denominated in Canadian Dollars or an ABR Loan denominated in Dollars. “Multiemployer Plan” means a multiemployer plan as defined in Section 4001(a)(3) of ERISA. “Net Proceeds” means, with respect to any event, (a) the cash proceeds received in respect of such event including (i) any cash received in respect of any non-cash proceeds (including any cash payments received by way of deferred payment of principal pursuant to a note or installment receivable or purchase price adjustment receivable or otherwise, but excluding any interest payments), but only as and when received, (ii) in the case of a casualty, insurance proceeds and (iii) in the case of a condemnation or similar event, condemnation awards and similar payments, net of (b) the sum of (i) all reasonable fees and out-of-pocket expenses paid to third parties (other than Affiliates) in connection with such event, (ii) in the case of a sale, transfer or other disposition of an asset (including pursuant to a Sale and Leaseback Transaction or a casualty or a condemnation or similar proceeding), the amount of all payments required to be made as a result of such event to repay Indebtedness (other than Loans) secured by such asset or otherwise subject to mandatory prepayment as a result of such event and (iii) the amount of all taxes paid (or reasonably estimated to be payable) and the amount of any reserves established to fund contingent liabilities reasonably estimated to be payable, in each case that are directly attributable to such event (as determined reasonably and in good faith by a Financial Officer); provided that on the date on which such reserve is no longer required to be maintained, the remaining amount of such reserve shall then be deemed to be Net Proceeds. “Net Short Lender” has the meaning specified in Section 9.02(f). “Non-Recourse Indebtedness” means Indebtedness: (a) as to which neither Parent nor any of the Restricted Subsidiaries (i) provides credit support of any kind (including any undertaking, agreement or instrument that would constitute Indebtedness) or (ii) is directly or indirectly liable as a guarantor or otherwise; and (b) as to which the lenders have been notified in writing that they will not have any recourse to the stock or assets of Parent or any of the Restricted Subsidiaries (other than the Equity Interests of an Unrestricted Subsidiary).

55 “Non-USD Multicurrency Tranche Revolving Credit Exposure” means, with respect to any Multicurrency Tranche Lender at any time, such Multicurrency Tranche Lender’s Multicurrency Tranche Revolving Credit Exposure with respect to Multicurrency Tranche Revolving Loans and Multicurrency Tranche Letters of Credit, in each case denominated in Agreed Currencies other than Dollars. “Non-USD Multicurrency Tranche Sublimit” means $200,000,000. “Non-U.S. Plan” means any plan, fund (including, without limitation, any superannuation fund) or other similar program established, contributed to (regardless of whether through direct contributions or through employee withholding) or maintained outside the United States of America by Parent or any one or more of its Subsidiaries primarily for the benefit of employees of Parent or such Subsidiaries residing outside the United States of America, which plan, fund or other similar program provides, or results in, retirement income, a deferral of income in contemplation of retirement or payments to be made upon termination of employment, and which plan is not subject to ERISA or the Code. “NYFRB” means the Federal Reserve Bank of New York. “NYFRB Rate” means, for any day, the greater of (a) the Federal Funds Effective Rate in effect on such day and (b) the Overnight Bank Funding Rate in effect on such day (or for any day that is not a Business Day, for the immediately preceding Business Day); provided that if none of such rates are published for any day that is a Business Day, the term “NYFRB Rate” means the rate for a federal funds transaction quoted at 11:00 a.m. on such day received to the Administrative Agent from a Federal funds broker of recognized standing selected by it; provided, further, that if any of the aforesaid rates shall be less than zero, such rate shall be deemed to be zero for purposes of this Agreement. “Obligations” means all unpaid principal of and accrued and unpaid interest on the Loans, all LC Exposure, all accrued and unpaid fees and all expenses, reimbursements, indemnities and other obligations and indebtedness (including interest accruing during the pendency of any bankruptcy, insolvency, receivership, examinership or other similar proceeding, regardless of whether allowed or allowable in such proceeding), obligations and liabilities of any of Parent, the Borrower and the other Loan Parties to any of the Lenders, the Administrative Agent, the Collateral Agent, the Issuing Bank or any indemnified party, individually or collectively, existing on the Effective Date or arising thereafter, direct or indirect, joint or several, absolute or contingent, matured or unmatured, liquidated or unliquidated, secured or unsecured, arising by contract, operation of law or otherwise, arising or incurred under this Agreement or any of the other Loan Documents or in respect of any of the Loans made or reimbursement or other obligations incurred or any of the Letters of Credit or other instruments at any time evidencing any thereof. “OFAC” means Office of Foreign Assets Control of the United States Department of the Treasury. “Other Applicable Indebtedness” means Indebtedness permitted hereunder that is secured on a pari passu basis with the Obligations.

56 “Other Connection Taxes” means, with respect to any Recipient, Taxes imposed as a result of a present or former connection between such Recipient and the jurisdiction imposing such Tax (other than connections arising from such Recipient having executed, delivered, become a party to, performed its obligations under, received payments under, received or perfected a security interest under, engaged in any other transaction pursuant to or enforced any Loan Document, or sold or assigned an interest in any Loan, Letter of Credit or Loan Document). “Other Refinancing Commitments” means the Other Refinancing Revolving Commitments and the Other Refinancing Term Commitments. “Other Refinancing Loans” means the Other Refinancing Revolving Loans and the Other Refinancing Term Loans. “Other Refinancing Revolving Commitments” means one or more Classes of Revolving Commitments hereunder or Extended Revolving Commitments that result from a Refinancing Amendment. “Other Refinancing Revolving Loans” means the Revolving Loans made pursuant to any Other Refinancing Revolving Commitment. “Other Refinancing Term Commitments” means one or more Classes of Term Loan Commitments hereunder that result from a Refinancing Amendment. “Other Refinancing Term Loans” means one or more Classes of Term Loans that result from a Refinancing Amendment. “Other Taxes” means any and all present or future stamp, court, recording, filing, intangible or documentary taxes or any other excise or property taxes, charges or similar levies arising from any payment made under, from the execution, delivery, performance, enforcement or registration of, from the receipt or perfection of a security interest under, or otherwise with respect to, this Agreement or any other Loan Document, except (a) any such Taxes that are Other Connection Taxes imposed with respect to an assignment (other than an assignment made pursuant to Section 2.19(b)) or (b) any such Taxes, charges or similar levies, incurred in Luxembourg as a result of a registration with the Administration de I'Enregistrement, des Domaines et de la TVA or other action by any or on behalf of any Lenders, the Administrative Agent or the Issuing Bank where such registration or action is not (i) mandatory and (ii) required to enforce the rights of the Lenders, the Administrative Agent or the Issuing Bank under this Agreement and any other Loan Document. “Other Term Loans” has the meaning set forth in Section 2.20(a). “Overnight Bank Funding Rate” means, for any day, the rate comprised of both overnight federal funds and overnight borrowings by U.S.-managed banking offices of depository institutions, as such composite rate shall be determined by the Federal Reserve Bank of New York as set forth on its public website from time to time, and published on the next succeeding Business Day by the Federal Reserve Bank of New York as an overnight bank funding rate (from and after such date as the Federal Reserve Bank of New York shall commence to publish such composite rate).

57 “Overnight Foreign Currency Rate” means, for any amount payable in a Foreign Currency, the rate of interest per annum as determined by the Administrative Agent at which overnight or weekend deposits in the relevant currency (or if such amount due remains unpaid for more than three (3) Business Days, then for such other period of time as the Administrative Agent may elect) for delivery in immediately available and freely transferable funds would be offered by the Administrative Agent to major banks in the interbank market upon request of such major banks for the relevant currency as determined above and in an amount comparable to the unpaid principal amount of the related Credit Event, plus any taxes, levies, imposts, duties, deductions, charges or withholdings imposed upon, or charged to, the Administrative Agent by any relevant correspondent bank in respect of such amount in such relevant currency. “Parent” has the meaning set forth in the Preamble. “Participant” has the meaning set forth in Section 9.04(c)(i). “Participant Register” has the meaning set forth in Section 9.04(c)(ii). “Payment” has the meaning set forth in Article VIII. “Payment Notice” has the meaning set forth in Article VIII. “PBGC” means the Pension Benefit Guaranty Corporation referred to and defined in ERISA and any successor entity performing similar functions. “Periodic Term CORRA Determination Day” has the meaning specified in the definition of “Term CORRA”. “Permitted Acquisition” means the purchase or other acquisition by Parent or any Restricted Subsidiary of Equity Interests in, or all or substantially all the assets of (or all or substantially all the assets constituting a business unit, division, product line (including rights in respect of any drug or other pharmaceutical product) or line of business of), any Person, or any Exclusive License of rights to a drug or other product line, in a single transaction or a series of related transactions if (a) (i) in the case of any purchase or other acquisition of Equity Interests in a Person, such Person (including each subsidiary of such Person to the extent such subsidiary was owned by such Person immediately prior to the purchase or acquisition), upon the consummation of such purchase or acquisition, will be a Restricted Subsidiary or (ii) in the case of any purchase, license or other acquisition of other assets, such assets will be owned and/or licensed by Parent or a Restricted Subsidiary; and (b) at the time of and immediately after giving effect (including pro forma effect) to any such purchase, license or other acquisition (subject to Section 1.04), no Event of Default shall have occurred and be continuing and either (x) Parent shall in compliance with the Financial Covenant or (y) the First Lien Net Leverage Ratio after giving effect thereto (including on a pro forma basis subject to Section 1.04) shall be no greater than the First Lien Net Leverage Ratio immediately prior thereto. “Permitted Bond Hedge” means any Swap Agreement that (i) is settled (after payment of any premium or any prepayment thereunder) through the delivery of cash and/or Equity Interests (other than Disqualified Equity Interests) of Parent or (ii) initially is settled (after payment of any premium or any prepayment thereunder) through the delivery of cash and/or

58 Equity Interests of any entity acquired in an acquisition permitted hereunder and in each case is entered into in connection with any Convertible Debt Securities or securities that became Convertible Debt Securities as a result of such acquisition, one of the purposes of which is, together with any Permitted Warrant entered into concurrently therewith, to provide for an effectively higher conversion premium. “Permitted Convertible Debt Hedge Transaction” means (i) any Permitted Bond Hedge and any Permitted Warrant or (ii) any capped call or similar transaction having substantially the same economic effect as the foregoing. “Permitted Encumbrances” means: (a) Liens imposed by law for taxes that are not yet due or payable or are being contested in compliance with Section 5.04 and Liens for unpaid utility charges; (b) carriers’, warehousemen’s, mechanics’, materialmen’s, repairmen’s and other like Liens imposed by law, arising in the ordinary course of business and securing obligations that are not overdue by more than sixty (60) days or are being contested in compliance with Section 5.04; (c) pledges and deposits made in the ordinary course of business in compliance with workers’ compensation, unemployment insurance and other social security laws or regulations or employment laws or to secure other public, statutory or regulatory obligations; (d) deposits and other liens to secure the performance of bids, trade contracts, leases, statutory obligations, surety and appeal bonds, performance bonds and other obligations of a like nature, in each case in the ordinary course of business; (e) judgment Liens in respect of judgments that do not constitute an Event of Default under clause (k) of Section 7.01 or securing appeal or surety bonds related to such judgments; (f) easements, zoning restrictions, rights-of-way and similar encumbrances on real property imposed by law or arising in the ordinary course of business that do not materially detract from the value of the affected property or interfere with the ordinary conduct of business of Parent or any Restricted Subsidiary; (g) banker’s liens, liens in favor of securities intermediaries, liens in favor of clearing agents, rights of setoff or similar rights and remedies as to deposit accounts or other funds maintained with depository institutions; provided that such deposit accounts or funds are not established or deposited for the purpose of providing collateral for any Indebtedness; (h) Liens arising by virtue of UCC financing statement filings (or similar filings under applicable law) regarding operating leases entered into by Parent and the Restricted Subsidiaries in the ordinary course of business;

59 (i) Canadian Statutory Liens in respect of any amount which may be overdue but the validity of which is being contested in good faith and in respect of which adequate reserves have been established in accordance with GAAP; (j) Liens or rights of distress reserved in or exercisable under any lease for rent not at the time overdue or for compliance with the terms of such lease not at the time in default; (k) any obligations or duties affecting any land due to any public utility or to any municipality or government, or to any statutory or public authority, with respect to any lease, franchise, grant, license or permit in good standing and any defects in title to structures or other facilities arising solely from the fact that such structures or facilities are constructed or installed on land under government permits, leases or other grants in good standing; which obligations, duties and defects in the aggregate do not materially impair the use of such property, structures or facilities for the purpose for which they are held; and (l) the reservations, limitations, provisions and conditions, if any, expressed in any original grant from His Majesty in Right of Canada or any province thereof of any real property located in Canada, provided they do not reduce the value of the assets of the Person or materially interfere with the use of such assets in the operation of the business of the Person. “Permitted Exchange” means an exchange of real property of Parent or any Restricted Subsidiary which qualifies as a like kind exchange pursuant to and in compliance with Section 1031 of the Code. “Permitted First Lien Indebtedness” means Indebtedness secured on a pari passu first lien basis with the Secured Obligations that is incurred after the Effective Date by Parent or any other Loan Party (and may in any case be co-borrowed or co-issued by any Borrower on a joint and several basis); provided that (i) both immediately prior to and after giving effect (including pro forma effect) thereto (subject to Section 1.04), no Event of Default shall exist or result therefrom, (ii) such Indebtedness shall not have a Weighted Average Life to Maturity that is less than the Weighted Average Life to Maturity of the then outstanding Class of Term Loans with the Latest Maturity Date that are secured on a pari passu basis with the Secured Obligations (other than such Indebtedness incurred under the Inside Maturity Basket), (iii) such Indebtedness is not guaranteed by Parent or any Restricted Subsidiary other than the Loan Parties, (iv) immediately after giving effect to the issuance, incurrence or assumption of such Indebtedness (subject to Section 1.04), the First Lien Net Leverage Ratio on a pro forma basis shall not be greater than 3.50 to 1.00, (v) the holders of such Indebtedness or their Designated Representative shall have entered into an Approved Intercreditor Agreement, (vi) if such Indebtedness consists of term loans, then the applicable Borrower shall comply with the “most favored nation” pricing provision in the proviso in Section 2.20(c)(v) as if such Indebtedness were Other Term Loans incurred under Section 2.20 (to the extent then applicable) and (vii) such Indebtedness may be incurred in the form of a bridge or other interim credit facility intended to be refinanced with long-term indebtedness (and such bridge or other interim credit facility shall be deemed to satisfy clause (ii) of this definition so long as (x) such credit facility includes customary “rollover” provisions and (y) assuming such credit facility were to be extended pursuant to such “rollover” provisions, such

60 extended credit facility would comply with clause (ii) above) and in which case, on or prior to the first anniversary of the incurrence of such “bridge” or other interim credit facility, nothing in this definition shall prohibit the inclusion of customary terms for “bridge” facilities, including customary mandatory prepayment, repurchase or redemption provisions. Permitted First Lien Indebtedness will include any Registered Equivalent Notes issued in exchange therefor. “Permitted Holders” means Golden Tree Asset Management LP, Silver Point Capital, L.P. or any of their respective Affiliates (other than portfolio companies). “Permitted Indebtedness” means Indebtedness (including Subordinated Indebtedness) that is incurred after the Effective Date by Parent or any Restricted Subsidiary (and may in any case be co-borrowed or co-issued by any Borrower on a joint and several basis); provided that (i) both immediately prior to and after giving effect (including pro forma effect) thereto (subject to Section 1.04), no Event of Default shall exist or result therefrom, (ii) such Indebtedness shall not have a Weighted Average Life to Maturity that is less than the Weighted Average Life to Maturity of the then outstanding Class of Term Loans with the Latest Maturity Date (other than such Indebtedness incurred under the Inside Maturity Basket), (iii) such Indebtedness is not guaranteed by Parent or any Restricted Subsidiary other than the Loan Parties, (iv) if such Indebtedness is unsecured or if such Indebtedness is incurred by a Restricted Subsidiary that is not a Loan Party, whether or not such Indebtedness is secured or unsecured, either (A) the Total Net Leverage Ratio would not exceed 6.00 to 1.00 (whether prior to or after giving effect (including pro forma effect) thereto and subject to Section 1.04) or (B) the Consolidated Interest Coverage Ratio would not be less than 2.00 to 1.00, (v) if such Indebtedness is to be secured, (A) the Secured Net Leverage Ratio shall not be greater than 5.00 to 1.00 (whether prior to or after giving effect (including pro forma effect) thereto and subject to Section 1.04) and (B) the holders of such Indebtedness or their Designated Representative shall have entered into an Approved Intercreditor Agreement and (vi) such Indebtedness may be incurred in the form of a bridge or other interim credit facility intended to be refinanced with long-term indebtedness (and such bridge or other interim credit facility shall be deemed to satisfy clause (ii) of this definition so long as (x) such credit facility includes customary “rollover” provisions and (y) assuming such credit facility were to be extended pursuant to such “rollover” provisions, such extended credit facility would comply with clause (ii) above) and in which case, on or prior to the first anniversary of the incurrence of such “bridge” or other interim credit facility, nothing in this definition shall prohibit the inclusion of customary terms for “bridge” facilities, including customary mandatory prepayment, repurchase or redemption provisions. Permitted Indebtedness will include any Registered Equivalent Notes issued in exchange therefor; provided that the aggregate principal amount of Permitted Indebtedness incurred by Restricted Subsidiaries that are not Loan Parties shall not exceed the greater of (x) $75,000,000 and (y) 10.0% of Consolidated EBITDA as of the end of the Reference Period. For the avoidance of doubt, any provision requiring an offer to purchase such Indebtedness as a result of a change of control, delisting, or asset sale or any provision permitting holders to convert such Indebtedness shall be deemed not to violate clause (ii). “Permitted Investments” means: (a) direct obligations of, or obligations the principal of and interest on which are unconditionally guaranteed by, the United States of America (or by any agency thereof

61 to the extent such obligations are backed by the full faith and credit of the United States of America), in each case maturing within one year from the date of acquisition thereof; (b) investments in commercial paper maturing within 270 days from the date of acquisition thereof and having, at such date of acquisition, a rating of P-2 (or higher) according to Moody’s or A-2 (or higher) according to S&P (or such similar equivalent rating or higher by at least one nationally recognized statistical rating organization (as defined in Rule 436 under the Securities Act)); (c) investments in certificates of deposit, banker’s acceptances and time deposits maturing within 180 days from the date of acquisition thereof issued or guaranteed by or placed with, and money market deposit accounts issued or offered by, any domestic office of any commercial bank organized under the laws of the United States of America or any State thereof which has a combined capital and surplus and undivided profits of not less than $500,000,000; (d) fully collateralized repurchase agreements with a term of not more than thirty (30) days for securities described in clause (a) above and entered into with a financial institution satisfying the criteria described in clause (c) above; (e) money market funds that (i) comply with the criteria set forth in SEC Rule 2a-7 under the Investment Company Act of 1940, (ii) are rated AAA by S&P and Aaa by Moody’s and (iii) have portfolio assets of at least $5,000,000,000; (f) in the case of Parent or any Foreign Subsidiary, other short-term investments that are analogous to the foregoing, are of comparable credit quality and are customarily used by companies in the jurisdiction of Parent or such Foreign Subsidiary for cash management purposes; (g) investments in auction rate securities to the extent held by Parent or any Restricted Subsidiary on the Effective Date; and (h) any other cash equivalent investments permitted by Parent’s investment policy as such policy is in effect and as disclosed to the Administrative Agent prior to the Effective Date and as such policy may be amended, restated, supplemented or otherwise modified from time to time with the consent of the Administrative Agent. “Permitted Junior Secured Refinancing Debt” means any secured Indebtedness incurred after the Effective Date by Parent or any Loan Party (and may in any case be co-borrowed or co-issued by any Borrower on a joint and several basis) in the form of one or more series of junior lien notes or junior lien loans; provided that (i) such Indebtedness is secured by all or a portion of the Collateral on a junior-priority basis with the Obligations and is not secured by any property or assets of Parent, Parent or any Subsidiary other than the Collateral, (ii) such Indebtedness constitutes Credit Agreement Refinancing Indebtedness, (iii) such Indebtedness does not mature or have scheduled amortization or scheduled payments of principal and is not subject to mandatory redemption, repurchase, prepayment or sinking fund obligation (other than customary offers to repurchase upon a change of control, asset sale or casualty event and customary acceleration rights after an event of default, in each case subject to and after giving effect to such

62 offers and rights under this Agreement) prior to the Latest Maturity Date at the time such Indebtedness is incurred (other than such Indebtedness incurred under the Inside Maturity Basket), (iv) the security agreements relating to such Indebtedness are substantially the same as the Collateral Documents (with such differences as are reasonably satisfactory to the Administrative Agent), (v) such Indebtedness is not guaranteed by Parent or any of its Subsidiaries other than the Loan Parties, (vi) a Designated Representative acting on behalf of the holders of such Indebtedness shall have become party to or otherwise subject to the provisions of an Approved Intercreditor Agreement; provided that if such Indebtedness is the initial Permitted Junior Secured Refinancing Debt incurred after the Effective Date, then Parent, the Borrower, the Subsidiary Guarantors, the Administrative Agent and the Designated Representative for such Indebtedness shall have executed and delivered an Approved Intercreditor Agreement and (vii) such Indebtedness may be incurred in the form of a bridge or other interim credit facility intended to be refinanced with long- term indebtedness (and such bridge or other interim credit facility shall be deemed to satisfy clause (iii) of this definition so long as (x) such credit facility includes customary “rollover” provisions and (y) assuming such credit facility were to be extended pursuant to such “rollover” provisions, such extended credit facility would comply with clause (iii) above) and in which case, on or prior to the first anniversary of the incurrence of such “bridge” or other interim credit facility, clauses (ii) and (iii) of this definition shall not prohibit the inclusion of customary terms for “bridge” facilities, including customary mandatory prepayment, repurchase or redemption provisions. Permitted Junior Secured Refinancing Debt will include any Registered Equivalent Notes issued in exchange therefor. “Permitted Pari Passu Secured Refinancing Debt” means any secured Indebtedness incurred after the Effective Date by Parent or any Loan Party in the form of one or more series of senior secured notes or senior secured loans; provided that (i) such Indebtedness is secured by all or a portion of the Collateral on a pari passu basis (but without regard to the control of remedies) with the Obligations and is not secured by any property or assets of Parent or any Subsidiary other than the Collateral, (ii) such Indebtedness constitutes Credit Agreement Refinancing Indebtedness, (iii) such Indebtedness does not mature or have scheduled amortization or scheduled payments of principal and is not subject to mandatory redemption, repurchase, prepayment or sinking fund obligation (other than customary offers to repurchase upon a change of control, asset sale or casualty event and customary acceleration rights after an event of default, in each case subject to and after giving effect to such offers and rights under this Agreement) prior to the Latest Maturity Date at the time such Indebtedness is incurred (other than such Indebtedness incurred under the Inside Maturity Basket), (iv) the security agreements relating to such Indebtedness are substantially the same as the Collateral Documents (with such differences as are reasonably satisfactory to the Administrative Agent), (v) such Indebtedness is not guaranteed by Parent or any of its Subsidiaries other than the Loan Parties, (vi) a Designated Representative acting on behalf of the holders of such Indebtedness shall have become party to or otherwise subject to the provisions of an Approved Intercreditor Agreement; provided that if such Indebtedness is the initial Permitted Pari Passu Secured Refinancing Debt incurred after the Effective Date, then Parent, the Borrower, the Subsidiary Guarantors, the Administrative Agent and the Designated Representative for such Indebtedness shall have executed and delivered an Approved Intercreditor Agreement and (vii) such Indebtedness may be incurred in the form of a bridge or other interim credit facility intended to be refinanced with long-term indebtedness (and such bridge or other interim credit facility shall be deemed to satisfy clause (iii) of this definition so long as (x) such credit facility includes customary “rollover” provisions and (y) assuming such credit facility were

63 to be extended pursuant to such “rollover” provisions, such extended credit facility would comply with clause (iii) above) and in which case, on or prior to the first anniversary of the incurrence of such “bridge” or other interim credit facility, clauses (ii) and (iii) of this definition shall not prohibit the inclusion of customary terms for “bridge” facilities, including customary mandatory prepayment, repurchase or redemption provisions. Permitted Pari Passu Secured Refinancing Debt will include any Registered Equivalent Notes issued in exchange therefor. “Permitted Receivables Facility” means any Receivables Facility (1) that meets the following conditions: (a) the Receivables Seller will have determined in good faith that such Receivables Facility (including financing terms, covenants, termination events and other provisions) is in the aggregate economically fair and reasonable to such Receivables Seller and (b) the sale, transfer, contribution or pledge of Receivables Assets to the applicable Person or Receivables Entity is made at fair market value (as reasonably determined in good faith by Parent) or (2) constituting a receivables financing facility. “Permitted Receivables Facility Assets” means any Receivables Assets sold, transferred, contributed or pledged in connection with a Permitted Receivables Facility. “Permitted Receivables Facility Documents” means each of the documents and agreements entered into in connection with any Permitted Receivables Facility, including all documents and agreements relating to the issuance, funding and/or purchase of certificates and purchased interests or the incurrence of loans, as applicable, as such documents and agreements may be amended, modified, supplemented, refinanced or replaced from time to time. “Permitted Refinancing Indebtedness” means any Indebtedness issued in exchange for, or the net proceeds of which are used to extend, refinance, renew, replace, defease or refund (collectively, to “Refinance”), other Indebtedness; provided that (a) the principal amount (or accreted value, if applicable) of such Permitted Refinancing Indebtedness does not exceed the principal amount (or accreted value, if applicable) of the Indebtedness so refinanced (plus unpaid accrued interest and premium (including tender premium) thereon, any committed or undrawn amounts and underwriting discounts, fees, commissions and expenses, associated with such Permitted Refinancing Indebtedness), (b) the final maturity date of such Permitted Refinancing Indebtedness is no earlier than the maturity date applicable to the Indebtedness being Refinanced (it being understood that, in each case, any provision requiring an offer to purchase such Indebtedness as a result of a change of control, delisting, asset sale or similar provision or any provision permitting holders to convert such Indebtedness shall not violate the foregoing restriction), (c) if the Indebtedness (including any Guarantee thereof) being Refinanced is by its terms subordinated in right of payment to the Secured Obligations, such Permitted Refinancing Indebtedness (including any Guarantee thereof) shall be subordinated in right of payment to the Secured Obligations on terms at least as favorable to the Lenders as those contained in the documentation governing the Indebtedness being Refinanced, taken as a whole (as determined in good faith by the board of directors of Parent), (d) such Permitted Refinancing Indebtedness contains mandatory redemption (or similar provisions), if any, covenants, if any, and events of default, if any, and is benefited by guarantees, if any, which are customary for Indebtedness of such type (reasonably determined in good faith by the board of directors of Parent), (e) no Permitted Refinancing Indebtedness shall have direct obligors or contingent obligors that were not the direct obligors or contingent obligors (or that would not have been required to become direct

64 obligors or contingent obligors) in respect of the Indebtedness being Refinanced, (f) if the Indebtedness being Refinanced is secured, such Permitted Refinancing Indebtedness may be secured on terms no less favorable, taken as a whole, to the Secured Parties than those contained in the documentation (including any intercreditor agreement) governing the Indebtedness being Refinanced (reasonably determined in good faith by Parent), (g) if the Indebtedness being refinanced was subject to an Approved Intercreditor Agreement, and if the respective Permitted Refinancing Indebtedness is to be secured by the Collateral, the Permitted Refinancing Indebtedness shall likewise be subject to Approved Intercreditor Agreement and (h) such Indebtedness may be incurred in the form of a bridge or other interim credit facility intended to be refinanced with long-term indebtedness (and such bridge or other interim credit facility shall be deemed to satisfy clause (b) of this definition so long as (x) such credit facility includes customary “rollover” provisions and (y) assuming such credit facility were to be extended pursuant to such “rollover” provisions, such extended credit facility would comply with clause (b) above) and in which case, on or prior to the first anniversary of the incurrence of such “bridge” or other interim credit facility, clause (d) of this definition shall not prohibit the inclusion of customary terms for “bridge” facilities, including customary mandatory prepayment, repurchase or redemption provisions. “Permitted Unsecured Refinancing Debt” means any unsecured Indebtedness incurred after the Effective Date by Parent or any Loan Party (and may in any case be co-borrowed or co-issued by any Borrower on a joint and several basis) in the form of one or more series of unsecured notes or loans; provided that (i) such Indebtedness is not secured by any property or assets of Parent or any Subsidiary, (ii) such Indebtedness constitutes Credit Agreement Refinancing Indebtedness, (iii) such Indebtedness does not mature or have scheduled amortization prior to the Latest Maturity Date at the time such Indebtedness is incurred (other than customary offers to repurchase upon a change of control or asset sale and customary acceleration rights after an event of default, in each case subject to and after giving effect to such offers and rights under this Agreement, and other than such Indebtedness incurred under the Inside Maturity Basket) (iv) such Indebtedness is not guaranteed by Parent or any of its Subsidiaries other than the Loan Parties and (v) such Indebtedness may be incurred in the form of a bridge or other interim credit facility intended to be refinanced with long-term indebtedness (and such bridge or other interim credit facility shall be deemed to satisfy clause (iii) of this definition so long as (x) such credit facility includes customary “rollover” provisions and (y) assuming such credit facility were to be extended pursuant to such “rollover” provisions, such extended credit facility would comply with clause (iii) above) and in which case, on or prior to the first anniversary of the incurrence of such “bridge” or other interim credit facility, clauses (ii) and (iii) of this definition shall not prohibit the inclusion of customary terms for “bridge” facilities, including customary mandatory prepayment, repurchase or redemption provisions. Permitted Unsecured Refinancing Debt will include any Registered Equivalent Notes issued in exchange therefor. “Permitted Warrant” means (i) one or more call options settled through the delivery of cash and/or Parent’s Equity Interests (not constituting Disqualified Equity Interests) or (ii) one or more call options initially settled through the delivery of cash and/or the Equity Interests of any entity acquired in an acquisition permitted hereunder, in each case, sold concurrently with the entry into one or more Permitted Bond Hedges and having an initial strike or exercise price (howsoever defined) that is greater than the strike or exercise price (howsoever defined) of such Permitted Bond Hedge.

65 “Person” means any natural person, corporation, limited liability company, trust, joint venture, association, company, partnership, Governmental Authority or other entity. “Plan” means any employee pension benefit plan (other than a Multiemployer Plan) subject to the provisions of Title IV of ERISA or Section 412 of the Code or Section 302 of ERISA, and in respect of which Parent or any ERISA Affiliate is (or, if such plan were terminated, would under Section 4069 of ERISA be deemed to be) an “employer” as defined in Section 3(5) of ERISA. “PLC” means Endo International plc, a company formed under the laws of Ireland. “Platform” has the meaning assigned to such term in the final paragraph of Section 5.01. “Pledge Subsidiary” means (i) each Loan Party that is a Domestic Subsidiary and (ii) subject to the Agreed Security Principles, each Irish Loan Party and Luxembourg Loan Party. “Pounds Sterling” means the lawful currency of the United Kingdom. “Prepayment Event” means: (a) any Asset Sale described in Section 6.03(a)(xx) (other than the Net Proceeds of which (x) individually, do not exceed $20,000,000 or (y) together with the aggregate amount of Net Proceeds received from all such Asset Sales described in Section 6.03(a)(xx) occurring in the same fiscal year of Parent, do not exceed $40,000,000); (b) any Sale and Leaseback Transaction described in Section 6.10 (other than any Sale and Leaseback Transaction the Net Proceeds of which, (x) individually do not exceed $20,000,000 or (y) together with the aggregate amount of Net Proceeds received from all such Sale and Lease Back Transactions occurring in the same fiscal year of Parent, do not exceed $40,000,000); (c) any casualty or other insured damage to, or any taking under power of eminent domain or by condemnation or similar proceeding of, any property or asset of Parent or any Restricted Subsidiary with a fair market value immediately prior to such event greater than $20,000,000 per event or $40,000,000 for all such events occurring in the same fiscal year of Parent; or (d) the incurrence by Parent or any Restricted Subsidiary of any Indebtedness, other than Indebtedness permitted under Section 6.01 (excluding Credit Agreement Refinancing Indebtedness required to be applied towards the prepayment of any Obligations pursuant to Section 2.11(c)(2)) or permitted by the Required Lenders pursuant to Section 9.02. “Prime Rate” last quoted by The Wall Street Journal as the “Prime Rate” in the U.S. or, if The Wall Street Journal ceases to quote such rate, the highest per annum interest rate published by the Federal Reserve Board in Federal Reserve Statistical Release H.15 (519)

66 (Selected Interest Rates) as the “bank prime loan” rate or, if such rate is no longer quoted therein, any similar rate quoted therein (as reasonably determined by the Administrative Agent) or any similar release by the Federal Reserve Board (as reasonably determined by the Administrative Agent).; each change in the Prime Rate shall be effective from and including the date such change is publicly announced as being effective. “Public Lender” has the meaning assigned to such term in in the final paragraph of Section 5.01. “Purchase Offer” has the meaning assigned to such term in Section 2.24(a). “Receivables” means accounts receivable, royalty or other revenue streams, including contract rights, lockbox accounts, records with respect to such accounts receivable, royalty or other revenue streams and other rights to payment and other assets related thereto created by or arising from sales of goods, leases of goods or the rendition of services rendered no matter how evidenced whether or not earned by performance (whether constituting accounts, general intangibles, chattel paper or otherwise). “Receivables Assets” means Receivables, the proceeds thereof and other revenue streams and other rights to payment customarily sold, transferred, contributed or pledged together with such Receivables in connection with a Receivables Facility. “Receivables Entity” means in connection with a Receivables Facility, any special purpose vehicle formed for the purpose of entering into a Receivables Facility and performing its duties and obligations (and exercising its rights) under the related Permitted Receivables Facility Documents, and that is not used for any other purpose or to engage in any other business or activity. For the avoidance of doubt, there may be more than one “Receivables Entity” with respect to any single Receivables Facility. “Receivables Facility” means a public or private transfer, sale, financing or pledge of Receivables Assets by which any Receivables Entity directly or indirectly securitizes a pool of specified Receivables Assets or pledges such specified Receivables Assets in a secured financing. “Receivables Sellers” means Parent and those Subsidiaries that are from time to time party to the Permitted Receivables Facility Documents (other than any Receivables Entity). “Recipient” means (a) the Administrative Agent, (b) any Lender or (c) any Issuing Bank, as applicable. “Reference Period” in effect at any time means the most recent period of four consecutive fiscal quarters of Parent ended on or prior to such time (taken as one accounting period) in respect of which, subject to Section 1.04, financial statements for each quarter or fiscal year in such period have been or are required to be delivered pursuant to Section 5.01(a) or (b), as applicable; provided that, prior to the first date that financial statements have been or are required to be delivered pursuant to Section 5.01(a) or (b), the Reference Period in effect shall be the period of four consecutive full fiscal quarters of PLC ended prior to the Effective Date for which financial statements would have been required to be delivered hereunder had the Effective Date occurred prior to the end of such period.

67 “Reference Time” with respect to any setting of the then-current Benchmark means (1) if such Benchmark is Term SOFR, 5:00 a.m. (Chicago time) on the day that is two U.S. Government Securities Business Days preceding the date of such setting, (2) if such Benchmark is the Adjusted Term CORRA Rate, 1:00 p.m. Toronto local time on the day that is two Business Days preceding the date of such setting or (3) if such Benchmark is none of Term SOFR or the Adjusted Term CORRA Rate, the time determined by the Administrative Agent in its reasonable discretion. “Refinanced Debt” has the meaning assigned to such term in the definition of “Credit Agreement Refinancing Indebtedness”. “Refinancing Amendment” means an amendment to this Agreement in form and substance reasonably satisfactory to the Administrative Agent and the Borrower executed by each of (a) Parent and the Borrower, (b) the Administrative Agent, (c) the Issuing Bank (in the case of Other Refinancing Revolving Commitments or Other Refinancing Revolving Loans) and (d) each Refinancing Lender and Lender that agrees to provide any portion of the Credit Agreement Refinancing Indebtedness being incurred pursuant thereto, in accordance with Section 2.25. “Refinancing Lender” means, at any time, any bank, other financial institution or institutional investor that, in any case, is not an existing Lender (and that is not Parent or any of its Subsidiaries or Affiliates) and that agrees to provide any portion of any Credit Agreement Refinancing Indebtedness pursuant to a Refinancing Amendment in accordance with Section 2.25; provided that each Refinancing Lender (other than any Person that is a Lender, an Affiliate of a Lender or an Approved Fund of a Lender at such time) shall be subject to the approval of the Administrative Agent and the Issuing Bank (in the case of Other Refinancing Revolving Commitments or Other Refinancing Revolving Loans) (such approval not to be unreasonably withheld or delayed), in each case to the extent any such consent would be required from the Administrative Agent and the Issuing Bank (in the case of Other Refinancing Revolving Commitments or Other Refinancing Revolving Loans) under Section 9.04(b)(i) for an assignment of Loans or Commitments to such Refinancing Lender. “Register” has the meaning set forth in Section 9.04(b)(iv). “Registered Equivalent Notes” means, with respect to any notes originally issued in a Rule 144A or other private placement transaction under the Securities Act, substantially identical notes (having the same Guarantees) issued in a dollar-for-dollar exchange therefor pursuant to an exchange offer registered with the SEC. “Related Indemnified Person” of an indemnified person means (a) any controlling person or controlled affiliate of such indemnified person, (b) the respective directors, officers, or employees of such indemnified person or any of its controlling persons or controlled affiliates and (c) the respective agents of such indemnified person or any of its controlling persons or controlled affiliates, in the case of this clause (c), acting at the instructions of such indemnified person, controlling person or such controlled affiliate; provided that each reference to a controlled affiliate or controlling person in this sentence pertains to a controlled affiliate or controlling person involved in the negotiation or syndication of this Agreement and the Loans.

68 “Related Parties” means, with respect to any specified Person, such Person’s Affiliates and the respective directors, officers, employees, agents and advisors of such Person and such Person’s Affiliates. “Release” means any release, spill, emission, discharge, leaking, pumping, injection, deposit, disposal, dispersal, leaching or migration of Hazardous Materials into the environment (including, without limitation, ambient air, surface water, groundwater and surface or subsurface strata). “Relevant Rate” means (i) with respect to any Borrowing denominated in Dollars, Term SOFR or (ii) with respect to any Borrowing denominated in Canadian Dollars, the Adjusted Term CORRA Rate, as applicable. “Reorganization Plan” means the Fourth Amended Joint Chapter 11 Plan of Reorganization of Endo International PLC and its Affiliated Debtors filed in the Chapter 11 Cases at Docket No. 3849. “Reorganization Plan Documents” means the Plan Documents (as defined in the Reorganization Plan). “Reorganization Plan Transactions” means (i) the acquisition of substantially all of the assets of PLC and certain of its affiliates as debtors and debtors in possession (collectively, the “Debtors”), and (ii) any other transactions expressly contemplated by and necessary to implement the Reorganization Plan as in effect on the Effective Date. “Repricing Event” means (a) the prepayment or refinancing of any of the Initial Term Loans or 2024 Refinancing Term Loans with the incurrence by any Loan Party of any Indebtedness incurred for the primary purpose (as reasonably determined by Parent) of lowering the Effective Yield of the Initial Term Loans or 2024 Refinancing Term Loans or (b) any effective reduction in the Effective Yield of the Initial Term Loans or 2024 Refinancing Term Loans (e.g., by way of amendment or waiver); provided that in no event shall any prepayment or repayment of the Initial Term Loans or 2024 Refinancing Term Loans in connection with a (i) Change in Control or (ii) an Enterprise Transformative Event constitute a Repricing Event. “Required Lenders” means, at any time, Lenders having Credit Exposures and unused Commitments representing more than 50% of the sum of the total Credit Exposures and unused Commitments at such time; provided that the unused Term Loan Commitment and unused Revolving Commitment of, and the portion of the Credit Exposure held or deemed held by, any Defaulting Lender or Net Short Lender shall be excluded for purposes of making a determination of Required Lenders; provided, further, that the Loans of any Affiliated Lender shall in each case be excluded for purposes of making a determination of Required Lenders unless the action in question affects such Affiliated Lender in a disproportionately adverse manner than its effect on the other Lenders. For all purposes of determining the Required Lenders hereunder, if any relevant Credit Exposures or unused Commitments are denominated in currencies other than Dollars, the respective Dollar Amounts (as determined in good faith by the Administrative Agent) thereof shall be utilized.

69 “Required Revolving Lenders” means, at any time, Lenders having Revolving Credit Exposures and unused Revolving Commitments representing more than 50% of the sum of the total Revolving Credit Exposures and unused Revolving Commitments at such time; provided that the unused Revolving Commitment of, and the portion of the Revolving Credit Exposure held or deemed held by, any Defaulting Lender or Net Short Lender shall be excluded for purposes of making a determination of Required Revolving Lenders; provided, further, that the Loans of any Affiliated Lender shall in each case be excluded for purposes of making a determination of Required Revolving Lenders unless the action in question affects such Affiliated Lender in a disproportionately adverse manner than its effect on the other Lenders. For all purposes of determining the Required Revolving Lenders hereunder, if any relevant Revolving Credit Exposures or unused Revolving Commitments are denominated in currencies other than Dollars, the respective Dollar Amounts (as determined in good faith by the Administrative Agent) thereof shall be utilized. “Resolution Authority” means an EEA Resolution Authority or, with respect to any UK Financial Institution, a UK Resolution Authority. “Responsible Officer” means, with respect to any Person, the chief executive officer, president, an executive vice president, senior vice president, manager, director, duly appointed attorney-in-fact or a Financial Officer. Unless otherwise specified, a Responsible Officer refers to a Responsible Officer of Parent. “Restricted Payment” means any dividend or other distribution (whether in cash, securities or other property) with respect to any Equity Interests in Parent or any Restricted Subsidiary, or any payment (whether in cash, securities or other property), including any sinking fund or similar deposit, on account of the purchase, redemption, retirement, acquisition, cancellation or termination of any such Equity Interests in Parent or any Restricted Subsidiary or any option, warrant or other right to acquire any such Equity Interests in Parent or any Restricted Subsidiary. For the avoidance of doubt, any interest payments with respect to Convertible Debt Securities shall not constitute Restricted Payments. “Retained Excess Cash Flow” means, for any period, an amount equal to (a) the cumulative amount of Excess Cash Flow (which amount shall not be less than zero) for such period minus (b) the amount that has been (or is required to be) applied to the prepayment of the Term Loans in accordance with Section 2.11(c)(3) for such period. “Restricted Subsidiary” means any Subsidiary of Parent (including the Borrower) other than an Unrestricted Subsidiary. “Revaluation Date” means (a)(i) with respect to any Loan denominated in any Foreign Currency, the date of the Borrowing of such Loan and (ii) with respect to any Term Benchmark Loan, each date of a conversion into or continuation of such Loan pursuant to the terms of this Agreement; (b) with respect to any Letter of Credit denominated in a Foreign Currency, each of the following: (i) the date on which such Letter of Credit is issued, (ii) the first Business Day of each calendar month and (iii) the date of any amendment of such Letter of Credit that has the effect of increasing the available balance thereof; and (c) any additional date as the

70 Administrative Agent may reasonably determine at any time when an Event of Default has occurred and is continuing. “Revolving Commitment” means a Dollar Tranche Commitment or a Multicurrency Tranche Commitment, as the context may require, and “Revolving Commitments” means, collectively, the Dollar Tranche Commitments and the Multicurrency Tranche Commitments. The aggregate principal amount of the Revolving Commitments on the Effective Date is $400,000,000. “Revolving Credit Exposure” means, with respect to any Revolving Lender at any time, the sum of the outstanding principal amount of such Revolving Lender’s Multicurrency Tranche Revolving Loans and Dollar Tranche Revolving Loans and its LC Exposure and Swingline Exposure at such time. “Revolving Facility” means the Revolving Commitments from time to time and the extensions of credit made thereunder. “Revolving Facility Obligations” means all Obligations in respect of the Revolving Facility. “Revolving Lender” means, as of any date of determination, each Lender that has a Revolving Commitment or, if the Revolving Commitments have terminated or expired, a Lender with Revolving Credit Exposure. “Revolving Loan” means any Multicurrency Tranche Revolving Loan or Dollar Tranche Revolving Loan, as the context may require, and “Revolving Loans” means, collectively, the Dollar Tranche Revolving Loans and the Multicurrency Tranche Revolving Loans. “Revolving Secured Obligations” means all Revolving Facility Obligations, together with (i) all Swap Obligations owing to any Person that is the Administrative Agent, Collateral Agent, Swingline Lender, Issuing Bank, a Lead Arranger, a Revolving Lender or an Affiliate of any of the foregoing or was the Administrative Agent, Collateral Agent, Swingline Lender, Issuing Bank, a Lead Arranger, a Revolving Lender or an Affiliate of any of the foregoing at the time the applicable Swap Agreement was entered into (excluding, in case of any Guarantor that is not an ECP, any Excluded Swap Obligations) and (ii) Banking Services Obligations owing to one or more Revolving Lenders or their respective Affiliates. “S&P” means Standard & Poor’s Ratings Services, a Standard & Poor’s Financial Services LLC business. “Sale and Leaseback Transaction” means any sale or other transfer of any property or asset by any Person with the intent to lease such property or asset as lessee. “Sanctioned Country” means, at any time, a country, region or territory which is itself the subject or target of any Sanctions (as of the Effective Date, Cuba, Iran, North Korea, Syria and the Crimea, the so-called Luhansk People’s Republic, and the so-called Donetsk People’s Republic regions of Ukraine).

71 “Sanctioned Person” means any Person listed in any Sanctions-related list of designated Persons maintained by the Office of Foreign Assets Control of the U.S. Department of the Treasury or the U.S. Department of State, the United Nations Security Council, the European Union, any Member State of the European Union or the United Kingdom, (b) any Person located, organized or ordinarily resident in a Sanctioned Country or (c) any person owned in the aggregate 50% or more or controlled by any such person. “Sanctions” means economic or financial sanctions or trade embargoes imposed, administered or enforced from time to time by the U.S. government, including those administered by the Office of Foreign Assets Control of the U.S. Department of the Treasury or the U.S. Department of State, the United Nations Security Council, the European Union, any Member State of the European Union or His Majesty’s Treasury of the United Kingdom. “Scheduled Principal Repayment Dates” means the last day of each March, June, September and December and the applicable Maturity Date. “SEC” means the United States Securities and Exchange Commission. “Secured Net Leverage Ratio” means the ratio of (a) Consolidated Secured Debt minus the aggregate amount of cash and Permitted Investments of Parent and its Restricted Subsidiaries on such date that (x) would not appear as “restricted” on a consolidated balance sheet of Parent and its Restricted Subsidiaries or (y) are restricted or secured in favor of the Indebtedness incurred under this Agreement or other Indebtedness secured by a pari passu or junior Lien on the Collateral as permitted under this Agreement to (b) Consolidated EBITDA of Parent and its Restricted Subsidiaries for such Reference Period, in each case on a pro forma basis with such pro forma adjustments as are appropriate and consistent with Section 1.04. “Secured Obligations” means, collectively, the Revolving Secured Obligations and the Term Loan Secured Obligations. “Secured Parties” means the holders of the Secured Obligations from time to time and shall include (i) each Lender and the Issuing Bank in respect of its Loans and LC Exposure respectively, (ii) the Administrative Agent, the Collateral Agent, the Issuing Bank and the Lenders in respect of all other present and future obligations and liabilities of Parent and each Restricted Subsidiary of every type and description arising under or in connection with this Agreement or any other Loan Document, (iii) with respect to any Swap Agreement, each Person that is the Administrative Agent, a Lead Arranger, a Lender or an Affiliate of any of the foregoing or was the Administrative Agent, a Lead Arranger, a Lender or an Affiliate of any of the foregoing at the time such Swap Agreement was entered into with such Person by Parent or any Restricted Subsidiary, (iv) each Lender and Affiliate of such Lender in respect of Banking Services Agreements entered into with such Person by Parent or any Restricted Subsidiary, (v) each indemnified party under Section 9.03 in respect of the obligations and liabilities of the Borrower to such Person hereunder and under the other Loan Documents, and (vi) their respective successors and (in the case of a Lender, permitted) transferees and assigns. “Securities Act” means the United States Securities Act of 1933, as amended from time to time and any successor statute.

72 “Senior Secured Notes” means the 8.500% senior secured notes due 2031 issued pursuant to the Senior Secured Notes Indenture. “Senior Secured Notes Indenture” means the Indenture, dated as of the Effective Date, among Endo Finance Holdings LP (f/k/a Endo Finance Holdings, Inc.), the guarantors named therein and Computershare Trust Company, as trustee and as collateral agent. “SOFR” means a rate per annum equal to the secured overnight financing rate as administered by the SOFR Administrator. “SOFR Administrator” means the NYFRB (or a successor administrator of the secured overnight financing rate). “SOFR Administrator’s Website” means the NYFRB’s website, currently at http://www.newyorkfed.org, or any successor source for the secured overnight financing rate identified as such by the SOFR Administrator from time to time. “SOFR Determination Date” has the meaning specified in the definition of “Daily Simple SOFR”. “SOFR Rate Day” has the meaning specified in the definition of “Daily Simple SOFR”. “SPC” has the meaning assigned to such term in Section 9.04(f). “Specified Transaction” means: (1) solely for the purposes of determining the applicable cash balance, any contribution of capital, including as a result of an issuance of Equity Interests, to Parent, in each case, in connection with an acquisition or Investment, (2) any designation of operations or assets of Parent or a Restricted Subsidiary as discontinued operations (as defined under GAAP), (3) any Investment that results in a Person becoming a Restricted Subsidiary, (4) any designation of a Subsidiary as a Restricted Subsidiary or an Unrestricted Subsidiary in compliance with this Agreement, (5) any purchase or other acquisition of a business of any Person, of assets constituting a business unit, line of business or division of any Person, (6) any Asset Sale (without regard to any de minimis thresholds set forth therein) (a) that results in a Restricted Subsidiary ceasing to be a Subsidiary of Parent or (b) of a business, business unit, line of business or division of Parent or a Restricted Subsidiary, in each case whether by merger, amalgamation, consolidation or otherwise,

73 (7) any operational changes identified by Parent that have been made by the Borrower Representative or any Restricted Subsidiary during the Reference Period, (8) any borrowing of Incremental Loans or Incremental Equivalent Debt (or establishment of Incremental Commitments), or (9) or any Restricted Payment or other transaction that by the terms of this Agreement requires a financial ratio to be calculated on a pro forma basis. “Subordinated Indebtedness” means any Indebtedness of Parent or any Restricted Subsidiary the payment of which is subordinated to payment of the obligations under the Loan Documents. “subsidiary” means, with respect to any Person at any date, any corporation, limited liability company, partnership, association or other entity the accounts of which would be consolidated with those of such Person in such Person’s consolidated financial statements if such financial statements were prepared in accordance with GAAP as of such date, as well as any other corporation, limited liability company, partnership, association or other entity of which securities or other ownership interests representing more than 50% of the equity or more than 50% of the ordinary voting power or, in the case of a partnership, more than 50% of the general partnership interests are, as of such date, owned, Controlled or held. “Subsidiary” means any subsidiary of Parent (unless a contrary intention appears herein). “Subsidiary Guarantor” means each (i) existing and subsequently acquired Domestic Subsidiary, (ii) direct or indirect wholly-owned Luxembourg Subsidiary existing on the Effective Date, (iii) direct or indirect wholly-owned Irish Subsidiary (x) existing on the Effective Date or (y) that owns any Material IP, and (iv) any other Restricted Subsidiary designated by Parent as a Subsidiary Guarantor, in each case, that is party to the Subsidiary Guaranty from time to time. Notwithstanding anything herein or in any other Loan Document to the contrary, no Receivables Entity or Excluded Subsidiary shall be required to be a Subsidiary Guarantor. “Subsidiary Guaranty” means that certain Guaranty dated as of the Effective Date (including any and all supplements thereto) and executed by each Subsidiary Guarantor, including any modification thereto or any separate Guarantee executed and delivered by any Foreign Loan Party in accordance with Section 5.09 and the Agreed Security Principles. “Swap Agreement” means any agreement with respect to any swap, forward, future or derivative transaction or option or similar agreement involving, or settled by reference to, one or more rates, currencies, commodities, equity or debt instruments or securities, or economic, financial or pricing indices or measures of economic, financial or pricing risk or value or any similar transaction or any combination of these transactions; provided that no phantom stock or similar plan providing for payments only on account of services provided by current or former directors, officers, employees or consultants of Parent or the Restricted Subsidiaries shall be a Swap Agreement.

74 “Swap Obligations” means any and all obligations of Parent or any Restricted Subsidiary, whether absolute or contingent and howsoever and whensoever created, arising, evidenced or acquired (including all renewals, extensions and modifications thereof and substitutions therefor), under (a) any and all Swap Agreements with the Administrative Agent, a Lead Arranger, a Lender or an Affiliate of any of the foregoing or a Person that was the Administrative Agent, a Lead Arranger, a Lender or an Affiliate of any of the foregoing at the time such Swap Agreement was entered into, and (b) any and all cancellations, buy backs, reversals, terminations or assignments of any such Swap Agreement transaction. “Swingline Exposure” means, at any time, the aggregate principal amount of all Swingline Loans outstanding at such time. The Swingline Exposure of any Lender at any time shall be its Dollar Tranche Percentage of the total Swingline Exposure at such time. “Swingline Lender” means Goldman Sachs Bank USA, in its capacity as lender of Swingline Loans hereunder. “Swingline Loan” means a Loan made pursuant to Section 2.05. “TARGET” means the real time gross settlement system operated by the Eurosystem (or, if such payment system ceases to be operative, such other payment system (if any) reasonably determined by the Administrative Agent to be a suitable replacement) for the settlement of payments in euro. “Taxes” means all present or future taxes, levies, imposts, duties, deductions, fees, assessments, charges or withholdings imposed by any Governmental Authority, including any interest, additions to tax or penalties applicable thereto. “Term Benchmark” when used in reference to any Loan or Borrowing, refers to whether such Loan, or the Loans comprising such Borrowing, are bearing interest at a rate determined by reference to (a) if such Loan or Borrowing is in Dollars, Term SOFR and (b) if such Loan or Borrowing is in Canadian Dollars, Adjusted Term CORRA Rate. “Term CORRA” means, for any calculation with respect to any Borrowing denominated in Canadian Dollars, the Term CORRA Reference Rate for a tenor comparable to the applicable Interest Period on the day (such day, the “Periodic Term CORRA Determination Day”) that is two (2) Business Days prior to the first day of such Interest Period, as such rate is published by the Term CORRA Administrator; provided, however, that if as of 1:00 p.m. (Toronto time) on any Periodic Term CORRA Determination Day the Term CORRA Reference Rate for the applicable tenor has not been published by the Term CORRA Administrator and a Benchmark Replacement Date with respect to the Term CORRA Reference Rate has not occurred, then Term CORRA will be the Term CORRA Reference Rate for such tenor as published by the Term CORRA Administrator on the first preceding Business Day for which such Term CORRA Reference Rate for such tenor was published by the Term CORRA Administrator so long as such first preceding Business Day is not more than five (5) Business Days prior to such Periodic Term CORRA Determination Day. “Term CORRA Administrator” means Candeal Benchmark Administration Services Inc., TSX Inc., or any successor administrator.

75 “Term CORRA Loan” when used in reference to any Loan or Borrowing, refers to a Loan, or the Loans comprising such Borrowing, bearing interest at a rate determined by reference to the Adjusted Term CORRA Rate. “Term CORRA Reference Rate” means the forward-looking term rate based on CORRA. “Term Lender” means any Initial Term Lender, any 2024 Refinancing Term Lender, any Incremental Term Lender and any Extending Term Lender. “Term Loan Commitments” means the Initial Term Loan Commitments, the 2024 Refinancing Term Loan Commitments and any Incremental Term Loan Commitments. “Term Loan Facility Obligations” means all Obligations in respect of the Term Loans. “Term Loan Secured Obligations” means all Term Loan Facility Obligations, together with (i) all Swap Obligations owing to any Person that is a Term Loan Lender or an Affiliate thereof or was a Term Loan Lender or an Affiliate thereof at the time the applicable Swap Agreement was entered into (excluding, in case of any Guarantor that is not an ECP, any Excluded Swap Obligations) and (ii) Banking Services Obligations owing to one or more Term Loan Lenders or their respective Affiliates. “Term Loans” means the Initial Term Loans, the 2024 Refinancing Term Loans, any Incremental Term Loan (including any Other Term Loan), any Other Refinancing Term Loans of the applicable Class or any Extended Term Loan. “Term SOFR” means, with respect to any Term Benchmark Borrowing and for any tenor comparable to the applicable Interest Period, the Term SOFR Reference Rate at approximately 5:00 a.m., Chicago time, two U.S. Government Securities Business Days prior to the commencement of such tenor comparable to the applicable Interest Period, as such rate is published by the CME Term SOFR Administrator; provided that if the Term SOFR as so determined would be less than the Floor, such rate shall be deemed to be equal to the Floor for the purposes of this Agreement. “Term SOFR Determination Day” has the meaning specified in the definition of “Term SOFR Reference Rate”. “Term SOFR Reference Rate” means, for any day and time (such day, the “Term SOFR Determination Day”), with respect to any Term Benchmark Borrowing denominated in Dollars and for any tenor comparable to the applicable Interest Period, the rate per annum published by the CME Term SOFR Administrator and identified by the Administrative Agent as the forward-looking term rate based on SOFR. If by 5:00 pm (New York City time) on such Term SOFR Determination Day, the “Term SOFR Reference Rate” for the applicable tenor has not been published by the CME Term SOFR Administrator and a Benchmark Replacement Date with respect to Term SOFR has not occurred, then, so long as such day is otherwise a U.S. Government Securities Business Day, the Term SOFR Reference Rate for such Term SOFR Determination Day

76 will be the Term SOFR Reference Rate as published in respect of the first preceding U.S. Government Securities Business Day for which such Term SOFR Reference Rate was published by the CME Term SOFR Administrator, so long as such first preceding U.S. Government Securities Business Day is not more than five (5) U.S. Government Securities Business Days prior to such Term SOFR Determination Day. “TEU” means a security (or combination of securities) that is composed of a prepaid stock purchase contract relating to the Equity Interest of Parent and an amortizing note. “Third Amendment” means that certain Third Amendment to this Agreement, dated as of November 3, 2025. “Third Amendment Effective Date” has the meaning assigned to such term in the Third Amendment. “Total Net Leverage Ratio” means the ratio of (i) Consolidated Total Indebtedness minus the aggregate amount of cash and Permitted Investments of Parent and its Restricted Subsidiaries on such date that (x) would not appear as “restricted” on a consolidated balance sheet of Parent and its Restricted Subsidiaries or (y) are restricted or secured in favor of the Indebtedness incurred under this Agreement or other Indebtedness secured by a pari passu or junior Lien on the Collateral as permitted under this Agreement to (ii) Consolidated EBITDA of Parent and its Restricted Subsidiaries for such Reference Period, in each case on a pro forma basis with such pro forma adjustments as are appropriate and consistent with Section 1.04. “Tranche” means a category of Commitments and extensions of credit thereunder. For purposes hereof, each of the following comprises a separate Tranche: (a) Multicurrency Tranche Commitments, Multicurrency Tranche Revolving Loans and Multicurrency Tranche Letters of Credit, (b) Dollar Tranche Commitments, Dollar Tranche Revolving Loans, Dollar Tranche Letters of Credit and Swingline Loans and (c) Term Loan Commitments and Term Loans. “Transactions” means (a) the execution, delivery and performance by the Loan Parties of this Agreement and the other Loan Documents to which they are a party, (b) the borrowing of Loans and other credit extensions, the use of the proceeds thereof and the issuance of Letters of Credit hereunder, (c) the execution, delivery and performance by the applicable Loan Parties of the Senior Secured Notes Indenture and the issuance of Senior Secured Notes pursuant thereto, (d) the granting of Liens pursuant to the Collateral Documents, (e) the Reorganization Plan Transactions, (e) any other transactions related to or entered into to implement any of the foregoing and (f) the payment of the fees and expenses incurred in connection with any of the foregoing. “Transaction Expenses” means any fees, expenses, costs or charges incurred or paid by Parent, any Borrower or any other Restricted Subsidiary in connection with the Transactions. “Type”, when used in reference to any Loan or Borrowing, refers to Loans or Borrowings in a single currency and whether the rate of interest on such Loan, or on the Loans comprising such Borrowing, is determined by reference to the Alternate Base Rate, the Canadian Prime Rate or the applicable Relevant Rate.

77 “UCC” means the Uniform Commercial Code as in effect from time to time in the State of New York or any other state the laws of which are required to be applied in connection with the perfection of security interests or any Collateral. “UK Financial Institution” means any BRRD Undertaking (as such term is defined under the PRA Rulebook (as amended form time to time) promulgated by the United Kingdom Prudential Regulation Authority) or any person falling within IFPRU 11.6 of the FCA Handbook (as amended from time to time) promulgated by the United Kingdom Financial Conduct Authority, which includes certain credit institutions and investment firms, and certain affiliates of such credit institutions or investment firms. “UK Resolution Authority” means the Bank of England or any other public administrative authority having responsibility for the resolution of any UK Financial Institution. “Unadjusted Benchmark Replacement” means the applicable Benchmark Replacement excluding the related Benchmark Replacement Adjustment. “Unliquidated Obligations” means, at any time, any Secured Obligations (or portion thereof) that are contingent in nature or unliquidated at such time, including any Secured Obligation that is: (i) an obligation to reimburse a bank for drawings not yet made under a letter of credit issued by it; (ii) any other obligation (including any guarantee) that is contingent in nature at such time; or (iii) an obligation to provide collateral to secure any of the foregoing types of obligations. “Unrestricted Subsidiary” means (a) each Subsidiary on the Effective Date which is noted on Schedule 3.01 hereof, (b) after the Effective Date, any additional Subsidiaries of Parent designated by the board of directors of Parent as an “Unrestricted Subsidiary” pursuant to Section 5.10, and (c) any Subsidiary of any of the foregoing. “Upfront Payments” means any upfront or similar payments made during the period of twelve months ending on the Effective Date or arising thereafter in connection with any drug or pharmaceutical product research and development or collaboration arrangements or the closing of any Drug Acquisition. “US Borrower” means any Borrower that is a “United States person” as defined in Section 7701(a)(30) of the Code. “US Borrowings” means any Borrowing of a US Borrower. “U.S. Government Securities Business Day” means any day except for (a) a Saturday, (b) a Sunday or (c) a day on which the Securities Industry and Financial Markets Association recommends that the fixed income departments of its members be closed for the entire day for purposes of trading in United States government securities. “US Security Agreement” means that certain Pledge and Security Agreement (including any and all supplements thereto), dated as of the Effective Date, initially between Parent, the Borrower Representative and each Domestic Subsidiary that is a Subsidiary Guarantor and the Collateral Agent, for the benefit of the Collateral Agent and the other Secured Parties (as

78 defined in the US Security Agreement), and any other pledge or security agreement entered into after the Effective Date by any other Loan Party that is a Domestic Subsidiary (as required by this Agreement or any other Loan Document) with the Collateral Agent. “USA Patriot Act” has the meaning assigned to such term in Section 9.14. “Weighted Average Life to Maturity” means, when applied to any Indebtedness at any date, the number of years obtained by dividing: (a) the sum of the products obtained by multiplying (i) the amount of each then remaining installment, sinking fund, serial maturity or other required payments of principal, including payment at final maturity, in respect thereof, by (ii) the number of years (calculated to the nearest one-twelfth) that will elapse between such date and the making of such payment; by (b) the then outstanding principal amount of such Indebtedness. “Withdrawal Liability” means liability to a Multiemployer Plan as a result of a complete or partial withdrawal from such Multiemployer Plan, as such terms are defined in Part I of Subtitle E of Title IV of ERISA. “Write-Down and Conversion Powers” means, (a) with respect to any EEA Resolution Authority, the write-down and conversion powers of such EEA Resolution Authority from time to time under the Bail-In Legislation for the applicable EEA Member Country, which write-down and conversion powers are described in the EU Bail-In Legislation Schedule, and (b) with respect to the United Kingdom, any powers of the applicable Resolution Authority under the Bail-In Legislation to cancel, reduce, modify or change the form of a liability of any UK Financial Institution or any contract or instrument under which that liability arises, to convert all or part of that liability into shares, securities or obligations of that person or any other person, to provide that any such contract or instrument is to have effect as if a right had been exercised under it or to suspend any obligation in respect of that liability or any of the powers under that Bail-In Legislation that are related to or ancillary to any of those powers. Section 1.02 Classification of Loans and Borrowings. For purposes of this Agreement, Loans may be classified and referred to by Class (e.g., a “Dollar Tranche Revolving Loan”) or by Type (e.g., a “Term Benchmark Loan”) or by Class and Type (e.g., a “Term SOFR Dollar Tranche Revolving Loan”). Borrowings also may be classified and referred to by Class (e.g., a “Dollar Tranche Revolving Borrowing”) or by Type (e.g., a “Term Benchmark Borrowing”) or by Class and Type (e.g., a “Dollar Tranche Term Benchmark Revolving Borrowing”). Section 1.03 Terms Generally. (a) The definitions of terms herein shall apply equally to the singular and plural forms of the terms defined. Whenever the context may require, any pronoun shall include the corresponding masculine, feminine and neuter forms. The words “include”, “includes” and “including” shall be deemed to be followed by the phrase “without limitation”. The word “will” shall be construed to have the same meaning and effect as the word “shall”. The word “law” shall be construed as referring to all statutes, rules, regulations, codes and other laws (including official rulings and interpretations thereunder having the force of law or with which affected Persons customarily comply), and all judgments, orders and decrees, of all Governmental Authorities. Unless the context requires otherwise (a) any definition of or reference to any agreement, instrument or other document herein shall be construed as referring to such agreement,

79 instrument or other document as from time to time amended, restated, supplemented or otherwise modified (subject to any restrictions on such amendments, restatements, supplements or modifications set forth herein), (b) any definition of or reference to any statute, rule or regulation shall be construed as referring thereto as from time to time amended, supplemented or otherwise modified (including by succession of comparable successor laws), (c) any reference herein to any Person shall be construed to include such Person’s successors and assigns (subject to any restrictions on assignment set forth herein) and, in the case of any Governmental Authority, any other Governmental Authority that shall have succeeded to any or all functions thereof, (d) the words “herein”, “hereof” and “hereunder”, and words of similar import, shall be construed to refer to this Agreement in its entirety and not to any particular provision hereof, (e) all references herein to Articles, Sections, Exhibits and Schedules shall be construed to refer to Articles and Sections of, and Exhibits and Schedules to, this Agreement and (f) the words “asset” and “property” shall be construed to have the same meaning and effect and to refer to any and all tangible and intangible assets and properties, including cash, securities, accounts and contract rights. Any references in this Agreement or any other Loan Document to “Permitted Encumbrances” is not intended to subordinate or postpone, and shall not be interpreted as subordination or postponing, or as any agreement to subordinate or postpone, any Lien created by any of the Loan Documents to any Permitted Encumbrance. (b) For purposes of determining compliance with any Section of Article VI, in the event that any Lien, Investment, Indebtedness, Asset Sale, Restricted Payment or affiliate transaction meets the criteria of one or more of the categories of transactions permitted pursuant to any clause of any one of such Sections, such transaction (or portion thereof) at any time, shall be permitted under one or more of such clauses of such Section as determined by the applicable Borrower in its sole discretion at such time. For purposes of determining compliance with the incurrence of any Credit Agreement Refinancing Indebtedness, Permitted Pari Passu Secured Refinancing Debt, Permitted Junior Secured Refinancing Debt, Permitted Refinancing Indebtedness and Permitted Unsecured Refinancing Debt that restricts the amount of such Indebtedness relative to the amount of Refinanced Debt, the Borrower may incur an incremental principal amount of such Credit Agreement Refinancing Indebtedness, Permitted Pari Passu Secured Refinancing Debt, Permitted Junior Secured Refinancing Debt, Permitted Refinancing Indebtedness or Permitted Unsecured Refinancing Debt to the extent that the excess portion of such Credit Agreement Refinancing Indebtedness, Permitted Pari Passu Secured Refinancing Debt, Permitted Junior Secured Refinancing Debt, Permitted Refinancing Indebtedness or Permitted Unsecured Refinancing Debt would otherwise be permitted to be incurred in accordance with this Agreement. For purposes of determining compliance with the incurrence of any Indebtedness under Revolving Commitments in reliance on compliance with any ratio, if on the date such Revolving Commitments are established, the applicable ratio is satisfied after giving pro forma effect to the incurrence of the entire committed amount of then proposed Indebtedness thereunder, then such committed amount under such Revolving Commitments may thereafter be borrowed and reborrowed, in whole or in part, from time to time, without further compliance with any ratio. (c) Notwithstanding anything to the contrary herein, with respect to any amounts incurred or transactions entered into (or consummated) in reliance on a provision of this Agreement under a restrictive covenant (including Section 2.20) that does not require compliance with a financial ratio or test (including, without limitation, any First Lien Net Leverage Ratio test,

80 any Secured Net Leverage Ratio test or any Total Net Leverage Ratio test) (any such amounts, the “Fixed Amounts”) substantially concurrently with any amounts incurred or transactions entered into (or consummated) in reliance on a provision of this Agreement in the same restrictive covenant (including Section 2.20) that requires compliance with any such financial ratio or test (any such amounts, the “Incurrence Based Amounts”), it is understood and agreed that the Fixed Amounts (and any cash proceeds thereof) shall be disregarded in the calculation of the financial ratio or test applicable to the Incurrence Based Amounts in connection with such substantially concurrent incurrence. (d) Notwithstanding anything to the contrary set forth herein, it is understood and agreed that in no event shall the Reorganization Plan Transactions or any other transactions expressly contemplated by the Reorganization Plan Documents, including any transactions directly related thereto or directly in connection therewith, result in the breach of any provision or covenant under this Agreement or any other Loan Document, or constitute a “Default” or “Event of Default” hereunder or thereunder, and such transactions shall be permitted for all purposes hereunder and under each other Loan Document. Section 1.04 Accounting Terms; GAAP; Pro Forma Calculations. (a) Except as otherwise expressly provided herein, all terms of an accounting or financial nature shall be construed in accordance with GAAP, as in effect from time to time; provided that, if Parent notifies the Administrative Agent that Parent requests an amendment to any provision hereof to eliminate the effect of any change occurring after the Effective Date in GAAP or in the application thereof on the operation of such provision (or if the Administrative Agent notifies Parent that the Required Lenders request an amendment to any provision hereof for such purpose), regardless of whether any such notice is given before or after such change in GAAP or in the application thereof, then such provision shall be interpreted on the basis of GAAP as in effect and applied immediately before such change shall have become effective until such notice shall have been withdrawn or such provision amended in accordance herewith. Notwithstanding any other provision contained herein, all terms of an accounting or financial nature used herein shall be construed, and all computations of amounts and ratios referred to herein shall be made (i) without giving effect to any election under Accounting Standards Codification 825-10-25 (or any other Accounting Standards Codification or Financial Accounting Standard having a similar result or effect) to value any Indebtedness or other liabilities of Parent or any Subsidiary at “fair value”, as defined therein, (ii) without giving effect to any treatment of Indebtedness in respect of convertible debt instruments under Accounting Standards Codification 470-20 (or any other Accounting Standards Codification or Financial Accounting Standard having a similar result or effect) to value any such Indebtedness in a reduced or bifurcated manner as described therein, and such Indebtedness shall at all times be valued at the full stated principal amount thereof and (iii) for the avoidance of doubt, except as provided in the definition of “Consolidated Net Income”, without giving effect to the financial condition, results and performance of the Unrestricted Subsidiaries. (b) Notwithstanding anything to the contrary herein, financial ratios and tests, including the First Lien Net Leverage Ratio, the Secured Net Leverage Ratio and the Total Net Leverage Ratio shall be calculated in the manner prescribed by this Section 1.04; provided that notwithstanding anything to the contrary in clauses (c), (d), (e) or (f) of this Section 1.04, when calculating the Total Net Leverage Ratio for purposes of (i) the definition of “Applicable Rate,” (ii) the Applicable Excess Cash Flow Percentage for the purposes of the mandatory

81 prepayment required by clause (3) of Section 2.11(c) and (iii) the Financial Covenant (other than for the purpose of determining pro forma compliance therewith), the events described in this Section 1.04 that occurred subsequent to the end of the applicable Reference Period shall not be given pro forma effect; provided however that voluntary prepayments made pursuant to Section 2.11(a) during any fiscal year (without duplication of any prepayments in such fiscal year that reduced the amount of Excess Cash Flow required to be repaid pursuant to Section 2.11(c) for any prior fiscal year) shall be given pro forma effect after such fiscal year-end and prior to the time any mandatory prepayment pursuant to Section 2.11(c) is due for purposes of calculating the First Lien Net Leverage Ratio for purposes of determining the Applicable Excess Cash Flow Percentage for such mandatory prepayment, if any. In addition, whenever a financial ratio or test is to be calculated on a pro forma basis, (1) the reference to “Reference Period” for purposes of calculating such financial ratio or test shall be deemed to be a reference to, and shall be based on, the most recently ended Reference Period for which Financials of Parent have been (or are required to be) delivered (it being understood that for purposes of determining pro forma compliance with the Financial Covenant, if no Reference Period with an applicable level cited in the Financial Covenant has passed, the applicable level shall be the level for the first Reference Period cited in the Financial Covenant with an indicated level) and (2) such calculation shall not net the cash proceeds of any Indebtedness being incurred at the time of such calculation. (c) For purposes of calculating any financial ratio or test (or Consolidated EBITDA or Consolidated Total Assets), Specified Transactions (and, subject to clause (e) below, the incurrence or repayment of any Indebtedness in connection therewith) that have been made (a) during the applicable Reference Period or (b) subsequent to such Reference Period and prior to or simultaneously with the event for which the calculation of any such ratio is made shall be calculated on a pro forma basis assuming that all such Specified Transactions (and any increase or decrease in Consolidated EBITDA and the component financial definitions used therein attributable to any Specified Transaction) had occurred on the first day of the applicable Reference Period (or, in the case of Consolidated Total Assets, on the last day of the applicable Reference Period). If since the beginning of any applicable Reference Period any Person that subsequently became a Restricted Subsidiary or was merged, amalgamated or consolidated with or into Parent or any Restricted Subsidiary since the beginning of such Reference Period shall have made any Specified Transaction that would have required adjustment pursuant to this Section 1.04, then such financial ratio or test (or Consolidated EBITDA or Consolidated Total Assets) shall be calculated to give pro forma effect thereto in accordance with this Section 1.04. (d) Whenever pro forma effect is to be given to a Specified Transaction, the pro forma calculations shall be made in good faith by a Financial Officer of Parent and may include, for the avoidance of doubt, the amount of “run-rate” cost savings, operating expense reductions and synergies projected by Parent in good faith to result from or relating to any Specified Transaction (including the Transactions and, for the avoidance of doubt, acquisitions occurring prior to the Effective Date) which is being given pro forma effect that have been realized or are expected to be realized and for which the actions necessary to realize such cost savings, operating expense reductions and synergies are taken, committed to be taken or with respect to which substantial steps have been taken or are expected to be taken (in the good faith determination of Parent) (calculated on a pro forma basis as though such cost savings, operating expense reductions and synergies had been realized on the first day of such period and as if such cost savings, operating expense reductions and synergies were realized during the entirety of such

82 period and “run-rate” means the full recurring benefit for a period that is associated with any action taken, committed to be taken or with respect to which substantial steps have been taken or are expected to be taken (including any savings expected to result from the elimination of a public target’s compliance costs with public company requirements), whether prior to or following the Effective Date, net of the amount of actual benefits realized during such period from such actions, and any such adjustments shall be included in the initial pro forma calculations of such financial ratios or tests and during any subsequent Reference Period in which the effects thereof are expected to be realized) relating to such Specified Transaction; provided that (a) such amounts are reasonably identifiable and factually supportable in the good faith judgment of Parent, (b) such actions are taken, committed to be taken or with respect to which substantial steps have been taken or are expected to be taken no later than eighteen (18) months after the date of such Specified Transaction (or actions undertaken or implemented prior to the consummation of such Specified Transaction), any such pro forma adjustments in respect of cost savings, synergies and operating expense reductions shall not exceed 20.0% of Consolidated EBITDA (prior to giving effect to such pro forma adjustments) as of the last day of the Reference Period and (c) no amounts shall be added to the extent duplicative of any amounts that are otherwise added back in computing Consolidated EBITDA (or any other components thereof), whether through a pro forma adjustment or otherwise, with respect to such period. (e) In the event that Parent or any Restricted Subsidiary incurs (including by assumption or guarantees), issues or repays (including by redemption, repurchase, repayment, retirement, discharge, defeasance or extinguishment) any Indebtedness (other than Indebtedness incurred or repaid under any revolving credit facility or line of credit unless such Indebtedness has been permanently repaid and not replaced) included in the calculations of any financial ratio or test, (i) during the applicable Reference Period or (ii) subsequent to the end of the applicable Reference Period and prior to or simultaneously with the event for which the calculation of any such ratio is made, then such financial ratio or test shall be calculated giving pro forma effect to such incurrence, issuance, repayment or redemption of Indebtedness to the extent required, as if the same had occurred on the last day of the applicable Reference Period. (f) If any Indebtedness bears a floating rate of interest and is being given pro forma effect, the interest on such Indebtedness shall be calculated as if the rate in effect on the date of determination had been the applicable rate for the entire period (taking into account any Swap Agreement applicable to such Indebtedness). Interest on a Capital Lease Obligation shall be deemed to accrue at an interest rate reasonably determined by a Financial Officer of Parent to be the rate of interest implicit in such Capital Lease Obligation in accordance with GAAP. Interest on Indebtedness that may optionally be determined at an interest rate based upon a factor of a prime or similar rate, an interbank offered rate, or other rate shall be determined to have been based upon the rate actually chosen, or if none, then based upon such optional rate chosen as Parent or applicable Restricted Subsidiary may designate. (g) Any determination of Consolidated Total Assets shall be made by reference to the last day of the Reference Period most recently ended for which Financials of Parent have been delivered on or prior to the relevant date of determination. (h) Notwithstanding anything in this Agreement or any Loan Document to the contrary, when (a) calculating any applicable ratio, Consolidated Net Income or

83 Consolidated EBITDA in connection with the incurrence of Indebtedness, the creation of Liens, the making of any Asset Sale, the making of an Investment or the making of a Restricted Payment, (b) determining compliance with any provision of this Agreement which requires that no Event of Default has occurred, is continuing or would result therefrom, (c) determining compliance with any provision of this Agreement which requires compliance with any representation or warranties set forth herein or (d) determining the satisfaction of all other conditions precedent to the incurrence of Indebtedness, the creation of Liens, the making of any Asset Sale, the making of an Investment or the making of a Restricted Payment, in each case in connection with a Limited Condition Transaction, the date of determination of such ratio or other provisions, determination of whether any Default or Event of Default has occurred, is continuing or would result therefrom, determination of compliance with any representations or warranties or the satisfaction of any other conditions shall, at the option of Parent (Parent’s election to exercise such option in connection with any Limited Condition Transaction, an “LCT Election,” which LCT Election may be in respect of one or more of clauses (a), (b), (c) and (d) above), be deemed to be the date the definitive agreements (or other relevant definitive documentation) for such Limited Condition Transaction are entered into (the “LCT Test Date”). If on a pro forma basis after giving effect to such Limited Condition Transaction and the other transactions to be entered into in connection therewith (including any incurrence or issuance of Indebtedness, and the use of proceeds thereof), with such ratios and other provisions calculated as if such Limited Condition Transaction or other transactions had occurred at the beginning of the most recent Reference Period ending prior to the LCT Test Date for which Financials have been (or are required to be) delivered, Parent could have taken such action on the relevant LCT Test Date in compliance with the applicable ratios or other provisions, such provisions shall be deemed to have been complied with, unless an Event of Default pursuant to Section 7.01(a), or, solely with respect to the Borrower, Section 7.01(h) shall be continuing on the date such Limited Condition Transaction is consummated. For the avoidance of doubt, (i) if, following the LCT Test Date, any of such ratios or other provisions are exceeded or breached as a result of fluctuations in such ratio (including due to fluctuations in Consolidated EBITDA or other components of such ratio) or other provisions at or prior to the consummation of the relevant Limited Condition Transactions, such ratios and other provisions will not be deemed to have been exceeded or failed to have been satisfied as a result of such fluctuations solely for purposes of determining whether the Limited Condition Transaction is permitted hereunder and (ii) such ratios and compliance with such conditions shall not be tested at the time of consummation of such Limited Condition Transaction or related Specified Transactions, unless, other than if an Event of Default pursuant to Section 7.01(a), or, solely with respect to the Borrower, Section 7.01(h), shall be continuing on such date, Parent elects, in its sole discretion, to test such ratios and compliance with such conditions on the date such Limited Condition Transaction or related Specified Transactions is consummated. If Parent has made an LCT Election for any Limited Condition Transaction, then in connection with any subsequent calculation of any ratio, basket availability or compliance with any other provision hereunder (other than actual compliance with the Financial Covenant) on or following the relevant LCT Test Date and prior to the earliest of the date on which such Limited Condition Transaction is consummated, the date that the definitive agreement for such Limited Condition Transaction is terminated or expires without consummation of such Limited Condition Transaction or the date Parent makes an election pursuant to clause (ii) of the immediately preceding sentence, any such ratio, basket or compliance with any other provision hereunder shall be calculated on a pro forma basis assuming such Limited Condition Transaction and other transactions in connection therewith

84 (including any incurrence or issuance of Indebtedness or Disqualified Equity Interests, and the use of proceeds thereof) had been consummated on the LCT Test Date. Section 1.05 Status of Obligations and Secured Obligations. In the event that Parent or any other Loan Party shall at any time issue or have outstanding any other Subordinated Indebtedness, Parent shall take or cause such other Loan Party to take all such actions as shall be necessary to cause the Secured Obligations to constitute senior indebtedness (however denominated) in respect of such Subordinated Indebtedness and to enable the Administrative Agent and the Lenders to have and exercise any payment blockage or other remedies available or potentially available to holders of senior indebtedness under the terms of such Subordinated Indebtedness. Without limiting the foregoing, the Secured Obligations are hereby designated as “senior indebtedness” and as “designated senior indebtedness” and words of similar import under and in respect of any indenture or other agreement or instrument under which such other Subordinated Indebtedness is outstanding and are further given all such other designations as shall be required under the terms of any such Subordinated Indebtedness in order that the Lenders may have and exercise any payment blockage or other remedies available or potentially available to holders of senior indebtedness under the terms of such Subordinated Indebtedness. Section 1.06 Special Luxembourg Provisions. Without prejudice to the generality of any provision of this Agreement, to the extent this Agreement relates to any Luxembourg Guarantor, a reference to: (a) bankruptcy, conservatorship, liquidation, insolvency, reorganization or dissolution includes, without limitation, bankruptcy (faillite), insolvency, voluntary or judicial liquidation (liquidation volontaire ou judiciaire), moratorium or reprieve from payment (sursis de paiement), administrative dissolution without liquidation (dissolution administrative sans liquidation), out-of-court mutual agreement (réorganisation extra-judiciaire par accord amiable), judicial reorganization in the form of a stay to enter into a mutual agreement (sursis en vue de la conclusion d'un accord amiable), judicial reorganization by collective agreement (réorganisation judiciaire par accord collectif), judicial reorganization by transfer of assets or activities (réorganisation judiciaire par transfert sous autorité de justice), conciliation (conciliation) general settlement with creditors, reorganization or any other similar proceedings affecting the rights of creditors generally under Luxembourg law, and shall be construed so as to include any equivalent or analogous liquidation or reorganization proceedings; (b) a receiver, liquidator, administrator, trustee, custodian, sequestrator, conservator or similar officer includes, without limitation, a juge-commissaire or curateur appointed under the Luxembourg Commercial Code, liquidateur appointed under articles 1100-1 to 1100-15 (inclusive) of the Luxembourg law dated 10 August 1915 on commercial companies, as amended (the “Luxembourg Law on Commercial Companies”), juge-commissaire or liquidateur appointed under article 1200-1 of the Luxembourg Law on Commercial Companies, mandataire judiciaire or conciliateur under the Luxembourg law of 7 August 2023 modernizing bankruptcy or any similar officer pursuant to any insolvency or similar proceedings; (c) a person being unable to pay its debts includes that person being in a state of cessation of payments (cessation de paiements) without having access to credit; (d) attachments or similar creditors process means an executory attachment (saisie exécutoire) or conservatory attachment (saisie conservatoire); (e) a guaranty includes any guaranty that is independent from the debt to which it relates and excludes any suretyship (cautionnement) within the meaning of Articles 2011 and seq. of the Luxembourg Civil Code; (f) a security, a security interest or a Lien includes, without limitation, any hypothèque, nantissement, gage, privilège, accord de transfert de propriété à titre de garantie, gage sur fonds de commerce, droit de rétention and any type of

85 security in rem (sûreté réelle) whatsoever whether granted or arising by operation of law; (g) Organizational Documents includes its up-to-date (restated) articles of association (statuts coordonnés); (h) an officer or a director includes an administrateur; (i) an agent includes a mandataire, (j) Capital Stock includes actions; (k) gross negligence means faute lourde; and (l) wilful misconduct means dol or faute dolosive. Section 1.07 Cashless Rollovers. Notwithstanding anything to the contrary contained in this Agreement or in any other Loan Document, to the extent that any Lender extends the maturity date of, or replaces, renews or refinances, any of its then-existing Loans with Incremental Loans, Extended Loans or Other Refinancing Loans or loans incurred under a new credit facility, in each case, to the extent such extension, replacement, renewal or refinancing is effected by means of a “cashless roll” by such Lender, such extension, replacement, renewal or refinancing shall be deemed to comply with any requirement hereunder or any other Loan Document that such payment be made “in Dollars”, “in immediately available funds”, “in cash” or any other similar requirement. Section 1.08 Divisions. For all purposes under the Loan Documents, in connection with any division or plan of division under the laws of the State of Delaware (or any comparable event under a different jurisdiction’s laws): (a) any reference to a merger, transfer, consolidation, amalgamation, consolidation, assignment, sale, disposition or transfer, or similar term, shall be deemed to apply to a division of or by a limited liability company, or an allocation of assets to a series of a limited liability company (or the unwinding of such a division or allocation), as if it were a merger, transfer, consolidation, amalgamation, consolidation, assignment, sale, disposition or transfer, or similar term, as applicable, to, of or with a separate Person and (b) any division of a limited liability company shall constitute a separate Person hereunder (and each division of any limited liability company that is a Subsidiary, joint venture or any other like term shall also constitute such a Person or entity). ARTICLE II THE CREDITS Section 2.01 Commitments and Loans. Subject to the terms and conditions set forth herein: (a) (i) each Initial Term Lender agrees, severally and not jointly, to make an Initial Term Loan to the Borrower Representative on the Effective Date in a principal amount not to exceed its Initial Term Loan Commitment listed on Schedule 2.01 and (ii) each 2024 Refinancing Term Lender agrees, severally and not jointly, to make a 2024 Refinancing Term Loan to the Borrower Representative on the First Amendment Effective Date as set forth in the First Amendment; (b) each Dollar Tranche Lender agrees to make Dollar Tranche Revolving Loans to the Borrower Representative or, subject to Section 9.18(a), any other Borrower, in Dollars from time to time during the Availability Period in an aggregate principal amount that will not result in (i) such Lender’s Dollar Tranche Revolving Credit Exposure exceeding such Lender’s Dollar Tranche Commitment, (ii) the sum of the total Dollar Tranche Revolving Credit Exposures exceeding the aggregate Dollar Tranche Commitments or (iii) subject

86 to Sections 2.04 and 2.11(b), the sum of the Dollar Amount of the total Revolving Credit Exposures exceeding the aggregate Revolving Commitments; and (c) each Multicurrency Tranche Lender agrees to make Multicurrency Tranche Revolving Loans to the Borrower Representative or, subject to Section 9.18(a), any other Borrower, in Agreed Currencies from time to time during the Availability Period in an aggregate principal amount that will not result in (i) subject to Sections 2.04 and 2.11(b), the Dollar Amount of such Lender’s Multicurrency Tranche Revolving Credit Exposure exceeding such Lender’s Multicurrency Tranche Commitment, (ii) subject to Sections 2.04 and 2.11(b), the sum of the Dollar Amount of the total Multicurrency Tranche Revolving Credit Exposures exceeding the aggregate Multicurrency Tranche Commitments, (iii) subject to Sections 2.04 and 2.11(b), the sum of the Dollar Amount of the total Revolving Credit Exposures exceeding the aggregate Revolving Commitments or (iv) subject to Sections 2.04 and 2.11(b), the sum of the Dollar Amount of the Non-USD Multicurrency Tranche Revolving Credit Exposures exceeding the Non-USD Multicurrency Tranche Sublimit. Within the foregoing limits and subject to the terms and conditions set forth herein, any Borrower may borrow, prepay and reborrow Dollar Tranche Revolving Loans and Multicurrency Tranche Revolving Loans. The full amount of each Class of Term Loan Commitments must be drawn in a single drawing on the closing date thereof and amounts repaid or prepaid in respect of Term Loans may not be reborrowed. Each Lender may, at its option, make any Loan available to the applicable Borrower by causing any foreign or domestic branch or Affiliate of such Lender to make such Loan (in which case such branch or Affiliate shall be treated as the “Lender” with respect to such Loan for all purposes of this Agreement); provided that (x) any exercise of such option shall not affect the obligation of the applicable Borrower to repay such Loan in accordance with the terms of this Agreement and (y) if the respective branch or Affiliate is a Foreign Lender, the same shall be capable of making the representation contained in the last sentence of Section 2.17(l) on the date it first becomes such a “Lender”. Section 2.02 Loans and Borrowings. (a) Each Loan (other than a Swingline Loan) shall be made as part of a Borrowing consisting of Loans of the same Class and Type made under a single Tranche and shall be made by the Lenders of such Class under such Tranche ratably in accordance with their respective Commitments in respect of the applicable Class and in respect of the applicable Tranche. The failure of any Lender to make any Loan required to be made by it shall not relieve any other Lender of its obligations hereunder; provided that the Commitments of the Lenders are several and no Lender shall be responsible for any other Lender’s failure to make Loans as required. Any Swingline Loan shall be made in accordance with the procedures set forth in Section 2.05. The Initial Term Loans and the 2024 Refinancing Term Loans shall amortize as set forth in Section 2.10. (b) Subject to Section 2.14, each Dollar Tranche Revolving Borrowing, each Multicurrency Tranche Revolving Borrowing (other than a Borrowing of Canadian Dollar Revolving Loans) and each Term Loan Borrowing shall be comprised entirely of ABR Loans or Term SOFR Loans as the Borrower may request in accordance herewith; provided that each ABR Loan shall only be made in Dollars. Subject to Section 2.14, each Multicurrency Tranche

87 Revolving Borrowing in Canadian Dollars shall be comprised entirely of Canadian Prime Rate Loans or Term CORRA Loans as the Borrower may request in accordance herewith. Each Swingline Loan shall be an ABR Loan. Each Lender at its option may make any Term Benchmark Loan by causing any domestic or foreign branch or Affiliate of such Lender to make such Loan (and in the case of an Affiliate, the provisions of Sections 2.14, 2.15, 2.16 and 2.17 shall apply to such Affiliate to the same extent as to such Lender); provided that any exercise of such option shall not affect the obligation of the respective Borrower to repay such Loan in accordance with the terms of this Agreement. (c) Each Canadian Dollar Revolving Loan shall be incurred and maintained as, and/or converted into one or more Borrowings of Term CORRA Loans, in each case on the terms and conditions provided for herein. (d) At the commencement of each Interest Period for any Borrowing of Term SOFR Revolving Loans and each Borrowing of Term CORRA Loans, such Borrowing shall be in an aggregate amount that is an integral multiple of $500,000 (or CAD $500,000) (or, if such Borrowing is denominated in a Foreign Currency, 500,000 units of such currency other than Japanese Yen and ¥50,000,000 in the case of Japanese Yen) and not less than $2,000,000 (or CAD $2,000,000) (or, if such Borrowing is denominated in a Foreign Currency, 2,000,000 units of such currency other than Japanese Yen and ¥200,000,000 in the case of Japanese Yen). At the time that each ABR Revolving Borrowing or Canadian Prime Rate Borrowing is made, such Borrowing shall be in an aggregate amount that is an integral multiple of $500,000 and not less than $1,000,000; provided that an ABR Revolving Borrowing or Canadian Prime Rate Borrowing may be in an aggregate amount that is equal to the entire unused balance of the aggregate Dollar Tranche Commitments of the relevant Class (with respect to any ABR Borrowing) or the aggregate Multicurrency Tranche Commitments of the relevant Class, as the case may be, or that is required to finance the reimbursement of an LC Disbursement as contemplated by Section 2.06(e). Each Swingline Loan shall be in an amount that is an integral multiple of $500,000 and not less than $1,000,000. Borrowings of more than one Type and Class and under more than one Tranche may be outstanding at the same time; provided that (x) there shall not at any time be more than a total of ten (10) Term SOFR Revolving Borrowings outstanding and (y) there shall not at any time be more than a total of ten (10) Borrowings of Term CORRA Loans outstanding. (e) Notwithstanding any other provision of this Agreement, the Borrower shall not be entitled to request, or to elect to convert or continue, any Borrowing of any Class if the Interest Period requested with respect thereto would end after the Maturity Date of such Class. Section 2.03 Requests for Borrowings. To request a Borrowing, a Borrower shall notify the Administrative Agent of such request (a) by irrevocable written notice (via a written Borrowing Request in a form approved by the Administrative Agent and signed by such Borrower, promptly followed by telephonic confirmation of such request) in the case of a Term Benchmark Borrowing, not later than 10:00 a.m., Local Time, three (3) Business Days (in the case of a Term Benchmark Borrowing denominated in Dollars) or by irrevocable written notice (via a written Borrowing Request in a form approved by the Administrative Agent and signed by such Borrower) not later than four (4) Business Days (in the case of a Term Benchmark Borrowing denominated in a Foreign Currency), in each case before the date of the proposed Borrowing (or, with respect to

88 Borrowings to be made on the Effective Date, such shorter time as the Administrative Agent may agree in its sole discretion) or (b) by telephone in the case of an ABR Borrowing or Canadian Prime Rate Borrowing, not later than 11:00 a.m., New York City time, one (1) Business Day before the date of the proposed Borrowing; provided that any such notice of an ABR Revolving Borrowing to finance the reimbursement of an LC Disbursement as contemplated by Section 2.06(e) may be given not later than 10:00 a.m., New York City time, on the date of the proposed Borrowing. Each such telephonic Borrowing Request shall be irrevocable and shall be confirmed promptly by hand delivery or telecopy to the Administrative Agent of a written Borrowing Request in a form approved by the Administrative Agent and signed by the applicable Borrower. Each such telephonic and written Borrowing Request shall specify the following information in compliance with Section 2.02: (a) the aggregate amount of the requested Borrowing; (b) the date of such Borrowing, which shall be a Business Day; (c) the Class of such Borrowing and whether such Borrowing is to be an ABR Borrowing, Canadian Prime Rate Borrowing, a Term SOFR Borrowing or a Borrowing of Term CORRA Loans and, if such Borrowing is a Revolving Borrowing, whether such Borrowing is to be a Dollar Tranche Revolving Borrowing or Multicurrency Tranche Revolving Borrowing; (d) in the case of a Term Benchmark Borrowing, the Agreed Currency (which shall comply with the limitation set forth in Section 2.01(c)(iv)) and initial Interest Period to be applicable thereto, which shall be a period contemplated by the definition of the term “Interest Period”; and (e) the location and number of the applicable Borrower’s account to which funds are to be disbursed, which shall comply with the requirements of Section 2.07. If no election as to the Type of Borrowing is specified, then, (x) in the case of a Borrowing denominated in Dollars, the requested Borrowing shall be an ABR Borrowing and (y) in the case of a Borrowing denominated in Canadian Dollars, the requested Borrowing shall be a Borrowing of Term CORRA Loans. If no Interest Period is specified with respect to any requested Term Benchmark Borrowing, then the applicable Borrower shall be deemed to have selected an Interest Period of one month’s duration. Promptly following receipt of a Borrowing Request in accordance with this Section 2.03, the Administrative Agent shall advise each applicable Lender of the details thereof and of the amount of such Lender’s Loan to be made as part of the requested Borrowing. Section 2.04 Determination of Dollar Amounts. The Administrative Agent will determine the Dollar Amount of: (a) each Multicurrency Tranche Revolving Borrowing and each Borrowing of Term CORRA Loans utilizing Revolving Commitments, in each case as of the date two (2) Business Days prior to the date of such Borrowing or, if applicable, the date of conversion/continuation of any such Borrowing as a Multicurrency Tranche Revolving Borrowing or a Borrowing of Term CORRA Loans (as the case may be),

89 (b) the LC Exposure as of the date of each request for the issuance, amendment or extension of any Letter of Credit, and (c) all outstanding Revolving Credit Exposure on and as of the last Business Day of each calendar quarter and, during the continuation of an Event of Default, on any other Business Day elected by the Administrative Agent in its discretion or upon instruction by the Required Lenders. Such Dollar Amount shall become effective as of such Revaluation Date and, if applicable, shall be the Dollar Amount of such amounts until the next Revaluation Date to occur. Section 2.05 Swingline Loans. (a) Subject to the terms and conditions set forth herein, the Swingline Lender agrees to make Swingline Loans in Dollars to the Borrower from time to time during the Availability Period, in an aggregate principal amount at any time outstanding that will not result in (i) the aggregate principal amount of outstanding Swingline Loans exceeding $50,000,000 (as such amount may be increased from time to time, but not above $75,000,000, with the consent of the Administrative Agent (such consent not to be unreasonably withheld) and the Swingline Lender), (ii) the Dollar Amount of the total Dollar Tranche Revolving Credit Exposures exceeding the aggregate Dollar Tranche Commitments or (iii) the Dollar Amount of the total Revolving Credit Exposures exceeding the aggregate Revolving Commitments; provided that the Swingline Lender shall not be required to make a Swingline Loan to refinance an outstanding Swingline Loan. Within the foregoing limits and subject to the terms and conditions set forth herein, the Borrower may borrow, prepay and reborrow Swingline Loans. (b) To request a Swingline Loan, a Borrower shall notify the Administrative Agent of such request by telephone (confirmed by telecopy), not later than 2:00 p.m., New York City time, on the day of a proposed Swingline Loan. Each such notice shall be irrevocable and shall specify the requested date (which shall be a Business Day) and amount of the requested Swingline Loan. The Administrative Agent will promptly advise the Swingline Lender of any such notice received from a Borrower. The Swingline Lender shall make each Swingline Loan available to the applicable Borrower by means of a credit to the general deposit account of such Borrower with the Swingline Lender (or, in the case of a Swingline Loan made to finance the reimbursement of an LC Disbursement as provided in Section 2.06(e), by remittance to the Issuing Bank) by 3:00 p.m., New York City time, on the requested date of such Swingline Loan. (c) The Swingline Lender may by written notice given to the Administrative Agent not later than 10:00 a.m., New York City time, on any Business Day require the Dollar Tranche Lenders to acquire participations on such Business Day in all or a portion of the Swingline Loans outstanding. Such notice shall specify the aggregate amount of Swingline Loans in which Dollar Tranche Lenders will participate. Promptly upon receipt of such notice, the Administrative Agent will give notice thereof to each Dollar Tranche Lender, specifying in such notice such Lender’s Dollar Tranche Percentage of such Swingline Loan or Loans. Each Dollar Tranche Lender hereby absolutely and unconditionally agrees, upon receipt of notice as provided above, to pay to the Administrative Agent, for the account of the Swingline Lender, such Dollar Tranche Lender’s Dollar Tranche Percentage (after giving effect to the reallocation provisions of paragraph (d) below) of such Swingline Loan or Loans. Each Dollar Tranche Lender

90 acknowledges and agrees that its obligation to acquire participations in Swingline Loans pursuant to this paragraph in an amount equal to its Dollar Tranche Percentage thereof is absolute and unconditional and shall not be affected by any circumstance whatsoever, including the occurrence and continuance of a Default or reduction or termination of the Dollar Tranche Commitments, and that each such payment shall be made without any offset, abatement, withholding or reduction whatsoever. Each Dollar Tranche Lender shall comply with its obligation under this paragraph by wire transfer of immediately available funds, in the same manner as provided in Section 2.07 with respect to Loans made by such Lender (and Section 2.07 shall apply, mutatis mutandis, to the payment obligations of the Dollar Tranche Lenders), and the Administrative Agent shall promptly pay to the Swingline Lender the amounts so received by it from the Dollar Tranche Lenders. The Administrative Agent shall notify the Borrower of any participations in any Swingline Loan acquired pursuant to this paragraph, and thereafter payments in respect of such Swingline Loan shall be made to the Administrative Agent and not to the Swingline Lender. Any amounts received by the Swingline Lender from a Borrower (or other party on behalf of such Borrower) in respect of a Swingline Loan after receipt by the Swingline Lender of the proceeds of a sale of participations therein shall be promptly remitted to the Administrative Agent; any such amounts received by the Administrative Agent shall be promptly remitted by the Administrative Agent to the applicable Dollar Tranche Lenders that shall have made their payments pursuant to this paragraph and to the Swingline Lender, as their interests may appear; provided that any such payment so remitted shall be repaid to the Swingline Lender or to the Administrative Agent, as applicable, if and to the extent such payment is required to be refunded to the Borrower for any reason. The purchase of participations in a Swingline Loan pursuant to this paragraph shall not relieve the Borrower of any default in the payment thereof. (d) Reallocations and Extensions. If the Maturity Date shall have occurred in respect of any Class of Revolving Commitments at a time when another Tranche or Tranches of any other Class of Revolving Commitments is or are in effect with a longer Maturity Date, then on the earliest occurring Maturity Date all then-outstanding Swingline Loans shall be repaid in full (and there shall be no adjustment to the participations in such Swingline Loans as a result of the occurrence of such earliest Maturity Date); provided, however, that if on the occurrence of such earliest Maturity Date (after giving effect to any repayments of Revolving Loans and any reallocation of Letter of Credit participations as contemplated in Section 2.06(k)), there shall exist sufficient unutilized Revolving Commitments of any other Class or Classes or Extended Revolving Commitments so that the respective outstanding Swingline Loans could be incurred pursuant to such Revolving Commitments of such other Class or Classes or Extended Revolving Commitments which will remain in effect after the occurrence of such earliest Maturity Date, then there shall be an automatic adjustment on such date of the risk participations of each Revolving Lender holding Revolving Commitments of such other Class or Classes or that is an Extending Revolving Lender and such outstanding Swingline Loans shall be deemed to have been incurred solely pursuant to the relevant Revolving Commitments of such other Class or Classes or Extended Revolving Commitments and such Swingline Loans shall not be so required to be repaid in full on such earliest Maturity Date. Section 2.06 Letters of Credit. (a) General. Subject to the terms and conditions set forth herein, the Borrower may request the issuance of Multicurrency Tranche Letters of Credit denominated in

91 Agreed Currencies and Dollar Tranche Letters of Credit denominated in Dollars, in each case for its own account, in a form reasonably acceptable to the Administrative Agent and the Issuing Bank, at any time and from time to time during the Availability Period. Any letters of credit issued (or deemed to be issued) by any Lender party hereto as set forth on Schedule 2.06 hereto and outstanding as of the Effective Date (the “Existing Letters of Credit”) shall continue to be “Letters of Credit” (constituting (x) Dollar Tranche Letters of Credit, if denominated in Dollars and (y) Multicurrency Tranche Letters of Credit, if denominated in any Agreed Currency) for all purposes of the Loan Documents. In the event of any inconsistency between the terms and conditions of this Agreement and the terms and conditions of any form of letter of credit application or other agreement submitted by a Borrower to, or entered into by such Borrower with, the Issuing Bank relating to any Letter of Credit, the terms and conditions of this Agreement shall control. (b) Notice of Issuance, Amendment, Extension; Certain Conditions. To request the issuance of a Letter of Credit (or an amendment to an outstanding Letter of Credit), a Borrower shall hand deliver or telecopy (or transmit by electronic communication, if arrangements for doing so have been approved by the Issuing Bank) to the Issuing Bank and the Administrative Agent (reasonably in advance of, but not less than five (5) Business Days prior to, the requested date of issuance) a notice in the form of Exhibit D requesting the issuance of a Letter of Credit or amendment and specifying the date of issuance (which shall be a Business Day), the date on which such Letter of Credit is to expire (which shall comply with paragraph (c) of this Section 2.06), the amount of such Letter of Credit, the Agreed Currency applicable thereto (subject to compliance with the limitation set forth in Section 2.01(c)(iv)), whether such Letter of Credit is a Multicurrency Tranche Letter of Credit or a Dollar Tranche Letter of Credit, the name and address of the beneficiary thereof and such other information as shall be necessary to prepare such Letter of Credit or amendment. A Borrower also shall submit a letter of credit application on the Issuing Bank’s standard form and related documents in connection with any request for a Letter of Credit and in connection with any request for a Letter of Credit amendment. A Letter of Credit or amendment shall be issued only (A) if (and upon issuance of each Letter of Credit and amendment, the applicable Borrower shall be deemed to represent and warrant that), after giving effect to such issuance (i) subject to Sections 2.04 and 2.11(b), the Dollar Amount of the LC Exposure shall not exceed $50,000,000 (as such amount may be increased from time to time, but not above $75,000,000, with the consent of the Administrative Agent (such consent not to be unreasonably withheld) and each Issuing Bank (other than an Issuing Bank solely with respect to Existing Letters of Credit)), (ii) subject to Sections 2.04 and 2.11(b), the sum of the Dollar Amount of the total Multicurrency Tranche Revolving Credit Exposures shall not exceed the aggregate Multicurrency Tranche Commitments, (iii) the sum of the Dollar Tranche Revolving Credit Exposure shall not exceed the aggregate Dollar Tranche Commitments and (iv) the sum of the total Revolving Credit Exposures shall not exceed the aggregate Revolving Commitments and (B) in accordance with the Issuing Bank’s usual and customary practices and policies applicable to letters of credit in general from time to time. (c) Expiration Date. Each Letter of Credit shall expire at or prior to the close of business on the earlier of (i) the date one year after the date of the issuance of such Letter of Credit (or, in the case of any extension thereof, one year after such extension) and (ii) the date that is five (5) Business Days prior to the Maturity Date with respect to the Revolving Commitments pursuant to which issued (or if any Extended Revolving Commitments, Incremental Revolving Commitments or Other Refinancing Revolving Commitments are outstanding, the last

92 Maturity Date applicable thereto (so long as the aggregate amount of such Letters of Credit are not in excess of such commitments)); provided that any Letter of Credit may contain customary automatic extension provisions agreed upon by the respective Borrower and the Issuing Bank pursuant to which the expiration date of such Letter of Credit (an “Auto-Extension Letter of Credit”) shall automatically be extended for consecutive periods of up to twelve (12) months (but not to a date later than the date set forth in clause (ii) above); provided that any such Auto- Extension Letter of Credit must permit the Issuing Bank to prevent any such extension at least once in each twelve month period (commencing with the date of issuance of such Letter of Credit) by giving prior notice to the beneficiary thereof not later than a day in each such twelve month period to be agreed upon at the time such Letter of Credit is issued. Unless otherwise directed by the Issuing Bank, the respective Borrower shall not be required to make a specific request to the Issuing Bank for any such extension. Once an Auto-Extension Letter of Credit has been issued, the Lenders shall be deemed to have authorized (but may not require) the Issuing Bank to permit the extension of such Letter of Credit at any time to an expiry date not later than such Maturity Date. (d) Participations. By the issuance of a Letter of Credit (or an amendment to a Letter of Credit increasing the amount thereof) and without any further action on the part of the Issuing Bank or any Revolving Lender in respect of the Tranche under which such Letter of Credit is issued (each such Revolving Lender, an “Applicable Lender”), the Issuing Bank hereby grants to each Applicable Lender, and each Applicable Lender hereby acquires from the Issuing Bank, a participation in such Letter of Credit equal to such Applicable Lender’s Applicable Percentage of the aggregate Dollar Amount available to be drawn under such Letter of Credit. In consideration and in furtherance of the foregoing, each Applicable Lender hereby absolutely and unconditionally agrees to pay to the Administrative Agent, for the account of the Issuing Bank, such Applicable Lender’s Applicable Percentage (after giving effect to the reallocation provisions of paragraph (k) below) of each LC Disbursement made by the Issuing Bank and not reimbursed by the Borrower on the date due as provided in paragraph (e) of this Section 2.06, or of any reimbursement payment required to be refunded to the Borrower for any reason. Each Revolving Lender acknowledges and agrees that its obligation to acquire participations pursuant to this paragraph in respect of Letters of Credit is absolute and unconditional and shall not be affected by any circumstance whatsoever, including any amendment, reinstatement or extension of any Letter of Credit or the occurrence and continuance of a Default or reduction or termination of the Commitments, and that each such payment shall be made without any offset, abatement, withholding or reduction whatsoever. (e) Reimbursement. If the Issuing Bank shall make any LC Disbursement in respect of a Letter of Credit, the respective Borrower shall reimburse such LC Disbursement by paying to the Issuing Bank in Dollars the Dollar Amount equal to such LC Disbursement, calculated as of the date the Issuing Bank made such LC Disbursement (or if the Issuing Bank shall so elect in its sole discretion by notice to the respective Borrower, in such other Agreed Currency which was paid by the Issuing Bank pursuant to such LC Disbursement in an amount equal to such LC Disbursement) not later than 12:00 noon, Local Time, on the date that such LC Disbursement is made, if a Borrower shall have received notice of such LC Disbursement prior to 10:00 a.m., Local Time, on such date, or, if such notice has not been received by a Borrower prior to such time on such date, then not later than 12:00 noon, Local Time, on the Business Day immediately following the day that a Borrower receives such notice, if such notice

93 is not received prior to such time on the day of receipt; provided that, if such LC Disbursement is not less than the Dollar Amount of $1,000,000, the Borrower may, subject to the conditions to borrowing set forth herein, request in accordance with Section 2.03 or 2.05 that such payment be financed with an ABR Revolving Borrowing or Swingline Loan in an equivalent Dollar Amount of such LC Disbursement and, to the extent so financed, the Borrower’s obligation to make such payment shall be discharged and replaced by the resulting ABR Revolving Borrowing or Swingline Loan. If the Borrower fails to make such payment when due, the Administrative Agent shall notify each Applicable Lender of the applicable LC Disbursement, the payment then due from the Borrower in respect thereof and such Lender’s Applicable Percentage thereof. Promptly following receipt of such notice, each Applicable Lender shall pay to the Administrative Agent its Applicable Percentage (after giving effect to the reallocation provisions of paragraph (k) below) of the payment then due from the Borrower, in the same manner as provided in Section 2.07 with respect to Loans made by such Lender (and Section 2.07 shall apply, mutatis mutandis, to the payment obligations of the Applicable Lenders), and the Administrative Agent shall promptly pay to the Issuing Bank the amounts so received by it from the Applicable Lenders. Promptly following receipt by the Administrative Agent of any payment from the Borrower pursuant to this paragraph, the Administrative Agent shall distribute such payment to the Issuing Bank or, to the extent that Applicable Lenders have made payments pursuant to this paragraph to reimburse the Issuing Bank, then to such Lenders and the Issuing Bank as their interests may appear. Any payment made by an Applicable Lender pursuant to this paragraph to reimburse the Issuing Bank for any LC Disbursement (other than the funding of ABR Revolving Loans or a Swingline Loan as contemplated above) shall not constitute a Loan and shall not relieve the Borrower of its obligation to reimburse such LC Disbursement. If the Borrower’s reimbursement of, or obligation to reimburse, any amounts in any Foreign Currency would subject the Administrative Agent, the Issuing Bank or any Multicurrency Tranche Lender to any stamp duty, ad valorem charge or similar tax that would not be payable if such reimbursement were made or required to be made in Dollars, the Borrower shall, at its option, either (x) pay the amount of any such tax requested by the Administrative Agent, the Issuing Bank or the relevant Multicurrency Tranche Lender or (y) reimburse each LC Disbursement made in such Foreign Currency in Dollars, in an amount equal to the Equivalent Amount, calculated using the applicable exchange rates, on the date such LC Disbursement is made, of such LC Disbursement. (f) Obligations Absolute. The Borrower’s obligation to reimburse LC Disbursements as provided in paragraph (e) of this Section 2.06 shall be absolute, unconditional and irrevocable, and shall be performed strictly in accordance with the terms of this Agreement under any and all circumstances whatsoever and irrespective of (i) any lack of validity or enforceability of any Letter of Credit or this Agreement, or any term or provision therein, (ii) any draft or other document presented under a Letter of Credit proving to be forged, fraudulent or invalid in any respect or any statement therein being untrue or inaccurate in any respect, (iii) payment by the Issuing Bank under a Letter of Credit against presentation of a draft or other document that does not comply with the terms of such Letter of Credit, or (iv) any other event or circumstance whatsoever, whether or not similar to any of the foregoing, that might, but for the provisions of this Section 2.06, constitute a legal or equitable discharge of, or provide a right of setoff against, the Borrower’s obligation hereunder. Neither the Administrative Agent, the Revolving Lenders nor the Issuing Bank, nor any of their Related Parties, shall have any liability or responsibility by reason of or in connection with the issuance or transfer of any Letter of Credit or any payment or failure to make any payment thereunder (irrespective of any of the

94 circumstances referred to in the preceding sentence), or any error, omission, interruption, loss or delay in transmission or delivery of any draft, notice or other communication under or relating to any Letter of Credit (including any document required to make a drawing thereunder), any error in interpretation of technical terms, any error in translation, or any consequence arising from causes beyond the control of the Issuing Bank; provided that the foregoing shall not be construed to excuse the Issuing Bank from liability to the respective Borrower to the extent of any direct damages (as opposed to consequential damages, claims in respect of which are hereby waived by the Borrower to the extent permitted by applicable law) suffered by the respective Borrower that are caused by the Issuing Bank’s failure to exercise care when determining whether drafts and other documents presented under a Letter of Credit comply with the terms thereof. The parties hereto expressly agree that, in the absence of gross negligence or willful misconduct on the part of the Issuing Bank (as finally determined by a court of competent jurisdiction), the Issuing Bank shall be deemed to have exercised care in each such determination. In furtherance of the foregoing and without limiting the generality thereof, the parties agree that, with respect to documents presented which appear on their face to be in substantial compliance with the terms of a Letter of Credit, the Issuing Bank may, in its sole discretion, either accept and make payment upon such documents without responsibility for further investigation, regardless of any notice or information to the contrary, or refuse to accept and make payment upon such documents if such documents are not in strict compliance with the terms of such Letter of Credit. (g) Disbursement Procedures. The Issuing Bank shall, promptly following its receipt thereof, examine all documents purporting to represent a demand for payment under a Letter of Credit. The Issuing Bank shall promptly notify the Administrative Agent and the respective Borrower by telephone (confirmed by telecopy) of such demand for payment if the Issuing Bank has made or will make an LC Disbursement thereunder; provided that any failure to give or delay in giving such notice shall not relieve the Borrower of its obligations to reimburse the Issuing Bank and the Applicable Lenders with respect to any such LC Disbursement. (h) Interim Interest. If the Issuing Bank shall make any LC Disbursement, then, unless the Borrower shall reimburse such LC Disbursement in full on the date such LC Disbursement is made, the unpaid amount thereof shall bear interest, for each day from and including the date such LC Disbursement is made to but excluding the date that the Borrower reimburse such LC Disbursement, at the rate per annum then applicable to ABR Revolving Loans (or in the case such LC Disbursement is denominated in a Foreign Currency, at the Overnight Foreign Currency Rate for such Agreed Currency plus the then effective Applicable Rate with respect to Term SOFR Revolving Loans); provided that, if the Borrower fails to reimburse such LC Disbursement when due pursuant to paragraph (e) of this Section 2.06, then Section 2.13(d) shall apply. Interest accrued pursuant to this paragraph shall be for the account of the Issuing Bank, except that interest accrued on and after the date of payment by any Applicable Lender pursuant to paragraph (e) of this Section 2.06 to reimburse the Issuing Bank shall be for the account of such Applicable Lender to the extent of such payment. (i) Replacement and Resignation of the Issuing Bank. (i) The Issuing Bank may be replaced at any time by written agreement among the Borrower, the Administrative Agent, the replaced Issuing Bank and the successor Issuing Bank. The Administrative Agent shall notify the Revolving Lenders

95 of any such replacement of the Issuing Bank. At the time any such replacement shall become effective, the Borrower shall pay all unpaid fees accrued for the account of the replaced Issuing Bank pursuant to Section 2.12(b). From and after the effective date of any such replacement, (x) the successor Issuing Bank shall have all the rights and obligations of the Issuing Bank under this Agreement with respect to Letters of Credit to be issued thereafter and (y) references herein to the term “Issuing Bank” shall be deemed to refer to such successor or to any previous Issuing Bank, or to such successor and all previous Issuing Banks, as the context shall require. After the replacement of an Issuing Bank hereunder, the replaced Issuing Bank shall remain a party hereto and shall continue to have all the rights and obligations of an Issuing Bank under this Agreement with respect to Letters of Credit then outstanding and issued by it prior to such replacement, but shall not be required to issue additional Letters of Credit. (ii) Subject to the appointment and acceptance of a successor Issuing Bank, the Issuing Bank may resign as an Issuing Bank at any time upon thirty days’ prior written notice to the Administrative Agent, the Borrower and the Lenders, in which case, the resigning Issuing Bank shall be replaced in accordance with Section 2.06(i)(i) above. (j) Cash Collateralization. If any Event of Default shall occur and be continuing, on the Business Day that a Borrower receives notice from the Administrative Agent or the Required Lenders (or, if the maturity of the Loans has been accelerated, the Required Revolving Lenders) demanding the deposit of cash collateral pursuant to this paragraph, the Borrower shall deposit in an account with the Administrative Agent, in the name of the Administrative Agent and for the benefit of the Revolving Lenders (the “LC Collateral Account”), an amount in cash equal to 103% of the Dollar Amount of the LC Exposure as of such date plus any accrued and unpaid interest thereon; provided that (i) the portions of such amount attributable to undrawn Foreign Currency Letters of Credit or LC Disbursements in a Foreign Currency that the Borrower is not late in reimbursing shall be deposited in the applicable Foreign Currencies in the actual amounts of such undrawn Letters of Credit and LC Disbursements and (ii) the obligation to deposit such cash collateral shall become effective immediately, and such deposit shall become immediately due and payable, without demand or other notice of any kind, upon the occurrence of any Event of Default with respect to the Borrower described in clause (h) or (i) of Section 7.01. For the purposes of this paragraph, the Foreign Currency LC Exposure shall be calculated using the applicable Exchange Rate on the date notice demanding cash collateralization is delivered to the Borrower. The Borrower also shall deposit cash collateral pursuant to this paragraph as and to the extent required by Section 2.11(b). Such deposit shall be held by the Administrative Agent as collateral for the payment and performance of the Secured Obligations. The Administrative Agent shall have exclusive dominion and control, including the exclusive right of withdrawal, over such account and the Borrower hereby grants the Administrative Agent, for the ratable benefit of the Secured Parties, a security interest in the LC Collateral Account. Other than any interest earned on the investment of such deposits, which investments shall be made at the option and sole discretion of the Administrative Agent and at the Borrower’s risk and expense, such deposits shall not bear interest. Interest or profits, if any, on such investments shall accumulate in such account. Moneys in such account shall be applied by the Administrative Agent to reimburse the Issuing Bank for LC Disbursements for which it has not been reimbursed and, to the extent not so applied, shall be held for the satisfaction of the reimbursement obligations of the Borrower for the LC

96 Exposure at such time or, if the maturity of the Loans has been accelerated (but subject to the consent of the Required Revolving Lenders), be applied to satisfy other Secured Obligations. If the Borrower is required to provide an amount of cash collateral hereunder as a result of the occurrence of an Event of Default, such amount (to the extent not applied as aforesaid) shall be returned to the Borrower within three (3) Business Days after all Events of Default have been cured or waived. If the Borrower is required to provide an amount of cash collateral hereunder pursuant to Section 2.11(b), such amount (to the extent not applied as aforesaid) shall be returned to the Borrower as and to the extent that, after giving effect to such return, the aggregate Revolving Credit Exposures would not exceed the aggregate Revolving Commitments and no Default shall have occurred and be continuing. (k) Reallocations and Extensions. If the Maturity Date in respect of any Class of Revolving Commitments occurs prior to the expiration of any Letter of Credit, then (i) if Extended Revolving Commitments or one or more other Tranches of Revolving Commitments of any other Class or Classes in respect of which the Maturity Date shall not have occurred are then in effect, such Letters of Credit shall automatically be deemed to have been issued (including for purposes of the obligations of the Revolving Lenders to purchase participations therein and to make Revolving Loans and payments in respect thereof pursuant to Section 2.06(e)) under (and ratably participated in by Revolving Lenders pursuant to) Extended Revolving Commitments or the Revolving Commitments of such other Class or Classes in respect of such non-terminating Extended Revolving Commitments or Tranches up to an aggregate amount not to exceed the aggregate principal amount of the unutilized Extended Revolving Commitments or Revolving Commitments of such other Class or Classes thereunder at such time (it being understood that no partial face amount of any Letter of Credit may be so reallocated) and (ii) to the extent not reallocated pursuant to immediately preceding clause (i), the respective Borrower shall Cash Collateralize any such Letter of Credit in accordance with Section 2.06(j). Except to the extent of reallocations of participations pursuant to clause (i) of the immediately preceding sentence, the occurrence of a Maturity Date with respect to a given Class of Revolving Commitments shall have no effect upon (and shall not diminish) the percentage participations of the Revolving Lenders in any Letter of Credit issued before such Maturity Date. (l) Issuing Bank Agreements. Each Issuing Bank (other than the Administrative Agent or its affiliates) agrees that, unless otherwise requested by the Administrative Agent, each Issuing Bank shall report in writing to the Administrative Agent (i) on the first Business Day of each week, the daily activity (set forth by day) in respect of Letters of Credit during the immediately preceding week, including all issuances, extensions and amendments, all expirations and cancellations and all disbursements and reimbursements, (ii) on or prior to each Business Day on which such Issuing Bank expects to issue, amend or extend any Letter of Credit, the date of such issuance, amendment or extension, and the currency and aggregate amount of the Letters of Credit to be issued, amended or extended by it and outstanding after giving effect to such issuance, amendment or extension occurred (and whether the amount thereof changed), it being understood that such Issuing Bank shall not permit any issuance, extension or amendment resulting in an increase in the amount of any Letter of Credit to occur without first obtaining written confirmation from the Administrative Agent that it is then permitted under this Agreement, (iii) on each Business Day on which such Issuing Bank makes any LC Disbursement, the date of such LC Disbursement and the amount of such LC Disbursement, (iv) on any Business Day on which the Borrower fails to reimburse an LC Disbursement required to

97 be reimbursed to such Issuing Bank on such day, the date of such failure and the amount and currency of such LC Disbursement and (v) on any other Business Day, such other information as the Administrative Agent shall reasonably request, as to the Letters of Credit issued by such Issuing Bank. Section 2.07 Funding of Borrowings. (a) Each Lender shall make each Loan to be made by it hereunder on the proposed date thereof by wire transfer of immediately available funds (i) in the case of Loans denominated in Dollars, by 12:00 noon, New York City time, to the account of the Administrative Agent most recently designated by it for such purpose by notice to the Lenders and (ii) in the case of each Loan denominated in a Foreign Currency, by 12:00 noon, Local Time, in the city of the Administrative Agent’s Foreign Currency Payment Office for such currency and at such Foreign Currency Payment Office for such currency; provided that Swingline Loans shall be made as provided in Section 2.05. The Administrative Agent will make such Loans available to the respective Borrower by promptly crediting the amounts so received, in like funds, to (x) an account of the applicable Borrower maintained with the Administrative Agent in New York City or Chicago and designated by such Borrower in the applicable Borrowing Request, in the case of Loans denominated in Dollars and (y) an account of the applicable Borrower in the relevant jurisdiction and designated by such Borrower in the applicable Borrowing Request, in the case of Loans denominated in a Foreign Currency; provided that ABR Revolving Loans made to finance the reimbursement of an LC Disbursement as provided in Section 2.06(e) shall be remitted by the Administrative Agent to the Issuing Bank. (b) Unless the Administrative Agent shall have received notice from a Lender prior to the proposed date of any Borrowing that such Lender will not make available to the Administrative Agent such Lender’s share of such Borrowing, the Administrative Agent may assume that such Lender has made such share available on such date in accordance with paragraph (a) of this Section 2.07 and may, in reliance upon such assumption, make available to the applicable Borrower a corresponding amount. In such event, if a Lender has not in fact made its share of the applicable Borrowing available to the Administrative Agent, then the applicable Lender and the applicable Borrower severally agree to pay to the Administrative Agent forthwith on demand such corresponding amount with interest thereon, for each day from and including the date such amount is made available to such Borrower to but excluding the date of payment to the Administrative Agent, at (i) in the case of such Lender, the greater of the Federal Funds Effective Rate and a rate determined by the Administrative Agent in accordance with banking industry rules on interbank compensation (including without limitation the Overnight Foreign Currency Rate in the case of Loans denominated in a Foreign Currency) or (ii) in the case of a Borrower, the interest rate applicable to the relevant Class of ABR Loans. If such Lender pays such amount to the Administrative Agent, then such amount shall constitute such Lender’s Loan included in such Borrowing. Section 2.08 Interest Elections. (a) Each Borrowing initially shall be of the Type, and under the applicable Tranche, specified in the applicable Borrowing Request and, in the case of a Term Benchmark Borrowing, shall have an initial Interest Period as specified in such Borrowing Request. Thereafter, subject to clause (e) below, the applicable Borrower may elect to convert such Borrowing to a different Type or to continue such Borrowing and, in the case of a Term Benchmark Borrowing, may elect Interest Periods therefor, all as provided in this Section 2.08. The applicable Borrower may elect different options with respect to different portions of the

98 affected Borrowing, in which case each such portion shall be allocated ratably among the Lenders holding the Loans comprising such Borrowing, and the Loans comprising each such portion shall be considered a separate Borrowing. This Section 2.08 shall not apply to Swingline Loans, which may not be converted or continued. Notwithstanding any other provision of this Section 2.08, the applicable Borrower shall not be permitted to change the Tranche or Class of any Borrowing. (b) To make an election pursuant to this Section 2.08, the applicable Borrower shall notify the Administrative Agent of such election (by telephone or irrevocable written notice in the case of a Borrowing denominated in Dollars or by irrevocable written notice (via an Interest Election Request in a form approved by the Administrative Agent and signed by such Borrower) in the case of a Borrowing denominated in a Foreign Currency) by the time that a Borrowing Request would be required under Section 2.03 if such Borrower were requesting a Borrowing of the Type and Class resulting from such election to be made on the effective date of such election. Each such telephonic Interest Election Request shall be irrevocable and shall be confirmed promptly by hand delivery or telecopy to the Administrative Agent of a written Interest Election Request in a form approved by the Administrative Agent and signed by the applicable Borrower. Notwithstanding any contrary provision herein, this Section 2.08 shall not be construed to permit the Borrower to (i) change the currency of any Borrowing, (ii) elect an Interest Period for Term Benchmark Loans that does not comply with Section 2.02(d) or (iii) convert any Borrowing to a Borrowing of a Type not available under the Class of Commitments or the Tranche pursuant to which such Borrowing was made. (c) Each telephonic and written Interest Election Request shall specify the following information in compliance with Section 2.02: (i) the Borrowing to which such Interest Election Request applies and, if different options are being elected with respect to different portions thereof, the portions thereof to be allocated to each resulting Borrowing (in which case the information to be specified pursuant to clauses (iii) and (iv) below shall be specified for each resulting Borrowing); (ii) the effective date of the election made pursuant to such Interest Election Request, which shall be a Business Day; (iii) whether the resulting Borrowing is to be an ABR Borrowing, a Term SOFR Borrowing, a Canadian Prime Rate Borrowing or a Borrowing of Term CORRA Loans and if such Borrowing is a Revolving Borrowing, whether the resulting Borrowing is to be a Dollar Tranche Borrowing or a Multicurrency Tranche Revolving Borrowing; and (iv) if the resulting Borrowing is a Term Benchmark Borrowing, the Interest Period and Agreed Currency (which shall comply with the limitation set forth in Section 2.01(c)(iv)) to be applicable thereto after giving effect to such election, which Interest Period shall be a period contemplated by the definition of the term “Interest Period”.

99 If any such Interest Election Request requests a Term Benchmark Borrowing but does not specify an Interest Period, then the applicable Borrower shall be deemed to have selected an Interest Period of one month’s duration. (d) Promptly following receipt of an Interest Election Request, the Administrative Agent shall advise each applicable Lender of the details thereof and of such Lender’s portion of each resulting Borrowing. (e) If the applicable Borrower fails to deliver a timely Interest Election Request with respect to a Term Benchmark Borrowing prior to the end of the Interest Period applicable thereto, then, unless such Borrowing is repaid as provided herein, at the end of such Interest Period (i) in the case of a Borrowing denominated in Dollars, such Borrowing shall be converted to an ABR Borrowing, (ii) in the case of a Borrowing denominated in Canadian Dollars, such Borrowing shall be converted into a Canadian Prime Rate Borrowing and (iii) in the case of a Borrowing denominated in a Foreign Currency (other than Canadian Dollars) in respect of which such Borrower shall have failed to deliver an Interest Election Request prior to the third (3rd) Business Day preceding the end of such Interest Period, such Borrowing shall automatically continue as a Term Benchmark Borrowing in the same Agreed Currency with an Interest Period of one month unless such Term Benchmark Borrowing is or was repaid in accordance with Section 2.11. Notwithstanding any contrary provision hereof, if an Event of Default has occurred and is continuing and the Administrative Agent, at the request of the Required Lenders, so notifies the applicable Borrower, then, so long as an Event of Default is continuing (i) no outstanding Borrowing may be converted to or continued as a Term Benchmark Borrowing and (ii) unless repaid, (A) each Term SOFR Borrowing shall be converted to an ABR Borrowing at the end of the Interest Period applicable thereto and (B) each Borrowing of Term CORRA Loans shall be converted at the end of the Interest Period applicable thereto to a Canadian Prime Rate Borrowing. Section 2.09 Termination and Reduction of Commitments. (a) Unless previously terminated, (i) the Revolving Commitment of each Revolving Lender shall automatically and permanently terminate on the relevant Maturity Date, (ii) the Initial Term Loan Commitments (other than any Initial Term Loan Commitments that constitute Incremental Term Loan Commitments) of each Initial Term Lender shall automatically and permanently terminate on the Effective Date (after giving effect to the incurrence of such Term Loans on such date) and (iii) the Additional 2024 Refinancing Term Loan Commitments of each Additional 2024 Refinancing Term Lender shall automatically and permanently terminate on the First Amendment Effective Date (after giving effect to the incurrence of such Additional 2024 Refinancing Term Loans on such date). (b) The Borrower may at any time terminate, or from time to time reduce, the Commitments of any Class; provided that (i) each reduction of such Commitments shall be in an amount that is an integral multiple of $5,000,000 and not less than $10,000,000 and (ii) the Borrower shall not terminate or reduce any Revolving Commitments if, after giving effect to any concurrent prepayment of the Revolving Loans of such Class in accordance with Section 2.11, the Dollar Amount of the sum of the total Revolving Credit Exposures in respect of such Class would exceed the aggregate Revolving Commitments of such Class.

100 (c) The Borrower shall notify the Administrative Agent of any election to terminate or reduce the Commitments of any Class under paragraph (b) of this Section 2.09 at least three (3) Business Days prior to the effective date of such termination or reduction, specifying such election and the effective date thereof. Promptly following receipt of any notice, the Administrative Agent shall advise the Lenders of the contents thereof. Each notice delivered by the Borrower pursuant to this Section 2.09 shall be irrevocable; provided that a notice of termination of the Commitments of any Class delivered by the Borrower may state that such notice is conditioned upon the effectiveness of other credit facilities or one or more other events specified therein, in which case such notice may be revoked by each applicable Borrower (by notice to the Administrative Agent on or prior to the specified effective date) if such condition is not satisfied. Any termination or reduction of the Commitments of any Class shall be permanent. Each reduction of the Commitments of any Class shall be made ratably among the applicable Lenders in accordance with their respective Commitments of such Class. Section 2.10 Repayment and Amortization of Loans; Evidence of Debt. (a) The Borrower hereby unconditionally promise to pay (i) to the Administrative Agent for the account of each Revolving Lender the then unpaid principal amount of each Revolving Loan on the Maturity Date in the currency of such Loan, (ii) to the Swingline Lender the then unpaid principal amount of each Swingline Loan on the earlier of the Latest Maturity Date with respect to any Revolving Commitments and the first date after such Swingline Loan is made that is the 15th or last day of a calendar month and is at least two (2) Business Days after such Swingline Loan is made; provided that on each date that a Dollar Tranche Revolving Borrowing is made, the Borrower shall repay all Swingline Loans then outstanding. (b) (i) Beginning on the last day of the second full fiscal quarter ending after the Effective Date, the Borrower shall repay principal of outstanding Initial Term Loans on each Scheduled Principal Repayment Date described below in the aggregate principal amount described opposite such Scheduled Principal Repayment Date (as adjusted from time to time pursuant to Sections 2.11(a), 2.11(d)(i), 2.20, 2.24, 2.25, 9.04(g) and 9.04(k)): Scheduled Principal Repayment Dates Amount Each Scheduled Principal Repayment Date 0.25% of the aggregate principal amount of Initial Term Loans incurred on the Effective Date Maturity Date All remaining outstanding principal of Initial Term Loans To the extent not previously repaid, all unpaid Initial Term Loans shall be paid in full in Dollars by the relevant Borrower on the applicable Maturity Date.

101 (ii) Beginning on the last day of the fiscal quarter ending after the First Amendment Effective Date, the Borrower shall repay principal of outstanding 2024 Refinancing Term Loans on each Scheduled Principal Repayment Date described below in the aggregate principal amount described opposite such Scheduled Principal Repayment Date (as adjusted from time to time pursuant to Sections 2.11(a), 2.11(d)(i), 2.20, 2.24, 2.25, 9.04(g) and 9.04(k)): Scheduled Principal Repayment Dates Amount Each Scheduled Principal Repayment Date 0.25% of the aggregate principal amount of 2024 Refinancing Term Loans incurred on the First Amendment Effective Date Maturity Date All remaining outstanding principal of 2024 Refinancing Term Loans To the extent not previously repaid, all unpaid 2024 Refinancing Term Loans shall be paid in full in Dollars by the Borrower on the applicable Maturity Date. To the extent specified in the applicable Extension Offer, amortization payments with respect to Extended Term Loans for periods prior to the then current Maturity Date for any applicable Term Loans may be reduced (but not increased) and amortization payments required with respect to Extended Term Loans for periods after such applicable Maturity Date shall be as specified in the applicable Extension Offer. (c) Each Lender shall maintain in accordance with its usual practice an account or accounts evidencing the indebtedness of the Borrower to such Lender resulting from each Loan made by such Lender, including the amounts of principal and interest payable and paid to such Lender from time to time hereunder. (d) The Administrative Agent shall maintain accounts in which it shall record (i) the amount of each Loan made hereunder, the Tranche under which it was made, the Class, Agreed Currency and Type thereof and the Interest Period applicable thereto, (ii) the amount of any principal or interest due and payable or to become due and payable from the respective Borrower to each Lender hereunder and (iii) the amount of any sum received by the Administrative Agent hereunder for the account of the Lenders and each Lender’s share thereof. (e) The entries made in the accounts maintained pursuant to paragraph (c) or (d) of this Section 2.10 shall be prima facie evidence of the existence and amounts of the obligations recorded therein; provided that the failure of any Lender or the Administrative Agent to maintain such accounts or any error therein shall not in any manner affect the obligation of the Borrower to repay the Loans in accordance with the terms of this Agreement. (f) Any Lender may request that Loans made by it be evidenced by a promissory note. In such event, the Borrower shall prepare, execute and deliver to such Lender promissory notes payable to such Lender (or, if requested by such Lender, to such Lender and its

102 registered assigns) and in a form approved by the Administrative Agent. Thereafter, the Loans evidenced by such promissory notes and interest thereon shall at all times (including after assignment pursuant to Section 9.04) be represented by one or more promissory notes in such form payable to the payee named therein (or, if such promissory note is a registered note, to such payee and its registered assigns). Section 2.11 Prepayment of Loans. (a) The Borrower shall have the right at any time and from time to time to prepay any Borrowing in whole or in part, without premium or penalty (except as set forth in Section 2.12(d)) but subject to break funding payments required by Section 2.16, subject to prior notice in accordance with the provisions of this Section 2.11(a). The applicable Borrower shall notify the Administrative Agent (and, in the case of prepayment of a Swingline Loan, the Swingline Lender) by telephone (confirmed by telecopy) of any prepayment hereunder (i) in the case of prepayment of a Term Benchmark Borrowing, not later than 11:00 a.m., Local Time, three (3) Business Days before the date of prepayment, (ii) in the case of prepayment of an ABR Borrowing or Canadian Prime Rate Borrowing, not later than 11:00 a.m., New York City time, one (1) Business Day before the date of prepayment or (iii) in the case of prepayment of a Swingline Loan, not later than 12:00 noon, New York City time, on the date of prepayment. Each such notice shall be irrevocable and shall specify the applicable Tranche prepayment date and the principal amount of each Borrowing or portion thereof to be prepaid; provided that a notice of prepayment may state that such notice is conditioned upon the effectiveness of other credit facilities or one or more events specified therein, in which case such notice may be revoked by each applicable Borrower by notice to the Administrative Agent on or prior to the specified effective date if such condition is not satisfied. Promptly following receipt of any such notice relating to a Borrowing, the Administrative Agent shall advise the Lenders of the contents thereof. Each partial prepayment of any Borrowing shall be in an amount that would be permitted in the case of an advance of a Borrowing of the same Type as provided in Section 2.02. Each prepayment of a Revolving Borrowing shall be applied ratably to the Revolving Loans included in the prepaid Revolving Borrowing, each voluntary prepayment of a Term Loan Borrowing shall be applied as directed by the Borrower and each mandatory prepayment of a Term Loan Borrowing shall be applied as directed by the Borrower (subject to Section 2.11(d)). Prepayments shall be accompanied by (i) accrued interest to the extent required by Section 2.13 and (ii) break funding payments pursuant to Section 2.16. (b) If at any time, (i) solely as a result of fluctuations in currency exchange rates, the sum of the aggregate principal Dollar Amount of all of the Multicurrency Tranche Revolving Credit Exposures (calculated, with respect to those Credit Events denominated in Foreign Currencies, as of the most recent Revaluation Date with respect to each such Credit Event) exceeds 105% of the aggregate Multicurrency Tranche Commitments, (ii) the sum of the aggregate principal Dollar Amount of all Non-USD Multicurrency Tranche Revolving Credit Exposure (calculated as of the most recent Revaluation Date) exceeds 105% of the aggregate Non- USD Multicurrency Tranche Sublimit or (iii) for any other reason, the sum of the aggregate principal Dollar Amount of all of the Revolving Credit Exposures of any Class (so calculated) exceeds the aggregate Commitments of such Class, the Borrower shall in each case immediately repay the applicable Borrowings or Cash Collateralize LC Exposure in an account with the Administrative Agent pursuant to Section 2.06(j), as applicable, in an aggregate principal amount sufficient to cause the aggregate Dollar Amount of all Revolving Credit Exposures (so calculated) of each Class to be less than or equal to the aggregate Commitments of such Class (or, in the case

103 of preceding clause (ii), cause the aggregate principal Dollar Amount of all Non-USD Multicurrency Tranche Revolving Credit Exposure to be less than or equal to the Non-USD Multicurrency Tranche Sublimit). (c) (1) In the event and on each occasion that any Net Proceeds are received by or on behalf of Parent or any of its Restricted Subsidiaries in respect of any Prepayment Event, the Borrower shall, within five (5) Business Days after such Net Proceeds are received, prepay (x) the Obligations and (y) Other Applicable Indebtedness (to the extent and if required by the terms of the documentation governing such Other Applicable Indebtedness), in each case, as set forth in Section 2.11(d)(i) below in an aggregate amount equal to 100% (with step downs to (i) 50% based upon the achievement and maintenance of a First Lien Net Leverage Ratio of less than or equal to 3.00 to 1.00 and (ii) 0% based upon the achievement and maintenance of a First Lien Net Leverage Ratio of less than or equal to 2.50 to 1.00 (each such step down, an “Asset Sale Step Down”)) of such Net Proceeds; provided that in the case of any event described in clause (a), (b) or (c) of the definition of the term “Prepayment Event”, the Borrower shall only be obligated to prepay, subject to the Asset Sale Step Down, the amount of the Net Proceeds received to the extent in excess of the applicable amounts set forth therein; provided, further, that in the case of any event described in clause (a), (b) or (c) of the definition of the term “Prepayment Event”, if Parent shall deliver to the Administrative Agent a certificate of a Financial Officer to the effect that Parent or its relevant Restricted Subsidiaries intend to apply the Net Proceeds from such event (or a portion thereof specified in such certificate), within 12 months after receipt of such Net Proceeds, to consummate a Permitted Acquisition or to otherwise acquire, replace, rebuild, maintain, develop, construct, improve, upgrade or repair property, equipment or other assets (excluding inventory) useful in the business of Parent and/or its Restricted Subsidiaries, and certifying that no Event of Default has occurred and is continuing, then no prepayment shall be required pursuant to this paragraph in respect of the Net Proceeds specified in such certificate; provided, further, that to the extent of any such Net Proceeds therefrom that have not been so applied by the end of such 12 month period (or committed to be applied by the end of the 12 month period and applied within 180 days after the end of such 12 month period), at which time a prepayment shall be required in an amount equal to such Net Proceeds that have not been so applied, (2) in the event and on each occasion that any Borrower incurs, issues or obtains any Credit Agreement Refinancing Indebtedness (other than solely by means of extending or renewing then existing Credit Agreement Refinancing Indebtedness without resulting in any Net Proceeds), the Borrower shall, on the date on which such Credit Agreement Refinancing Indebtedness is incurred, issued or obtained, prepay the applicable Refinanced Debt as set forth in Section 2.11(d)(ii) below in an aggregate amount equal to 100% of the Net Proceeds of such Credit Agreement Refinancing Indebtedness and (3) on each Excess Cash Payment Date the Borrower shall prepay the Obligations as set forth in Section 2.11(d)(i) below in an amount equal to the Applicable Excess Cash Flow Percentage of the Excess Cash Flow for the applicable fiscal year (but only if such amount exceeds $25,000,000 in the aggregate); provided that repayments of principal of Loans made as a voluntary prepayment pursuant to Section 2.11(a) (other than with the proceeds of long-term Indebtedness) (but in the case of a voluntary prepayment of Revolving Loans or Swingline Loans, only to the extent accompanied by a voluntary reduction to the Revolving Commitments in an amount equal to such prepayment) during the applicable fiscal year shall reduce on a dollar-for-dollar basis the amount of such mandatory repayment otherwise required on the applicable Excess Cash Payment Date pursuant to this clause (3).

104 (d) Subject to Sections 2.11(e) and 2.11(f) below and except as set forth in the applicable Incremental Amendment, Extension Amendment and Refinancing Amendment, (i) all such amounts pursuant to Sections 2.11(c)(1) and 2.11(c)(3) shall be applied to each Class of Term Loans on a pro rata basis and to the scheduled payments of each such Class as directed by Parent (and absent such direction, in direct order of maturity); provided that, if at the time that prepayment would be required pursuant to Sections 2.11(c)(1) and 2.11(c)(3), Parent or any Restricted Subsidiary is required to prepay or offer to redeem or repurchase any Other Applicable Indebtedness pursuant to the terms of the documentation governing such Other Applicable Indebtedness with such amounts, then Parent or such Restricted Subsidiary may apply such amounts on a pro rata basis (determined on the basis of the aggregate outstanding principal amount of the Term Loans and such Other Applicable Indebtedness at such time) to prepay the Term Loans and prepay, redeem or repurchase such Other Applicable Indebtedness; provided, further, that (A) any prepayment, redemption or repurchase of such Other Applicable Indebtedness shall be at par (or less than par), (B) the portion of such prepayment amount allocated to such Other Applicable Indebtedness shall not exceed the amount required to be allocated to such Other Applicable Indebtedness pursuant to the terms thereof, (C) the amount of prepayment of the Term Loans that would otherwise have been required pursuant to this clause (d) shall be reduced accordingly and (D) to the extent the holders of such Other Applicable Indebtedness decline to have such Indebtedness prepaid, redeemed or repurchased, the declined amount shall promptly (and in any event within ten (10) Business Days after the date of such rejection) be applied to prepay the Term Loans in accordance with the terms hereof and (ii) all such amounts pursuant to Section 2.11(c)(2) shall be applied to prepay an aggregate principal amount of the applicable Refinanced Debt equal to the Net Proceeds of the applicable Credit Agreement Refinancing Indebtedness (and to the extent the applicable Refinanced Debt is not repaid in full, such Net Proceeds shall reduce the remaining scheduled principal repayments of such Refinanced Debt on a pro rata basis). (e) Notwithstanding any other provisions of this Section 2.11 to the contrary, with respect to any prepayment required pursuant to Section 2.11(c)(1), if at the time of such prepayment, the Restricted Subsidiary receiving the Net Proceeds (i) is prohibited, restricted or delayed by applicable local law from repatriating such Net Proceeds to Parent or the Borrower, the portion of such Net Proceeds so affected will not be required to be applied to repay Term Loans at the times provided in Section 2.11(c)(1) but may be retained by the applicable Restricted Subsidiary so long, but only so long, as the applicable local law will not permit repatriation to Parent or the Borrower, and once such repatriation of any of such affected Net Proceeds is permitted under the applicable local law, such repatriation will be effected and such repatriated Net Proceeds will be promptly applied (net of additional taxes payable or reserved against as a result thereof) to the repayment of the Term Loans pursuant to Section 2.11(d) to the extent provided therein or (ii) cannot repatriate such funds to Parent or the Borrower without (in the good faith determination of Parent) the repatriation of such Net Proceeds (or a portion thereof) that would otherwise be required to be applied pursuant to Section 2.11(c)(1) resulting in material adverse tax consequences, the Net Proceeds (or portion thereof) so affected may be retained by the applicable Restricted Subsidiary (Parent and the Borrower hereby agreeing to cause the applicable Restricted Subsidiary to promptly use commercially reasonable efforts to take all actions within the reasonable control of Parent and the Borrower that are reasonably required to eliminate such tax effects) until such time as such material adverse tax consequences would not apply to the repatriation thereof, at which time the mandatory prepayments otherwise required by Section 2.11(c)(1) with respect to such Net Proceeds shall be made.

105 (f) The Borrower shall notify the Administrative Agent in writing of any mandatory prepayment of Term Loans required to be made pursuant to Section 2.11(c)(1) or 2.11(c)(3) at least three (3) Business Days prior to the date of such prepayment. Each such notice shall specify the date of such prepayment and provide a reasonably detailed calculation of the amount of such prepayment. The Administrative Agent will promptly notify each Term Lender of the contents of any such prepayment notice and of such Term Lender’s ratable portion of such prepayment (based on such Lender’s Applicable Percentage of each relevant Tranche of the Term Loans). Any Term Lender (a “Declining Term Lender,” and any Term Lender which is not a Declining Term Lender, an “Accepting Term Lender”) may elect, by delivering written notice to the Administrative Agent and the applicable Borrower no later than 5:00 p.m. one (1) Business Day after the date of such Term Lender’s receipt of notice from the Administrative Agent regarding such prepayment, that the full amount of any mandatory prepayment otherwise required to be made with respect to the Term Loans held by such Term Lender pursuant to Section 2.11(c)(1) or 2.11(c)(3) not be made (the aggregate amount of such prepayments declined by the Declining Term Lenders, the “Declined Prepayment Amount”). If a Term Lender fails to deliver notice setting forth such rejection of a prepayment to the Administrative Agent within the time frame specified above or such notice fails to specify the principal amount of the Term Loans to be rejected, any such failure will be deemed an acceptance of the total amount of such mandatory prepayment of Term Loans. In the event that the Declined Prepayment Amount related to a prepayment under Section 2.11(c)(1) is greater than $0, the Administrative Agent will promptly notify each Accepting Term Lender of the amount of such Declined Prepayment Amount and of any such Accepting Term Lender’s ratable portion of such Declined Prepayment Amount (based on such Lender’s Applicable Percentage in respect of the and Term Loans (excluding the Applicable Percentage of Declining Term Lenders), as applicable). In the event that the Declined Prepayment Amount related to a prepayment under Section 2.11(c)(3) is greater than $0, the Administrative Agent will promptly notify each Accepting Term Lender of the amount of such Declined Prepayment Amount and of any such Accepting Term Lender’s ratable portion of such Declined Prepayment Amount (based on such Lender’s Applicable Percentage in respect of the Term Loans (excluding the Applicable Percentage of Declining Term Lenders), as applicable). Any such Accepting Term Lender may elect, by delivering, no later than 5:00 p.m. (New York time) one (1) Business Day after the date of such Accepting Term Lender’s receipt of notice from the Administrative Agent regarding such additional prepayment, a written notice, that such Accepting Term Lender’s ratable portion of such Declined Prepayment Amount not be applied to repay such Accepting Term Lender’s Term Loans, in which case the portion of such Declined Prepayment Amount which would otherwise have been applied to such Term Loans of the Declining Term Lenders shall instead be retained by the Borrower. For the avoidance of doubt, the Borrower may, at their option, apply any amounts retained in accordance with the immediately preceding sentence to prepay loans in accordance with Section 2.11(a) above. Section 2.12 Fees. (a) The Borrower jointly and severally agree to pay to the Administrative Agent for the account of each Revolving Lender a commitment fee, which shall accrue at the Applicable Rate on the daily amount of the Available Revolving Commitment of such Revolving Lender during the period from and including the Effective Date to but excluding the date on which the last of the Revolving Commitments (or Extended Revolving Commitments) of such Revolving Lender terminates. Accrued commitment fees shall be payable in arrears on the last Business Day of March, June, September and December of each year and on the date on which the last of the Revolving Commitments terminate, commencing on the first such date to

106 occur after the Effective Date; provided that any commitment fees accruing after the date on which such Revolving Commitments terminate shall be payable on demand. All commitment fees shall be computed on the basis of a year of 360 days and shall be payable for the actual number of days elapsed (including the first day but excluding the last day). (b) The Borrower agrees to pay (i) to the Administrative Agent for the account of each Revolving Lender a participation fee with respect to its participations in Letters of Credit, which shall accrue at the same Applicable Rate used to determine the interest rate applicable to Term SOFR Revolving Loans on the average daily Dollar Amount of such Revolving Lender’s LC Exposure (excluding any portion thereof attributable to unreimbursed LC Disbursements) during the period from and including the Effective Date to but excluding the later of the date on which the last of such Revolving Lender’s Revolving Commitment terminates and the date on which such Revolving Lender ceases to have any LC Exposure and (ii) to the Issuing Bank for its own account a fronting fee, which shall accrue at the rate per annum separately agreed upon by the Borrower and the Issuing Bank (including, for the avoidance of doubt, with respect to any Existing Letters of Credit) on the average daily Dollar Amount of the LC Exposure (excluding any portion thereof attributable to unreimbursed LC Disbursements) attributable to Letters of Credit issued by the Issuing Bank during the period from and including the Effective Date to but excluding the later of the date of termination of the last of the Revolving Commitments and the date on which there ceases to be any LC Exposure, as well as the Issuing Bank’s standard fees and commissions with respect to the issuance, amendment, cancellation, negotiation, transfer, presentment or extension of any Letter of Credit or processing of drawings thereunder. Unless otherwise specified above, participation fees and fronting fees accrued through and including the last day of March, June, September and December of each year shall be payable on the third (3rd) Business Day following such last day, commencing on the first such date to occur after the Effective Date; provided that all such fees shall be payable on the date on which the last of the Revolving Commitments terminate and any such fees accruing after the date on which the such Revolving Commitments terminate shall be payable on demand. Any other fees payable to the Issuing Bank pursuant to this paragraph shall be payable within ten (10) days after demand. All participation fees and fronting fees shall be computed on the basis of a year of 360 days and shall be payable for the actual number of days elapsed (including the first day but excluding the last day). (c) The Borrower Representative agrees to pay to the Administrative Agent, for its own account, fees payable in the amounts and at the times agreed in the Administrative Agent Fee Letter. (d) (i) If any Repricing Event occurs prior to the date occurring sixth months after the Effective Date, the Borrower agrees to pay to the Administrative Agent, for the ratable account of each Lender with Initial Term Loans that are subject to such Repricing Event (including any Lender which is replaced pursuant to Section 9.02(e) as a result of its refusal to consent to an amendment giving rise to such Repricing Event), a fee in an amount equal to 1.00% of the aggregate principal amount of the Initial Term Loans subject to such Repricing Event. Such fees shall be earned, due and payable upon the date of the occurrence of the respective Repricing Event.

107 (ii) If any Repricing Event occurs prior to the date occurring sixth months after the First Amendment Effective Date, the Borrower agrees to pay to the Administrative Agent, for the ratable account of each Lender with 2024 Refinancing Term Loans that are subject to such Repricing Event (including any Lender which is replaced pursuant to Section 9.02(e) as a result of its refusal to consent to an amendment giving rise to such Repricing Event), a fee in an amount equal to 1.00% of the aggregate principal amount of the 2024 Refinancing Term Loans subject to such Repricing Event. Such fees shall be earned, due and payable upon the date of the occurrence of the respective Repricing Event. (e) All fees payable hereunder shall be paid on the dates due, in Dollars and immediately available funds, to the Administrative Agent (or to the Issuing Bank, in the case of fees payable to it) for distribution, in the case of commitment fees and participation fees, to the applicable Revolving Lenders. Fees paid shall not be refundable under any circumstances. Section 2.13 Interest. (a) The Loans comprising each ABR Borrowing (including each Swingline Loan) shall bear interest at the Alternate Base Rate plus the Applicable Rate. The Loans comprising each Borrowing of Canadian Prime Rate Loans shall bear interest at the Canadian Prime Rate plus the Applicable Rate. (b) The Loans comprising each Term SOFR Borrowing shall bear interest at Term SOFR for the Interest Period in effect for such Borrowing plus the Applicable Rate. (c) The Loans comprising each Term CORRA Borrowing shall bear interest at the Adjusted Term CORRA Rate for the Interest Period in effect for such Borrowing plus the Applicable Rate. (d) Notwithstanding the foregoing, if any principal of or interest on any Loan or any fee or other amount payable by the Borrower hereunder is not paid when due, whether at stated maturity, upon acceleration or otherwise, such overdue amount shall bear interest, after as well as before judgment, at a rate per annum equal to (i) in the case of overdue principal of any Loan, 2% plus the rate otherwise applicable to such Loan as provided in the preceding paragraphs of this Section 2.13 or (ii) in the case of any other amount, 2% plus the rate applicable to ABR Loans (or the Canadian Prime Rate, in the case of Canadian Prime Rate Loans) as provided in paragraph (a) of this Section 2.13. (e) Accrued interest on each Loan shall be payable in arrears on each Interest Payment Date for such Loan and, in the case of Revolving Loans, upon termination of the applicable Revolving Commitments; provided that (i) interest accrued pursuant to paragraph (d) of this Section 2.13 shall be payable on demand, (ii) in the event of any repayment or prepayment of any Loan of any Class (other than a prepayment of an ABR Revolving Loan or Canadian Prime Rate Revolving Loan prior to the end of the Availability Period), accrued interest on the principal amount repaid or prepaid shall be payable on the date of such repayment or prepayment and (iii) in the event of any conversion of any Term Benchmark Loan prior to the end of the current Interest Period therefor, accrued interest on such Loan shall be payable on the effective date of such conversion.

108 (f) All interest hereunder shall be computed on the basis of a year of 360 days, except that (i) interest computed by reference to the Alternate Base Rate at times when the Alternate Base Rate is based on the Prime Rate, as well as all Canadian Dollar denominated Loans, shall be computed on the basis of a year of 365 days (or 366 days in a leap year) and (ii) for Borrowings denominated in Pounds Sterling, interest shall be computed on the basis of a year of 365 days, and in each case shall be payable for the actual number of days elapsed (including the first day but excluding the last day). The applicable Alternate Base Rate, Canadian Prime Rate or Relevant Rate shall be determined by the Administrative Agent, and such determination shall be conclusive absent manifest error. (g) Notwithstanding anything to the contrary set forth herein, the Borrower shall not be permitted to request a Term CORRA Loan at any time a Default has occurred and is continuing (and upon such event, any outstanding Term CORRA Loans shall be converted into Canadian Prime Rate Loans on the maturity thereof). Section 2.14 Alternate Rate of Interest. (a) Subject to clauses (b), (c), (d), (e) and (f) of this Section 2.14, if prior to the commencement of any Interest Period for a Term Benchmark Borrowing: (i) the Administrative Agent determines (which determination shall be conclusive absent manifest error) that adequate and reasonable means do not exist for ascertaining Term SOFR or the Adjusted Term CORRA Rate, as applicable (including because the Term SOFR Reference Rate or the Term CORRA Reference Rate is not available or published on a current basis), for the applicable Agreed Currency and such Interest Period; or (ii) the Administrative Agent is advised by the Required Lenders that Term SOFR or the Adjusted Term CORRA Rate, as applicable, for such Interest Period will not adequately and fairly reflect the cost to such Lenders (or Lender) of making or maintaining their Loans (or its Loan) included in such Borrowing for the applicable Agreed Currency and such Interest Period; then the Administrative Agent shall give notice thereof to the Borrower Representative and the Lenders by telephone, telecopy or electronic mail as promptly as practicable thereafter and, until (x) the Administrative Agent notifies the Borrower Representative and the Lenders that the circumstances giving rise to such notice no longer exist with respect to the relevant Benchmark and (y) the Borrower delivers a new Interest Election Request in accordance with the terms of Section 2.08 or a new Borrowing Request in accordance with the terms of Section 2.03, (A) any Interest Election Request that requests the conversion of any Borrowing to, or continuation of any Borrowing as, a Term SOFR Borrowing and any Borrowing Request that requests a Term SOFR Borrowing shall instead be deemed to be an Interest Election Request or a Borrowing Request, as applicable, for a Daily Simple SOFR Borrowing, or, if Daily Simple SOFR is unavailable, an ABR Borrowing and (B) any Interest Election Request that requests the conversion of any Borrowing to, or continuation of any Borrowing as, a Term CORRA Borrowing and any Borrowing Request that requests a Term CORRA Borrowing shall instead be deemed to be an Interest Election Request or a Borrowing Request, as applicable, for a Canadian Prime Rate Borrowing; provided that if the

109 circumstances giving rise to such notice affect only one Type of Borrowings, then all other Types of Borrowings shall be permitted. Furthermore, if any Term Benchmark Loan is outstanding on the date of the Borrower’s receipt of the notice from the Administrative Agent referred to in this Section 2.14(a) with respect to a Relevant Rate applicable to such Term Benchmark Loan, then until (x) the Administrative Agent notifies the Borrower Representative and the Lenders that the circumstances giving rise to such notice no longer exist with respect to the relevant Benchmark and (y) the Borrower Representative delivers a new Interest Election Request in accordance with the terms of Section 2.07 or a new Borrowing Request in accordance with the terms of Section 2.03, (1)(A) any Term SOFR Loan shall, on the last day of the Interest Period applicable to such Loan, be converted by the Administrative Agent to, and shall constitute, a Daily Simple SOFR Borrowing, or, if Daily Simple SOFR is unavailable, an ABR Borrowing, and (B) any Term CORRA Loan shall, on the last day of the Interest Period applicable to such Loan, be converted by the Administrative Agent to, and shall constitute, a Canadian Prime Rate Borrowing, in each case, on such day. (b) Notwithstanding anything to the contrary herein or in any other Loan Document, if a Benchmark Transition Event and its related Benchmark Replacement Date have occurred prior to the Reference Time in respect of any setting of the then-current Benchmark, then (x) if a Benchmark Replacement is determined in accordance with clause (1) or clause (2) of the definition of “Benchmark Replacement” for such Benchmark Replacement Date, such Benchmark Replacement will replace such Benchmark for all purposes hereunder and under any Loan Document in respect of such Benchmark setting and subsequent Benchmark settings without any amendment to, or further action or consent of any other party to, this Agreement or any other Loan Document and (y) if a Benchmark Replacement is determined in accordance with clause (3) of the definition of “Benchmark Replacement” for such Benchmark Replacement Date, such Benchmark Replacement will replace such Benchmark for all purposes hereunder and under any Loan Document in respect of any Benchmark setting at or after 5:00 p.m. (New York City time) on the fifth (5th) Business Day after the date notice of such Benchmark Replacement is provided to the Lenders without any amendment to, or further action or consent of any other party to, this Agreement or any other Loan Document so long as the Administrative Agent has not received, by such time, written notice of objection to such Benchmark Replacement from Lenders comprising the Required Lenders. (c) In connection with the implementation, administration or adoption of a Benchmark Replacement, the Administrative Agent will have the right, in consultation with the Borrower, to make Benchmark Replacement Conforming Changes from time to time and, notwithstanding anything to the contrary herein or in any other Loan Document, any amendments implementing such Benchmark Replacement Conforming Changes will become effective without any further action or consent of any other party to this Agreement or any other Loan Document. (d) The Administrative Agent will promptly notify the Borrower Representative and the Lenders of (i) any occurrence of a Benchmark Transition Event and its related Benchmark Replacement Date, (ii) the implementation of any Benchmark Replacement, (iii) the effectiveness of any Benchmark Replacement Conforming Changes, (iv) the removal or reinstatement of any tenor of a Benchmark pursuant to clause (e) below and (v) the commencement or conclusion of any Benchmark Unavailability Period. Any determination, decision or election that may be made by the Administrative Agent or, if applicable, any Lender (or group of Lenders)

110 pursuant to this Section 2.14, including any determination with respect to a tenor, rate or adjustment or of the occurrence or non-occurrence of an event, circumstance or date and any decision to take or refrain from taking any action or any selection, will be conclusive and binding absent manifest error and may be made in its or their sole discretion and without consent from any other party to this Agreement or any other Loan Document, except, in each case, as expressly required pursuant to this Section 2.14. (e) Notwithstanding anything to the contrary herein or in any other Loan Document, at any time (including in connection with the implementation of a Benchmark Replacement), (i) if the then-current Benchmark is a term rate (including Term SOFR) and either (A) any tenor for such Benchmark is not displayed on a screen or other information service that publishes such rate from time to time as selected by the Administrative Agent in its reasonable discretion or (B) the regulatory supervisor for the administrator of such Benchmark has provided a public statement or publication of information announcing that any tenor for such Benchmark is or will be no longer representative, then the Administrative Agent may modify the definition of “Interest Period” for any Benchmark settings at or after such time to remove such unavailable or non-representative tenor and (ii) if a tenor that was removed pursuant to clause (i) above either (A) is subsequently displayed on a screen or information service for a Benchmark (including a Benchmark Replacement) or (B) is not, or is no longer, subject to an announcement that it is or will no longer be representative for a Benchmark (including a Benchmark Replacement), then the Administrative Agent may modify the definition of “Interest Period” for all Benchmark settings at or after such time to reinstate such previously removed tenor. (f) Upon the Borrower’s receipt of notice of the commencement of a Benchmark Unavailability Period, the applicable Borrower may revoke any request for a Term Benchmark Borrowing of, conversion to or continuation of Term Benchmark Loans to be made, converted or continued during any Benchmark Unavailability Period and, failing that, (x) such Borrower will be deemed to have converted any request for (1) a Term Benchmark Borrowing denominated in Dollars into a request for a Borrowing of or conversion to Daily Simple SOFR Loans, or, if Daily Simple SOFR is unavailable, ABR Loans, (2) a Term CORRA Borrowing into a request of or conversion to Canadian Prime Rate Loans and (y) any Term Benchmark Borrowing denominated in a Foreign Currency other than Canadian Dollars shall be ineffective. During any Benchmark Unavailability Period or at any time that a tenor for the then-current Benchmark is not an Available Tenor, the component of ABR or the Canadian Prime Rate, as applicable, based upon the then-current Benchmark or such tenor for such Benchmark, as applicable, will not be used in any determination of ABR or the Canadian Prime Rate, as applicable. Furthermore, if any Term Benchmark Loan in any Agreed Currency is outstanding on the date of the Borrower’s receipt of notice of the commencement of a Benchmark Unavailability Period with respect to the Relevant Rate applicable to such Term Benchmark Loan, then (i) if such Term Benchmark Loan is denominated in Dollars, then on the last day of the Interest Period applicable to such Loan (or the next succeeding Business Day if such day is not a Business Day), such Loan shall be converted by the Administrative Agent to, and shall constitute, a Daily Simple SOFR Loan denominated in Dollars or, if Daily Simple SOFR is unavailable, an ABR Loan denominated in Dollars, in each case, on such day, (ii) if such Term Benchmark Loan is denominated in Canadian Dollars, then on the last day of the Interest Period applicable to such Term Benchmark Loan (or the next succeeding Business Day if such day is not a Business Day), such Term Benchmark Loan shall be converted by the Administrative Agent to, and shall constitute, a Canadian Prime Rate Loan on such day and

111 (iii) if such Term Benchmark Loan is denominated in any Foreign Currency other than Canadian Dollars, then such Loan shall, on the last day of the Interest Period applicable to such Loan (or the next succeeding Business Day if such day is not a Business Day), at the Borrower’s election prior to such day: (A) be prepaid by the Borrower on such day or (B) be converted by the Administrative Agent to, and (subject to the remainder of this subclause (B)) shall constitute, a Daily Simple SOFR Loan, or, if Daily Simple SOFR is unavailable, an ABR Loan, in each case denominated in Dollars (in an amount equal to the Dollar Amount of such Foreign Currency) on such day (it being understood and agreed that if the Borrower does not so prepay such Loan on such day by 12:00 noon, local time, the Administrative Agent is authorized to effect such conversion of such Term Benchmark Loan into a Daily Simple SOFR Loan or ABR Loan, as applicable, denominated in Dollars), and, in the case of such subclause (B), upon any subsequent implementation of a Benchmark Replacement in respect of such Foreign Currency pursuant to this Section 2.14, such Daily Simple SOFR Loan or ABR Loan, as applicable, denominated in Dollars shall then be converted by the Administrative Agent to, and shall constitute, a Term Benchmark Loan denominated in such original Foreign Currency on the day of such implementation, giving effect to such Benchmark Replacement in respect of such Foreign Currency. Section 2.15 Increased Costs. (a) If any Change in Law shall: (i) impose, modify or deem applicable any reserve, special deposit, compulsory loan, insurance charge or similar requirement against assets of, deposits with or for the account of, or credit extended or participated in by, any Lender or the Issuing Bank; (ii) impose on any Lender or the Issuing Bank or the applicable offshore interbank market for the applicable Agreed Currency any other condition, cost or expense (other than Taxes) affecting this Agreement or Loans made by such Lender or any Letter of Credit or participation therein; or (iii) subject the Administrative Agent, any Lender or the Issuing Bank to any Taxes (other than (A) Indemnified Taxes, (B) Other Taxes and (C) Connection Income Taxes) with respect to this Agreement, or any Loan made by it or any Letter of Credit or participation therein; and the result of any of the foregoing shall be to increase the cost to the Administrative Agent or such Lender of making or maintaining, continuing or converting any Loan or of maintaining its obligation to make any such Loan (including, without limitation, pursuant to any conversion of any Borrowing denominated in an Agreed Currency into a Borrowing denominated in any other Agreed Currency) or to increase the cost to the Administrative Agent, such Lender or the Issuing Bank of participating in, issuing or maintaining any Letter of Credit (including, without limitation, pursuant to any conversion of any Borrowing denominated in an Agreed Currency into a Borrowing denominated in any other Agreed Currency) or to reduce the amount of any sum received or receivable by the Administrative Agent, such Lender or the Issuing Bank hereunder, whether of principal, interest or otherwise (including, without limitation, pursuant to any conversion of any Borrowing denominated in an Agreed Currency into a Borrowing denominated in any other Agreed Currency), then the applicable Borrower will pay to the Administrative Agent, such Lender or the Issuing Bank, as the case may be, such additional amount or amounts as will

112 compensate such Lender or the Issuing Bank, as the case may be, for such additional costs incurred or reduction suffered. (b) If any Lender or the Issuing Bank determines that any Change in Law regarding capital or liquidity requirements has or would have the effect of reducing the rate of return on such Lender’s or the Issuing Bank’s capital or on the capital of such Lender’s or the Issuing Bank’s holding company, if any, as a consequence of this Agreement or the Loans made by, or participations in Letters of Credit held by, such Lender, or the Letters of Credit issued by the Issuing Bank, to a level below that which such Lender or the Issuing Bank or such Lender’s or the Issuing Bank’s holding company could have achieved but for such Change in Law (taking into consideration such Lender’s or the Issuing Bank’s policies and the policies of such Lender’s or the Issuing Bank’s holding company with respect to capital adequacy and liquidity), then from time to time the Borrower will pay to such Lender or the Issuing Bank, as the case may be, such additional amount or amounts as will compensate such Lender or the Issuing Bank or such Lender’s or the Issuing Bank’s holding company for any such reduction suffered. (c) A certificate of a Lender or the Issuing Bank setting forth, in reasonable detail, the basis and calculation of the amount or amounts necessary to compensate such Lender or the Issuing Bank or its holding company, as the case may be, as specified in paragraph (a) or (b) of this Section 2.15 shall be delivered to the applicable Borrower and shall be conclusive absent manifest error. The applicable Borrower shall pay such Lender or the Issuing Bank, as the case may be, the amount shown as due on any such certificate within ten (10) days after receipt thereof. (d) Failure or delay on the part of any Lender or the Issuing Bank to demand compensation pursuant to this Section 2.15 shall not constitute a waiver of such Lender’s or the Issuing Bank’s right to demand such compensation; provided that the Borrower shall not be required to compensate a Lender or the Issuing Bank pursuant to this Section 2.15 for any increased costs or reductions incurred more than 180 days prior to the date that such Lender or the Issuing Bank, as the case may be, notifies the applicable Borrower of the Change in Law giving rise to such increased costs or reductions and of such Lender’s or the Issuing Bank’s intention to claim compensation therefor; provided further that, if the Change in Law giving rise to such increased costs or reductions is retroactive, then the 180-day period referred to above shall be extended to include the period of retroactive effect thereof. (e) Notwithstanding anything contained herein to the contrary, a Lender shall not be entitled to any compensation pursuant to this Section 2.15 unless such Lender certifies in its reasonable good faith determination that it is imposing such charges or requesting such compensation from borrowers (similarly situated to the Borrower hereunder) under comparable syndicated credit facilities as a matter of general practice and policy pursuant to a certificate delivered to the applicable Borrower. Section 2.16 Break Funding Payments. In the event of (a) the payment of any principal of any Term Benchmark Loan other than on the last day of an Interest Period applicable thereto (including as a result of an Event of Default or as a result of any prepayment pursuant to Section 2.11), (b) the conversion of any Term Benchmark Loan other than on the last day of the Interest Period applicable thereto, (c) the failure to borrow, convert, continue or prepay any Term

113 Benchmark Loan on the date specified in any notice delivered pursuant hereto (regardless of whether such notice may be revoked under Section 2.11(a) and is revoked in accordance therewith) or (d) the assignment of any Term Benchmark Loan other than on the last day of the Interest Period applicable thereto as a result of a request by a Borrower pursuant to Section 2.19, then, in any such event, the Borrower shall compensate each Lender for the actual loss, cost and expense attributable to such event. A certificate of any Lender setting forth any amount or amounts that such Lender is entitled to receive pursuant to this Section 2.16, and setting forth in reasonable detail the calculations used by such Lender to determine such amount or amounts, shall be delivered to the Borrower and shall be conclusive absent manifest error. The Borrower shall pay such Lender the amount shown as due on any such certificate within ten (10) days after receipt thereof; provided that the Borrower shall not be required to compensate a Lender pursuant to this Section 2.16 for any amounts under this Section 2.16 incurred more than 180 days prior to the date that such Lender notifies the Borrower Representative of such amount and of such Lender’s intention to claim compensation therefor. Section 2.17 Taxes. (a) Any and all payments by or on account of any obligation of the Borrower or Guarantors, as the case may be, hereunder or under any other Loan Document shall be made free and clear of and without deduction for any Taxes except as required by applicable law; provided that if any Borrower, Guarantor or the Administrative Agent, as the case may be, shall be required (as determined in the good faith discretion of the payor) to deduct any Taxes from such payments, then (i) if such Taxes are Indemnified Taxes, the sum payable by the Borrower or Guarantors shall be increased as necessary so that after making all required deductions (including deductions applicable to additional sums payable under this Section 2.17) the applicable Recipient receives an amount equal to the sum it would have received had no such deductions been made, (ii) the Borrower, Guarantor, or the Administrative Agent, as the case may be, shall make such deductions, and (iii) the applicable Borrower, Guarantor, or the Administrative Agent, as the case may be, shall pay the full amount deducted to the relevant Governmental Authority in accordance with applicable law. (b) Without duplication of other amounts payable by the Borrower or Guarantors under Section 2.17(a), the Borrower or Guarantors, as the case may be, shall timely pay any Other Taxes imposed by the relevant Governmental Authority in accordance with applicable law or, at the option of a Recipient, timely reimburse such Recipient for any Other Taxes paid. (c) The Borrower and Guarantors shall jointly and severally indemnify each Recipient, within ten (10) days after written demand therefor, for the full amount of any Indemnified Taxes or Other Taxes paid by such Recipient on or with respect to any payment by or on account of any obligation of a Borrower or Guarantor, as the case may be, hereunder or under any other Loan Document (including Indemnified Taxes or Other Taxes imposed or asserted on or attributable to amounts payable under this Section 2.17) and any reasonable expenses arising therefrom or with respect thereto, whether or not such Indemnified Taxes or Other Taxes were correctly or legally imposed or asserted by the relevant Governmental Authority. A certificate setting forth in reasonable detail the basis for and calculation of such payment or liability delivered to the applicable Borrower or Guarantor, as the case may be, by a Lender or the Issuing Bank, or by the Administrative Agent on its own behalf or on behalf of a Lender or the Issuing Bank, shall be conclusive absent manifest error.

114 (d) As soon as practicable after any payment of Indemnified Taxes or Other Taxes by a Borrower or Guarantor, as the case may be, to a Governmental Authority, such Borrower or Guarantor, as the case may be, shall deliver to the Administrative Agent the original or a certified copy of a receipt issued by such Governmental Authority evidencing such payment, a copy of the return reporting such payment or other evidence of such payment reasonably satisfactory to the Administrative Agent. (e) Any Lender that is entitled to an exemption from or reduction of withholding Tax with respect to payments made hereunder or under any other Loan Document shall deliver to the Borrower or Guarantors and the Administrative Agent, at the time or times reasonably requested by the Borrower or Guarantors or the Administrative Agent, such properly completed and executed documentation reasonably requested by the Borrower or Guarantors or the Administrative Agent as will permit such payments to be made without withholding or at a reduced rate of withholding. In addition, any Lender, if reasonably requested by the Borrower or Guarantors or the Administrative Agent, shall deliver such other properly completed and executed documentation prescribed by applicable law or reasonably requested by the Borrower or Guarantors or the Administrative Agent as will enable the Borrower or Guarantors or the Administrative Agent to determine whether or not such Lender is subject to backup withholding or information reporting requirements. Notwithstanding anything to the contrary in the preceding two sentences, the completion, execution and submission of such documentation (other than such documentation set forth in Section 2.17(f), (g) and (k) below) shall not be required if in the Lender’s reasonable judgment such completion, execution or submission would subject such Lender to any material unreimbursed cost or expense or would materially prejudice the legal or commercial position of such Lender. (f) Without limiting the generality of Section 2.17(e), each Lender and Administrative Agent that is a “United States person,” as defined in Section 7701(a)(30) of the Code, shall deliver on or about the date on which such Lender or Administrative Agent becomes a Lender or Administrative Agent under this Agreement (and from time to time thereafter upon the reasonable request of the Borrower or the Administrative Agent), to the Borrower Representative and the Administrative Agent (as applicable), a properly completed and duly executed copy of United States Internal Revenue Form W-9 or any successor form, certifying that such Lender or Administrative Agent, as applicable, is exempt from United States backup withholding Tax on payments made hereunder. (g) Without limiting the generality of Section 2.17(e), each Foreign Lender shall, to the extent it is legally entitled to do so, deliver to the Borrower and the Administrative Agent (in such number of copies as shall be requested by the recipient) on or about the date on which such Foreign Lender becomes a Lender under this Agreement (and from time to time thereafter upon the reasonable request of the Borrower or the Administrative Agent), whichever of the following is applicable: (i) in the case of a Foreign Lender claiming the benefits of an income tax treaty to which the United States is a party (x) with respect to payments of interest under any Loan Document, executed copies of IRS Form W-8BEN or IRS Form W-8BEN-E establishing an exemption from, or reduction of, U.S. federal withholding Tax pursuant to the “interest” article of such tax treaty and (y) with respect to any other

115 applicable payments under any Loan Document, executed copies of IRS Form W-8BEN or IRS Form W-8BEN-E establishing an exemption from, or reduction of, U.S. federal withholding Tax pursuant to the “business profits” or “other income” article of such tax treaty; (ii) executed copies of IRS Form W-8ECI; (iii) in the case of a Foreign Lender claiming the benefits of the exemption for portfolio interest under Section 881(c) of the Code, (x) a certificate substantially in the form of Exhibit F-1 to the effect that such Foreign Lender is not a “bank” within the meaning of Section 881(c)(3)(A) of the Code, a “10-percent shareholder” of the Borrower within the meaning of Section 871(h)(3)(B) of the Code, or a “controlled foreign corporation” related to the Borrower as described in Section 881(c)(3)(C) of the Code (a “U.S. Tax Compliance Certificate”) and (y) executed copies of IRS Form W-8BEN or IRS Form W-8BEN-E; or (iv) to the extent a Foreign Lender is not the beneficial owner, executed copies of IRS Form W-8IMY, accompanied by IRS Form W-8ECI, IRS Form W- 8BEN, IRS Form W 8BEN-E, a U.S. Tax Compliance Certificate substantially in the form of Exhibit F-2 or Exhibit F-3, IRS Form W-9, and/or other certification documents from each beneficial owner, as applicable; provided that if the Foreign Lender is a partnership and one or more direct or indirect partners of such Foreign Lender are claiming the portfolio interest exemption, such Foreign Lender may provide a U.S. Tax Compliance Certificate substantially in the form of Exhibit F-4 on behalf of each such direct and indirect partner. (h) Each Foreign Lender shall, to the extent it is legally entitled to do so, deliver to the Borrower and the Administrative Agent (in such number of copies as shall be requested by the recipient) on or about the date on which such Foreign Lender becomes a Lender under this Agreement (and from time to time thereafter upon the reasonable request of the Borrower or the Administrative Agent), executed copies of any other form prescribed by applicable law as a basis for claiming exemption from or a reduction in withholding Tax, duly completed, together with such supplementary documentation as may be prescribed by applicable law to permit the Borrower or the Administrative Agent to determine the withholding or deduction required to be made. (i) If the Administrative Agent or a Lender determines, in its sole discretion, that it (on a standalone or an affiliated group basis) has received a refund of any Taxes as to which it has been indemnified by any Borrower or Guarantor, as the case may be, or with respect to which any Borrower or Guarantor, as the case may be, has paid additional amounts pursuant to this Section 2.17, it shall pay over such refund to the applicable Borrower or Guarantor (but only to the extent of indemnity payments made, or additional amounts paid, by such Borrower or Guarantor under this Section 2.17 with respect to the Taxes giving rise to such refund), net of all out-of-pocket expenses (including Taxes) of the Administrative Agent or such Lender and without interest (other than any interest paid by the relevant Governmental Authority with respect to such refund); provided that the applicable Borrower or Guarantor, as the case may be, upon the request of the Administrative Agent or such Lender, agrees to repay the amount paid over to such

116 Borrower or Guarantor (plus any penalties, interest or other charges imposed by the relevant Governmental Authority) to the Administrative Agent or such Lender in the event the Administrative Agent or such Lender is required to repay such refund to such Governmental Authority. Notwithstanding anything to the contrary in this paragraph (i), in no event will the Administrative Agent or a Lender be required to pay any amount to any Borrower or Guarantor pursuant to this paragraph (i) the payment of which would place the Administrative Agent or Lender, as the case may be, in a less favorable net after-Tax position than the Administrative Agent or Lender would have been in if the Tax subject to indemnification and giving rise to such refund had not been deducted or otherwise imposed and the indemnification payments or additional amounts with respect to such Tax had never been paid. This paragraph (i) shall not be construed to require the Administrative Agent or any Lender to make available its tax returns (or any other information relating to its taxes which it deems confidential) to any Borrower, any Guarantor or any other Person. (j) Each Lender shall severally indemnify (A) the Administrative Agent for (i) any Indemnified Taxes or Other Taxes attributable to such Lender (but only to the extent that any Borrower or Guarantor, as the case may be, has not already indemnified the Administrative Agent for such Indemnified Taxes or Other Taxes and without limiting the obligation of the Borrower and each Guarantor to do so) and (ii) any Taxes attributable to such Lender’s failure to comply with the provisions of Section 9.04(c) relating to the maintenance of a Participant Register, and (B) any Borrower or Guarantor, or the Administrative Agent, as the case may be, for any Excluded Taxes attributable to such Lender, in each case, that are paid or payable by any Borrower or Guarantor, or the Administrative Agent, as the case may be, in connection with any Loan Document and any reasonable expenses arising therefrom or with respect thereto, whether or not such Taxes were correctly or legally imposed or asserted by the relevant Governmental Authority. The indemnity under this Section 2.17(j) shall be paid within ten (10) days after the Administrative Agent or a Borrower or Guarantor (as applicable) delivers to the applicable Lender a certificate stating the amount of Taxes so paid or payable by the Administrative Agent or such Borrower or Guarantor (as applicable). Such certificate shall be conclusive of the amount so paid or payable absent manifest error. Each Lender hereby authorizes the Administrative Agent to set off and apply any and all amounts at any time owing to such Lender under any Loan Document or otherwise payable by the Administrative Agent to the Lender from any other source against any amount due to the Administrative Agent under this paragraph (j). (k) If a payment made to a Lender under any Loan Document would be subject to deduction or withholding for any Tax imposed pursuant to FATCA if such Lender were to fail to comply with the applicable reporting requirements of FATCA (including those contained in Section 1471(b) or 1472(b) of the Code, as applicable), such Lender shall deliver to the applicable Borrower or Guarantor and the Administrative Agent at the time or times prescribed by law and at such time or times reasonably requested by such Borrower or Guarantor or the Administrative Agent such documentation prescribed by applicable law (including as prescribed by Section 1471(b)(3)(C)(i) of the Code) and such additional documentation reasonably requested by such Borrower or Guarantor or the Administrative Agent as may be necessary for the Borrower or Guarantor and the Administrative Agent to comply with their obligations under FATCA and to determine that such Lender has complied with such Lender’s obligations under FATCA or to determine the amount to deduct and withhold from such payment. If a Lender confirms to a Borrower or Guarantor or the Administrative Agent pursuant to this clause (k) that it is entitled to

117 receive payments free from any deduction or withholding for any Tax imposed pursuant to FATCA, and it subsequently becomes aware that it is not or has ceased to be so entitled, that Lender shall notify that other party reasonably promptly. Solely for purposes of this Section 2.17(k), “FATCA” shall include any amendments made to FATCA after the Effective Date. (l) EACH FOREIGN LENDER LISTED ON SCHEDULE 2.01 REPRESENTS AND WARRANTS THAT, AS OF THE EFFECTIVE DATE, ASSUMING COMPLIANCE WITH PROCEDURAL FORMALITIES, AMOUNTS PAYABLE TO SUCH FOREIGN LENDER PURSUANT TO THIS AGREEMENT ARE EXEMPT FROM U.S. FEDERAL WITHHOLDING TAX. EACH FOREIGN LENDER WHICH BECOMES A LENDER AFTER THE EFFECTIVE DATE HEREBY REPRESENTS AND WARRANTS THAT, ON THE DATE SUCH FOREIGN LENDER FIRST BECAME A LENDER HEREUNDER, ASSUMING COMPLIANCE WITH PROCEDURAL FORMALITIES, AMOUNTS PAYABLE TO SUCH FOREIGN LENDER PURSUANT TO THIS AGREEMENT ARE EXEMPT FROM U. S. FEDERAL WITHHOLDING TAX OR WOULD BE SO EXEMPT BUT FOR ONE OR MORE CHANGES IN LAW WHICH HAVE OCCURRED AFTER THE EFFECTIVE DATE. (m) Each party’s obligations under this Section 2.17 shall survive the resignation or replacement of the Administrative Agent or any assignment of rights by, or the replacement of, a Lender, the termination of the Commitments and the repayment, satisfaction or discharge of all obligations under any Loan Document. Section 2.18 Payments Generally; Allocations of Proceeds; Pro Rata Treatment; Sharing of Set-offs. (a) The Borrower shall make each payment required to be made by it hereunder (whether of principal, interest, fees or reimbursement of LC Disbursements, or of amounts payable under Section 2.15, 2.16 or 2.17, or otherwise) prior to (i) in the case of payments denominated in Dollars, 12:00 noon, New York City time and (ii) in the case of payments denominated in a Foreign Currency, 12:00 noon, Local Time, in the city of the Administrative Agent’s Foreign Currency Payment Office for such currency, in each case on the date when due, in immediately available funds, without set-off or counterclaim. Any amounts received after such time on any date may, in the discretion of the Administrative Agent, be deemed to have been received on the next succeeding Business Day for purposes of calculating interest thereon. All such payments shall be made (i) in the same currency in which the applicable Credit Event was made (or where such currency has been converted to euro, in euro) and (ii) to the Administrative Agent at its offices at 2001 Ross Ave, 37th Floor Dallas, TX 75201 or such other office as the Administrative Agent may hereafter designate in writing as such to the other parties hereto, or, in the case of a Credit Event denominated in a Foreign Currency, the Administrative Agent’s Foreign Currency Payment Office for such currency, except payments to be made directly to the Issuing Bank or Swingline Lender as expressly provided herein and except that payments pursuant to Sections 2.15, 2.16, 2.17 and 9.03 shall be made directly to the Persons entitled thereto. The Administrative Agent shall distribute any such payments denominated in the same currency received by it for the account of any other Person to the appropriate recipient promptly following receipt thereof. If any payment hereunder shall be due on a day that is not a Business Day, the date for payment shall be extended to the next succeeding Business Day, and, in the case of any payment accruing interest, interest thereon shall be payable for the period of such extension. Notwithstanding the foregoing

118 provisions of this Section 2.18, if, after the making of any Credit Event in any Foreign Currency, currency control or exchange regulations are imposed in the country which issues such currency with the result that the type of currency in which the Credit Event was made (the “Original Currency”) no longer exists or the respective Borrower is not able to make payment to the Administrative Agent for the account of the applicable Lenders in such Original Currency, then all payments to be made by the respective Borrower hereunder in such currency shall instead be made when due in Dollars in an amount equal to the Dollar Amount (as of the date of repayment) of such payment due, it being the intention of the parties hereto that the respective Borrower takes all risks of the imposition of any such currency control or exchange regulations. (b) Notwithstanding anything herein or in any other Loan Document to the contrary, if (i) an Event of Default has occurred and is continuing, (ii) the Collateral Agent is taking action at any time to enforce rights in respect of any Collateral pursuant to the Loan Documents or applicable law, (iii) any distribution is made to or proceeds are received by any Secured Party on account of any Collateral during such Event of Default or in connection with any enforcement in respect thereof or in connection with any Insolvency or Liquidation Proceeding of the Loan Parties (including in the form of any adequate protection payments) or (iv) any payment is received by any Secured Party pursuant to the Intercreditor Agreement or any other Approved Intercreditor Agreement with respect to any Collateral, then the proceeds of any sale, collection or other liquidation of the Collateral by the Administrative Agent or any other Secured Party (or at the direction or with the consent of the Required Revolving Lenders or the Required Lenders, as applicable) and the proceeds of any distribution or payment shall be applied as follows: (i) while the Intercreditor Agreement is in effect, subject to and as set forth therein; and (ii) any amounts received pursuant to the Intercreditor Agreement or otherwise shall be applied to the Secured Obligations in the following order: first, to pay that portion of the Secured Obligations constituting fees, indemnities, expense reimbursements or other amounts (but not principal or interest) then due to the Administrative Agent or Collateral Agent (in their respective capacities as such), second, to pay that portion of Revolving Facility Obligations constituting fees (including commitment, fronting and participation fees), indemnities, or expense reimbursements then due to the Revolving Lenders, Swingline Lenders or Issuing Banks (in their respective capacities as such) ratably among them in proportion to the amounts described in this second clause, third, to pay that portion of Revolving Facility Obligations constituting accrued and unpaid interest (including post-petition interest, whether or not an allowed claim in any Insolvency or Liquidation Proceeding) on the Revolving Loans and Swingline Loans then due to the Revolving Lenders or Swingline Lenders (in their respective capacities as such) ratably among them in proportion to the amounts described in this third clause,

119 fourth, (w) to repay that portion of Revolving Secured Obligations constituting unpaid principal on the Revolving Loans and Swingline Loans and unreimbursed LC Disbursements, (x) to Cash Collateralize all undrawn Letters of Credit in an amount equal to one hundred five percent (105%) of the aggregate undrawn face amount of such outstanding Letters of Credit, (y) to pay that portion of Revolving Secured Obligations constituting unpaid Banking Services Obligations and (z) to pay that portion of Revolving Secured Obligations constituting unpaid Swap Obligations, in each case, then due and payable to the Secured Parties ratably among them in proportion to the amounts described in this fourth clause, fifth, to pay in full all other outstanding Revolving Secured Obligations then due to the Secured Parties ratably among them in accordance with this Agreement and the other Loan Documents, sixth, to pay that portion of Term Loan Facility Obligations constituting fees (including commitment, fronting, and participation fees), indemnities, expense reimbursements or other amounts (but not principal or interest) then due to the Term Lenders (in their capacities as such) ratably among them in proportion to the amounts described in this sixth clause, seventh, to pay that portion of Term Loan Facility Obligations constituting accrued and unpaid interest (including post-petition interest, whether or not an allowed claim in any Insolvency or Liquidation Proceeding) on the Term Loans then due to the Term Lenders (in their capacities as such) ratably among them in proportion to the amounts described in this seventh clause, eighth, (x) to repay that portion of Term Loan Secured Obligations constituting unpaid principal on the Term Loans, (y) to pay that portion of Term Loan Secured Obligations constituting unpaid Banking Services Obligations and (z) to pay that portion of Term Loan Secured Obligations constituting unpaid Swap Obligations, in each case, then due and payable to the Secured Parties ratably among them in proportion to the amounts described in this eighth clause, ninth, to pay in full all other outstanding Term Loan Secured Obligations then due to the Secured Parties ratably among them in accordance with this Agreement and the other Loan Documents, and last, the balance, if any, to the Borrower or as otherwise required by law. Subject to Section 2.6, amounts used to Cash Collateralize the aggregate undrawn amount of Letters of Credit pursuant to clause fourth above shall be applied to satisfy drawings under such Letters of Credit as they occur. If any amount remains on deposit as Cash Collateral after all Letters of Credit have either been fully drawn or expired, such remaining amount shall be applied to the other Secured Obligations, if any, in the order set forth above and, if no Secured Obligations remain outstanding, to the Borrower.

120 Notwithstanding the foregoing, (a) amounts received from the Borrower or any Guarantor that is not a ECP shall not be applied to the obligations that are Excluded Swap Obligations (it being understood, that in the event that any amount is applied to Secured Obligations other than Excluded Swap Obligations as a result of this clause (a), to the extent permitted by applicable law, the Administrative Agent shall make such adjustments as it determines are appropriate to distributions pursuant to clauses fourth or eighth above (as applicable) from amounts received from ECP to ensure, as nearly as possible, that the proportional aggregate recoveries with respect to obligations described in clauses fourth or eighth above (as applicable) by the holders of any Excluded Swap Obligations are the same as the proportional aggregate recoveries with respect to other obligations pursuant to clauses fourth or eighth above (as applicable)) and (b) Banking Services Obligations and Swap Obligations shall be excluded from the application described above if the Administrative Agent has not received written notice thereof, together with such supporting documentation as the Administrative Agent may request, from the applicable holder of such obligations. Notwithstanding anything to the contrary contained in this Agreement, unless so directed by the respective Borrower, or unless a Default is in existence, none of the Administrative Agent or any Lender shall apply any payment which it receives to any Term Benchmark Loan of a Class, except (a) on the expiration date of the Interest Period or maturity date (as applicable) applicable to any such Term Benchmark Loan or (b) in the event, and only to the extent, that there are no outstanding ABR Loans or Canadian Prime Rate Loans (as applicable) of the same Class and, in any event, the Borrower shall pay the break funding payment required in accordance with Section 2.16. The Administrative Agent and the Lenders shall have the continuing and exclusive right to apply and reverse and reapply any and all such received proceeds and payments to any portion of the Secured Obligations. (c) At the election of the Administrative Agent, all payments of principal, interest, LC Disbursements, fees, premiums, reimbursable expenses (including, without limitation, all reimbursement for fees and expenses pursuant to Section 9.03), and other sums due and payable under the Loan Documents, may be paid from the proceeds of Borrowings made hereunder pursuant to Section 2.02. (d) If any Lender shall, by exercising any right of set-off or counterclaim or otherwise, obtain payment in respect of any principal of or interest on any of its Loans or participations in LC Disbursements or Swingline Loans resulting in such Lender receiving payment of a greater proportion of the aggregate amount of its Loans and participations in LC Disbursements and Swingline Loans and accrued interest thereon than the proportion received by any other Lender, then the Lender receiving such greater proportion shall purchase (for cash at face value) participations in the Loans and participations in LC Disbursements and Swingline Loans of other Lenders to the extent necessary so that the benefit of all such payments shall be shared by the Lenders ratably in accordance with the aggregate amount of principal of and accrued interest on their respective Loans and participations in LC Disbursements and Swingline Loans; provided that (i) if any such participations are purchased and all or any portion of the payment giving rise thereto is recovered, such participations shall be rescinded and the purchase price restored to the extent of such recovery, without interest, (ii) the provisions of this paragraph shall not be construed to apply to any payment made by the Borrower pursuant to and in accordance with the express terms of this Agreement, any Incremental Amendment, Extension Amendment or Refinancing Amendment or any payment obtained by a Lender as consideration

121 for the assignment of or sale of a participation in any of its Loans or participations in LC Disbursements and Swingline Loans to any assignee or participant, other than, except as provided in Sections 2.24, 9.04(g) or 9.04(k), to Parent or any Subsidiary thereof (as to which the provisions of this paragraph shall apply) and (iii) nothing in this Section 2.18(d) shall be construed to limit the applicability of Section 2.18(b) in the circumstances where Section 2.18(b) is applicable in accordance with its terms. The Borrower consent to the foregoing and agree, to the extent they may effectively do so under applicable law, that any Lender acquiring a participation pursuant to the foregoing arrangements may exercise against the Borrower right of set-off and counterclaim with respect to such participation as fully as if such Lender were a direct creditor of the Borrower in the amount of such participation. (e) Unless the Administrative Agent shall have received notice from a Borrower prior to the date on which any payment is due to the Administrative Agent for the account of the relevant Lenders or the Issuing Bank hereunder that such Borrower will not make such payment, the Administrative Agent may assume that such Borrower has made such payment on such date in accordance herewith and may, in reliance upon such assumption, distribute to the relevant Lenders or the Issuing Bank, as the case may be, the amount due. In such event, if such Borrower has not in fact made such payment, then each of the relevant Lenders or the Issuing Bank, as the case may be, severally agrees to repay to the Administrative Agent forthwith on demand the amount so distributed to such Lender or Issuing Bank with interest thereon, for each day from and including the date such amount is distributed to it to but excluding the date of payment to the Administrative Agent, at the greater of the Federal Funds Effective Rate and a rate determined by the Administrative Agent in accordance with banking industry rules on interbank compensation (including without limitation the Overnight Foreign Currency Rate in the case of Loans denominated in a Foreign Currency). (f) Subject to Section 2.22, if any Lender shall fail to make any payment required to be made by it pursuant to Section 2.05(c), 2.06(d) or (e), 2.07(b), 2.18(e) or 9.03(c), then the Administrative Agent may, in its discretion (notwithstanding any contrary provision hereof), (i) apply any amounts thereafter received by the Administrative Agent for the account of such Lender and for the benefit of the Administrative Agent, the Swingline Lender or the Issuing Bank to satisfy such Lender’s obligations to it under such Section 2.18 until all such unsatisfied obligations are fully paid and/or (ii) hold any such amounts in a segregated account as cash collateral for, and application to, any future funding obligations of such Lender under any such Section 2.18; in the case of each of clauses (i) and (ii) above, in any order as determined by the Administrative Agent in its discretion. (g) Without limiting the generality of the foregoing, Section 2.18 is intended to constitute and shall be deemed to constitute a “subordination agreement” within the meaning of Section 510(a) of the Bankruptcy Code and is intended to be and shall be interpreted to be enforceable to the maximum extent permitted pursuant to applicable nonbankruptcy law. Amounts applied pursuant to Section 2.18(b) are to be applied, for the avoidance of doubt, in the order required by such clause until the payment in full in cash of the applicable Secured Obligations referred to in the applicable clause. (h) If any Secured Party collects or receives any amounts received on account of the Secured Obligations of which it is not entitled under Section 2.18(b), such Secured

122 Party shall hold the same in trust for the applicable Secured Parties entitled thereto and shall forthwith deliver the same to the Administrative Agent, for the account of such Secured Parties, to be applied in accordance with Section 2.18(b) hereof, in each case until the prior payment in full of cash of the applicable Secured Obligations due and owing to Secured Parties in accordance with Section 2.18(b). Section 2.19 Mitigation Obligations; Replacement of Lenders. (a) If any Lender requests compensation under Section 2.15, if the Borrower is required to pay any additional amount to any Lender or any Governmental Authority for the account of any Lender pursuant to Section 2.17 or if any Lender delivers a notice pursuant to Section 2.26, then such Lender shall use reasonable efforts to designate a different lending office for funding or booking its Loans hereunder or to assign its rights and obligations hereunder to another of its offices, branches or affiliates, if, in the judgment of such Lender, such designation or assignment would (i) eliminate or reduce amounts payable pursuant to Section 2.15 or 2.17, as the case may be, in the future and (ii) not subject such Lender to any material unreimbursed cost or expense and would not otherwise be materially disadvantageous to such Lender. The Borrower hereby jointly and severally agrees to pay all reasonable costs and expenses incurred by any Lender in connection with any such designation or assignment. (b) If (i) any Lender requests compensation under Section 2.15, or (ii) the Borrower is required to pay any additional amount to any Lender or any Governmental Authority for the account of any Lender pursuant to Section 2.17 or if any Lender delivers a notice pursuant to Section 2.26, or (iii) any Lender becomes a Defaulting Lender, then the Borrower may, at its sole expense and effort, upon notice to such Lender and the Administrative Agent, require such Lender to assign and delegate, without recourse (in accordance with and subject to the restrictions contained in Section 9.04), all its interests, rights and obligations (other than its existing rights to payment pursuant to Sections 2.15 and 2.17) under the Loan Documents to an assignee that shall assume such obligations (which assignee may be another Lender, if a Lender accepts such assignment); provided that (i) the Borrower shall have received the prior written consent of the Administrative Agent (and if a Revolving Commitment is being assigned, the Issuing Bank), which consent shall not unreasonably be withheld, (ii) such Lender shall have received payment of an amount equal to the outstanding principal of its Loans and participations in LC Disbursements and Swingline Loans, accrued interest thereon, accrued fees and all other amounts payable to it hereunder (including any amounts under Section 2.16), from the assignee (to the extent of such outstanding principal and accrued interest and fees) or each applicable Borrower (in the case of all other amounts), (iii) in the case of any such assignment resulting from a claim for compensation under Section 2.15 or payments required to be made pursuant to Section 2.17, such assignment will result in a reduction in such compensation or payments and (iv) such assignment does not conflict with applicable law. A Lender shall not be required to make any such assignment and delegation if, prior thereto, as a result of a waiver by such Lender or otherwise, the circumstances entitling the Borrower to require such assignment and delegation cease to apply. Section 2.20 Incremental Credit Extensions. (a) Any Borrower, may, by written notice to the Administrative Agent (whereupon the Administrative Agent shall promptly deliver a copy to each of the Lenders) from time to time after the Effective Date, request Incremental Term Loan Commitments and/or Incremental Revolving Commitments, as applicable, in an aggregate amount

123 not to exceed (when aggregated with any Incremental Equivalent Debt) the Incremental Amount from one or more Incremental Term Lenders and/or Incremental Revolving Lenders (which, in each case, may include any existing Lender, but shall be required to be Persons which would qualify as assignees of a Lender in accordance with Section 9.04) willing to provide such Incremental Term Loans and/or Incremental Revolving Commitments, as the case may be, in their own discretion. Each notice provided pursuant to this Section 2.20 shall set forth (i) the amount of the Incremental Term Loan Commitments and/or Incremental Revolving Commitments being requested (which shall be in minimum increments of $10,000,000 and a minimum amount of $25,000,000 or equal to the remaining Incremental Amount), (ii) the date on which such Incremental Term Loan Commitments and/or Incremental Revolving Commitments are requested to become effective, (iii) in the case of Incremental Revolving Commitments, whether such Incremental Revolving Commitments are to constitute an increase to the Dollar Tranche Commitments or Multicurrency Tranche Commitments; provided that, where multiple Classes of Revolving Commitments exist with different Maturity Dates, any Incremental Revolving Commitments shall constitute and increase to the Class of Revolving Commitments with the Latest Maturity Date; provided, further that the aggregate amount of all Incremental Revolving Commitments established hereunder shall not exceed $100,000,000 and (iv) in the case of Incremental Term Loan Commitments, whether such Incremental Term Loan Commitments are commitments to make term loans with the same interest rates, amortization, maturity and other terms as the Initial Term Loans made on the Effective Date or commitments to make term loans with interest rates and/or amortization and/or maturity and/or other terms different from the Initial Term Loans (“Other Term Loans”). (b) [reserved]. (c) The applicable Borrower and each Incremental Term Lender shall execute and deliver to the Administrative Agent an Incremental Amendment and such customary other documentation as the Administrative Agent shall reasonably specify to evidence the Incremental Term Loan Commitment of such Incremental Term Lender. Each Incremental Amendment providing for Incremental Term Loans shall specify the terms of the applicable Incremental Term Loans; provided that (i) except as to pricing, amortization, mandatory prepayments and final maturity date (which shall, subject to clause (ii), (iii) and (iv) of this proviso, be determined by such Borrower and the Incremental Term Lenders in their sole discretion), the Other Term Loans shall have (x) the same terms as the Initial Term Loans or (y) such other terms as shall be reasonably satisfactory to the Administrative Agent, (ii) the final maturity date of any Other Term Loan shall be no earlier than the Latest Maturity Date with respect to then-existing Term Loans (except for Other Term Loans incurred pursuant to the Inside Maturity Basket), (iii) the Weighted Average Life to Maturity of any Other Term Loans shall be no shorter than the remaining Weighted Average Life to Maturity of the Class of Term Loans with the Latest Maturity Date (except for Other Term Loans incurred pursuant to the Inside Maturity Basket), (iv) the prepayment provisions of any Other Term Loans shall not be more favorable than the prepayment provisions applicable to the Initial Term Loans that will remain outstanding after giving effect to the incurrence of such Other Term Loans and use of proceeds thereof and (v) the Effective Yield of any Other Term Loans may exceed the Effective Yield then applicable to the Initial Term Loans; provided that the Effective Yield for the Initial Term Loans shall be increased to the extent necessary (without any further action by any party or any amendment hereto) so that the Effective Yield for such Initial Term Loans is not less than the Effective Yield of any such Other Term

124 Loans minus 0.50%; provided, however that the foregoing proviso (x) shall only be effective until the date that is twelve months after the Effective Date, (y) shall not apply to any Other Term Loans or Incremental Equivalent Debt incurred in reliance on the Incremental Ratio Basket and (z) shall not apply to any Other Term Loans (or Incremental Equivalent Debt) with a final maturity date at least 24 months after the Maturity Date then applicable to the Initial Term Loans and a Weighted Average Life to Maturity longer by 24 months or more than the Weighted Average Life to Maturity then applicable to the Initial Term Loans. The Incremental Term Loans shall rank pari passu or junior in right of payment and of security with the Initial Term Loans and shall not be (x) secured by any property or assets of Parent or any Restricted Subsidiary other than the Collateral or (y) guaranteed by Parent or any of its Restricted Subsidiaries other than the Guarantors; provided that, if such Other Term Loans rank junior in right of security with the Initial Term Loans, such Other Term Loans will be established as a separate Tranche from the Initial Term Loans. In the case of any junior lien Incremental Term Loans, such Indebtedness shall be subject to the terms of an Approved Intercreditor Agreement. (d) The Borrower and each Incremental Revolving Lender shall execute and deliver to the Administrative Agent an Incremental Amendment and such other customary documentation as the Administrative Agent shall reasonably specify to evidence the Incremental Revolving Commitment of such Incremental Revolving Lender. Any Incremental Revolving Commitment established hereunder shall have terms identical to (and shall form part of) such Class of Revolving Commitments with the Latest Maturity Date existing on the Effective Date, it being understood that the Borrower and the Administrative Agent may make (without the consent of or notice to any other party) any amendment to reflect such increase in the Revolving Commitments. (e) Notwithstanding the foregoing, no Incremental Term Loan Commitment or Incremental Revolving Commitment shall become effective under this Section 2.20 unless, subject to Section 1.04, at the time that any such Incremental Term Loan or Incremental Revolving Commitment is made (and after giving effect thereto), (A) no Event of Default shall exist or would exist after giving effect thereto (including on a pro forma basis) and (B) the representations and warranties of the Borrower set forth in this Agreement shall be true and correct in all material respects (other than to the extent qualified by materiality or “Material Adverse Effect”, in which case, such representations and warranties shall be true and correct); provided that, in the event that the tranche of Incremental Term Loans is used to finance an acquisition or other Investment permitted by this Agreement and to the extent the Incremental Term Lenders participating in such tranche of Incremental Term Loans agree, the foregoing clause (B) shall be limited to customary “specified representations” and those representations included in the agreement related to such acquisition or other Investment that are material to the interests of the Lenders and only to the extent that Parent or its applicable Subsidiary has the right to terminate its obligations under such agreement as a result of a breach of such representations. The Administrative Agent shall promptly notify each Lender as to the effectiveness of each Incremental Amendment. Each of the parties hereto hereby agrees that, upon the effectiveness of any Incremental Amendment, this Agreement shall be amended to the extent (but only to the extent) necessary to reflect the existence and terms of the Incremental Term Loan Commitments and/or Incremental Revolving Commitments evidenced thereby. Any such deemed amendment may be memorialized in writing by the Administrative Agent with the Borrower’s consent (not to be unreasonably withheld, delayed or conditioned) and furnished to the other parties hereto. Each Incremental Amendment shall be delivered to the Administrative Agent, together with such

125 documents (including local law confirmations as to Collateral) and legal opinions substantially consistent with those delivered on the Effective Date (other than changes to such legal opinions resulting from a Change in Law, change in fact or change to counsel’s form of opinion) as to such matters as are reasonably requested by the Administrative Agent. (f) The Incremental Amendment may, without the consent of the Administrative Agent, the Collateral Agent or Lenders, effect such amendments to this Agreement and the other Loan Documents as may be necessary or appropriate, in the reasonable opinion of the Administrative Agent and the Borrower, to effect the provisions of this Section 2.20, including those required by the Incremental Term Loan Lenders or Incremental Revolving Lenders, as applicable (and not adverse to any existing Lender after giving effect to the Incremental Loans made pursuant to such amendment). The Borrower will use the proceeds of the Incremental Term Loans and Incremental Revolving Loans for their general corporate purposes (including loans and other Investments in Parent and its Subsidiaries as permitted herein). Incremental Term Loans and Incremental Revolving Commitments may be made by any existing Lender (but no existing Lender will have any obligation to make or provide any portion of any Incremental Term Loan or Incremental Revolving Commitments) or by any other bank or other financial institution; provided that any bank or financial institution (including any new or existing Lenders) providing Incremental Revolving Commitments shall be reasonably satisfactory to the Administrative Agent, each Issuing Bank and the Borrower. No Lender shall be obligated to provide any Incremental Term Loans or Incremental Revolving Commitments, unless it so agrees. (g) This Section 2.20 shall supersede any provisions in Section 2.18 or 9.02 to the contrary. Section 2.21 Judgment Currency. If for the purposes of obtaining judgment in any court it is necessary to convert a sum due from a Borrower hereunder in the currency expressed to be payable herein (the “specified currency”) into another currency, the parties hereto agree, to the fullest extent that they may effectively do so, that the rate of exchange used shall be that at which in accordance with normal banking procedures the Administrative Agent could purchase the specified currency with such other currency at the Administrative Agent’s main New York City office on the Business Day preceding that on which final, non-appealable judgment is given. The obligations of the Borrower in respect of any sum due to any Lender or the Administrative Agent hereunder shall, notwithstanding any judgment in a currency other than the specified currency, be discharged only to the extent that on the Business Day following receipt by such Lender or the Administrative Agent (as the case may be) of any sum adjudged to be so due in such other currency such Lender or the Administrative Agent (as the case may be) may in accordance with normal, reasonable banking procedures purchase the specified currency with such other currency. If the amount of the specified currency so purchased is less than the sum originally due to such Lender or the Administrative Agent, as the case may be, in the specified currency, the applicable Borrower agrees, to the fullest extent that it may effectively do so, as a separate obligation and notwithstanding any such judgment, to indemnify such Lender or the Administrative Agent, as the case may be, against such loss, and if the amount of the specified currency so purchased exceeds (a) the sum originally due to any Lender or the Administrative Agent, as the case may be, in the specified currency and (b) any amounts shared with other Lenders as a result of allocations of such excess as a disproportionate payment to such Lender under Section 2.18, such Lender or the Administrative Agent, as the case may be, agrees to remit such excess to such Borrower.

126 Section 2.22 Defaulting Lenders. Notwithstanding any provision of this Agreement to the contrary, if any Revolving Lender becomes a Defaulting Lender, then the following provisions shall apply for so long as such Revolving Lender is a Defaulting Lender: (a) fees shall cease to accrue on the unfunded portion of the Revolving Commitment of such Defaulting Lender pursuant to Section 2.12(a); (b) the unused Commitment and Revolving Credit Exposure of such Defaulting Lender shall not be included in determining whether the Required Lenders or the Required Revolving Lenders, as applicable, have taken or may take any action hereunder (including any consent to any amendment, waiver or other modification pursuant to Section 9.02); provided that this clause (b) shall not apply to the vote of a Defaulting Lender in the case of an amendment, waiver or other modification requiring the consent of such Lender or each Lender affected thereby under Section 9.02; (c) if any Swingline Exposure or LC Exposure exists at the time such Revolving Lender becomes a Defaulting Lender then: (i) so long as no Default has occurred and is continuing: (1) all or any part of the Swingline Exposure of such Defaulting Lender shall be reallocated among the non-Defaulting Dollar Tranche Lenders in accordance with their respective Dollar Tranche Percentages (after giving effect to the reallocation provisions of Section 2.05(d)) but only to the extent (A) the sum of all non-Defaulting Lenders’ Dollar Tranche Revolving Credit Exposures plus such Defaulting Lender’s Swingline Exposure does not exceed the total of all non-Defaulting Dollar Tranche Lenders’ Dollar Tranche Commitments and (B) each non-Defaulting Lender’s Dollar Tranche Revolving Credit Exposure in respect of any Class does not exceed such non-Defaulting Lender’s Dollar Tranche Commitment in respect of such Class; and (2) all or any part of the Dollar Tranche LC Exposure of such Defaulting Lender shall be reallocated among the non-Defaulting Dollar Tranche Lenders in accordance with their respective Dollar Tranche Percentages (after giving effect to the reallocation provisions of Section 2.06(k)) but only to the extent (A) the sum of all non-Defaulting Lenders’ Dollar Tranche Revolving Credit Exposures plus such Defaulting Lender’s Dollar Tranche LC Exposure does not exceed the total of all non-Defaulting Dollar Tranche Lenders’ Dollar Tranche Commitments and (B) each non- Defaulting Lender’s Dollar Tranche Revolving Credit Exposure in respect of any Class does not exceed such non-Defaulting Lender’s Dollar Tranche Commitment in respect of such Class; and all or any part of the Multicurrency Tranche LC Exposure of such Defaulting Lender shall be reallocated among the non-Defaulting Multicurrency Tranche Lenders in accordance with their respective Multicurrency Tranche Percentages but only to the extent (E) the sum of all non-Defaulting Lenders’ Multicurrency Tranche Revolving Credit Exposures plus such Defaulting Lender’s Multicurrency Tranche LC Exposure does not exceed the total of all non-Defaulting Multicurrency Tranche Lenders’ Multicurrency Tranche Commitments and (F) each non-Defaulting Lender’s Multicurrency Tranche Revolving Credit Exposure in respect of any Class does not exceed such non-Defaulting Lender’s Multicurrency Tranche Commitment in respect of such Class;

127 (ii) if the reallocations described in clause (i) above cannot, or can only partially, be effected, the respective Borrower shall within one (1) Business Day following notice by the Administrative Agent (x) first, prepay such Swingline Exposure and (y) second, cash collateralize for the benefit of the Issuing Bank only the respective Borrower’s obligations corresponding to such Defaulting Lender’s LC Exposure (after giving effect to any partial reallocation pursuant to clause (i) above) in accordance with the procedures set forth in Section 2.06(j) for so long as such LC Exposure is outstanding; (iii) if the Borrower cash collateralizes any portion of such Defaulting Lender’s LC Exposure pursuant to clause (ii) above, the Borrower shall not be required to pay any fees to such Defaulting Lender pursuant to Section 2.12(b) with respect to such Defaulting Lender’s LC Exposure during the period (and to the extent) such Defaulting Lender’s LC Exposure is cash collateralized; (iv) if the LC Exposure of the non-Defaulting Lenders is reallocated pursuant to clause (ii) above, then the fees payable to the Lenders pursuant to Section 2.12(a) and Section 2.12(b) shall be adjusted in accordance with such non- Defaulting Lenders’ Applicable Percentages (after giving effect to the reallocation provisions of Sections 2.05(d) and 2.06(k)); and (v) if all or any portion of such Defaulting Lender’s LC Exposure is neither reallocated nor cash collateralized pursuant to clause (i) or (ii) above, then, without prejudice to any rights or remedies of the Issuing Bank or any other Lender hereunder, all letter of credit fees payable under Section 2.12(b) with respect to such Defaulting Lender’s LC Exposure shall be payable to the Issuing Bank until and to the extent that such LC Exposure is reallocated and/or cash collateralized; and (d) so long as such Lender is a Defaulting Lender, the Swingline Lender shall not be required to fund any Swingline Loan and the Issuing Bank shall not be required to issue, amend or increase any Letter of Credit, unless it is satisfied that the related exposure and the Defaulting Lender’s then outstanding LC Exposure will be 100% covered by the Revolving Commitments of the non-Defaulting Lenders and/or cash collateral will be provided by the Borrower in accordance with Section 2.22(c), and participating interests in any such newly made Swingline Loan or any newly issued or increased Letter of Credit shall be allocated among non- Defaulting Lenders in a manner consistent with Section 2.22(c)(i) (and such Defaulting Lender shall not participate therein). If (i) a Bankruptcy Event with respect to a Holding Company of any Lender shall occur following the Effective Date and for so long as such event shall continue or (ii) the Swingline Lender or the Issuing Bank has a good faith belief that any Lender has defaulted in fulfilling its obligations under one or more other agreements in which such Lender commits to extend credit, the Swingline Lender shall not be required to fund any Swingline Loan and the Issuing Bank shall not be required to issue, amend or increase any Letter of Credit, unless the Swingline Lender or the Issuing Bank, as the case may be, shall have entered into arrangements with the Borrower or such Lender, reasonably satisfactory to the Swingline Lender or the Issuing Bank, as the case may be, to defease any risk to it in respect of such Lender hereunder.

128 In the event that the Administrative Agent, the Borrower, the Issuing Bank and the Swingline Lender each agrees that a Defaulting Lender has adequately remedied all matters that caused such Lender to be a Defaulting Lender, then the Swingline Exposure and LC Exposure of the Lenders shall be readjusted to reflect the inclusion of such Lender’s Commitment and on such date such Lender shall purchase at par such of the Dollar Tranche Revolving Loans of any Class (other than Swingline Loans) and/or Multicurrency Tranche Revolving Loans of any Class of the other Lenders as the Administrative Agent shall determine may be necessary in order for such Lender to hold such Loans in accordance with its Applicable Percentage; provided that no adjustments will be made retroactively with respect to fees accrued or payments made by or on behalf of the Borrower while that Lender was a Defaulting Lender; provided, further, that, subject to Section 9.19, except to the extent otherwise expressly agreed by the affected parties, no change hereunder from Defaulting Lender to Lender will constitute a waiver or release of any claim of any party hereunder arising from that Lender’s having been a Defaulting Lender. Section 2.23 Extensions of Loans and Commitments. (a) Notwithstanding anything to the contrary in this Agreement, pursuant to one or more offers (each, an “Extension Offer”) made from time to time by the applicable Borrower(s) to all Term Lenders of Term Loans with a like Maturity Date, all Incremental Term Lenders of Incremental Term Loans with a like Maturity Date, all Lenders of Other Term Loans with a like Maturity Date, all Lenders of Other Refinancing Term Loans with a like Maturity Date, all Incremental Revolving Lenders of Incremental Revolving Commitments with a like Maturity Date, all Revolving Lenders with Revolving Commitments with a like Maturity Date or all Lenders with Other Refinancing Revolving Commitments with a like Maturity Date, in each case on a pro rata basis (based on the aggregate outstanding principal amount of the respective Loans or the aggregate amount of the Commitments with the same Maturity Date, as the case may be, and using Dollar Amounts in the case of any amounts denominated in an Agreed Currency other than Dollars) and on the same terms to each such Lender, the Borrower may from time to time offer to extend the Maturity Date for any such Term Loans, Incremental Term Loans, Other Term Loans, Other Refinancing Term Loans, Revolving Commitments, Incremental Revolving Commitments and/or Other Refinancing Revolving Commitments and otherwise modify the terms of such Loans and/or Commitments pursuant to the terms of the relevant Extension Offer (including by increasing the interest rate or fees payable in respect of such Loans and/or Commitments (and related outstandings) and/or modifying the amortization schedule in respect of such Lender’s Loans) (each, an “Extension”, and each group of Loans or Commitments, as applicable, in each case of a given Tranche as so extended, as well as the original Loans and Commitments of the original respective Tranche (in each case not so extended), shall (for the avoidance of doubt) be part of a single Tranche; and any Extended Term Loans, extended Incremental Term Loans or extended Other Term Loans shall constitute a separate Class of Term Loans from the Class of Term Loans from which they were converted, and any Extended Revolving Commitments shall constitute a separate Class of Revolving Commitments from the Class of Revolving Commitments from which they were converted), so long as the following terms are satisfied: (i) no Event of Default shall have occurred and be continuing at the time an Extension Offer is delivered to the Lenders or at the time of the Extension; (ii) except as to interest rates, fees and final maturity (which shall, subject to the requirements of this Section 2.23, be determined by Borrower and set

129 forth in the relevant Extension Offer), the Revolving Commitment, the Incremental Revolving Commitment or Other Refinancing Revolving Commitment of any Revolving Lender (an “Extending Revolving Lender”) extended pursuant to an Extension (an “Extended Revolving Commitment”), and the related outstandings, shall be a Revolving Commitment, Incremental Revolving Commitment or Other Refinancing Revolving Commitment (or related outstandings, as the case may be) with the same terms as the original Revolving Commitments of the same Class, the Incremental Revolving Commitments or Other Refinancing Revolving Commitments (and related outstandings); provided that (x) subject to the provisions of Sections 2.05(d) and 2.06(k) to the extent dealing with Letters of Credit and Swingline Loans which mature or expire after a Maturity Date when there exist Extended Revolving Commitments with a longer Maturity Date, all Letters of Credit and Swingline Loans shall be participated in on a pro rata basis by all Lenders with Revolving Commitments and Incremental Revolving Commitments in accordance with their pro rata share of the aggregate Revolving Commitments and Incremental Revolving Commitments (and except as provided in Sections 2.05(d) and 2.06(k), without giving effect to changes thereto on an earlier Maturity Date with respect to Swingline Loans and Letters of Credit theretofore incurred or issued) and all borrowings under Revolving Commitments of such Class and any related Incremental Revolving Commitments or Extended Revolving Commitments and repayments thereunder shall be made on a pro rata basis (except for (A) payments of interest and fees at different rates on Extended Revolving Commitments (and related outstandings) and (B) repayments required upon the Maturity Date for the non-extending Revolving Commitments of the same Class, or any related Incremental Revolving Commitments or Extended Revolving Commitments) and (y) at no time shall there be Revolving Commitments, Extended Revolving Commitments, Incremental Revolving Commitments and/or Other Refinancing Revolving Commitments hereunder (including Extended Revolving Commitments and any original Revolving Commitments) which have more than three different Maturity Dates; (iii) [reserved]; (iv) except as to interest rates, fees, amortization, final maturity date, premium, required prepayment dates and participation in prepayments (which shall, subject to the succeeding clauses (v), (vi) and (vii), be determined by the applicable Borrower and set forth in the relevant Extension Offer), the Term Loans of any Term Lender (an “Extending Term Lender”) extended pursuant to any Extension (“Extended Term Loans”) shall have the same terms as the Tranche of Term Loans subject to such Extension Offer; (v) the final maturity date for any Extended Term Loans shall be no earlier than the then Latest Maturity Date for Term Loans, respectively, hereunder and the amortization schedules applicable to Extended Term Loans pursuant to Section 2.10(b) for periods prior to the applicable Maturity Date may not be increased; (vi) the Weighted Average Life to Maturity of any Extended Term Loans shall be no shorter than the remaining Weighted Average Life to Maturity of the Term Loans extended thereby;

130 (vii) any Extended Term Loans may participate on a pro rata basis or a less than pro rata basis (but not greater than a pro rata basis) in any voluntary or mandatory repayments or prepayments hereunder, in each case as specified in the respective Extension Offer; (viii) if the aggregate principal amount of applicable Term Loans (calculated on the face amount thereof), Revolving Commitments, Incremental Revolving Commitments or Other Refinancing Revolving Commitments, as the case may be, in respect of which applicable Term Lenders or Revolving Lenders, as the case may be, shall have accepted the relevant Extension Offer shall exceed the maximum aggregate principal amount of applicable Term Loans, Revolving Commitments, Incremental Revolving Commitments or Other Refinancing Revolving Commitments, as the case may be, offered to be extended by the Borrower pursuant to such Extension Offer, then the applicable Term Loans, Revolving Loans, Incremental Revolving Loans or Other Refinancing Loans, as the case may be, of the applicable Term Lenders or Revolving Lenders, as the case may be, shall be extended ratably up to such maximum amount based on the respective principal amounts (but not to exceed actual holdings of record) with respect to which such Term Lenders or Revolving Lenders, as the case may be, have accepted such Extension Offer; (ix) all documentation in respect of such Extension shall be consistent with the foregoing, (x) the Extension shall not become effective unless, on the proposed effective date of the Extension, (x) the Borrower shall deliver to the Administrative Agent one or more legal opinions reasonably satisfactory to the Administrative Agent and a certificate of an authorized officer of each Loan Party dated the applicable date of the Extension and executed by an authorized officer of such Loan Party certifying and attaching the resolutions adopted by such Loan Party approving or consenting to such Extension and (y) the conditions set forth in Section 4.02 shall be satisfied (with all references in such Section 4.02 to any Credit Event being deemed to be references to the Extension on the applicable date of the Extension) and the Administrative Agent shall have received a certificate to that effect dated the applicable date of the Extension and executed by a Financial Officer of Parent; (xi) any applicable Minimum Extension Condition shall be satisfied unless waived by the Borrower; and (xii) the Minimum Tranche Amount shall be satisfied unless waived by the Administrative Agent. (b) With respect to all Extensions consummated by the Borrower pursuant to this Section 2.23, (i) such Extensions shall not constitute voluntary or mandatory payments or prepayments for purposes of Section 2.11 and (ii) no Extension Offer is required to be in any minimum amount or any minimum increment; provided that (A) the Borrower may at its election specify as a condition (a “Minimum Extension Condition”) to consummating any such Extension that a minimum amount (to be determined and specified in the relevant Extension Offer in Borrower’s sole discretion and may be waived by Borrower) of Term Loans, Other Refinancing

131 Term Loans or Revolving Commitments, Incremental Revolving Commitments or Other Refinancing Revolving Commitments (as applicable) of any or all applicable Tranches and Classes be tendered and (B) no Tranche of Extended Loans shall be in an amount (taking the Dollar Amount of any amounts denominated in Agreed Currencies other than Dollars) of less than $100,000,000 (the “Minimum Tranche Amount”), unless such Minimum Tranche Amount is waived by the Administrative Agent. Subject to compliance with the terms of this Section 2.23, the Administrative Agent, the Issuing Bank and the Lenders hereby consent to the Extensions and the other transactions contemplated by this Section 2.23 (including, for the avoidance of doubt, payment of any interest, fees or premium in respect of any Extended Term Loans and/or Extended Revolving Commitments on such terms as may be set forth in the relevant Extension Offer) and hereby waive the requirements of any provision of this Agreement (including, without limitation, Sections 2.11 and 2.18) or any other Loan Document that may otherwise prohibit any such Extension or any other transaction contemplated by this Section 2.23. (c) No consent of any Lender, the Issuing Bank or the Administrative Agent shall be required to effectuate any Extension, other than the consent of each Lender agreeing to such Extension with respect to one or more of its Term Loans of any Class, Other Refinancing Term Loans, Revolving Commitments of any Class, Incremental Revolving Commitments and/or Other Refinancing Revolving Commitments (or a portion thereof); provided that the consent of the Issuing Bank shall be required to effect an Extension of Revolving Commitments. All Extended Term Loans, Extended Revolving Commitments and all obligations in respect thereof shall be Secured Obligations under this Agreement and the other Loan Documents that are secured by all or a portion of the Collateral on a pari passu or junior lien basis with all other applicable Obligations under this Agreement and the other Loan Documents; provided that, if such Extended Term Loans or Extended Revolving Commitments rank junior in right of security with any other Loans or Commitments hereunder, such Extended Term Loans or Extended Revolving Commitments will be subject to the terms of an Approved Intercreditor Agreement. The Lenders hereby irrevocably authorize the Administrative Agent to enter into amendments to this Agreement and the other Loan Documents with the Borrower as may be necessary in order to establish new Tranches or sub-tranches in respect of Revolving Commitments or Term Loans so extended and such technical amendments as may be necessary or appropriate in the reasonable opinion of the Administrative Agent and the Borrower in connection with the establishment of such new Tranches or subtranches, in each case on terms consistent with this Section 2.23. Without limiting the foregoing, in connection with any Extensions the respective Loan Parties shall (at their expense) amend (and the Administrative Agent is hereby directed to amend) any Mortgage that has a maturity date prior to the then Latest Maturity Date so that such maturity date is extended to the then Latest Maturity Date (or such later date as may be advised by local counsel to the Administrative Agent). (d) In connection with any Extension, the Borrower shall provide the Administrative Agent at least ten (10) days (or such shorter period as may be agreed by the Administrative Agent) prior written notice thereof, and shall agree to such procedures, if any, as may be established by, or acceptable to, the Administrative Agent, in each case acting reasonably to accomplish the purposes of this Section 2.23. (e) Notwithstanding anything to the contrary contained herein, no Lender shall be required to accept an Extension Offer.

132 Section 2.24 Loan Repurchases. (a) (1) So long as no Event of Default has occurred and is continuing, the applicable Borrower may purchase its outstanding Term Loans on a non-pro rata basis through open market purchases consisting solely of cash (subject to 9.04(k)) and (2) subject to the terms and conditions set forth or referred to below, the applicable Borrower may from time to time, at its discretion, conduct modified Dutch auctions in order to purchase its Term Loans of one or more Classes (as determined by the applicable Borrower) (each, a “Purchase Offer”), each such Purchase Offer to be managed exclusively by the Administrative Agent (or such other financial institution chosen by Parent and reasonably acceptable to the Administrative Agent) (in such capacity, the “Auction Manager”), so long as the following conditions are satisfied: (i) each Purchase Offer shall be conducted in accordance with the procedures, terms and conditions set forth in this Section 2.24 and the Auction Procedures; (ii) no Event of Default shall have occurred and be continuing on the date of the delivery of each notice of an auction and at the time of (and immediately after giving effect to) the purchase of any Term Loans in connection with any Purchase Offer; (iii) the principal amount (calculated on the face amount thereof) of each and all Classes of Term Loans that the applicable Borrower offers to purchase in any such Purchase Offer shall be no less than U.S. $25,000,000 (unless another amount is agreed to by the Administrative Agent) (across all such Classes); (iv) the aggregate principal amount (calculated on the face amount thereof) of all Term Loans of the applicable Class or Classes so purchased by the Borrower shall automatically be cancelled and retired by the Borrower on the settlement date of the relevant purchase (and may not be resold), and in no event shall the Borrower be entitled to any vote hereunder in connection with such Term Loans; (v) no more than one Purchase Offer with respect to any Class may be ongoing at any one time; (vi) any Purchase Offer with respect to any Class shall be offered to all Term Lenders holding Term Loans of such Class on a pro rata basis; and (vii) no purchase of any Term Loans shall be made from the proceeds of any Revolving Loan or Swingline Loan. (b) The applicable Borrower must terminate any Purchase Offer if it fails to satisfy one or more of the conditions set forth above which are required to be met at the time which otherwise would have been the time of purchase of Term Loans pursuant to such Purchase Offer. If the applicable Borrower commences any Purchase Offer (and all relevant requirements set forth above which are required to be satisfied at the time of the commencement of such Purchase Offer have in fact been satisfied), and if at such time of commencement the applicable Borrower reasonably believes that all required conditions set forth above which are required to be satisfied at the time of the consummation of such Purchase Offer shall be satisfied, then the applicable Borrower shall have no liability to any Term Lender for any termination of

133 such Purchase Offer as a result of its failure to satisfy one or more of the conditions set forth above which are required to be met at the time which otherwise would have been the time of consummation of such Purchase Offer, and any such failure shall not result in any Event of Default hereunder. With respect to all purchases of Term Loans of any Class or Classes made by the applicable Borrower pursuant to this Section 2.24, (x) the applicable Borrower shall pay on the settlement date of each such purchase all accrued and unpaid interest (except to the extent otherwise set forth in the relevant offering documents), if any, on the purchased Term Loans of the applicable Class or Classes up to the settlement date of such purchase and (y) such purchases (and the payments made by the applicable Borrower and the cancellation of the purchased Loans, in each case in connection therewith) shall not constitute voluntary or mandatory payments or prepayments for purposes of Section 2.11 hereof. (c) The Administrative Agent and the Lenders hereby consent to the Purchase Offers and the other transactions effected pursuant to and in accordance with the terms of this Section 2.24; provided that, notwithstanding anything to the contrary contained herein, no Lender shall have an obligation to participate in any such Purchase Offer. For the avoidance of doubt, it is understood and agreed that the provisions of Sections 2.16, 2.18 and 9.04 will not apply to the purchases of Term Loans made pursuant to and in accordance with the provisions of this Section 2.24. The Auction Manager acting in its capacity as such hereunder shall be entitled to the benefits of the provisions of Article VIII and Section 9.03 to the same extent as if each reference therein to the “Administrative Agent” were a reference to the Auction Manager, and the Administrative Agent shall cooperate with the Auction Manager as reasonably requested by the Auction Manager in order to enable it to perform its responsibilities and duties in connection with each Purchase Offer. (d) This Section 2.24 shall supersede any provisions in Section 2.18 or 9.02 to the contrary. Section 2.25 Refinancing Amendment. At any time after the Effective Date, the Borrower may obtain, from any Lender or any Refinancing Lender, Credit Agreement Refinancing Indebtedness in respect of all or any portion of any Class of Loans or Commitments of such Borrower then outstanding under this Agreement (which for purposes of this Section 2.25 will be deemed to include any then outstanding Other Refinancing Loans, Other Refinancing Commitments, Incremental Loans, Incremental Commitments, Extended Loans or Extended Commitments), in the form of Other Refinancing Loans or Other Refinancing Commitments in each case pursuant to a Refinancing Amendment; provided that such Credit Agreement Refinancing Indebtedness (i) will rank pari passu or junior in right of payment and of security with the other Loans and Commitments hereunder; provided that, if such Credit Agreement Refinancing Indebtedness ranks junior in right of security with any other Loans or Commitments hereunder, such Credit Agreement Refinancing Indebtedness will be subject to the terms of an Approved Intercreditor Agreement, (ii) will have such pricing, premiums and optional prepayment or redemption terms as may be agreed by the applicable Borrower and the Lenders thereof; (iii) will have a maturity date no earlier than, and will have a Weighted Average Life to Maturity equal to or greater than, the Loans or Commitments being refinanced (other than such Credit Agreement Refinancing Indebtedness incurred under the Inside Maturity Basket) and (iv) will have terms and conditions that are substantially identical to, or (taken as a whole) are no more favorable to the lenders or holders providing such Credit Agreement Refinancing Indebtedness than those

134 applicable to the Loans or Commitments being refinanced; provided, further, that the terms and conditions applicable to such Credit Agreement Refinancing Indebtedness may provide for any additional or different financial or other covenants or other provisions that are agreed between the applicable Borrower and the Lenders thereof and applicable only during periods after the Latest Maturity Date that is in effect on the date such Credit Agreement Refinancing Indebtedness is issued, incurred or obtained. Any Other Refinancing Loans or Other Refinancing Commitments, as applicable, may participate on a pro rata basis or on a less than pro rata basis (but not on a greater than pro rata basis) in any voluntary or mandatory prepayments hereunder, as specified in the applicable Refinancing Amendment. The effectiveness of any Refinancing Amendment shall be subject to the satisfaction on the date thereof of each of the conditions set forth in Section 4.02 and, to the extent reasonably requested by the Administrative Agent, receipt by the Administrative Agent of legal opinions, board resolutions, officers’ certificates and/or reaffirmation agreements generally consistent with those delivered on the Effective Date pursuant to Sections 4.01(b) and (f) (other than changes to such legal opinions resulting from a change in law, change in fact or change to counsel’s form of opinion reasonably satisfactory to the Administrative Agent). Each Credit Agreement Refinancing Indebtedness incurred under this Section 2.25 shall be in an aggregate principal amount that is not less than $100,000,000. The Administrative Agent shall promptly notify each Lender as to the effectiveness of each Refinancing Amendment. Each of the parties hereto hereby agrees that, upon the effectiveness of any Refinancing Amendment, this Agreement shall be deemed amended to the extent (but only to the extent) necessary to reflect the existence and terms of the Credit Agreement Refinancing Indebtedness incurred pursuant thereto (including any amendments necessary to treat the Loans and Commitments subject thereto as Other Refinancing Loans and/or Other Refinancing Commitments). Any Refinancing Amendment may, without the consent of any other Lenders, effect such amendments to this Agreement and the other Loan Documents as may be necessary or appropriate, in the reasonable opinion of the Administrative Agent and the Borrower, to effect the provisions of this Section 2.25. This Section 2.25 shall supersede any provisions in Section 2.18 or 9.02 to the contrary. Section 2.26 Illegality. If any Lender determines that any law has made it unlawful, or that any Governmental Authority has asserted that it is unlawful, for any Lender or its applicable lending office to make, maintain or fund Loans whose interest is determined by reference to Term SOFR or Term CORRA, or to determine or charge interest rates based upon Term SOFR or Term CORRA, or any Governmental Authority has imposed material restrictions on the authority of such Lender to purchase or sell, or to take deposits of, any Agreed Currency in the applicable offshore interbank market for the applicable currency then, on notice thereof by such Lender to the Borrower through the Administrative Agent, (i) any obligation of such Lender to make or continue Term Benchmark Loans in the affected Agreed Currency or Agreed Currencies or to convert ABR Loans to Term SOFR Loans or Term CORRA Loans to Canadian Prime Rate Loans (as applicable) shall be suspended, (ii) if such notice asserts the illegality of such Lender making or maintaining ABR Loans the interest rate of which is determined by reference to the clause (c) of the definition of “Alternate Base Rate”, the interest rate for ABR Loans of such Lender shall, if necessary to avoid such illegality, be determined by the Administrative Agent without reference to clause (c) of such definition and (iii) if such notice asserts the illegality of such Lender making or maintaining Canadian Prime Rate Loans the interest rate of which is determined by reference to clause (ii) of the definition of “Canadian Prime Rate”, the interest rate for Canadian Prime Rate Loans of such Lender shall, if necessary to avoid such illegality, be determined by the Administrative Agent without reference to clause (ii) of such definition, in each case until such

135 Lender notifies the Administrative Agent and the Borrower that the circumstances giving rise to such determination no longer exist. Upon receipt of such notice, (x) the Borrower shall, upon demand from such Lender (with a copy to the Administrative Agent), prepay or, if applicable, convert (A) all Term SOFR Loans of such Lender to ABR Loans (the interest rate on which ABR Loans of such Lender shall, if necessary to avoid such illegality, be determined by the Administrative Agent without reference to the clause (c) of the definition of “Alternate Base Rate”), either on the last day of the Interest Period therefor, if such Lender may lawfully continue to maintain such Term SOFR Loans to such day, or immediately, if such Lender may not lawfully continue to maintain such Term SOFR Loan and/or (B) all Term CORRA Loans of such Lender to Canadian Prime Rate Loans (the interest rate on which Canadian Prime Rate Loans of such Lender shall, if necessary to avoid such illegality, be determined by the Administrative Agent without reference to clause (ii) of the definition of “Canadian Prime Rate”), on the maturity date therefor, if such Lender may lawfully continue to maintain such Term CORRA Loans to such day, or immediately, if such Lender may not lawfully continue to maintain such Term CORRA Loans and (y) if such notice asserts the illegality of such Lender determining or charging interest rates based upon Term SOFR, the Administrative Agent shall during the period of such suspension compute the Alternate Base Rate applicable to such Lender without reference to the Term SOFR component thereof until the Administrative Agent is advised in writing by such Lender that it is no longer illegal for such Lender to determine or charge interest rates based upon Term SOFR. Upon any such prepayment or conversion, the applicable Borrower shall also pay accrued interest on the amount so prepaid or converted. ARTICLE III REPRESENTATIONS AND WARRANTIES In order to induce the Administrative Agent and the Lenders to (A) enter into this Agreement on the Effective Date and (B) make each Loan or other extension of credit to be made hereunder on each applicable Credit Event, each of Parent and the Borrower represents and warrants to the Administrative Agent and Lenders that, on the Effective Date and on the date of each other Credit Event, that each of the following statements are true and correct in all material respects: Section 3.01 Organization; Powers; Subsidiaries. Each of Parent and its Material Subsidiaries is duly organized, incorporated (in the case of each Material Subsidiary incorporated under the laws of Ireland) and validly existing and (to the extent the concept is applicable in such jurisdiction) in good standing under the laws of the jurisdiction of its organization, has all requisite power and authority to carry on its business as now conducted and, except where the failure to do so, individually or in the aggregate, could not reasonably be expected to result in a Material Adverse Effect, is qualified to do business in, and (to the extent the concept is applicable in such jurisdiction) is in good standing in, every jurisdiction where such qualification is required. Schedule 3.01 hereto identifies each Subsidiary (other than Subsidiaries in respect of which Parent and its Subsidiaries own less than 50% of the Equity Interests thereof) as of the Effective Date, noting whether such Subsidiary is a Material Subsidiary, whether such Subsidiary is a Guarantor, whether such Subsidiary is an Unrestricted Subsidiary, the jurisdiction of its incorporation or organization, as the case may be, the percentage of issued and outstanding shares of each class of its capital stock or other equity interests owned by Parent and the Subsidiaries and, if such

136 percentage is not 100% (excluding directors’ qualifying shares as required by law), a description of each class issued and outstanding. All of the outstanding shares of capital stock and other equity interests of each Material Subsidiary are validly issued and outstanding and fully paid and non- assessable and all such shares and other equity interests owned by Parent or any Material Subsidiary are owned, beneficially and of record, by Parent or such Material Subsidiary free and clear of all Liens, other than Liens created under the Loan Documents and Liens permitted by Section 6.02. As of the Effective Date, there are no outstanding commitments or other obligations of any Material Subsidiary to issue, and no options, warrants or other rights of any Person to acquire, any shares of any class of capital stock or other equity interests of any Material Subsidiary, except as expressly disclosed in the Reorganization Plan Documents. Section 3.02 Authorization; Enforceability. The Transactions are within each Loan Party’s corporate or other organizational powers and have been duly authorized by all necessary corporate or other organizational actions and, if required, actions by shareholders, members or equity holders. The Loan Documents to which each Loan Party is a party have been duly executed and delivered by such Loan Party and constitute a legal, valid and binding obligation of such Loan Party, enforceable in accordance with its terms, subject to applicable bankruptcy, insolvency, reorganization, moratorium or other laws affecting creditors’ rights generally and subject to general principles of equity, regardless of whether considered in a proceeding in equity or at law. Section 3.03 Governmental Approvals; No Conflicts. The Transactions (a) do not require any consent or approval of, registration or filing with, or any other action by, any Governmental Authority, except as expressly contemplated by the Reorganization Plan Documents, and such as have been obtained or made and are in full force and effect and except for filings or registrations necessary to perfect Liens created pursuant to the Loan Documents, (b) will not violate any applicable law or regulation (except such non-compliance that, individually or in the aggregate, could not reasonably be expected to result in a Material Adverse Effect) or the charter, by-laws or other constitutional or organizational documents of Parent or any of its Material Subsidiaries or any order of any Governmental Authority, (c) will not violate in any material respect or result in a default under any indenture, material agreement or other material instrument binding upon Parent or any of its Material Subsidiaries or its assets, or give rise to a right thereunder to require any payment to be made by Parent or any of its Material Subsidiaries, and (d) will not result in the creation or imposition of any Lien on any asset of Parent or any of its Material Subsidiaries, other than Liens created under the Loan Documents and under the Senior Secured Notes Indenture. Section 3.04 Financial Condition; No Material Adverse Change. (a) PLC has heretofore furnished to the Lenders the consolidated balance sheet and statements of income, stockholders equity and cash flows as of and for the fiscal year ended December 31, 2023, reported on by PricewaterhouseCoopers LLP. Such financial statements present fairly, in all material respects, the financial position and results of operations and cash flows of PLC and its consolidated Subsidiaries as of such dates and for such periods in accordance with GAAP. (b) Since the Effective Date (as defined in the Reorganization Plan), there has been no material adverse change in the business, assets, operations or condition, financial or otherwise, of Parent and its Subsidiaries, taken as a whole.

137 Section 3.05 Properties. (a) Each of Parent and its Material Subsidiaries has good title to, or (to the knowledge of Parent and the Borrower) valid leasehold interests in, all its real and personal property (excluding Intellectual Property, which is considered in Section 3.05(b)) material to its business, except for minor defects in title that do not interfere with its ability to conduct its business as currently conducted or to utilize such properties for their intended purposes. (b) Each of Parent and its Restricted Subsidiaries owns, or is licensed (or otherwise has the rights) to use, all trademarks, tradenames, copyrights, patents and other Intellectual Property used in or necessary to its business, and the use thereof by Parent and its Restricted Subsidiaries does not infringe upon the rights of any other Person, except for any such infringements (or ownership or license issues) that, individually or in the aggregate, could not reasonably be expected to result in a Material Adverse Effect. Section 3.06 Litigation, Environmental and Labor Matters. (a) Except as set forth in Schedule 3.06 hereto, the Reorganization Plan Documents and in PLC’s Annual Report on Form 10-K for the year ended December 31, 2023, there are no actions, suits, proceedings or investigations by or before any arbitrator or Governmental Authority pending against or, to the knowledge of Parent, threatened against or affecting Parent or any of its Restricted Subsidiaries that could reasonably be expected, individually or in the aggregate, to result in a Material Adverse Effect. (b) Except with respect to matters that, individually or in the aggregate, could not reasonably be expected to result in a Material Adverse Effect, neither Parent nor any of its Restricted Subsidiaries (i) has failed to comply with any Environmental Law or to obtain, maintain or comply with any permit, license or other approval required under any Environmental Law, (ii) is subject to any Environmental Liability or (iii) has received notice of any claim with respect to any Environmental Liability. (c) There are no strikes, lockouts or slowdowns against Parent or any of its Restricted Subsidiaries pending or, to their knowledge, threatened that have resulted in, or could reasonably be expected to result in, a Material Adverse Effect. The hours worked by and payments made to employees of Parent and its Restricted Subsidiaries have not been in violation of the Fair Labor Standards Act or any other applicable Federal, state, local or foreign law relating to such matters that has resulted in, or could reasonably be expected to result in, a Material Adverse Effect. All material payments due from Parent or any of its Restricted Subsidiaries, or for which any claim may be made against Parent or any of its Restricted Subsidiaries, on account of wages and employee health and welfare insurance and other benefits, have been paid or accrued as liabilities on the books of Parent or such Restricted Subsidiary except to the extent that the failure to do so has not resulted in, and could not reasonably be expected to result in, a Material Adverse Effect. The consummation of the Transactions will not give rise to any right of termination or right of renegotiation on the part of any union under any collective bargaining agreement under which Parent or any of its Material Subsidiaries is bound. Section 3.07 Compliance with Laws and Agreements. Except as set forth in Schedule 3.07 hereto, each of Parent and its Restricted Subsidiaries is in compliance with all laws, regulations and orders of any Governmental Authority applicable to it or its property and all indentures, agreements and other instruments binding upon it or its property, except where the

138 failure to do so, individually or in the aggregate, could not reasonably be expected to result in a Material Adverse Effect. Section 3.08 Investment Company Status. Neither Parent nor any of its Restricted Subsidiaries is an “investment company” as defined in, or subject to regulation under, the Investment Company Act of 1940. Section 3.09 Taxes. Each of Parent and its Restricted Subsidiaries has filed or caused to be filed all federal Tax returns and all other material Tax returns and reports required to have been filed and has paid or caused to be paid all Taxes required to have been paid by it, except (a) Taxes that are being contested in good faith by appropriate proceedings and for which Parent or such Restricted Subsidiary, as applicable, has set aside on its books adequate reserves or (b) to the extent that the failure to do so could not reasonably be expected to result in a Material Adverse Effect. Section 3.10 Benefit Plans. (a) No ERISA Event has occurred or is reasonably expected to occur that, in each case, when taken together with all other such ERISA Events for which liability is reasonably expected to occur, could reasonably be expected to result in a Material Adverse Effect. (b) Each Non-U.S. Plan has been maintained in compliance with its terms and with the requirements of any and all applicable laws, statutes, rules, regulations and orders and has been maintained, where required, in good standing with applicable regulatory authorities, except as would not reasonably be expected to result in a Material Adverse Effect. All contributions required to be made by Parent or any Restricted Subsidiary with respect to a Non- U.S. Plan have been timely made, except as would not reasonably be expected to result in a Material Adverse Effect. Neither Parent nor any of its Restricted Subsidiaries has incurred any obligation in connection with the termination of, or withdrawal from, any Non-U.S. Plan, except as would not reasonably be expected to result in a Material Adverse Effect. Section 3.11 Disclosure. As of the Effective Date, all written or formally presented information, including any Information Memorandum, other than any projections and information of a general economic or general industry nature, furnished by or on behalf of, Parent or any Subsidiary to the Administrative Agent, any of its Affiliates or any Lender pursuant to or in connection with this Agreement or any other Loan Document, taken as a whole together with all other written information so delivered on or prior to the Effective Date, together with all information contained in regular or periodic reports filed by or on behalf of Parent or the Borrower with the SEC, the United States Bankruptcy Court for the Southern District of New York or, in each case, any similar Governmental Authority on or prior to such date is complete and correct in all material respects and does not contain any untrue statement of material fact or omit to state a material fact necessary in order to make the statements contained therein not materially misleading in light of the circumstances under which such statements are made; provided that, with respect to forecasts or projected financial information, Parent and the Borrower represents only that such information was prepared in good faith based upon assumptions believed by it to be reasonable at the time so furnished (it being understood by the Administrative Agent and the Lenders that any such projections are subject to significant uncertainties and contingencies, many of which are

139 beyond the control of Parent or its Subsidiaries, that no assurances can be given that such projections will be realized and that actual results may differ materially from such projections). Section 3.12 Federal Reserve Regulations. No part of the proceeds of any Loan have been used or will be used, whether directly or indirectly, for any purpose that entails a violation of any of the Regulations of the Board, including Regulations T, U and X. Section 3.13 Security Interest in Collateral. To the extent the US Security Agreement has been executed and delivered by the parties thereto and are then in effect, such US Security Agreement will create in favor of the Collateral Agent, for the benefit of the Secured Parties, a valid and enforceable security interest in the Collateral covered thereby and (i) when the Collateral constituting certificated securities (as defined in the UCC) is delivered to the Collateral Agent, together with instruments of transfer duly endorsed in blank, the Liens under such US Security Agreement will constitute a fully perfected security interest in all right, title and interest of the pledgors thereunder in such Collateral, prior and superior in right to any other Person, and (ii) when financing statements (or short-form intellectual property security agreements) in appropriate form are filed in the applicable filing offices (including the United States Patent and Trademark Office and the United States Copyright Office, as applicable), the security interest created under such US Security Agreement will constitute a fully perfected security interest in all right, title and interest of the Loan Parties in the remaining Collateral to the extent perfection can be obtained by filing UCC financing statements (or short-form intellectual property security agreements), prior and superior to the rights of any other Person, except, in each case, for (x) Liens permitted by Section 6.02 and Liens securing the obligations under the Senior Secured Notes and (y) any requirement under Luxembourg law, including the foreign lex rei sitae, referred to under Luxembourg international private law, with respect to any Collateral which (1) under Luxembourg law, would be located or deemed located in Luxembourg or (2) would be granted by a Loan Party formed under the laws of the Grand Duchy of Luxembourg. As to any Collateral, the representations and the warranties with respect thereto contained in the relevant Collateral Documents shall be true and correct. Section 3.14 Solvency. As of the Effective Date, (a) the fair value of the assets of Parent and its Subsidiaries on a consolidated basis will exceed their consolidated debts and liabilities, subordinated, contingent or otherwise; (b) the present fair saleable value of the property of Parent and its Subsidiaries on a consolidated basis will be greater than the amount that will be required to pay the probable liability on their debts and other liabilities, subordinated, contingent or otherwise, as such debts and other liabilities become absolute and matured; (c) Parent and its Subsidiaries on a consolidated basis will not have incurred any debts and liabilities, subordinated, contingent or otherwise, that they do not believe that they will be able to pay as such debts and liabilities become absolute and matured; and (d) Parent and its Subsidiaries on a consolidated basis will not have unreasonably small capital with which to conduct the business in which they are engaged as such business is now conducted and is proposed to be conducted following the Effective Date. Section 3.15 Sanctions; Anti-Corruption. (a) Neither Parent nor any of its Restricted Subsidiaries or, to the knowledge of Parent, any of its Affiliates over which any of the foregoing exercises management control (each, a “Controlled Affiliate”) is a Sanctioned Person, and Parent, its Restricted Subsidiaries and, to the knowledge of Parent, such Controlled Affiliates are in compliance in all material respects with all applicable orders, rules and regulations of OFAC.

140 (b) Neither Parent nor any of its Restricted Subsidiaries or, to the knowledge of the Parent, any of its Controlled Affiliates, is a Sanctioned Person. (c) Parent and its Restricted Subsidiaries and, to the knowledge of Parent and its Restricted Subsidiaries and with respect to the business of Parent and its Restricted Subsidiaries, their respective officers, directors and employees are in compliance with Anti- Corruption Laws in all material respects. Section 3.16 Beneficial Ownership Certificate. The information included in the Beneficial Ownership Certificate last delivered with respect to the Borrower (solely to the extent such Borrower qualifies as a “legal entity customer” under the Beneficial Ownership Regulation) is true and correct in all material respects. Section 3.17 Affected Financial Institutions. No Loan Party is an Affected Financial Institution. Section 3.18 Luxembourg Regulatory Matters. Each Luxembourg Guarantor is in compliance with all requirements of the Luxembourg legislation and regulations on the domiciliation of companies, and, to the extent applicable, in particular with the Luxembourg Act dated May 31, 1999 on the domiciliation of companies, as amended from time to time, except where failure to comply with any such requirement could not reasonably be expected to result in a Material Adverse Effect. No Luxembourg Guarantor has filed a request with any competent court seeking that such Luxembourg Guarantor be declared subject to bankruptcy (faillite), insolvency, voluntary or judicial liquidation (liquidation volontaire ou judiciaire), or administrative dissolution without liquidation proceedings (procédure de dissolution administrative sans liquidation), judicial reorganization proceeding (procedure de reorganisation judiciaire), reprieve from payment (sursis de paiement) out-of-court mutual agreement (réorganisation extra-judiciaire par accord amiable), judicial reorganisation in the form of a stay to enter into a mutual agreement (sursis en vue de la conclusion d'un accord amiable), judicial reorganisation by collective agreement (réorganisation judiciaire par accord collectif), judicial reorganisation by transfer of assets or activities (réorganisation judiciaire par transfert sous autorité de justice), conciliation (conciliation), general settlement with creditors, reorganization or similar laws affecting the rights of creditors generally and no application has been made or is to be made by its manager or, as far as it is aware, by any other person for the appointment of a commissaire, juge-commissaire, liquidateur, curateur or similar officer pursuant to any voluntary or judicial insolvency, winding-up, liquidation or similar proceedings or analogous procedures according to Regulation (EU) 2015/848 of the European Parliament and of the Council of 20 May 2015 on insolvency proceedings (recast) (the “Insolvency Regulation”). The head office (administration centrale), the place of effective management (siège de direction effective) and (for the purposes of the Insolvency Regulation) the center of main interests (centre des intérêts principaux) of each of the Luxembourg Guarantors in Luxembourg is located at the place of its registered office (siège statutaire) in Luxembourg.

141 ARTICLE IV CONDITIONS Section 4.01 Effective Date. The obligations of the Lenders to extend Loans in respect of the Commitments on the date of the first Credit Event hereunder are subject to the satisfaction (or waiver) of the following conditions precedent: (a) Execution. The Administrative Agent shall have received (i) this Agreement, executed and delivered by Parent, the Borrower Representative and each of the Lenders, (ii) the Subsidiary Guaranty, executed and delivered by each Subsidiary Guarantor, (iii) the US Security Agreement, executed and delivered by each applicable Loan Party, (iv) the Irish Debenture, executed and delivered by each Irish Loan Party, (v) the Irish Share Pledge executed by Endo US Holdings Luxembourg I S.a.r.l. and (vi) the Luxembourg Pledge Agreement executed by Endo Enterprise, Inc. (b) Organizational Documents and Necessary Consents. The Administrative Agent shall have received (i) a copy of the certificate or articles of incorporation or other formation or constitutional documents, including all amendments thereto, of each Loan Party as of the Effective Date, certified (to the extent available and customary in any non-U.S. jurisdiction) as of a recent date by the Secretary of State of the state of its organization (or similar Governmental Authority in any foreign jurisdiction with respect to any such Loan Party organized outside the United States of America) or, in the case of any Luxembourg Loan Party, by an authorized signatory of such Luxembourg Loan Party, and (to the extent available and customary in a non-U.S. jurisdiction) a certificate as to the good standing of each such Loan Party as of a recent date, from such Secretary of State (or similar Governmental Authority in any foreign jurisdiction (to the extent available in that foreign jurisdiction) with respect to any Loan Party organized outside the United States of America (including, in the case of any Irish Loan Party, a letter of status from the Irish Companies Registration Office) and in the case of any Luxembourg Loan Party, an electronic copy of an extract (extrait) issued by the Luxembourg Register of Commerce and Companies (R.C.S. Luxembourg) not earlier than one Business Day before the Effective Date with respect to such Luxembourg Loan Party and an electronic copy of a negative certificate (certificat de non-inscription d'une décision judiciarie) issued by the Luxembourg Register of Commerce and Companies (R.C.S. Luxembourg) as of the Effective Date with respect to such Luxembourg Loan Party, together the “Lux Extracts”); (ii) a certificate of the secretary or assistant secretary (to the extent customary in a non-U.S. jurisdiction) of each Loan Party as of the Effective Date (or, of a manager or director, if applicable and customary, in the case of any Foreign Loan Party) dated the Effective Date and certifying (A) that attached thereto is a true and complete copy of the by-laws or constitutional documents (or similar governing documentation) of such Loan Party as in effect on the Effective Date and at all times since the date of the resolutions described in clause (B) below, (B) that attached thereto is a true and complete copy of resolutions duly adopted by the board of directors or similar governing body of such Loan Party authorizing the execution, delivery and performance of the Loan Documents to which such Person is a party, (in the case of the Borrower) the borrowings hereunder, (in the case of each such Loan Party) the granting of the Liens contemplated to be granted by it under the Collateral Documents and (in the case of each Guarantor) the Guaranteeing of the Secured Obligations as contemplated by this Agreement or the Subsidiary Guaranty, as applicable, and that such resolutions have not been

142 modified, rescinded or amended and are in full force and effect, (C) in relation to a Luxembourg Loan Party, that the attached hereto are true and complete copies of the Lux Extracts, (D) if applicable, that the certificate or articles of incorporation or other formation or constitutional documents of such Loan Party have not been amended since the date of the last amendment thereto shown on the certificate of good standing furnished pursuant to clause (i) above or where a certificate of good standing is not applicable in its jurisdiction of incorporation that attach a true, up to date and correct copy of the certificate or articles of incorporation or other formation or constitutional documents of each Loan Party duly certified as being true, up to date and correct and (E) unless delivery is not customary in the jurisdiction of any Foreign Loan Party, as to the incumbency and specimen signature of each officer executing any Loan Document or any other document delivered in connection herewith on behalf of such Loan Party; and (iii) a certificate of another officer as to the incumbency and specimen signature of the secretary or assistant secretary (or manager or director, if applicable) executing the certificate pursuant to (ii) above. (c) USA Patriot Act. To the extent requested by the Lenders at least ten Business Days prior to the Effective Date, the Borrower Representative shall have delivered a Beneficial Ownership Certificate and each Person which shall become a Loan Party on the Effective Date shall have provided the documentation and other information to the Lenders that are required by regulatory authorities under the applicable “know-your-customer” rules and regulations and anti-money laundering rules and regulations, including the USA Patriot Act. (d) Guarantees; Collateral. (i) The Guaranty with respect to Parent and each Subsidiary Guarantor (and any confirmation thereof) shall have been executed and be in full force and effect, and (ii) all documents and instruments required to perfect the Collateral Agent’s security interest in (A) all of the issued and outstanding Equity Interests of each Subsidiary Guarantor constituting Collateral and (B) subject to the Agreed Security Principles (in the case of any Foreign Subsidiary), substantially all of the assets of each Subsidiary Guarantor (in each case, to the extent included in the Collateral) shall have been executed and delivered and, if applicable, be in proper form for filing (excluding, in any event, any obligations identified on Schedule 5.12 and Mortgages). (e) Opinions of Counsel. The Administrative Agent shall have received, on behalf of itself, the Lenders and the Issuing Bank, a written opinion of (i) Skadden, Arps, Slate, Meagher & Flom LLP, New York counsel for the Loan Parties, (ii) A&L Goodbody, Irish counsel for the Loan Parties, (iii) Elvinger Hoss Prussen, Luxembourg counsel for the Loan Parties and (iv) NautaDutilh Avocats Luxembourg S.à r.l, Luxembourg counsel for the Administrative Agent, in each case, in form and substance reasonably acceptable to the Administrative Agent. (f) Solvency Certificate. The Administrative Agent shall have received a certificate of Parent, signed by an authorized signatory of Parent, in substantially the form attached hereto as Exhibit E. (g) Fees. To the extent invoiced at least two Business Days prior to the Effective Date, all costs, fees, expenses (including, without limitation, legal fees and expenses) and other compensation contemplated by the Engagement Letter or as otherwise agreed by the

143 parties thereto, payable to each Lead Arranger, the Administrative Agent and the Lenders, shall have been paid to the extent due. (h) Exit Transactions. The Exit Transactions shall have been consummated, or shall be consummated, substantially concurrently with the initial funding of the Initial Term Loans hereunder. Each Borrowing, and each issuance, amendment or extension of a Letter of Credit, in each case on the Effective Date shall be deemed to constitute a representation and warranty by Parent and the Borrower on such date as to the satisfaction of the matters specified above in this Section 4.01 (except that no representation shall be deemed made as to whether any item is required to be acceptable or satisfactory to the Administrative Agent is acceptable or satisfactory to it). Section 4.02 Each Credit Event. The obligation of each Lender to make a Loan on the occasion of any Borrowing, and of the Issuing Bank to issue, amend or extend any Letter of Credit, is subject to the satisfaction of the following conditions: (a) Subject to Sections 1.04 and 2.20(e) (except with respect to any Borrowing made on the Effective Date), the representations and warranties of Parent and the Borrower set forth in this Agreement shall be true and correct in all material respects (other than to the extent qualified by materiality or “Material Adverse Effect”, in which case, such representations and warranties shall be true and correct) on and as of the date of such Borrowing or the date of issuance, amendment or extension of such Letter of Credit, as applicable, except in the case of any such representation and warranty that expressly relates to an earlier date, in which case such representation and warranty shall be true and correct in all material respects, other than to the extent qualified by materiality or “Material Adverse Effect”, in which case such representation and warranty shall be true and correct on and as of such earlier date. (b) Subject to Sections 1.04 and 2.20(e) (except with respect to any Borrowing made on the Effective Date), at the time of and immediately after giving effect to such Borrowing or the issuance, amendment or extension of such Letter of Credit, as applicable, no Default shall have occurred and be continuing. Each Borrowing and each issuance, amendment or extension of a Letter of Credit shall be deemed to constitute a representation and warranty by Parent and the Borrower on the date thereof as to the matters specified in paragraphs (a) and (b) of this Section 4.02. ARTICLE V AFFIRMATIVE COVENANTS Until the Commitments have expired or been terminated and the principal of and interest on each Loan and all fees payable hereunder shall have been paid in full and all Letters of Credit shall have expired or terminated (in each case, without any pending drawings) or been Cash Collateralized, and all LC Disbursements shall have been reimbursed, each of Parent and the Borrower covenants and agrees with the Lenders that:

144 Section 5.01 Financial Statements and Other Information. Parent will furnish to the Administrative Agent, on behalf of each Lender: (a) within ninety (90) days after the end of each fiscal year of Parent (commencing with the fiscal year ending December 31, 2024), (i) anannual audited consolidated balance sheet and relatedfinancial statements of operations, stockholders’ equity and cash flows for Parent and its consolidated Subsidiaries as of the end of and for such year, setting forth in each case in comparative form the figures for the previous fiscal year (if any), with such audited balance sheet and relateda “Management’s Discussion and Analysis of Financial Condition and Results of Operations” with respect to the periods presented and a report on the annual audited consolidated financial statements reported on by PricewaterhouseCoopers or other independent public accountants of recognized national standing (without a “going concern” or like qualification or exception and without any qualification or exception as to the scope of such audit, except to the extent solely due to the scheduled occurrence of a Maturity Date within one year from the date of such audit or the potential inability to satisfy the Financial Covenant) to the effect that such consolidated financial statements present fairly in all material respects the financial condition and results of operations of Parent and its consolidated Subsidiaries on a consolidated basis in accordanceconsistent with GAAP consistently applied and (ii) to the extent there exist any Unrestricted Subsidiaries, a consolidateding balance sheet and related statements of operations, stockholders’ equity and cash flowsinformation that explains in reasonable detail the differences between such information for Parent and its consolidated Subsidiaries, on the one hand, and Parent and its consolidated Restricted Subsidiaries as of the end of and for such year, setting forth in each case in comparative form the figures for the previous fiscal year (if any)on a standalone basis, on the other hand, certified by one of Parent’s Financial Officers as presenting fairly in all material respects the financial condition and results of operations of Parent and its consolidated Restricted Subsidiaries on a consolidated basis in accordance with GAAP consistently applied; (b) within forty-five (45) days (or, solely with respect to the fiscal quarter ending in September 2025, within sixty (60) days) after the end of each of the first three fiscal quarters of each fiscal year of Parent (commencing with the fiscal quarter ending June 30, 2024), (i) a consolidated balance sheet and related statements of operations, stockholders’ equity and cash flows for Parent and its consolidated Subsidiaries as of the end of and for such fiscal quarter and the then elapsed portion of the fiscal year, setting forth in each case in comparative form the figures for the corresponding period or periods of (or, in the case of the balance sheet, as of the end of) the previous fiscal year (if any) and (ii) to the extent there exist any Unrestricted Subsidiaries, a consolidated balance sheet and related statements of operations, stockholders’ equity and cash flows for Parent and its consolidated Restrictedunaudited quarterly condensed consolidated interim financial statements of Parent and its Subsidiaries as of the end of and for such fiscal quarter and the then elapsed portion of the fiscal year, setting forth in each case in comparative form the figures for the corresponding period or periods of (or, in the case of the balance sheet, as of the end of) the previous fiscal year (if any), in each case all certified by one of itsParent’s Financial Officers as presenting fairly in all material respects the financial condition and results of operations of Parent and its consolidated Subsidiaries (or on a basis consistent with GAAP and (ii) to the extent there exist any Unrestricted Subsidiaries, consolidating information that explains in reasonable detail the differences between such information for Parent and its consolidated Subsidiaries, on the one hand, and Parent and its consolidated Restricted Subsidiaries, as applicable) on a consolidated basis in accordance with GAAP consistently applied, subject to

145 normal year-end audit adjustments and the absence of footnotes; on a standalone basis, on the other hand, certified by one of Parent’s Financial Officers; (c) concurrently with any delivery of financial statements under clause (a) or (b) above, a certificate of a Financial Officer of Parent (i) certifying as to whether a Default has occurred and, if a Default has occurred, specifying the details thereof and any action taken or proposed to be taken with respect thereto, (ii) if the Financial Covenant is required to be tested pursuant to Section 6.11, setting forth reasonably detailed calculations demonstrating compliance with the Financial Covenant (including compliance on a consolidated basis without giving effect to the Unrestricted Subsidiaries) and (iii) stating whether any change in GAAP or in the application thereof has occurred since the date of the audited financial statements referred to in Section 3.04 and, if any such change has occurred, specifying the effect of such change on the financial statements accompanying such certificate; (d) [reserved];Notwithstanding the foregoing, (i) Parent will be deemed to have furnished the reports referred to in Sections 5.01(a) and 5.01(b) to the Administrative Agent if Parent or any direct or indirect parent of Parent has filed or furnished such reports via a filing system and such reports are publicly available and (ii) Parent may satisfy its obligations in Sections 5.01(a) and 5.01(b) with respect to financial information relating to Parent and its consolidated Subsidiaries (or Parent and its consolidated Restricted Subsidiaries, as applicable) by furnishing financial information relating to any direct or indirect parent of Parent; provided that if and for so long as such parent has Independent Assets or Operations, the same is accompanied by consolidating information that explains in reasonable detail the differences between the information relating to such parent, on the one hand, and the information relating to Parent and its consolidated Subsidiaries (or Parent and its consolidated Restricted Subsidiaries, as applicable) on a standalone basis, on the other hand; (e) concurrently with the delivery of the certificate of a Financial Officer of Parent under clause (c) above, an updated version of Exhibit B to the US Security Agreement (provided that if there have been no changes to any such exhibits since the previous updating required thereby, Parent shall indicate that there has been “no change” to the applicable exhibit(s)); (f) [reserved]; (g) as soon as available, but in any event not more than ninety (90) days after the end of each fiscal year of Parent (commencing with the fiscal year ending December 31, 2024), a copy of the plan and forecast (including a projected consolidated balance sheet, income statement and funds flow statement) of Parent for each month of the fiscal year following such fiscal year in form reasonably satisfactory to the Administrative Agent (without giving effect to any Unrestricted Subsidiaries); (h) promptly after the same become publicly available, copies of all periodic and other reports, proxy statements and other materials filed by Parent or any Restricted Subsidiary with the SEC, or any Governmental Authority succeeding to any or all of the functions of the SEC, or with any national securities exchange, or distributed by Parent to its respective shareholders generally, as the case may be; and

146 (i) promptly after any request therefor, such other information regarding the operations, business affairs and financial condition of Parent or any Restricted Subsidiary, or compliance with the terms of any Loan Document, as may be reasonably requested by the Administrative Agent or by any Lender through the Administrative Agent (including any information that any Lender reasonably requests in order to comply with its obligations under the USA Patriot Act and the Beneficial Ownership Regulation). Information required to be delivered pursuant to Sections 5.01(a), 5.01(b) and 5.01(h) shall be deemed to have been delivered if such information, or one or more annual, quarterly or other periodic reports containing such information, shall have been posted by the Administrative Agent on an IntraLinks or similar site to which the Lenders have been granted access or shall be available on the website of the SEC at http://www.sec.gov. Information required to be delivered pursuant to this Section 5.01 may also be delivered by electronic communications pursuant to procedures approved by the Administrative Agent. In the event any financial statements delivered under clause (a) or (b) above shall be restated, Parent shall deliver, promptly after such restated financial statements become available, revised compliance certificates required by clause (c) of this Section 5.01 with respect to the periods covered thereby that give effect to such restatement, signed by a Financial Officer of Parent. Parent and the Borrower hereby acknowledges that (a) the Administrative Agent and/or the Lead Arrangers will make available to the Lenders materials and/or information provided by or on behalf of Parent and the Borrower hereunder (collectively, “Borrower Materials”) by posting the Borrower Materials on IntraLinks or another similar electronic system (the “Platform”) and (b) certain of the Lenders (each, a “Public Lender”) may have personnel who do not wish to receive material non-public information with respect to Parent, the Borrower or their respective Subsidiaries, or the respective securities of any of the foregoing, and who may be engaged in investment and other market-related activities with respect to such Persons’ securities. Parent and the Borrower hereby agrees that (w) all Borrower Materials that are to be made available to Public Lenders shall be clearly and conspicuously marked “PUBLIC” which, at a minimum, shall mean that the word “PUBLIC” shall appear prominently on the first page thereof; (x) by marking Borrower Materials “PUBLIC,” Parent and the Borrower shall be deemed to have authorized the Administrative Agent, the Lead Arrangers and the Lenders to treat such Borrower Materials as not containing any material non-public information with respect to Parent, Parent, the Borrower or their respective securities for purposes of United States Federal and state securities laws (provided, however, that to the extent such Borrower Materials constitute Information, they shall be treated as set forth in Section 9.12); (y) all Borrower Materials marked “PUBLIC” are permitted to be made available through a portion of the Platform designated “Public Side Information”; and (z) the Administrative Agent and the Lead Arrangers shall be entitled to treat any Borrower Materials that are not marked “PUBLIC” as being suitable only for posting on a portion of the Platform not designated “Public Side Information”. Section 5.02 Notices of Material Events. Parent and the Borrower will, upon knowledge thereof by a Responsible Officer, furnish to the Administrative Agent prompt written notice of the following: (a) the occurrence of any Default;

147 (b) the filing or commencement of any action, suit or proceeding by or before any arbitrator or Governmental Authority against or affecting Parent or any Subsidiary or Affiliate thereof that could reasonably be expected to result in a Material Adverse Effect; (c) the occurrence of any ERISA Event that, alone or together with any other ERISA Events that have occurred, could reasonably be expected to result in a Material Adverse Effect; (d) (i) any contribution required to be made with respect to a Non-U.S. Plan has not been timely made; (ii) Parent or any Restricted Subsidiary has incurred any obligation in connection with the termination of, or withdrawal from, any Non-U.S. Plan; or (iii) Parent or any Restricted Subsidiary may incur any material liability pursuant to any Non-U.S. Plan, in each case, to the extent that such event could reasonably be expected to result in a Material Adverse Effect; and (e) any other development that results in, or could reasonably be expected to result in, a Material Adverse Effect. Each notice delivered under this Section 5.02 shall be accompanied by a statement of a Responsible Officer of Parent setting forth the details of the event or development requiring such notice and any action taken or proposed to be taken with respect thereto. Information required to be delivered pursuant to clause (b) of this Section 5.02 shall be deemed to have been delivered if such information, or one or more annual or quarterly or other periodic reports containing such information, shall have been posted by the Administrative Agent on an IntraLinks or similar site to which the Lenders have been granted access or shall be available on the website of the SEC at http://www.sec.gov. Information required to be delivered pursuant to this Section 5.02 may also be delivered by electronic communications pursuant to procedures approved by the Administrative Agent. Section 5.03 Existence; Conduct of Business. Parent will, and will cause its Material Subsidiaries to, do or cause to be done all things necessary to preserve, renew and keep in full force and effect its legal existence and the rights, qualifications, licenses, permits, privileges, franchises, governmental authorizations and Intellectual Property rights material to the conduct of its business, and maintain all requisite authority to conduct its business in each jurisdiction in which its business is conducted; provided that (i) the foregoing shall not prohibit any merger, amalgamation, consolidation, liquidation or dissolution permitted under Section 6.03 and (ii) neither Parent nor its Material Subsidiaries shall be required to preserve any right, license, permit, privilege, franchise, patent, copyright, trademark, trade name or other Intellectual Property rights if Parent or such Material Subsidiary shall determine, in its reasonable judgment, that the preservation thereof is no longer desirable in the conduct of business of Parent or such Material Subsidiary, as the case may be, and that the loss thereof is not disadvantageous in any material respect to Parent, such Material Subsidiary or the Lenders. Section 5.04 Payment of Obligations. Parent will, and will cause each of its Restricted Subsidiaries to, pay its obligations, including Tax liabilities, that, if not paid, could result in a Material Adverse Effect before the same shall become delinquent or in default, except where (a) the validity or amount thereof is being contested in good faith by appropriate proceedings, (b)

148 Parent or such Restricted Subsidiary has set aside on its books adequate reserves with respect thereto in accordance with GAAP and (c) the failure to make payment pending such contest could not reasonably be expected to result in a Material Adverse Effect. Section 5.05 Maintenance of Properties; Insurance. Parent will, and will cause each of its Material Subsidiaries to, (a) keep and maintain all tangible property material to the conduct of its business in good working order and condition, ordinary wear and tear excepted, and (b) maintain with financially sound and reputable carriers (i) insurance in such amounts (with no greater risk retention) and against such risks and such other hazards, as is customarily maintained by companies of established repute engaged in the same or similar businesses operating in the same or similar locations and (ii) all insurance required pursuant to the Collateral Documents. Parent will furnish to the Lenders, upon request of the Administrative Agent, information in reasonable detail as to the insurance so maintained. Parent shall deliver to the Administrative Agent endorsements (x) to all “All Risk” physical damage insurance policies on all of the Loan Parties’ tangible personal property and assets and business interruption insurance policies naming the Collateral Agent as lender loss payee, and (y) to all general liability and other liability policies naming the Administrative Agent an additional insured. In the event Parent or any of its Material Subsidiaries at any time or times hereafter shall fail to obtain or maintain any of the policies or insurance required herein or to pay any premium in whole or in part relating thereto, then the Administrative Agent, without waiving or releasing any obligations or resulting Default hereunder, may at any time or times thereafter (but shall be under no obligation to do so) obtain and maintain such policies of insurance and pay such premiums and take any other action with respect thereto which the Administrative Agent deems advisable. All sums so disbursed by the Administrative Agent shall constitute part of the Obligations, payable as provided in this Agreement. Section 5.06 Books and Records; Inspection Rights. Parent will, and will cause each of the Material Subsidiaries to, keep proper books of record and account in which full, true and correct entries in conformity with GAAP and applicable law are made of all material financial dealings and transactions in relation to its business and activities. Parent will, and will cause each of its Material Subsidiaries to, permit any representatives designated by the Administrative Agent or any Lender (pursuant to a request made through the Administrative Agent), at reasonable times upon reasonable prior notice (but not more than once annually if no Event of Default shall exist), to visit and inspect its properties, to examine and make extracts from its books and records, including examination of its environmental assessment reports and Phase I or Phase II studies, and to discuss its affairs, finances and condition with its officers and independent accountants, all at such reasonable times and as often as reasonably requested. Parent acknowledges that the Administrative Agent, after exercising its rights of inspection, may prepare and distribute to the Lenders certain reports pertaining to Parent and its Material Subsidiaries’ assets for internal use by the Administrative Agent and the Lenders. Notwithstanding anything to the contrary in this Section 5.06, none of Parent or any of its Restricted Subsidiaries will be required to disclose, permit the inspection, examination or making copies or abstracts of, or discussion of, any document, information or other matter that (a) constitutes non-financial trade secrets or non- financial proprietary information, (b) in respect of which disclosure to the Administrative Agent or any Lender (or their respective representatives or contractors) is prohibited by law or any binding agreement (not entered into in contemplation hereof) or (c) is subject to attorney-client or similar privilege or constitutes attorney work product.

149 Section 5.07 Compliance with Laws and Material Contractual Obligations. Parent will, and will cause each of its Restricted Subsidiaries to, (i) comply with all laws, rules, regulations and orders of any Governmental Authority applicable to it or its property (including without limitation Environmental Laws) and (ii) perform in all material respects its obligations under material agreements to which it is a party, in each case except where the failure to do so, individually or in the aggregate, could not reasonably be expected to result in a Material Adverse Effect. Section 5.08 Use of Proceeds. (a) The Borrower Representative shall use the proceeds of the Initial Term Loans for (a) the consummation of the Reorganization Plan Transactions, (b) the payment of Transaction Expenses and (c) general corporate purposes. The Borrower Representative shall use the proceeds of the 2024 Refinancing Term Loans as set forth in the First Amendment. (b) The proceeds of the Revolving Loans will be used for general corporate purposes of Parent and its Subsidiaries. (c) No part of the proceeds of any Loan will be used, whether directly or knowingly indirectly, (i) in furtherance of an offer, payment, promise to pay, or authorization of the payment or giving of money, or anything else of value, to any Person in violation of any Anti-Corruption Laws, (ii) for any purpose that entails a violation of any of the Regulations of the Board, including Regulations T, U and X, (iii) in violation of the USA Patriot Act, (iv) for the purpose of funding, financing or facilitating any activities, business or transaction of or with any Sanctioned Person, or in any Sanctioned Country, to the extent such activities, business or transaction would be prohibited by applicable Sanctions or (v) in any manner in violation of any applicable Sanctions. Any provision of this Section 5.08(c) shall not apply to or in favor of any person incorporated in a member state of the European Union or the United Kingdom if and to the extent that it would result in a breach, by or in respect of that person, of any applicable Blocking Law. “Blocking Law” means any provision of Council Regulation (EC) No 2271/1996 of 22 November 1996 (or any law or regulation implementing such Regulation in any member state of the European Union) or any similar blocking or anti-boycott law in any Member State of the European Union or the United Kingdom. Section 5.09 Subsidiary Guarantors; Pledges; Additional Collateral; Further Assurances. (a) As promptly as possible but in any event within forty-five (45) days (or such later date as may be agreed upon by the Administrative Agent) after any Person becomes a Material Subsidiary (other than an Excluded Subsidiary) or any Subsidiary qualifies independently as a Material Subsidiary (other than an Excluded Subsidiary) or is designated by Parent as a Subsidiary Guarantor, Parent shall provide the Administrative Agent with written notice thereof and shall (subject to the Agreed Security Principles, in the case of any Foreign Subsidiary) cause each such Material Subsidiary to deliver to the Administrative Agent a supplement to the Subsidiary Guaranty and the US Security Agreement and/or each other applicable Collateral Document (in each case in the form contemplated thereby and modified as required in order to comply with local laws in accordance with the Agreed Security Principles, if applicable) pursuant to which such Subsidiary agrees to be bound by the terms and provisions thereof, the Subsidiary Guaranty, the US Security Agreement and/or other applicable Collateral Document, as applicable, to be accompanied by appropriate corporate resolutions, other corporate documentation and legal

150 opinions as may be reasonably requested by, and in form and substance reasonably satisfactory to, the Administrative Agent and its counsel. (b) Subject to the Agreed Security Principles (except in the case of any Loan Party organized under the laws of the United States) and Section 5.09(f), Parent will cause, and will cause each other Loan Party to cause, all of its owned property (whether real, personal, tangible, intangible, or mixed but excluding Excluded Assets) to be subject at all times to perfected Liens in favor of the Collateral Agent for the benefit of the Secured Parties to secure the Secured Obligations in accordance with the terms and conditions of the Collateral Documents on a first priority basis, subject to no other Liens other than Liens permitted by Section 6.02. Without limiting the generality of the foregoing, and subject to the Agreed Security Principles (where applicable) and Section 5.09(f), Parent (i) will cause the issued and outstanding Equity Interests of each Pledge Subsidiary directly or indirectly owned by the Borrower or any other Loan Party (other than Excluded Assets) to be subject at all times to a first priority, perfected Lien in favor of the Collateral Agent to secure the Secured Obligations in accordance with the terms and conditions of the Collateral Documents or such other pledge, collateral and security documents as the Administrative Agent shall reasonably request and (ii) will, and will cause each other Loan Party to, deliver Mortgages and Mortgage Instruments with respect to real property (excluding Excluded Assets) owned by the Borrower or such Loan Party to the extent, and within such time period as is, reasonably required by the Administrative Agent. (c) Without limiting the foregoing, but subject to the Agreed Security Principles (except in the case of any Loan Party organized under the laws of the United States) and Section 5.09(f), Parent will, and will cause each other Loan Party to, execute and deliver, or cause to be executed and delivered, to the Collateral Agent such documents, agreements and instruments, and will take or cause to be taken such further actions (including the filing and recording of financing statements, fixture filings, Mortgages, deeds of trust and other documents and such other actions or deliveries of the type required by Section 4.01, as applicable), which may be required by law or which the Administrative Agent may, from time to time, reasonably request to carry out the terms and conditions of this Agreement and the other Loan Documents and to ensure perfection and priority of the Liens created or intended to be created by the Collateral Documents, all at the expense of the Borrower; provided that, in connection with any real property subject to this Section 5.09(c), the Borrower will comply with the National Flood Insurance Reform Act of 1994 and related legislation and regulations. (d) Subject to the Agreed Security Principles (except in the case of any Loan Party organized under the laws of the United States) and Section 5.09(f), other than with respect to such Loan Parties as expressly provided in the final proviso to the definition of Agreed Security Principles), if any assets (including any real property or improvements thereto or any interest therein) are acquired by a Loan Party (other than Excluded Assets and assets constituting Collateral that become subject to the Lien in favor of the Administrative Agent upon acquisition thereof), Parent will notify the Administrative Agent thereof, and, if requested by the Administrative Agent, Parent will cause such assets to be subjected to a Lien securing the Secured Obligations and will take, and cause the other Loan Parties to take, such actions as shall be necessary or reasonably requested by the Administrative Agent to grant and perfect such Liens, including actions described in paragraph (b) of this Section 5.09, all at the expense of Parent.

151 (e) Concurrently with the designation of any Subsidiary as a guarantor under any other Material Indebtedness of the Borrower after the Effective Date, the Borrower shall cause each such Subsidiary to deliver to the Administrative Agent a duly executed copy of the Subsidiary Guaranty (or supplement thereto) pursuant to which such Subsidiary agrees to be bound by the terms and provisions of the Subsidiary Guaranty and, in the case of a Foreign Subsidiary, modified as required in order to comply with local laws in accordance with the Agreed Security Principles, and such Subsidiary Guaranty (or supplement thereto) shall be accompanied by appropriate officer’s certificates, resolutions, organizational documents and legal opinions of counsel as the Administrative Agent may reasonably request, all in form and substance reasonably satisfactory to the Administrative Agent and its counsel. (f) (i) Notwithstanding anything in this Agreement to the contrary, in no event shall any Mortgage be required to be executed and delivered with respect to any real property constituting Collateral, unless and until the Administrative Agent has so requested (and the conditions set forth in this Section 5.09(f) and in Section 5.09(c) have been met). The Administrative Agent shall not deliver such request with respect to any such real property located in the United States and its territories until (x) a date that is at least 45 Business Days after the Administrative Agent has delivered to the Lenders (A) written notice of its intention to request delivery and execution of the applicable Mortgage and (B) (1) a completed standard “life of loan” flood hazard determination form and such other documents as any Lender may reasonably request to complete its flood insurance due diligence with respect to the applicable real property; (2) if the improvements to the applicable real property are determined to have special flood hazards by the Federal Emergency Management Agency, a notification to the applicable Loan Party (“Loan Party Notice”) and (if applicable) notification to the applicable Loan Party that flood insurance coverage under the National Flood Insurance Program (“NFIP”) is not available because the community where such real property is located does not participate in the NFIP; (3) documentation evidencing the applicable Loan Party’s receipt of the Loan Party Notice; and (4) if the Loan Party Notice is required to be given and, to the extent flood insurance is required by any applicable requirement of law or any Lender’s written regulatory or compliance procedures and flood insurance is available in the community in which such real property is located, evidence of a flood insurance policy in compliance with the Flood Insurance Laws (including without limitation, in an amount required under the Flood Insurance Laws) and (y) all Lenders shall have consented to the making of such request; provided that a Lender shall be deemed to have so consented unless such Lender objects to the execution and delivery of such Mortgage in writing to the Administrative Agent no later than 45 Business Days after delivery of the documentation and written notice described in clauses (x)(A) and (B) above. (ii) Within 120 days of the satisfaction of the conditions set forth in clause (i) above (which may be extended in the Administrative Agent’s sole discretion) with respect to a parcel of real property constituting Collateral located in the United States owned by any Domestic Subsidiary that is a Loan Party, Parent shall procure the execution and delivery of, and deliver to the Administrative Agent, Mortgages and Mortgage Instruments related thereto reasonably required by the Administrative Agent. (g) Notwithstanding anything to the contrary herein or in any other Loan Document, no Loan Party shall have any obligation to (i) perfect through control agreements or “control” with respect to any assets (other than in respect of promissory notes in excess of

152 $10,000,000 and certificated Equity Interests constituting Collateral that are required to be pledged pursuant to the Collateral Documents), (ii) perfect any security interest or lien in any Intellectual Property included in the Collateral in any jurisdiction other than in the United States or Ireland, (iii) enter into any Guarantees governed by the laws of any non-U.S. jurisdiction, (iv) obtain any landlord waivers, estoppels or collateral access letters, and (v) perfect a security interest in any letter of credit rights (other than by the filing of a UCC or similar financing statement). Section 5.10 Designation of Subsidiaries. Parent may, at any time from and after the Effective Date, designate any Restricted Subsidiary as an Unrestricted Subsidiary or any Unrestricted Subsidiary as a Restricted Subsidiary; provided that (i) immediately before and after such designation, no Default or Event of Default shall have occurred and be continuing and (ii) immediately after giving effect to such designation (including giving effect on a pro forma basis subject to Section 1.04), the Total Net Leverage Ratio shall be no greater than 6.00 to 1.00. The designation of any Restricted Subsidiary as an Unrestricted Subsidiary after the Effective Date shall constitute an Investment by the applicable Loan Party therein at the date of designation in an amount equal to the fair market value of the applicable Loan Party’s (or any of its Restricted Subsidiaries’) investment therein. The designation of any Unrestricted Subsidiary as a Restricted Subsidiary after the Effective Date shall constitute (i) the incurrence at the time of designation of any Investment, Indebtedness or Liens of such Subsidiary existing at such time and (ii) a return on any Investment by the applicable Loan Party in Unrestricted Subsidiaries pursuant to the preceding sentence in an amount equal to the fair market value at the date of such designation of such Loan Party’s Investment in such Subsidiary. Notwithstanding the foregoing, (i) no Borrower nor any parent company of any Borrower shall be permitted to be an Unrestricted Subsidiary and (ii) no Restricted Subsidiary that owns or exclusively licenses any Material IP shall be permitted to be designated as an Unrestricted Subsidiary. Notwithstanding anything to the contrary contained in this Agreement (including, without limitation, in Section 6.02, Section 6.03, Section 6.04, Section 6.06 and Section 6.07), no sale, transfer of legal title or exclusive license (other than an exclusive license for a specific country that is not material to the business of the Borrower and its Restricted Subsidiaries, taken as a whole, where such Loan Party retains, and does not exclusively license to any party that is not a Loan Party, all other worldwide rights with respect thereto) of Material IP may be made by any Loan Party to (i) any Unrestricted Subsidiary or (ii) any Restricted Subsidiary that is not a Loan Party. Section 5.11 Ratings. Until the Term Loans are paid in full and terminated in accordance with this Agreement, Parent shall use commercially reasonable efforts to cause (x) S&P and Moody’s to issue, and maintain, ratings for the Term Loans, (y) Moody’s to issue, and maintain, a corporate family rating (or the equivalent thereof) of Parent and (z) S&P to issue, and maintain, a corporate credit rating (or the equivalent thereof) of Parent (it being understood, in each case, that such obligation shall not require Parent to maintain a specific rating). Section 5.12 Post-Closing Obligations. As soon as practicable but in any event within the time periods set forth on Schedule 5.12 (or such later date that the Administrative Agent in its reasonable discretion may permit), Parent shall take or cause its Restricted Subsidiaries to take the actions set forth on Schedule 5.12. Notwithstanding anything in this Agreement or in the other Loan Documents to the contrary, to the extent any representation and warranty in any Loan Document would not be true because the actions set forth on Schedule 5.12 were not taken on the Effective Date, the respective representation and warranty shall not be required to be true and

153 correct in all material respects until the time the respective action is taken (or was required to be taken) in accordance with Schedule 5.12. ARTICLE VI NEGATIVE COVENANTS Until the Commitments have expired or terminated and the principal of and interest on each Loan and all fees payable hereunder have been paid in full and all Letters of Credit have expired or terminated (in each case, without any pending drawings) or been Cash Collateralized, and all LC Disbursements shall have been reimbursed, each of Parent and the Borrower covenants and agrees with the Lenders that: Section 6.01 Indebtedness. Parent will not, and will not permit any Restricted Subsidiary to, create, incur, assume or permit to exist any Indebtedness, except: (a) the Secured Obligations; (b) Indebtedness existing on the Effective Date and, with respect to any item of Indebtedness in an aggregate outstanding principal amount in excess of $5.0 million, set forth in Schedule 6.01 and any refinancing, extensions, renewals or replacements of any such Indebtedness that does not increase the outstanding principal amount thereof (other than with respect to unpaid accrued interest and premium thereon, any committed or undrawn amounts and underwriting discounts, fees, commissions and expenses, associated with such Indebtedness); (c) Indebtedness (i) under the Senior Secured Notes and (ii) any Permitted Refinancing Indebtedness in respect thereof; (d) Indebtedness of Parent to any Restricted Subsidiary and of any Restricted Subsidiary to Parent or any other Restricted Subsidiary; provided that (x) Indebtedness of any Restricted Subsidiary that is not a Loan Party to any Loan Party shall be subject to the limitations set forth in Section 6.04(d) and (y) any Indebtedness owing by any Loan Party to a Restricted Subsidiary that is not a Loan Party shall be unsecured and subordinated in right of payment to the Secured Obligations on a basis, and pursuant to an agreement, reasonably satisfactory to the Administrative Agent; (e) Guarantees by Parent or any Restricted Subsidiary of Indebtedness or other obligations of Parent or any Restricted Subsidiary; provided that the aggregate amount of Indebtedness and other payment obligations (other than in respect of any overdrafts and related liabilities arising in the ordinary course of business from treasury, depository and cash management services or in connection with any automated clearing house transfer of funds) of Restricted Subsidiaries that are not Loan Parties that is Guaranteed by any Loan Party shall be subject to the limitations set forth in Section 6.04(d); (f) Indebtedness (1) of Parent or any Restricted Subsidiary incurred to finance the acquisition, construction or improvement of any fixed or capital assets, including Capital Lease Obligations and any Indebtedness assumed in connection with the acquisition of any such assets or secured by a Lien on any such assets prior to the acquisition thereof; provided that

154 (i) such Indebtedness is incurred prior to or within 270 days after such acquisition or the completion of such construction or improvement and (ii) the aggregate principal amount of Indebtedness incurred under this clause (f) shall not exceed the greater of (x) $125,000,000 and (y) 20.0% of Consolidated EBITDA as of the end of the Reference Period or (2) constituting Permitted Refinancing Indebtedness in respect of Indebtedness theretofore outstanding (and permitted to be outstanding) pursuant to this clause (f); (g) Indebtedness of Parent or any Restricted Subsidiary as an account party in respect of commercial letters of credit; (h) Indebtedness owed in respect of any Banking Services and any other netting services, overdrafts and related liabilities arising from treasury, depository and cash management services or in connection with any automated clearing-house transfers of funds; (i) Indebtedness under bid bonds, performance bonds, surety bonds and similar obligations, in each case, incurred by Parent or any of its Restricted Subsidiaries in the ordinary course of business, including guarantees or obligations with respect to letters of credit supporting such bid bonds, performance bonds, surety bonds and similar obligations; (j) Swap Agreements permitted under Section 6.05; (k) Indebtedness of Restricted Subsidiaries that are not Loan Parties and Foreign Subsidiaries, and guarantees thereof by other such Restricted Subsidiaries; provided that the aggregate principal amount of such Indebtedness shall not exceed, on a pro forma basis in accordance with Section 1.04, immediately after giving effect to the issuance or incurrence of such Indebtedness the greater of (x) $75,000,000 and (y) 10.0% of Consolidated EBITDA as of the end of the Reference Period; (l) Guarantees of Indebtedness of directors, officers, employees, agents and advisors of Parent, or any of its Restricted Subsidiaries in respect of expenses of such Persons in connection with relocations and other ordinary course of business purposes; provided that the aggregate amount of Indebtedness so guaranteed, when added to the aggregate amount of unreimbursed payments theretofore made in respect of such guarantees and the amount of loans and advances then outstanding under Section 6.04(u), shall not at any time exceed the greater of (x) $30,000,000 and (y) 5.0% of Consolidated EBITDA as of the end of the Reference Period; (m) Indebtedness arising from agreements providing for indemnification, adjustment of purchase price or similar obligations, or from guaranties, surety bonds or performance bonds securing the performance of Parent or any of its Restricted Subsidiaries pursuant to such agreements, in connection with Permitted Acquisitions or permitted Dispositions; (n) Indebtedness representing installment insurance premiums owing in the ordinary course of business; (o) Indebtedness representing deferred compensation, severance, pension, and health and welfare retirement benefits or the equivalent to current and former

155 employees of Parent and its Restricted Subsidiaries incurred in the ordinary course of business or existing on the Effective Date; (p) unsecured Indebtedness arising out of judgments not constituting an Event of Default; (q) Indebtedness of Parent or any of its Restricted Subsidiaries incurred in connection with a Permitted Acquisition, so long as (i) subject to Section 1.04, no Event of Default shall have occurred and be continuing or would exist immediately after giving effect (including giving effect on a pro forma basis) to such incurrence, (ii) such Indebtedness is not scheduled to mature prior to the date that is 91 days after the Latest Maturity Date (other than such Indebtedness incurred under the Inside Maturity Basket) and (iii)(a) immediately after giving effect thereto (including on a pro forma basis subject to Section 1.04), if such Indebtedness is secured on a junior basis with the Secured Obligations, the Secured Net Leverage Ratio shall be no greater than 5.00 to 1.00, (b) if such Indebtedness is secured on a pari passu basis with the Secured Obligations, the First Lien Net Leverage Ratio shall be no greater than 3.50 to 1.00 and (c) if such Indebtedness is unsecured or if such Indebtedness is incurred by a Restricted Subsidiary that is not a Loan Party, whether or not such Indebtedness is secured or unsecured, either (x) the Total Net Leverage Ratio shall be no greater than 6.00 to 1.00 or (y) the Consolidated Interest Coverage Ratio shall be no less than 2.00 to 1.00; provided that the aggregate principal amount of Indebtedness incurred under this clause (q) by Restricted Subsidiaries that are not Loan Parties, together with the aggregate principal amount of Indebtedness incurred pursuant to Section 6.01(r) by such Restricted Subsidiaries shall not exceed the greater of (x) $75,000,000 and (y) 10.0% of Consolidated EBITDA as of the end of the Reference Period; (r) Indebtedness (x) of any Person that becomes a Restricted Subsidiary (or of any Person not previously a Subsidiary that is merged or consolidated with or into a Restricted Subsidiary in a transaction permitted hereunder) after the Effective Date (except by way of designation of an Unrestricted Subsidiary as a Restricted Subsidiary), or Indebtedness of any Person that is assumed by any Restricted Subsidiary in connection with an acquisition of assets by such Restricted Subsidiary in a Permitted Acquisition; provided that (A) such Indebtedness exists at the time such Person becomes a Restricted Subsidiary (or is so merged or consolidated) or such assets are acquired and is not created in contemplation of or in connection with such Person becoming a Restricted Subsidiary (or such merger or consolidation) or such assets being acquired, and (B) immediately prior to and after giving effect (including giving effect on a pro forma basis subject to Section 1.04), to the assumption of such Indebtedness or making of such Guarantee, as the case may be, (i) if such Indebtedness is secured on a junior basis with the Secured Obligations, the Secured Net Leverage Ratio shall be no greater than 5.00 to 1.00, (ii) if such Indebtedness is secured on a pari passu basis with the Secured Obligations, the First Lien Net Leverage Ratio shall be no greater than 3.50 to 1.00 and (iii) if such Indebtedness is unsecured or if such Indebtedness is incurred by a Restricted Subsidiary that is not a Loan Party, whether or not such Indebtedness is secured or unsecured, either (x) the Total Net Leverage Ratio shall be no greater than 6.00 to 1.00, or (y) the Consolidated Interest Coverage Ratio shall be no less than 2.00 to 1.00; or (y) constituting Permitted Refinancing Indebtedness in respect of Indebtedness theretofore outstanding (and permitted to be outstanding) pursuant to this clause (r); provided that the aggregate principal amount of Indebtedness incurred under this clause (r) by Restricted Subsidiaries that are not Loan Parties, together with the aggregate principal amount of

156 Indebtedness incurred pursuant to Section 6.01(q) by such Restricted Subsidiaries, shall not exceed the greater of (x) $75,000,000 and (y) 10.0% of Consolidated EBITDA as of the end of the Reference Period; (s) Permitted Indebtedness and Permitted First Lien Indebtedness and any Permitted Refinancing Indebtedness in respect thereof; (t) other Indebtedness of Parent and its Subsidiaries; provided that the aggregate outstanding principal amount of such Indebtedness shall not at any time exceed the greater of (x) $230,000,000 and (y) 30.0% of Consolidated EBITDA as of the end of the Reference Period; (u) Indebtedness of joint ventures and/or any Indebtedness incurred on behalf thereof or representing guarantees of Indebtedness of joint ventures; provided that the aggregate principal amount of such Indebtedness shall not exceed, on a pro forma basis in accordance with Section 1.04, immediately after giving effect to the issuance or incurrence of such Indebtedness, the greater of (x) $75,000,000 and (y) 10.0% of Consolidated EBITDA as of the end of the Reference Period; (v) (i) Permitted Pari Passu Secured Refinancing Debt, (ii) Permitted Junior Secured Refinancing Debt and (iii) Permitted Unsecured Refinancing Debt, and any Permitted Refinancing Indebtedness in respect thereof; (w) Indebtedness (x) of the Borrower Representative or any other Loan Party in respect of (1) one or more series of senior unsecured notes or senior secured notes that will be secured by all or a portion of the Collateral on a pari passu or junior basis with the Secured Obligations, and/or (2) one or more series of term loans that will be unsecured or secured by all or a portion of the Collateral on a pari passu or junior basis with the Secured Obligations, in each case that are issued or made in lieu of Incremental Revolving Loans and/or Incremental Term Loans; provided that (A) such Indebtedness is not scheduled to mature prior to the Latest Maturity Date (other than such Indebtedness incurred under the Inside Maturity Basket), (B) the aggregate principal amount of all such Indebtedness issued or incurred pursuant to this sub-clause (x) shall not, when aggregated with all Incremental Revolving Loans and Incremental Term Loans, exceed the Incremental Amount, (C) such Indebtedness shall not be subject to any Guarantee by Parent or any Restricted Subsidiary other than a Loan Party, (D) in the case of any such Indebtedness that is secured, the obligations in respect thereof shall not be secured by any Lien on any asset of Parent or any of its Restricted Subsidiaries other than any asset constituting Collateral, (E) [reserved], (F) if such Indebtedness is secured, the security agreements relating to such Indebtedness shall be substantially the same as the Collateral Documents (with such differences as are reasonably satisfactory to the Administrative Agent), (G) no Event of Default shall have occurred and be continuing or would exist immediately after giving effect (including giving effect on a pro forma basis) to such incurrence (subject to Section 1.04), (H) if such Indebtedness is secured, such Indebtedness shall be subject to an Approved Intercreditor Agreement, (I) if such Indebtedness consists of term loans secured on a pari passu basis with the Secured Obligations hereunder, then the applicable Borrower shall comply with the “most favored nation” pricing provision in the proviso in Section 2.20(c)(v) as if such Indebtedness were Other Term Loans incurred pursuant to Section 2.20 (to the extent then applicable), (J) the terms and conditions of such Indebtedness

157 (excluding pricing, fees, prepayment or redemption premiums and terms) are (in the reasonable judgment of Parent), when taken as a whole, (1) not materially more favorable to the lenders or holders providing such Indebtedness than those applicable to the Obligations when taken as a whole (other than covenants or other provisions applicable only to periods after the Latest Maturity Date at the time of incurrence of such Indebtedness) or (2) otherwise on current market terms for such type of Indebtedness and (K) such Indebtedness may be incurred in the form of a bridge or other interim credit facility intended to be refinanced with long-term indebtedness (and such bridge or other interim credit facility shall be deemed to satisfy clause (A) of this section so long as (x) such credit facility includes customary “rollover” provisions and (y) assuming such credit facility were to be extended pursuant to such “rollover” provisions, such extended credit facility would comply with clause (A) above) and in which case, on or prior to the first anniversary of the incurrence of such “bridge” or other interim credit facility, clause (J) in this definition shall not prohibit the inclusion of customary terms for “bridge” facilities, including customary mandatory prepayment, repurchase or redemption provisions; and (y) Permitted Refinancing Indebtedness in respect of Indebtedness previously incurred pursuant to, and then outstanding pursuant to, this clause (w) (with any Indebtedness outstanding pursuant to this clause (w) from time to time being herein called the “Incremental Equivalent Debt”); (x) Indebtedness of Parent or any Restricted Subsidiary incurred pursuant to Permitted Receivables Facilities, provided that the Attributable Receivables Indebtedness thereunder shall not exceed an aggregate amount of (x) $380,000,000 and (y) 50.0% of Consolidated EBITDA as of the end of the Reference Period at any time outstanding; (y) Indebtedness in an aggregate outstanding principal amount not to exceed 100% of the amount of Net Proceeds received by Parent from the issuance or sale of Equity Interests (other than Disqualified Equity Interests) to the extent the relevant Net Proceeds was not previously (and is not concurrently being) applied in determining the permissibility of a transaction under the Loan Documents where such permissibility was or is (or may have been) contingent on receipt of such amount or utilization of such amount for a specified purpose, and (ii) any Permitted Refinancing Indebtedness in respect thereof; (z) customer deposits and advance payments received in the ordinary course of business from customers for goods and services purchased in the ordinary course of business; (aa) Indebtedness in respect of any letter of credit or bank guarantee issued in favor of any Issuing Bank to support any Defaulting Lender’s participation in Letters of Credit issued; (bb) the incurrence of Indebtedness by Parent or any Restricted Subsidiary undertaken in connection with cash management (including netting services, automatic clearinghouse arrangements, overdraft protections, employee credit card programs and related or similar services or activities) with respect to Parent, any Subsidiaries or any joint venture in the ordinary course of business or consistent with industry practice; and (cc) Indebtedness of Parent or any Restricted Subsidiary to the extent that 100% of such Indebtedness is supported by any Letter of Credit.

158 For purposes of determining compliance with any Dollar-denominated restriction on the incurrence of Indebtedness, the Dollar-equivalent principal amount of Indebtedness denominated in a foreign currency shall be calculated based on the relevant currency exchange rate in effect on the date such Indebtedness was incurred, in the case of term debt, or first committed, in the case of revolving credit debt; provided that if such Indebtedness is incurred to extend, replace, refund, refinance, renew or defease other Indebtedness denominated in a foreign currency, and such extension, replacement, refunding, refinancing, renewal or defeasance would cause the applicable Dollar-denominated restriction to be exceeded if calculated at the relevant currency exchange rate in effect on the date of such extension, replacement, refunding, refinancing, renewal or defeasance, such Dollar-denominated restriction shall be deemed not to have been exceeded so long as the principal amount of such refinancing Indebtedness does not exceed the principal amount of such Indebtedness being extended, replaced, refunded, refinanced, renewed or defeased, plus the aggregate amount of fees, underwriting discounts, premiums and other costs and expenses incurred in connection with such refinancing. For purposes of determining compliance with this Section 6.01: (1) in the event that an item of Indebtedness (or any portion thereof) at any time, whether at the time of incurrence or upon the application of all or a portion of the proceeds thereof or subsequently, meets the criteria of more than one of the categories of permitted Indebtedness described in clauses (a) through (cc) above, Parent, in its sole discretion, may divide and classify and may subsequently redivide and reclassify, such item of Indebtedness (or any portion thereof) and will only be required to include the amount and type of such Indebtedness (or a portion thereof) in such of the above clauses (a) through (cc) under Section 6.01 as determined by Parent at such time; provided that all Indebtedness (x) incurred or established hereunder on the Effective Date and (y) represented by the Senior Secured Notes and related Guarantees on the Effective Date will, at all times, be treated as incurred on the Effective Date under Sections 6.01(a) and (c), respectively, and may not be reclassified; (2) Parent is entitled to divide and classify an item of Indebtedness in more than one of the types of Indebtedness described in Sections 6.01(a) through (cc), subject to the proviso to the preceding clause (1); (3) the principal amount of Indebtedness outstanding under any clause of this Section 6.01 will be determined after giving effect to the application of proceeds of any such Indebtedness to refinance any such other Indebtedness; and (4) guarantees of, or obligations in respect of letters of credit relating to, Indebtedness that are otherwise included in the determination of a particular amount of Indebtedness will not be included in the determination of such amount of Indebtedness; provided that the incurrence of the Indebtedness represented by such guarantee or letter of credit, as the case may be, was incurred in compliance with this Section 6.01.

159 The accrual of interest, the accretion of accreted value and the payment of interest in the form of additional Indebtedness shall not be deemed to be an incurrence of Indebtedness for purposes of this Section 6.01. The principal amount of any non-interest bearing Indebtedness or other discount security constituting Indebtedness at any date shall be the principal amount thereof that would be shown on a balance sheet of Parent dated such date prepared in accordance with GAAP. Notwithstanding anything to the contrary contained in this Agreement, other than pursuant to Section 2.20, neither Parent nor any Loan Party shall incur Indebtedness for borrowed money secured by all or any portion of the Collateral where the payments from or on account of the Collateral are on a pari passu or senior basis with the Revolving Facility Obligations without the prior written consent of the Required Revolving Lenders. Section 6.02 Liens. Parent will not, and will not permit any Restricted Subsidiary to, create, incur, assume or permit to exist any Lien on any property or asset now owned or hereafter acquired by it, or assign or sell any income or revenues (including accounts receivable) or rights in respect of any thereof, except: (a) Liens created pursuant to any Loan Document; (b) Permitted Encumbrances; (c) any Lien on any property or asset of Parent or any Restricted Subsidiary existing on the Effective Date and set forth in Schedule 6.02 and any modifications, renewals and extensions thereof and any Lien granted as a replacement or substitute therefor; provided that (i) such Lien shall not apply to any other property or asset of Parent or any Restricted Subsidiary other than improvements thereon or proceeds from the disposition of such asset and (ii) such Lien shall secure only those obligations which it secures on the Effective Date and any refinancings, extensions, renewals or replacements thereof that do not increase the outstanding principal amount thereof (other than as permitted by Section 6.01); (d) any Lien existing on any property or asset prior to the acquisition thereof by Parent or any Restricted Subsidiary or existing on any property or asset of any Person that becomes a Restricted Subsidiary after the Effective Date prior to the time such Person becomes a Restricted Subsidiary and any modifications, replacements, renewals or extensions thereof; provided that (i) such Lien is not created in contemplation of or in connection with such acquisition or such Person becoming a Restricted Subsidiary, as the case may be, (ii) such Lien shall not apply to any other property or assets of Parent or any Restricted Subsidiary and (iii) such Lien shall secure only those obligations which it secures on the date of such acquisition or the date such Person becomes a Restricted Subsidiary, as the case may be, and any refinancing, extensions, renewals or replacements thereof that do not increase the outstanding principal amount thereof (other than as permitted by Section 6.01); (e) Liens on fixed or capital assets acquired, constructed or improved by Parent or any Restricted Subsidiary; provided that (i) such Liens secure Indebtedness permitted by clause (f) of Section 6.01, (ii) such Liens and the Indebtedness secured thereby are initially incurred prior to or within 270 days after such acquisition or the completion of such construction or improvement, (iii) the Indebtedness secured thereby does not exceed the cost of acquiring,

160 constructing or improving such fixed or capital assets and (iv) such Liens shall not apply to any other property or assets of Parent or any Restricted Subsidiary other than improvements thereon or proceeds from the disposition of such property or assets except that individual financings provided by a Person or its Affiliates may be cross collateralized to other financings provided by such Person or its Affiliates; (f) in connection with the sale or transfer of any assets in a transaction permitted under Section 6.03, customary rights and restrictions contained in agreements relating to such sale or transfer pending the completion thereof; (g) any encumbrance or restriction (including put, call arrangements, tag, drag, right of first refusal and similar rights) with respect to Equity Interests of any joint venture or similar arrangement pursuant to any joint venture or similar agreement; (h) any interest or title of a lessor under any lease or sublease entered into by Parent or any Restricted Subsidiary in the ordinary course of its business and other statutory and common law landlords’ liens under leases; (i) any interest or title of a licensor under any license or sublicense entered into by Parent or any Restricted Subsidiary as a licensee or sublicensee (A) existing on the Effective Date or (B) in the ordinary course of its business not materially interfering with the business of Parent and the Restricted Subsidiaries taken as a whole; (j) licenses, sublicenses, leases or subleases granted to other Persons permitted under Section 6.03 or otherwise existing on or prior to the Effective Date; (k) Liens on earnest money deposits of cash or cash equivalents made in connection with any Permitted Acquisition or other Investment permitted pursuant to Section 6.04; (l) Liens in the nature of the right of setoff in favor of counterparties to contractual agreements with Parent or any Restricted Subsidiaries in the ordinary course of business; (m) Liens arising out of conditional sale, title retention, consignment or similar arrangements for the sale of goods entered into by Parent or any Restricted Subsidiary in the ordinary course of business in accordance with the past practices of Parent or such Restricted Subsidiary; (n) Liens in favor of customs and revenue authorities arising as a matter of law to secure payment of customs duties in connection with the importation of goods in the ordinary course of business; (o) Liens on the assets and equity interests of Foreign Subsidiaries that are not Loan Parties provided that such Liens shall secure only Indebtedness or other obligations of such Foreign Subsidiaries permitted hereunder;

161 (p) Liens on insurance policies and the proceeds thereof securing Indebtedness permitted by Section 6.01(n); (q) Dispositions and other sales of assets permitted under Section 6.03; (r) Liens on deposits or other amounts held in escrow to secure contractual payments (contingent or otherwise) payable by Parent or its Restricted Subsidiaries to a seller after the consummation of a Permitted Acquisition; (s) Liens securing Indebtedness incurred under Section 6.01(q) and Section 6.01(r) and any Permitted Refinancing Indebtedness in respect thereof; provided that, such Liens provided under this Section 6.02(s) shall be subject to an Approved Intercreditor Agreement; (t) Liens on all or a portion of the Collateral securing Permitted Indebtedness; provided that (i) such Liens are junior to the Liens securing the Secured Obligations, (ii) such Indebtedness shall not be secured by any Lien on any asset of Parent or any of its Restricted Subsidiaries other than any asset constituting Collateral, (iii) the security agreements relating to such Indebtedness shall be substantially the same as the Collateral Documents (with such differences as are reasonably satisfactory to the Administrative Agent) and (iv) such Indebtedness shall be subject to an Approved Intercreditor Agreement; and Liens securing Permitted Refinancing Indebtedness in respect of the foregoing, in accordance with the definition of Permitted Refinancing Indebtedness contained herein; (u) Liens securing Permitted Pari Passu Secured Refinancing Debt, Permitted Junior Secured Refinancing Debt and Indebtedness incurred under Section 6.01(w), and any Permitted Refinancing Indebtedness in respect thereof; (v) Liens securing Permitted First Lien Indebtedness; provided that (i) such Indebtedness may be secured by all or a portion of the Collateral on a pari passu basis (except as otherwise provided in the Intercreditor Agreement) with the Secured Obligations, (ii) such Indebtedness shall not be secured by any Lien on any asset of Parent or any of its Restricted Subsidiaries other than any asset constituting Collateral, (iii) the security agreements relating to such Indebtedness shall be substantially the same as the Collateral Documents (with such differences as are reasonably satisfactory to the Administrative Agent) and (iv) such Indebtedness shall be subject to an Approved Intercreditor Agreement; and Liens securing Permitted Refinancing Indebtedness in respect of the foregoing, in accordance with the definition of Permitted Refinancing Indebtedness contained herein; (w) Liens on deposits or other amounts held in escrow to secure payments (contingent or otherwise) payable by Parent or any of its Restricted Subsidiaries with respect to settlements related to any litigation disclosed in public filings; (x) Liens on Permitted Receivables Facility Assets of Parent and its Restricted Subsidiaries arising under Permitted Receivables Facilities; (y) Liens on assets of Parent and its Restricted Subsidiaries not otherwise permitted above; provided that (i) the aggregate amount of obligations subject to any such Liens shall not immediately after giving effect to the incurrence of such obligations exceed

162 the greater of (x) $230,000,000 and (y) 30.0% of Consolidated EBITDA at the end of the Reference Period and (ii) to the extent such Lien is on all or a portion of the Collateral and securing Indebtedness for borrowed money, such Indebtedness shall be subject to an Approved Intercreditor Agreement for Indebtedness secured on a junior basis to the Secured Obligations; (z) Liens securing obligations in respect of Indebtedness or other obligations of a Restricted Subsidiary owing to Parent or another Restricted Subsidiary permitted to be incurred in accordance with Section 6.01; (aa) Liens on equipment or vehicles of Parent or any Restricted Subsidiary granted in the ordinary course of business or consistent with industry practice; (bb) receipt of progress payments and advances from customers in the ordinary course of business or consistent with industry practice to the extent the same creates a Lien on the related inventory and proceeds thereof and Liens on property or assets under construction arising from progress or partial payments by a third party relating to such property or assets; (cc) Liens on the proceeds of Escrow Debt and any interest thereof, securing the applicable Escrow Debt; and (dd) Liens securing the Senior Secured Notes and any Permitted Refinancing Indebtedness in respect thereof (subject to an Approved Intercreditor Agreement). For purposes of determining compliance with this Section 6.02, (A) a Lien need not be incurred solely by reference to one category described in this Section 6.02, but is permitted to be incurred in part under any combination thereof and of any other available exemption and (B) in the event that a Lien (or any portion thereof) meets the criteria of one or more of the categories of Liens permitted hereunder, Parent will, in its sole discretion, be entitled to divide, classify or reclassify, in whole or in part, any such Lien (or any portion thereof) among one or more of such categories or clauses in any manner. The expansion of Liens by virtue of accretion or amortization of original issue discount, the payment of dividends in the form of Indebtedness, and increases in the amount of Indebtedness outstanding solely as a result of fluctuations in the exchange rate of currencies will not be deemed to be an incurrence of Liens for purposes of this Section 6.02. Section 6.03 Fundamental Changes and Asset Sales. (a) Parent will not, and will not permit any Restricted Subsidiary to, merge into, amalgamate with or consolidate with any other Person, or permit any other Person to merge into, amalgamate with or consolidate with it, or sell, transfer, lease, Exclusively License or otherwise dispose of (in one transaction or in a series of transactions) any of its assets (including pursuant to a Sale and Leaseback Transaction), or any of the Equity Interests of any of its Subsidiaries (in each case, whether now owned or hereafter acquired), or liquidate, dissolve or wind-up, except that: (i) any Person (other than any Borrower) may merge into, amalgamate with or consolidate with Parent in a transaction in which Parent is the surviving corporation;

163 (ii) (x) any Person (other than Parent, any Borrower or any Intermediate Parent Entity) may merge into, amalgamate with or consolidate with any Restricted Subsidiary of Parent in a transaction in which the surviving entity is a Restricted Subsidiary, (y) any Person (including any Intermediate Parent Entity) may merge into, amalgamate with or consolidate with any other Intermediate Parent Entity in a transaction in which the surviving entity is an Intermediate Parent Entity and (z) any Borrower may merge into, amalgamate with or consolidate with Parent, any Intermediate Parent Entity or any other Restricted Subsidiary so long as such Borrower is the surviving entity or the surviving entity assumes all the obligations of such Borrower under this Agreement and the other Loan Documents and the successor Borrower is organized in (x) the same jurisdiction as such Borrower, (y) the same jurisdiction as a co-Borrower on the same Class of Loan or (z) a jurisdiction reasonably agreed to by the Administrative Agent and each materially and adversely affected Lender; (iii) any Restricted Subsidiary (other than any Borrower or any Intermediate Parent Entity) may merge into, amalgamate with or consolidate with any Person in a transaction permitted under clauses (xv), (xix) and (xx) hereunder in which the surviving entity is not a Subsidiary; (iv) (x) any Restricted Subsidiary (other than any Borrower or any Intermediate Parent Entity) may dispose of all or substantially all of its assets (upon voluntary liquidation, dissolution, winding-up or otherwise) to Parent or any other Restricted Subsidiary of Parent; provided that (i) the foregoing shall not permit the voluntary liquidation, dissolution of winding up of any Borrower and (ii) any such Disposition made by a Loan Party to a Restricted Subsidiary that is not a Loan Party shall be made in compliance with Section 6.04 and (y) any Intermediate Parent Entity may dispose of any or all of its assets (upon voluntary liquidation, dissolution, winding up or otherwise) to any other Intermediate Parent Entity or to Parent; (v) any Restricted Subsidiary (other than any Borrower) may liquidate, dissolve or wind-up if Parent determines in good faith that such liquidation or dissolution is in the best interests of Parent and is not materially disadvantageous to the Lenders; (vi) sales, transfers and other dispositions of inventory, used, worn out, obsolete or surplus property, cash and Permitted Investments in the ordinary course of business and the assignment, cancellation, abandonment or other disposition of Intellectual Property that is, in the reasonable judgment of Parent, no longer economically practicable to maintain or useful in the conduct of the business of Parent and the Restricted Subsidiaries, taken as a whole; (vii) Dispositions (or any license or sublicense of Intellectual Property) to Parent or any Restricted Subsidiary; provided that any such Disposition (or any license or sublicense of Intellectual Property) made by a Loan Party to a Restricted Subsidiary that is not a Loan Party shall be made in compliance with Section 6.04;

164 (viii) the discount or sale, in each case without recourse and in the ordinary course of business, of past due receivables arising in the ordinary course of business, but only in connection with the compromise or collection thereof consistent with customary industry practice (and not as part of any bulk sale or financing of receivables); (ix) leases, subleases, licenses or sublicenses of property to other Persons in the ordinary course of business, in each case, not materially interfering with the business of Parent and the Restricted Subsidiaries taken as a whole; (x) Liens incurred in compliance with Section 6.02; (xi) Investments permitted by Section 6.04; (xii) subject to Section 2.11(c)(1), dispositions of property as a result of a casualty event involving such property or any disposition of real property to a Governmental Authority as a result of a condemnation of such real property; (xiii) Permitted Exchanges; (xiv) Dispositions of investments in joint ventures, to the extent required by, or made pursuant to buy/sell arrangements between the joint venture parties set forth in joint venture arrangements and similar binding arrangements; provided that the consideration received shall be in an amount at least equal to the fair market value thereof (determined in good faith by the board of directors of Parent); (xv) sales or other Dispositions of non-core assets acquired in a Permitted Acquisition; provided that such sales shall be consummated within 360 days of such Permitted Acquisition; provided, further, that (i) the consideration received for such assets shall be in an amount at least equal to the fair market value thereof (determined in good faith by the board of directors of Parent) and (ii) no less than 75% of the consideration received for such assets shall be paid in cash or Permitted Investments (provided that, for purposes of satisfying the requirements of this clause (ii), Parent shall be permitted to designate, pursuant to a certificate executed by a Financial Officer of Parent and delivered to the Administrative Agent, non-cash consideration received for any such Disposition as cash consideration in an amount not to exceed $10,000,000 for each such Disposition); (xvi) any Immaterial Asset Sale; (xvii) any lease or sublease by Parent or any Restricted Subsidiary of a portion of its interest in its headquarters located in Malvern, Pennsylvania; (xviii) Parent or any Restricted Subsidiary may transfer, sell and/or pledge Receivables and Permitted Receivables Facility Assets under Permitted Receivables Facilities; (xix) Dispositions of assets that are not permitted by any other clause of this Section 6.03; provided that the Disposition Consideration of all assets sold, transferred, leased or otherwise disposed of, and of all assets Exclusively Licensed in

165 reliance on this clause (xix) shall not at the time of and immediately after giving effect to any such transaction exceed in any fiscal year the greater of (x) $275,000,000 and (y) 35.0% of Consolidated EBITDA at the end of the immediately preceding fiscal year of Parent; (xx) Dispositions of assets (but not Equity Interests in any Restricted Subsidiary unless such Restricted Subsidiary is not a Borrower (or a direct or indirect holding company thereof)) that are not permitted by any other clause of this Section 6.03; provided that (x) the consideration received for such assets shall be in an amount at least equal to the fair market value thereof (determined in good faith by the board of directors of Parent) and (y) no less than 75% of the consideration received for such assets shall be paid in cash or Permitted Investments (provided that, for purposes of satisfying the requirements of this clause (y), Parent shall be permitted to designate, pursuant to a certificate executed by a Financial Officer of Parent and delivered to the Administrative Agent, non-cash consideration received for any such Disposition as cash consideration in an amount not to exceed, in the aggregate for all such Dispositions, the greater of (1) $150,000,000 and (2) 20.0% of Consolidated EBITDA as of the end of the Reference Period); (xxi) the issuance of Equity Interests by a Restricted Subsidiary that represents all or a portion of the consideration paid by Parent or a Restricted Subsidiary in connection with any Investment permitted by Section 6.04, including in connection with the formation of a joint venture with a Person other than a Restricted Subsidiary; (xxii) Dispositions of Equity Interests (I) deemed to occur upon the exercise of stock options, warrants or other equity derivatives or settlement of convertible securities if such Equity Interests represent (i) a portion of the exercise price thereof or (ii) withholding incurred in connection with such exercise or (II) upon the exercise of any Permitted Warrant; (xxiii) Dispositions of the Equity Interests of, or the assets or securities of, Unrestricted Subsidiaries; and (xxiv) Dispositions of assets (including Equity Interests of any Subsidiary) that would constitute a Restricted Payment permitted by Section 6.07. (b) Parent will not, and will not permit any of its Restricted Subsidiaries to, engage to any material extent in any business other than businesses of the type conducted by Parent and its Restricted Subsidiaries on the Effective Date and businesses reasonably related thereto or similar or complementary thereto or reasonable extensions thereof (including, but not limited to the business of diagnostics, medical devices, delivery technologies and biotechnology). (c) Parent will not change its fiscal year from the basis applicable to Parent prior to the Effective Date; provided that Parent shall be permitted to change its fiscal year from the basis applicable to Parent prior to the Effective Date to the basis applicable to Mallinckrodt plc as in effect on the Third Amendment Effective Date.

166 Section 6.04 Investments, Loans, Advances, Guarantees and Acquisitions. Parent will not, and will not permit any Restricted Subsidiary to, (i) purchase, hold or acquire (including pursuant to any merger or consolidation with any Person that was not a wholly owned Restricted Subsidiary prior to such merger) any capital stock, evidence of indebtedness or other securities (including any option, warrant or other right to acquire any of the foregoing) of, make or permit to exist any loans or advances to, Guarantee any obligations of, or make or permit to exist any investment or any other interest in, any other Person, or (ii) purchase or otherwise acquire (in one transaction or a series of transactions) substantially all the assets of any Person or any assets of any other Person constituting a business unit, division, product line (including rights in respect of any drug or other pharmaceutical product) or line of business of such Person, or (iii) acquire an exclusive long-term license of rights to a drug or other product line of any Person (each, an “Investment”) except: (a) cash and Permitted Investments; (b) Permitted Acquisitions; (c) Investments by Parent and its Restricted Subsidiaries existing on the Effective Date and set forth on Schedule 6.04 and any modification, replacement, renewal or extension thereof to the extent not involving any additional Investment; (d) Investments made by Parent in or to any Restricted Subsidiary and made by any Restricted Subsidiary in or to Parent or any other Restricted Subsidiary and Guarantees by Parent or any Restricted Subsidiary of obligations of any other Restricted Subsidiary, provided that the amount of any Investment by a Loan Party to a Restricted Subsidiary that is not a Loan Party or constituting a Guarantee of obligations of any Restricted Subsidiary that is not a Loan Party shall not exceed, together with the aggregate amount of all other Investments pursuant to this proviso, the greater of (x) $300,000,000 and (y) 40.0% of Consolidated EBITDA as of the end of the Reference Period at any time outstanding; (e) Guarantees constituting Indebtedness permitted by Section 6.01; (f) Investments received in connection with the bankruptcy or reorganization of, or settlement of delinquent accounts and disputes with, customers and suppliers, in each case in the ordinary course of business; (g) Investments made as a result of the receipt of non-cash consideration from a Disposition, of any asset in compliance with Section 6.03; (h) Investments in the form of Swap Agreements permitted by Section 6.05; (i) payroll, travel and similar advances to directors, officers and employees of Parent or any Restricted Subsidiary that are made in the ordinary course of business; (j) extensions of trade credit in the ordinary course of business;

167 (k) Investments to the extent the consideration paid therefor consists of Equity Interests (other than Disqualified Equity Interests) of Parent; (l) Investments of any Person in existence at the time such Person becomes a Restricted Subsidiary; provided such Investment was not made in connection with or anticipation of such Person becoming a Restricted Subsidiary and any modification, replacement, renewal or extension thereof; (m) licenses, sublicenses or transfers of rights with respect to one or more products or technologies under development to joint ventures with third parties or to other entities where Parent or a Restricted Subsidiary retains rights to acquire such joint ventures or other entities or otherwise repurchase such products or technologies; (n) any customary upfront milestone, marketing or other funding payment in the ordinary course of business to another Person in connection with obtaining a right to receive royalty or other payments in the future; (o) [reserved]; (p) exclusive licenses from a Foreign Subsidiary to Parent or a Domestic Subsidiary of rights to a drug or other pharmaceutical products, diagnostics, delivery technologies, medical devices or biotechnology businesses acquired by such Foreign Subsidiary in an acquisition permitted by Section 6.03; (q) Investments in joint ventures and acquisitions of Equity Interests that would constitute Permitted Acquisitions but for the fact that Persons in which such Equity Interests are acquired do not become wholly owned Subsidiaries of Parent; provided that the sum of the aggregate amount of such Investments, plus the aggregate consideration paid in all such acquisitions, made under this clause (q) shall not exceed the greater of (x) $300,000,000 and (y) 40.0% of Consolidated EBITDA as of the end of the Reference Period, in each case, at any time outstanding. For purposes of this clause (q), the aggregate consideration payable for any Investment shall be the cash amount (and the fair market value of any non-cash consideration, as determined in good faith by Parent) paid on or prior to the consummation of such Investment and, except in the case of Milestone Payments, shall not include any purchase price adjustment, royalty, earnout, contingent payment or any other deferred payment of a similar nature that may be payable in connection therewith; (r) any Investment made by any Restricted Subsidiary that is not a Loan Party to the extent that such Investment is financed with the proceeds received by such Restricted Subsidiary from an Investment in such Restricted Subsidiary permitted under this Agreement; (s) [Reserved]; (t) Investments consisting of Liens made in accordance with Section 6.02; (u) loans or advances to directors and employees of Parent or any Restricted Subsidiary made in the ordinary course of business; provided that the aggregate

168 outstanding amount of such loans and advances, when aggregated with the Guarantees then outstanding under Section 6.01(l), at any time shall not exceed the greater of (x) $30,000,000 (y) 5.0% of Consolidated EBITDA as of the end of the Reference Period; (v) any Investment in an aggregate amount, when aggregated with the aggregate amount of Restricted Payments made pursuant to Section 6.07(g), not to exceed at any time the aggregate amount of net cash proceeds received by Parent from sales or issuances of Equity Interests of Parent (other than Disqualified Equity Interests) after the Effective Date; (w) (i) Investments made by any Restricted Subsidiary in or to any Unrestricted Subsidiary and (ii) any purchase or other acquisition by any Restricted Subsidiary of all or substantially all of the assets constituting a business unit, division, product line (including rights in respect of any drug or other pharmaceutical product) or line of business of any Unrestricted Subsidiary; provided that (x) any such Investment, purchase or other acquisition shall be made on terms and conditions (A) not materially less favorable to such Restricted Subsidiary than it would obtain on an arm’s-length basis from a Person that is not an Affiliate or (B) otherwise reasonably acceptable to the Administrative Agent, and (y) the aggregate fair market value of all such Investments, purchases and other acquisitions made pursuant to this clause (w), or the consideration payable in connection therewith, shall not exceed the greater of $275,000,000 and 35.0% of Consolidated EBITDA as of the end of the Reference Period; provided, further that any Investment in or to any Unrestricted Subsidiary shall only be permitted to be made pursuant to this clause (w); (x) Parent or any Restricted Subsidiary may make contributions of Permitted Receivables Facility Assets to any Receivables Seller, Receivables Entity or other Person in connection with a Permitted Receivables Facility; (y) any Investment made solely in exchange for the substantially contemporaneous issuance of Equity Interests (other than Disqualified Equity Interests) of Parent; (z) Investments in Restricted Subsidiaries in connection with reorganizations or other activities related to tax planning; provided that, after giving effect to any such reorganization or other activity related to tax planning, the security interest (including as to priority and value) of the Administrative Agent in the Collateral, taken as a whole, is not materially impaired; (aa) any other Investments so long as the aggregate amount of all such Investments does not exceed the greater of $380,000,000 and 50.0% of Consolidated EBITDA as of the end of the Reference Period at any time outstanding. For purposes of this clause (aa), the aggregate consideration payable for any Investment shall be the cash amount (and the fair market value of any non-cash consideration, as determined in good faith by Parent) paid on or prior to the consummation of such Investment and, except in the case of Milestone Payments, shall not include any purchase price adjustment, royalty, earnout, contingent payment or any other deferred payment of a similar nature that may be payable in connection therewith;

169 (bb) Investments made to fund the settlement of mesh device related claims, litigation, arbitration or other disputes and judgments, orders, fees and expenses related thereto; (cc) any other Investments in an amount not to exceed the Available Amount on such date; so long as subject to Section 1.04, no Event of Default described in Sections 7.01(a) or (b) or, solely with respect to the Borrower, Sections 7.01(h) or (i) has occurred and is continuing or would arise after giving effect (including pro forma effect) thereto; and (dd) any other Investments so long as on a pro forma basis after giving effect thereto (subject to Section 1.04) the Total Net Leverage Ratio is no greater than 3.25 to 1.00. For purposes of covenant compliance with this Section 6.04, (A) the amount of any Investment shall be the amount actually invested, without adjustment for subsequent increases or decreases in the value of such Investment, less any amount paid, repaid, returned, distributed or otherwise received in cash in respect of such Investment and (B) in the event that an Investment (or any portion thereof) meets the criteria of one or more of the categories of permitted Investments (or any portion thereof) described in Sections 6.04(a) through (dd), Parent may, in its sole discretion, classify or reclassify, or later divide, classify or reclassify (as if made at such later time), such Investment (or any portion thereof) in any manner that complies with this Section 6.04 and will be entitled to only include the amount and type of such Investment (or any portion thereof) in one or more (as relevant) of the above clauses (or any portion thereof) and such Investment (or any portion thereof) shall be treated as having been made or existing pursuant to only such clause or clauses (or any portion thereof); provided, that all Investments described in Section 6.04(b), Schedule 6.04 and Section 6.04(d) shall be deemed outstanding under Section 6.04(b), 6.04(c) and Section 6.04(d), respectively. Section 6.05 Swap Agreements. Parent will not, and will not permit any of its Restricted Subsidiaries to, enter into any Swap Agreement, except (a) Swap Agreements entered into to hedge or mitigate risks to which Parent or any Restricted Subsidiary has actual or anticipated exposure (other than those in respect of Equity Interests of Parent or any of its Restricted Subsidiaries but without giving effect to any other Indebtedness convertible into Equity Interests in Parent), (b) Swap Agreements entered into in order to effectively cap, collar or exchange interest rates (from fixed to floating rates, from one floating rate to another floating rate or otherwise) with respect to any interest-bearing liability or investment of Parent or any Restricted Subsidiary, (c) any Swap Agreement constituting part of a TEU and (d) Permitted Convertible Debt Hedge Transactions. Section 6.06 Transactions with Affiliates. Parent will not, and will not permit any of its Restricted Subsidiaries to, sell, lease or otherwise transfer any property or assets to, or purchase, lease or otherwise acquire any property or assets from, or otherwise engage in any other transactions with, any of its Affiliates (other than Parent or any Restricted Subsidiary) involving aggregate payments or consideration in excess of the greater of (x) $60,000,000 and (y) 7.50% of Consolidated EBITDA as of the end of the Reference Period, except (a) transactions that are on terms and conditions not materially less favorable to Parent or such Restricted Subsidiary than it would obtain on an arm’s-length basis from a Person that is not an Affiliate or, if in the good faith judgment of the board of directors of Parent no comparable transaction is available with which to compare such transaction, such transaction is otherwise fair to Parent or such Restricted Subsidiary

170 from a financial point of view, (b) any Restricted Payment permitted by Section 6.07, (c) customary fees and indemnifications paid to directors of Parent and its Restricted Subsidiaries, (d) transactions undertaken in good faith for the purpose of improving the consolidated tax efficiency of Parent and its Subsidiaries and not for the purpose of circumventing any covenant set forth in this Agreement, (e) compensation and indemnification of, and other employment agreements and arrangements, employee benefit plans, and stock incentive plans with directors, officers and employees of Parent or any Restricted Subsidiary entered in the ordinary course of business, (f) Intellectual Property licenses to Restricted Subsidiaries in existence on the Effective Date, (g) loans and advances and other transactions to the extent permitted by Sections 6.01 and 6.04, (h) leases or subleases of property in the ordinary course of business not materially interfering with the business of Parent and the Restricted Subsidiaries taken as a whole, (i) transactions between or among Parent and/or any Restricted Subsidiary and any entity that becomes a Restricted Subsidiary as a result of such transaction, (j) transactions permitted by Section 6.03(a)(xvii), (k) transactions in the ordinary course of business between or among Parent and/or any Restricted Subsidiary and any Unrestricted Subsidiary, (l) sales or issuances of Equity Interests of Parent to Affiliates of Parent which are otherwise permitted or not restricted by the Loan Documents; (m) any agreement between any Person and an Affiliate of such Person existing at the time such Person is acquired by or merged into such Parent or its Restricted Subsidiaries pursuant to the terms of this Agreement; provided that such agreement was not entered into in contemplation of such acquisition or merger, or any amendment thereto (so long as any such amendment is not disadvantageous to the Lenders in any material respect in the good faith judgment of Parent when taken as a whole as compared to such agreement as in effect on the date of such acquisition or merger), (n) any other transactions with an Affiliate, which is approved by a majority of disinterested members of the board of directors (or equivalent governing body) of Parent in good faith, (o) transactions, pursuant to or permitted by the Loan Documents or Senior Secured Notes Indenture, with Affiliated Lenders or Debt Fund Affiliates (in each case, in their respective capacities as Lenders or bondholders, as the case may be) and (p) the Transactions. Section 6.07 Restricted Payments. Parent will not, and will not permit any of its Restricted Subsidiaries to, declare or make, or agree to pay or make, directly or indirectly, any Restricted Payment, except: (a) Parent may declare and pay dividends or make other Restricted Payments with respect to its Equity Interests payable solely in additional Equity Interests of Parent (other than Disqualified Equity Interests); (b) Parent may repurchase its Equity Interests (i) upon the exercise of stock options, warrants or other equity derivatives or settlement of convertible securities if such Equity Interests represent a portion of the exercise price of such options, warrants or other equity derivatives or the settlement price of such convertible securities or (ii) upon the exercise of any Permitted Bond Hedge; (c) Parent may make cash payments in lieu of the issuance of fractional shares in connection with the exercise of warrants, options or other securities convertible into or exchangeable for Equity Interests in Parent;

171 (d) Restricted Subsidiaries may (x) make Restricted Payments ratably with respect to their Equity Interests; provided that any payments to other Restricted Subsidiaries or Persons may be made on a greater than ratable basis to the extent such payments would not be materially adverse to the Lenders and (y) make Restricted Payments to Parent and any other Restricted Subsidiaries; (e) Parent may make any dividend or other distribution (whether in cash, securities or other property) with respect to its Equity Interests or any payment (whether in cash, securities or other property), including any sinking fund or similar deposit, on account of the purchase, redemption, retirement, acquisition, cancellation or termination of any such Equity Interests in Parent or any option, warrant or other right to acquire any such Equity Interests in Parent pursuant to and in accordance with stock incentive plans or other employee benefit plans for directors, officers or employees of Parent and its Restricted Subsidiaries; (f) Parent may purchase Equity Interests from future, present or former employees, directors, officers, members of management, consultants or independent contractors (or their respective Affiliates or immediate family members or any permitted transferees thereof) of Parent or any Subsidiary upon the death, disability, retirement or termination of employment or service of such officer, director or employee, in an aggregate amount not exceeding the greater of (x) $25,000,000 and (y) 3.0% Consolidated EBITDA as of the end of the Reference Period in any fiscal year of Parent; provided that unused amounts in any fiscal year may be carried over to succeeding fiscal years; (g) Parent may make Restricted Payments in an aggregate amount not to exceed, when aggregated with the aggregate amount of Investments made pursuant to Section 6.04(v), the aggregate amount of net cash proceeds received from sales or issuances of Equity Interests of Parent (other than Disqualified Equity Interests) after the Effective Date; (h) repurchase of Equity Interests deemed to occur upon the non-cash exercise of Equity Interests to pay taxes; (i) the payment of any dividend or distribution, or the consummation of any irrevocable redemption, within 60 days after the date of declaration of the dividend or distribution or giving of the redemption notice, as the case may be, if at such date of declaration or redemption notice such dividend, distribution or redemption, as the case may be, would have complied with this Section 6.07; (j) Parent and its Restricted Subsidiaries may make any other Restricted Payment after the Effective Date in an amount not to exceed the Available Amount on such date, so long as subject to Section 1.04, no Event of Default described in Sections 7.01(a) or (b) or, solely with respect to the Borrower, Sections 7.01(h) or (i) has occurred and is continuing or would arise after giving effect (including pro forma effect) thereto; (k) Restricted Payments made (A) in connection with (including, without limitation, purchases of) any Permitted Convertible Debt Hedge Transaction (B) to settle any Permitted Warrant (i) by delivery of common stock of Parent or any of its direct or indirect parent companies, (ii) by set-off against the related Permitted Bond Hedge or (iii) with cash

172 payments in an aggregate amount not to exceed the aggregate amount of any payments received pursuant to the settlement of any related Permitted Bond Hedge (subject to any increase in the price of the underlying common stock since the settlement of such Permitted Bond Hedge) or (C) to terminate any Permitted Warrant; (l) any other Restricted Payments so long as (i) after giving pro forma effect thereto (including pro forma effect in accordance with Section 1.04), the Total Net Leverage Ratio shall be no greater than 3.00 to 1.00 and (ii) subject to Section 1.04, no Event of Default described in Sections 7.01(a) or (b) or, solely with respect to the Borrower, Sections 7.01(h) or (i) has occurred and is continuing or would arise after giving effect (including pro forma effect) thereto; (m) any other Restricted Payments in an aggregate amount not to exceed the greater of $230,000,000 and 30.0% of Consolidated EBITDA as of the end of the Reference Period; and (n) any Restricted Payments made with the proceeds of amounts received by Parent, the Borrower Representative or any of their respective Restricted Subsidiaries from the administrator of the Reorganization Plan. Section 6.08 Restrictive Agreements. Parent will not, and will not permit any of its Restricted Subsidiaries to, directly or indirectly, enter into, incur or permit to exist any agreement or other arrangement that prohibits, restricts or imposes any condition upon (a) the ability of Parent or any Loan Party to create, incur or permit to exist any Lien upon any of its property or assets to the extent such Lien is required to be granted in favor of the Secured Parties pursuant to the Loan Documents or (b) the ability of any Restricted Subsidiary to pay dividends or other distributions to Parent or any Restricted Subsidiary or to make or repay loans or advances to Parent or any other Restricted Subsidiary or to Guarantee the Obligations; provided that (i) the foregoing limitations in clauses (a) and (b) shall not apply to (A) restrictions and conditions imposed any law, by any Loan Document, any Permitted Receivables Facility Documents or any Swap Agreements to the extent permitted by Section 6.05, (B) restrictions and conditions existing on the Effective Date identified on Schedule 6.08, (C) restrictions and conditions imposed by agreements relating to Indebtedness of any Restricted Subsidiary in existence at the time such Restricted Subsidiary became a Restricted Subsidiary and any amendments or modifications thereof that do not materially expand the scope of any such restriction or condition taken as a whole; provided that such restrictions and conditions apply only to such Restricted Subsidiary, (D) any agreement or other instrument of a Person, or relating to Indebtedness or Equity Interests of a Person, acquired by or merged, amalgamated or consolidated with and into Parent or any Restricted Subsidiary or an Unrestricted Subsidiary that is designated as a Restricted Subsidiary, or any other transaction entered into in connection with any such acquisition, merger, consolidation or amalgamation in existence at the time of such acquisition or at the time it merges, amalgamates or consolidates with or into Parent or any Restricted Subsidiary or an Unrestricted Subsidiary that is designated as a Restricted Subsidiary or assumed in connection with the acquisition of assets from such Person (but, in any such case, not created in contemplation thereof), which encumbrance or restriction is not applicable to any Person, or the properties or assets of any Person, other than the Person so acquired and its Subsidiaries, or the property or assets of the Person so acquired or designated and its Subsidiaries or the property or assets so acquired or designated; (E) customary restrictions and

173 conditions contained in agreements relating to the sale of a Subsidiary pending such sale to the extent such sale is permitted hereunder, (F) any restriction arising under or in connection with any agreement or instrument of any joint venture (including with respect to Equity Interests therein), (G) customary restrictions and conditions contained in any agreement relating to the Disposition of any property permitted by Section 6.03 pending the consummation of such Disposition, (H) restrictions or conditions upon the transfers of assets encumbered by a Lien permitted by Section 6.02, (I) restrictions or conditions set forth in the Senior Secured Notes (including, in each case, the indentures and other agreements and documents related thereto), (J) customary restrictions or conditions set forth in any agreement governing Indebtedness permitted by Section 6.01; provided that such restrictions or conditions are no more restrictive, taken as a whole, than the comparable restrictions and conditions set forth in this Agreement as determined in the good faith judgment of Parent, (K) customary restrictions or provisions restricting assignments of any agreement, (L) restrictions on cash or other deposits (including escrowed funds) or net worth imposed under contracts entered into in the ordinary course of business or consistent with industry practice, (M) restrictions or conditions contained in any trading, netting, operating, construction, service, supply, purchase, sale or other agreement to which Parent or any Restricted Subsidiary is a party entered into in the ordinary course of business or consistent with industry practice; provided that such agreement prohibits the encumbrance of solely the property or assets of Parent or such Restricted Subsidiary that are subject to such agreement; (N) restrictions or conditions imposed by any amendments, modifications, restatements, renewals, increases, supplements, refundings, replacements or refinancings of the contracts, instruments or obligations referred to in clauses (A) through (N) of this Section 6.08; provided that such amendments or refinancings do not materially expand the scope of any such restriction or condition; and (ii) clause (a) of the foregoing shall not apply to (1) restrictions or conditions imposed by any agreement relating to secured Indebtedness permitted by this Agreement if such restrictions or conditions apply only to the property or assets securing such Indebtedness, (2) customary provisions in leases, subleases, licenses, sublicenses and other agreements entered into in the ordinary course of business and (3) customary provisions in purchase money obligations and capital lease obligations on the property acquired pursuant thereto. Section 6.09 Amendments to Subordinated Indebtedness. Parent will not, and will not permit any Restricted Subsidiary to, amend, modify or waive any of its rights under any agreement or instrument governing or evidencing any Subordinated Indebtedness to the extent such amendment, modification or waiver could reasonably be expected to be adverse in any material respect to the Lenders unless the respective amendment, modification or waiver is reasonably satisfactory to the Administrative Agent. Section 6.10 Sale and Leaseback Transactions. Neither Parent nor any Restricted Subsidiary will enter into any Sale and Leaseback Transaction unless (a) the sale or transfer of the property thereunder is permitted by Section 6.03, (b) any Capital Lease Obligations arising in connection therewith are permitted by Section 6.01 and (c) any Liens arising in connection therewith (including Liens deemed to arise in connection with any such Capital Lease Obligations) are permitted by Section 6.02. Section 6.11 Financial Covenant.

174 Parent covenants and agrees that it will not permit the First Lien Net Leverage Ratio to exceed 6.10 to 1.00 on any Compliance Date. The provisions of this Section 6.11 are for the benefit of the Revolving Lenders only and the Lenders constituting the Required Revolving Lenders only may amend, waive or otherwise modify this Section 6.11 or the defined terms or calculation provisions used in or with respect to any determination under this Section 6.11 (solely in respect of the use of such defined terms in or in respect of any determination under this Section 6.11) or waive any Default or Event of Default resulting from a breach of this Section 6.11 without the consent of any Lenders other than the Required Revolving Lenders. ARTICLE VII EVENTS OF DEFAULT Section 7.01 Events of Default. If any of the following events (“Events of Default”) shall occur: (a) a Borrower shall fail to pay any principal of any Loan or any reimbursement obligation in respect of any LC Disbursement when and as the same shall become due and payable, whether at the due date thereof or at a date fixed for prepayment thereof or otherwise; (b) a Borrower shall fail to pay any interest on any Loan or any fee or any other amount (other than an amount referred to in clause (a) of this Section 7.01) payable under this Agreement or any other Loan Document, when and as the same shall become due and payable, and such failure shall continue unremedied for a period of five (5) Business Days; (c) any representation or warranty made or deemed made by or on behalf of Parent or any other Loan Party in or in connection with this Agreement or any other Loan Document or any amendment or modification hereof or thereof or waiver hereunder or thereunder, or in any report, certificate, financial statement or other document furnished pursuant to or in connection with this Agreement or any other Loan Document or any amendment or modification thereof or waiver thereunder, shall prove to have been incorrect in any material respect when made or deemed made; (d) Parent or any other Loan Party shall fail to observe or perform any covenant, condition or agreement contained in Section 5.02, 5.03 (with respect to the existence of Parent or a Borrower), 5.08, 5.09 or in Article VI; provided that Parent’s failure to comply with the Financial Covenant (a “Financial Covenant Event of Default”) shall not constitute an Event of Default with respect to any Term Loans or Term Loan Commitments unless and until the Required Revolving Lenders have actually terminated the Revolving Commitments and/or declared all Obligations with respect thereto to be immediately due and payable pursuant to this Section 7.01 as a result of such failure to comply (and such declaration has not been rescinded as of the applicable date); provided further that any Financial Covenant Event of Default is subject to cure pursuant to Section 7.02;

175 (e) Parent or any other Loan Party, as applicable, shall fail to observe or perform any covenant, condition or agreement contained in this Agreement (other than those specified in clause (a), (b) or (d) of this Section 7.01) or any other Loan Document, and such failure shall continue unremedied for a period of thirty (30) days after notice thereof from the Administrative Agent to Parent; (f) Parent or any Restricted Subsidiary shall fail to make any payment (whether of principal or interest and regardless of amount) in respect of any Material Indebtedness, when and as the same shall become due and payable after the expiration of any applicable grace period; (g) any event or condition occurs that results in any Material Indebtedness becoming due prior to its scheduled maturity or that enables or permits, after the expiration of any applicable grace period provided in the applicable agreement or instrument under which such Indebtedness was created, the holder or holders of any Material Indebtedness or any trustee or agent on its or their behalf to cause any Material Indebtedness to become due, or to require the prepayment, repurchase, redemption or defeasance thereof, prior to its scheduled maturity; provided that this clause (g) shall not apply to (i) secured Indebtedness that becomes due as a result of the voluntary sale or transfer of the property or assets securing such Indebtedness, (ii) any redemption, repurchase, conversion or settlement with respect to any Convertible Debt Security pursuant to its terms unless such redemption, repurchase, conversion or settlement results from a default thereunder or an event of the type that constitutes an Event of Default, or (iii) any early payment requirement or unwinding or termination with respect to any Swap Agreement. (h) an involuntary case or application or proceeding shall be commenced or an involuntary petition shall be filed seeking (i) liquidation, reorganization, winding-up, dissolution, compromise, arrangement or other relief in respect of Parent or any Material Subsidiary or its debts, or of a substantial part of its assets, under any Federal, state or foreign bankruptcy, insolvency, receivership, examinership or similar law now or hereafter in effect or (ii) the appointment of a receiver, receiver and manager, trustee, custodian, sequestrator, conservator, examiner, liquidator or similar official for Parent or any Material Subsidiary or for a substantial part of its assets, and, in any such case, such case or application or proceeding or petition shall continue undismissed for sixty (60) days or an order or decree approving or ordering any of the foregoing shall be entered; (i) Parent or any Material Subsidiary shall (i) voluntarily commence any proceeding or file any petition seeking liquidation, reorganization winding-up, dissolution, compromise, arrangement or other relief under any Federal, state or foreign bankruptcy, insolvency, receivership or similar law now or hereafter in effect (except in a transaction expressly permitted by Section 6.03), (ii) consent to the institution of, or fail to contest in a timely and appropriate manner, any proceeding or petition described in clause (h) of this Section 7.01, (iii) apply for or consent to the appointment of a receiver, receiver and manager, trustee, custodian, sequestrator, conservator or similar official for, Parent or any Material Subsidiary or for a substantial part of its assets, (iv) file an answer admitting the material allegations of a petition filed against it in any such proceeding, (v) make a general assignment for the benefit of creditors or (vi) take any action for the purpose of effecting any of the foregoing;

176 (j) Parent or any Material Subsidiary shall become unable, admit in writing its inability or fail generally to pay its debts as they become due; (k) one or more judgments for the payment of money in an aggregate amount in excess of $150,000,000 (or the equivalent amount in any other currency) shall be rendered by a court of competent jurisdiction against Parent or any Restricted Subsidiary or any combination thereof and the same shall remain unpaid or undischarged for a period of sixty (60) consecutive days after such judgment has become final and non-appealable and during which execution shall not be effectively stayed, or any action shall be legally taken by a judgment creditor to attach or levy upon any assets of, Parent or any Restricted Subsidiary to enforce any such judgment; provided that any such amount shall be calculated after deducting from the sum so payable any amount of such judgment or order that is covered by a valid and binding policy of insurance in favor of, Parent or such Restricted Subsidiary (but only if the applicable insurer shall have been advised of such judgment and of the intent of Parent or such Restricted Subsidiary to make a claim in respect of any amount payable by it in connection therewith and such insurer shall not have disputed coverage); (l) an ERISA Event shall have occurred that when taken together with all other ERISA Events that have occurred, could reasonably be expected to result in a Material Adverse Effect; (m) (i) a contribution required to be made with respect to a Non-U.S. Plan has not been timely made, or Parent or any Restricted Subsidiary has incurred liabilities pursuant to one or more Non-U.S. Plans; or that Parent or any Restricted Subsidiary has incurred any obligation in connection with the termination of, or withdrawal from, any Non-U.S. Plan; (ii) there shall result from any such event or events the imposition of a lien, the granting of a security interest, or a liability or a material risk of incurring a liability; and (iii) with respect to clauses (i) and (ii) above, such lien, security interest, contribution failure or liability, individually, or in the aggregate, has had, or could reasonably be expected to have, a Material Adverse Effect; (n) a Change in Control shall occur; (o) any material provision of any Loan Document for any reason ceases to be valid, binding and enforceable in accordance with its terms (or Parent or any other Loan Party shall challenge the enforceability of any Loan Document or shall assert in writing, or engage in any action or inaction based on any such assertion, that any provision of any of the Loan Documents has ceased to be or otherwise is not valid, binding and enforceable in accordance with its terms); or (p) any Collateral Document shall for any reason fail to create a valid and perfected first priority security interest in any material portion of the Collateral purported to be covered thereby, except as permitted by the terms of any Loan Document or the Agreed Security Principles, or as a result of the gross negligence or willful misconduct of the Administrative Agent so long as not resulting from the breach or non-compliance with any Loan Document by any Loan Party;

177 then, and in every such event (other than an event with respect to Parent or any Borrower described in clause (h) or (i) of this Section 7.01), and at any time thereafter during the continuance of such event, the Administrative Agent may, and (x) with respect to clause (i) below, at the request of the Required Revolving Lenders, shall, (y) at any time any of the Priority Revolving Credit Obligations remains outstanding with respect to clause (ii) below, at the request of the Required Revolving Lenders, shall, and (z) at any time on and after the Discharge of the Priority Revolving Credit Obligations with respect to clause (ii) below, at the request of the Required Lenders, shall, by notice to the Borrower, take either or both of the following actions, at the same or different times: (i) terminate the Revolving Commitments of such Class, and thereupon such Revolving Commitments shall terminate immediately, and (ii) declare the Loans then outstanding to be due and payable in whole (or in part, in which case any principal not so declared to be due and payable may thereafter be declared to be due and payable), and thereupon the principal of the Loans so declared to be due and payable, together with accrued interest thereon and all fees and other Secured Obligations accrued hereunder and under the other Loan Documents, shall become due and payable immediately, without presentment, demand, protest or other notice of any kind, all of which are hereby waived by the Borrower; and in case of any event with respect to Parent or any Borrower described in clause (h) or (i) of this Section 7.01, the Revolving Commitments shall automatically terminate and the principal of the Loans then outstanding, together with accrued interest thereon and all fees and other Secured Obligations accrued hereunder and under the other Loan Documents, shall automatically become due and payable, without presentment, demand, protest or other notice of any kind, all of which are hereby waived by the Borrower. Upon the occurrence and during the continuance of an Event of Default, the Administrative Agent may, and the Administrative Agent shall, at the request of (i) any time any of the Priority Revolving Credit Obligations remains outstanding, the Required Revolving Lenders and (ii) any time after the Discharge of the Priority Revolving Credit Obligations, the Required Lenders, exercise any rights and remedies provided to the Administrative Agent under the Loan Documents or at law or equity, including all remedies provided under the UCC, in each case, subject to and in accordance with the Intercreditor Agreement. Notwithstanding anything to the contrary contained herein, any “Default” under this Section 7.01 will not constitute an “Event of Default” until the Loan Parties do not cure such “Default” within the time period (if any) specified in the applicable clauses of this Section 7.01 after receipt of any required notice (if any) provided for therein to the extent such clauses of Section 7.01 provide for such cure periods or required notice; provided that, the Administrative Agent shall not be entitled to provide such notice, take any enforcement action and/or seek remedies in respect of a Default under this Section 7.01 for actions taken and reported by the Borrower to the Administrative Agent and the Lenders pursuant to a notice of Default provided by the Borrower to the Administrative Agent as of the date that is two years after delivery of such notice of Default and no Default or Event of Default can occur as a result thereof; provided further that, such two year limitation shall not apply if (i) the Administrative Agent has commenced (or been directed to commence) any remedial action in respect of any such Event of Default or has been stayed from so commencing by operation law or (ii) any Loan Party had actual knowledge of such Default or Event of Default and failed to notify to Administrative Agent as required hereby. Section 7.02 Right to Cure.

178 (a) Notwithstanding anything to the contrary contained in Section 7.01, but subject to Sections 7.02(b) and (c), for the purpose of determining whether an Event of Default under the Financial Covenant has occurred, Parent may on one or more occasions designate any portion of the Net Proceeds from any sale or issuance of any Equity Interests (other than Disqualified Equity Interests) of Parent (or from any other contribution to capital or sale or issuance of any other Equity Interests on terms reasonably satisfactory to the Administrative Agent) (the “Cure Amount”) as an increase to Consolidated EBITDA of Parent for the applicable fiscal quarter; provided that (i) such amounts to be designated are actually received by Parent (i) on and after the first Business Day of the applicable fiscal quarter and (ii) on and prior to the tenth (10th) Business Day after the date on which financial statements are required to be delivered with respect to such applicable fiscal quarter (the “Cure Expiration Date”), (ii) such amounts to be designated do not exceed the maximum aggregate amount necessary to cure any Event of Default under the Financial Covenant as of such date and (iii) Parent will have provided notice to the Administrative Agent on the date such amounts are designated as a “Cure Amount” (it being understood that to the extent such notice is provided in advance of delivery of a Compliance Certificate for the applicable period, the amount of such Net Proceeds that is designated as the Cure Amount may be lower than specified in such notice to the extent that the amount necessary to cure any Event of Default under the Financial Covenant is less than the full amount of such originally designated amount). The Cure Amount used to calculate Consolidated EBITDA for any fiscal quarter will be used and included when calculating Consolidated EBITDA for each Reference Period that includes such fiscal quarter. The parties hereby acknowledge that this Section 7.02(a) may not be relied on for purposes of calculating any financial ratios other than as applicable to the Financial Covenant (and may not be included for purposes of determining pricing, mandatory prepayments and the availability or amount permitted pursuant to any covenant under Article VI) and may not result in any adjustment to any amounts (including the amount of Indebtedness) or increase in cash with respect to the fiscal quarter with respect to which such Cure Amount was received other than the amount of the Consolidated EBITDA referred to in the immediately preceding sentence (but for the avoidance of doubt may be applied to prepay Indebtedness in a subsequent fiscal quarter). Notwithstanding anything to the contrary contained in Section 7.01, (A) upon designation of the Cure Amount by Parent in an amount necessary to cure any Event of Default under the Financial Covenant, the Financial Covenant will be deemed satisfied and complied with as of the end of the relevant fiscal quarter with the same effect as though there had been no failure to comply with the Financial Covenant and no Event of Default under the Financial Covenant (and any other Default as a result thereof) will be deemed to have occurred for purposes of the Loan Documents, (B) from and after the date that Parent delivers a written notices to the Administrative Agent that it intends to exercise its cure right under this Section 7.02 neither the Administrative Agent nor any Lender may exercise any rights or remedies under Section 7.01 (or under any other Loan Document) on the basis of any actual or purported Event of Default under the Financial Covenant (and any other

179 Default as a result thereof) until and unless the Cure Expiration Date has occurred without the Cure Amount having been designated and (C) the Loan Parties shall not be able to obtain any Borrowing hereunder until receipt by the Administrative Agent of the notice described in Section 7.02(a)(iii) from Parent. (b) In each period of four consecutive fiscal quarters, there shall be no more than two (2) fiscal quarters in which the cure right set forth in Section 7.02(a) is exercised. (c) There shall be no more than five (5) fiscal quarters in which the cure rights set forth in Section 7.02(a) are exercised during the term of this Agreement; provided that, so long as the Revolving Commitments incurred on the Effective Date have matured or been terminated, there may be an additional fiscal quarter after the Maturity Date applicable to such Revolving Commitments in which the cure rights set forth in this Section 7.02 are exercised during the term of any other Revolving Commitments. ARTICLE VIII The Administrative Agent and the Collateral Agent Each of the Lenders and the Issuing Bank hereby irrevocably appoints Goldman Sachs Bank USA as its administrative agent and authorizes Goldman Sachs Bank USA to take such actions on its behalf, including execution of the other Loan Documents, and to exercise such powers as are delegated to the Administrative Agent by the terms of the Loan Documents, together with such actions and powers as are reasonably incidental thereto, and Goldman Sachs Bank USA hereby accepts such appointment. The banks serving as the Administrative Agent hereunder shall have the same rights and powers in its capacity as a Lender as any other Lender and may exercise the same as though it were not the Administrative Agent, and such banks and their Affiliates may accept deposits from, lend money to and generally engage in any kind of business with Parent or any Subsidiary or other Affiliate thereof as if they were not an Agent hereunder. The Administrative Agent shall not have any duties or obligations except those expressly set forth in the Loan Documents. Without limiting the generality of the foregoing, (a) the Administrative Agent shall not be subject to any fiduciary or other implied duties, regardless of whether a Default has occurred and is continuing, (b) the Administrative Agent shall not have any duty to take any discretionary action or exercise any discretionary powers, except discretionary rights and powers expressly contemplated by the Loan Documents that the Administrative Agent is required to exercise in writing as directed by the Required Lenders (or such other number or percentage of the Lenders as shall be necessary under the circumstances as provided in Section 9.02), and (c) except as expressly set forth in the Loan Documents, the Administrative Agent shall not have any duty to disclose, and shall not be liable for the failure to disclose, any information relating to Parent or any of its Subsidiaries that is communicated to or obtained by any bank serving as the Administrative Agent or any of its Affiliates in any capacity. The Administrative Agent shall not be liable for any action taken or not taken by it with the consent or at the request of the Required Lenders (or such other number or percentage of the Lenders as shall be necessary under the circumstances as provided in Section 9.02 or elsewhere in the Loan

180 Documents, including without limitation the Required Revolving Lenders pursuant hereto and pursuant to the Intercreditor Agreement) or in the absence of its own gross negligence or willful misconduct. The Administrative Agent shall be deemed not to have knowledge of any Default unless and until written notice thereof is given to the Administrative Agent by Parent or a Lender, and no Administrative Agent shall be responsible for or have any duty to ascertain or inquire into (i) any statement, warranty or representation made in or in connection with any Loan Document, (ii) the contents of any certificate, report or other document delivered hereunder or in connection with any Loan Document, (iii) the performance or observance of any of the covenants, agreements or other terms or conditions set forth in any Loan Document, (iv) the validity, enforceability, effectiveness or genuineness of any Loan Document or any other agreement, instrument or document, (v) the creation, perfection or priority of Liens on the Collateral or the existence of the Collateral or (vi) the satisfaction of any condition set forth in Article IV or elsewhere in any Loan Document, other than to confirm receipt of items expressly required to be delivered to such Agent. The Administrative Agent shall be entitled to rely upon, and shall not incur any liability for relying upon, any notice, request, certificate, consent, statement, instrument, document or other writing believed by it to be genuine and to have been signed or sent by the proper Person. The Administrative Agent also may rely upon any statement made to it orally or by telephone and believed by it to be made by the proper Person, and shall not incur any liability for relying thereon. The Administrative Agent may consult with legal counsel (who may be counsel for Parent or the Borrower), independent accountants and other experts selected by it, and shall not be liable for any action taken or not taken by it in accordance with the advice of any such counsel, accountants or experts. The Administrative Agent may perform any and all its duties and exercise their rights and powers by or through any one or more sub-agents appointed by the respective Agent. The Administrative Agent and any such sub-agent may perform any and all its duties and exercise its rights and powers through their respective Related Parties. The exculpatory provisions of the preceding paragraphs shall apply to any such sub-agent and to the Related Parties of the Administrative Agent and any such sub-agent, and shall apply to their respective activities in connection with the syndication of the credit facilities provided for herein as well as activities as an Agent. Subject to the appointment and acceptance of a successor Administrative Agent as provided in this paragraph, the Administrative Agent may resign at any time by notifying the Lenders, the Issuing Bank and the Borrower. Any such resignation by the Administrative Agent hereunder shall also constitute its resignation as an Issuing Bank and the Swingline Lender, as applicable, in which case the resigning Administrative Agent (x) shall not be required to issue any further Letters of Credit or make any additional Swingline Loans hereunder and (y) shall maintain all of its rights as Issuing Bank or Swingline Lender, as the case may be, with respect to any Letters of Credit issued by it, or Swingline Loans made by it, prior to the date of such resignation. Upon any such resignation, the Required Lenders and the Required Revolving Lenders shall have the right (with the consent of the Borrower (such consent not to be unreasonably withheld or delayed), provided that no consent of the Borrower shall be required if an Event of Default under clause (a), (b), (h), (i) or (j) of Section 7.01 has occurred and is continuing) to appoint a successor. If no successor shall have been so appointed by the Required Lenders and the Required Revolving Lenders and shall have accepted such appointment within thirty (30) days after the retiring

181 Administrative Agent gives notice of its resignation, then the retiring Administrative Agent may, on behalf of the Lenders and the Issuing Bank, appoint a successor Administrative Agent which shall be a bank with an office in New York, New York, or an Affiliate of any such bank. Upon the acceptance of its appointment as Administrative Agent hereunder by a successor, such successor shall succeed to and become vested with all the rights, powers, privileges and duties of the retiring Administrative Agent, and the retiring Administrative Agent shall be discharged from its duties and obligations hereunder. The fees payable by the Borrower to a successor Administrative Agent shall be the same as those payable to its predecessor unless otherwise agreed between the Borrower and such successor. After the Administrative Agent’s resignation hereunder, the provisions of this Article VIII and Section 9.03 shall continue in effect for the benefit of such retiring Administrative Agent, its sub-agents and their respective Related Parties in respect of any actions taken or omitted to be taken by any of them while it was acting as Administrative Agent. Each Lender acknowledges that it has, independently and without reliance upon the Administrative Agent or any other Lender and based on such documents and information as it has deemed appropriate, made its own credit analysis and decision to enter into this Agreement. Each Lender also acknowledges that it will, independently and without reliance upon the Administrative Agent or any other Lender and based on such documents and information as it shall from time to time deem appropriate, continue to make its own decisions in taking or not taking action under or based upon this Agreement, any other Loan Document or any related agreement or any document furnished hereunder or thereunder. None of the Lenders, if any, identified in this Agreement as a Lead Arranger shall have any right, power, obligation, liability, responsibility or duty under this Agreement other than those applicable to all Lenders as such. Without limiting the foregoing, none of such Lenders shall have or be deemed to have a fiduciary relationship with any Lender. Each Lender hereby makes the same acknowledgments with respect to the relevant Lenders in their capacity as a Lead Arranger as it makes with respect to the Administrative Agent in the preceding paragraph. The Lenders are not partners or co-venturers, and no Lender shall be liable for the acts or omissions of, or (except as otherwise set forth herein in case of the Administrative Agent) authorized to act for, any other Lender. The Administrative Agent shall have the exclusive right on behalf of the Lenders to enforce the payment of the principal of and interest on any Loan after the date such principal or interest has become due and payable pursuant to the terms of this Agreement. Each of the Administrative Agent, Lenders and the Issuing Bank hereby irrevocably appoints Goldman Sachs Bank USA as its collateral agent and authorizes Goldman Sachs Bank USA to take such actions on its behalf, including execution of the Collateral Documents, and to exercise such powers as are delegated to the Collateral Agent by the terms of the Loan Documents, together with such actions and powers as are reasonably incidental thereto, and Goldman Sachs Bank USA hereby accepts such appointment. In its capacity, the Collateral Agent is “representative” of the Secured Parties within the meaning of the term “secured party” as defined in the New York UCC. Each Lender authorizes the Collateral Agent to (i) enter into each of the Collateral Documents to which it is a party and to

182 take all action contemplated by such documents, (ii) act as collateral agent for each Secured Party for purposes of the acquiring, perfection, holding and enforcing of all Liens created by such agreements (together with such powers and discretion as are reasonably incidental thereto) and all other purposes stated therein and be irrevocably authorized to enter into the Loan Documents in its capacity as Collateral Agent and to take the action and to exercise the rights that are expressly or by implication delegated to it by a Loan Document and any other action or rights that are reasonably incidental thereto, (iii) take such other action as is necessary or desirable to maintain the perfection and priority of the Liens created or purported to be created by the Loan Documents, (iv) except as may be otherwise specified in any Loan Document, exercise all remedies given to the Collateral Agent and the other Secured Parties with respect to the Collateral, whether under the Loan Documents, applicable law or otherwise. Each Lender agrees that no Secured Party (other than the Collateral Agent) shall have the right individually to seek to realize upon the security granted by any Collateral Document, it being understood and agreed that such rights and remedies may be exercised solely by the Collateral Agent for the benefit of the Secured Parties upon the terms of the Collateral Documents. In the event that any Collateral is hereafter pledged by any Person as collateral security for the Secured Obligations, the Collateral Agent is hereby authorized, and hereby granted a power of attorney, to execute and deliver on behalf of the Secured Parties any Loan Documents necessary or appropriate to grant and perfect a Lien on such Collateral in favor of the Collateral Agent on behalf of the Secured Parties. The Lenders hereby authorize the Collateral Agent to release any Lien granted to or held by the Collateral Agent upon any Collateral as described in Section 9.13 or the US Security Agreement and the Administrative Agent is hereby authorized to provide confirmation of such authorization if requested by the Collateral Agent. The Administrative Agent and the Collateral Agent is hereby authorized to enter into the Intercreditor Agreement and any Approved Intercreditor Agreement (and any amendments, amendments and restatements, restatements or waivers of or supplements to or other modifications to, and extensions, restructuring, renewals, replacements of, such agreements) in connection with the incurrence by any Loan Party of any Permitted First Lien Indebtedness, Permitted Junior Secured Refinancing Debt, Permitted Pari Passu Secured Refinancing Debt or Permitted Refinancing Indebtedness with respect thereto, or any other Indebtedness permitted by the terms of this Agreement to be secured by the Collateral on a pari passu or junior priority secured basis, in each case in order to permit such Indebtedness to be secured by a valid, perfected Lien (with such priority as may be designated by such Loan Party to the extent such priority is permitted by the Loan Documents), and the parties hereto acknowledge that the Intercreditor Agreement and each Approved Intercreditor Agreement is (if entered into) binding upon them. Each Lender (a) understands, acknowledges and agrees that Liens may be created on the Collateral pursuant to the documentation relating to any Indebtedness incurred as permitted by this Agreement which is (in accordance with the terms hereof) to be secured thereby, on a pari passu, priority or junior, secured basis to the Liens securing the Secured Obligations, which Liens securing any such other Indebtedness shall be subject to the terms and conditions of the Intercreditor Agreement and each Approved Intercreditor Agreement executed and delivered as required hereby, (b) hereby agrees that it will be bound by and will take no actions contrary to the provisions of the Intercreditor Agreement and any Approved Intercreditor Agreement (if entered into) and (c) hereby authorizes and instructs the Administrative Agent and the Collateral Agent to enter into the Intercreditor Agreement and each Approved Intercreditor Agreement (and any

183 amendments, amendments and restatements, restatements or waivers of or supplements to or other modifications to, such agreements) in connection with the incurrence by any Loan Party of any secured Indebtedness as contemplated above, in order to permit such Indebtedness to be secured by a valid, perfected Lien (with such priority as may be designated by the Borrower or such Loan Party, to the extent such priority is permitted by the Loan Documents), and to subject the Liens on the Collateral securing the Secured Obligations to the provisions thereof. Each Lender and Issuing Bank hereby agrees that (x) if the Administrative Agent notifies such Lender or Issuing Bank that the Administrative Agent has determined in its sole discretion that any funds received by such Lender or Issuing Bank from the Administrative Agent or any of its Affiliates (whether as a payment, prepayment or repayment of principal, interest, fees or otherwise; individually and collectively, a “Payment”) were erroneously transmitted to such Lender or Issuing Bank (whether or not known to such Lender or Issuing Bank), and demands the return of such Payment (or a portion thereof), such Lender or Issuing Bank shall promptly, but in no event later than one Business Day thereafter, return to the Administrative Agent the amount of any such Payment (or portion thereof) as to which such a demand was made in same day funds, together with interest thereon in respect of each day from and including the date such Payment (or portion thereof) was received by such Lender or Issuing Bank to the date such amount is repaid to the Administrative Agent at the greater of the NYFRB Rate and a rate determined by the Administrative Agent in accordance with banking industry rules on interbank compensation from time to time in effect, and (y) to the extent permitted by applicable law, such Lender or Issuing Bank shall not assert, and hereby waives, as to the Administrative Agent, any claim, counterclaim, defense or right of set-off or recoupment with respect to any demand, claim or counterclaim by the Administrative Agent for the return of any Payment received, including without limitation any defense based on “discharge for value” or any similar doctrine. Any notice from the Administrative Agent to any Lender or Issuing Bank under this paragraph shall be conclusive, absent manifest error. Each Lender and Issuing Bank hereby further agrees that if it receives a Payment from the Administrative Agent or any of its Affiliates (x) that is in a different amount than, or on a different date from, that specified in a notice of payment sent by the Administrative Agent (or any of its Affiliates) with respect to such Payment (a “Payment Notice”) or (y) that was not preceded or accompanied by a Payment Notice, it shall be on notice, in each such case, that an error has been made with respect to such Payment. Each Lender and Issuing Bank agrees that, in each such case, or if it otherwise becomes aware a Payment (or portion thereof) may have been sent in error, such Lender or Issuing Bank shall promptly notify the Administrative Agent of such occurrence and, upon demand from the Administrative Agent, it shall promptly, but in no event later than one Business Day thereafter, return to the Administrative Agent the amount of any such Payment (or portion thereof) as to which such a demand was made in same day funds, together with interest thereon in respect of each day from and including the date such Payment (or portion thereof) was received by such Lender or Issuing Bank to the date such amount is repaid to the Administrative Agent at the greater of the NYFRB Rate and a rate determined by the Administrative Agent in accordance with banking industry rules on interbank compensation from time to time in effect. The Borrower and each other Loan Party hereby agree that in the event an erroneous Payment (or portion thereof) is not recovered from any Lender or Issuing Bank that has received

184 such Payment (or portion thereof) for any reason, the Administrative Agent shall be subrogated to all of the rights of such Lender or Issuing Bank with respect to such amount. Each party’s obligations under this paragraph and the previous three paragraphs of this Article VIII shall survive the resignation or replacement of the Administrative Agent or any transfer of rights or obligations by, or the replacement of, a Lender or Issuing Bank, the termination of the Commitments or the repayment, satisfaction or discharge of all Obligations under any Loan Document. ARTICLE IX MISCELLANEOUS Section 9.01 Notices. (a) Except in the case of notices and other communications expressly permitted to be given by telephone (and subject to paragraph (b) below), all notices and other communications provided for herein shall be in writing and shall be delivered by hand or overnight courier service, mailed by certified or registered mail or sent by telecopy, as follows: (i) if to Parent or the Borrower, to it at 1400 Atwater Drive, Malvern, Pennsylvania 19355440 Route 22 East, Suite 302, Bridgewater, NJ 08807, Attention of Treasurer (Telecopy No. 484-216-3002; Telephone No. 484-216-7909); (ii) if to the Administrative Agent, to it at Goldman Sachs Bank USA, 2001 Ross Ave, 37th Floor Dallas, Texas 75201, Attention of SBD Operations (Telephone No. 972-368-2323; Facsimile No. 646-769-7829; E-mail: gs-dallas- adminagency@ny.email.gs.com and gs-sbdagencyborrowernotices@ny.email.gs.com), with copy to Goldman Sachs Bank USA, 200 West Street, New York, New York 10282, Attention of Bank Debt Portfolio Group (Telephone No. 212-902-5717; E-mail: Luke.Qiu@gs.com), or such other office or person as the Administrative Agent may hereafter designate in writing as such to the other parties hereto; (iii) if to the Collateral Agent, to it at Goldman Sachs Bank USA, 200 West Street, New York, New York 10282, Attention of Bank Debt Portfolio Group (Telephone No. 212-902-5717; E-mail: Luke.Qiu@gs.com), or such other office or person as the Collateral Agent may hereafter designate in writing as such to the other parties hereto; (iv) if to the Issuing Bank, to it at Goldman Sachs Bank USA, 2001 Ross Ave, 37th Floor Dallas, Texas 75201, Attention of Letter of Credit Department (Telephone No. 972-368-2790; Facsimile No. 646-769-7829; E-mail: gs-loc- operations@ny.email.gs.com), with copy to Goldman Sachs Bank USA, 200 West Street, New York, New York 10282, Attention of Bank Debt Portfolio Group (Telephone No. 212-902-5717; E-mail: Luke.Qiu@gs.com), or such other office or person as the Issuing Bank may hereafter designate in writing as such to the other parties hereto; (v) if to the Swingline Lender, to it at Goldman Sachs Bank USA, 2001 Ross Ave, 37th Floor Dallas, Texas 75201, Attention of SBD Operations (Telephone No. 972-368-2323; Facsimile No. 646-769-7829; E-mail: gs-dallas-

185 adminagency@ny.email.gs.com and gs-sbdagencyborrowernotices@ny.email.gs.com), with copy to Goldman Sachs Bank USA, 200 West Street, New York, New York 10282, Attention of Bank Debt Portfolio Group (Telephone No. 212-902-5717; E-mail: Luke.Qiu@gs.com), or such other office or person as the Swingline Lender may hereafter designate in writing as such to the other parties hereto; and (vi) if to any other Lender, to it at its address (or telecopy number) set forth in its Administrative Questionnaire. (b) Notices and other communications to the Lenders hereunder may be delivered or furnished by electronic communications pursuant to procedures approved by the Administrative Agent; provided that the foregoing shall not apply to notices pursuant to Article II unless otherwise agreed by the Administrative Agent and the applicable Lender. The Administrative Agent or the Borrower may, in their discretion, agree to accept notices and other communications to it hereunder by electronic communications pursuant to procedures approved by it; provided that approval of such procedures may be limited to particular notices or communications. (c) The Platform. THE PLATFORM IS PROVIDED “AS IS” AND “AS AVAILABLE”. THE AGENT PARTIES (AS DEFINED BELOW) DO NOT WARRANT THE ACCURACY OR COMPLETENESS OF THE BORROWER MATERIALS OR THE ADEQUACY OF THE PLATFORM, AND EXPRESSLY DISCLAIM LIABILITY FOR ERRORS IN OR OMISSIONS FROM THE BORROWER MATERIALS. NO WARRANTY OF ANY KIND, EXPRESS, IMPLIED OR STATUTORY, INCLUDING ANY WARRANTY OF MERCHANTABILITY, FITNESS FOR A PARTICULAR PURPOSE, NON-INFRINGEMENT OF THIRD PARTY RIGHTS OR FREEDOM FROM VIRUSES OR OTHER CODE DEFECTS, IS MADE BY ANY AGENT PARTY IN CONNECTION WITH THE BORROWER MATERIALS OR THE PLATFORM. In no event shall the Administrative Agent or any of its Related Parties or any Lead Arranger (collectively, the “Agent Parties”) have any liability to any Borrower, any Lender, or any other Person for losses, claims, damages, liabilities or expenses of any kind (whether in tort, contract or otherwise) arising out of Parent’s or any Borrower’s or the Administrative Agent’s transmission of Borrower Materials through the Internet, except to the extent that such losses, claims, damages, liabilities or expenses are determined by a court of competent jurisdiction by a final and nonappealable judgment to have resulted from the gross negligence or willful misconduct of such Agent Party; provided, however, that in no event shall any Agent Party have any liability to Parent or any Borrower, any Lender or any other Person for indirect, special, incidental, consequential or punitive damages (as opposed to direct or actual damages). (d) Any party hereto may change its address or telecopy number for notices and other communications hereunder by notice to the other parties hereto. All notices and other communications given to any party hereto in accordance with the provisions of this Agreement shall be deemed to have been given on the date of receipt. Furthermore, each Public Lender agrees to cause at least one individual at or on behalf of such Public Lender to at all times have selected the “Private Side Information” or similar designation on the content declaration screen of the Platform in order to enable such Public Lender or its delegate, in accordance with such Public Lender’s compliance procedures and applicable Law, including United States Federal

186 and state securities Laws, to make reference to Borrower Materials that are not made available through the “Public Side Information” portion of the Platform and that may contain material non- public information with respect to Parent, the Borrower or their respective subsidiaries and its or their securities for purposes of United States Federal or state securities laws. Section 9.02 Waivers; Amendments. (a) No failure or delay by the Administrative Agent, the Collateral Agent, the Issuing Bank or any Lender in exercising any right or power hereunder or under any other Loan Document shall operate as a waiver thereof, nor shall any single or partial exercise of any such right or power, or any abandonment or discontinuance of steps to enforce such a right or power, preclude any other or further exercise thereof or the exercise of any other right or power. The rights and remedies of the Administrative Agent, the Collateral Agent, the Issuing Bank and the Lenders hereunder and under the other Loan Documents are cumulative and are not exclusive of any rights or remedies that they would otherwise have. No waiver of any provision of any Loan Document or consent to any departure by any Borrower therefrom shall in any event be effective unless the same shall be permitted by paragraph (b) of this Section 9.02, and then such waiver or consent shall be effective only in the specific instance and for the purpose for which given. Without limiting the generality of the foregoing, the making of a Loan or issuance of a Letter of Credit shall not be construed as a waiver of any Default, regardless of whether the Administrative Agent, the Collateral Agent, any Lender or the Issuing Bank may have had notice or knowledge of such Default at the time. (b) Except as provided in Section 2.20 with respect to an Incremental Amendment, Section 2.23 with respect to an Extension Amendment and Section 2.25 with respect to a Refinancing Amendment, and except as otherwise expressly provided in Section 9.19, neither this Agreement, any other Loan Document nor any provision hereof or thereof may be waived, amended or modified except pursuant to an agreement or agreements in writing entered into by the Borrower and the Required Lenders (other than as expressly provided below) or by the Borrower and the Administrative Agent with the consent of the Required Lenders (other than as expressly provided below); provided that no such agreement shall (i) increase or reinstate the Commitment of any Lender without the written consent of such Lender, (ii) reduce the principal amount of any Loan or LC Disbursement or reduce the rate of interest thereon or reduce any fees payable hereunder, without the written consent of each Lender directly affected thereby; provided that (x) any amendment to the financial definitions in this Agreement shall not constitute a reduction in the rate of interest for purposes of this clause (ii) even if the effect of such amendment would be to reduce the rate of interest on any Loan or any LC Disbursement or to reduce any fee payable hereunder and (y) only the consent of the Required Lenders (or, with respect to any default rate payable in respect of the Revolving Facility, Majority in Interest of Revolving Lenders, shall be necessary to waive any obligation of the Borrower to pay interest at the default rate) shall be necessary to reduce or waive any obligation of the Borrower to pay interest or fees at the applicable default rate set forth in Section 2.13(d), (iii) postpone the scheduled date of payment of the principal amount of any Loan or LC Disbursement (other than any reduction of the amount of, or any extension of the payment date for, the mandatory prepayments required under Section 2.11, in each case which shall only require the approval of the Required Lenders), or any interest thereon (other than interest payable at the applicable default rate of interest set forth in Section 2.13(d)), or any fees payable hereunder, or reduce the amount of, waive or excuse any such payment, or postpone the scheduled Maturity Date or scheduled date of expiration of any Commitment, without the written consent of each Lender directly affected thereby, (iv) change Section 2.18(b) or (d) in

187 a manner that would alter the pro rata sharing or the priority or scope of the application of payments, distributions or proceeds required thereby, without the written consent of each Lender directly and adversely affected thereby, (v) change the application of payments, distributions or proceeds with respect to the Revolving Secured Obligations set forth in Section 2.18(b), in Section 2.01 of the Intercreditor Agreement or in any other Approved Intercreditor Agreement, in each case, without the written consent of each Revolving Lender; provided, however, that the amendments, waivers and other modifications described in this clause (v) shall not require the consent of any Lenders other than each Revolving Lender, (vi) change any provision in the Intercreditor Agreement or in any other Approved Intercreditor Agreement that would alter definition of “Controlling Collateral Agent” (as defined therein) or similar such term or change any provision of this Agreement or any other Loan Document that would alter the right of the Required Revolving Lenders to direct the exercise of rights and remedies under the Loan Documents, in each case, without the written consent of each Lender directly and adversely affected thereby, (vii) change any of the provisions of this Section 9.02 or the definitions of “Required Lenders” or “Majority in Interest” or any other provision hereof specifying the number or percentage of Lenders required to waive, amend or modify any rights hereunder or make any determination or grant any consent hereunder, without the written consent of each Lender (it being understood that, solely with the consent of the parties prescribed by Section 2.20 to be parties to an Incremental Amendment, Incremental Term Loans may be included in the determination of Required Lenders on substantially the same basis as the Term Loans are included on the Effective Date), (viii) change the definition of “Required Revolving Lenders” without the written consent of each Revolving Lender; provided, however, that the amendments, waivers and other modifications described in this clause (viii) shall not require the consent of any Lenders other than each Revolving Lender, (ix) release all or substantially all of the Subsidiary Guarantors from their obligations under the Subsidiary Guaranty without the written consent of each Lender, (x) except as provided in Section 9.13 or in any Collateral Document in each case as in effect on the Effective Date, release all or substantially all of the Collateral, without the written consent of each Lender, (xi) amend, waive or otherwise modify the Financial Covenant or any definition or calculation provisions related thereto (solely in respect of the use of such defined terms or calculation provisions in or in any determination with respect to the Financial Covenant) or waive any Default or Event of Default resulting from a failure to perform or observe the Financial Covenant without the written consent of the Required Revolving Lenders; provided, however, that the amendments, waivers and other modifications described in this clause (xi) shall not require the consent of any Lenders other than the Required Revolving Lenders, (xii) amend, waive or modify any provisions of any Loan Document in a manner that by its terms adversely affects the rights in respect of payments due to, or the remedies of, Lenders holding Loans of any Class differently than those holding Loans of any other Class without the written consent of Lenders representing a Majority in Interest of each adversely affected Class (and in the case of multiple Classes which are affected, Majority in Interest with respect to all such Classes shall consent together as one Class); provided, however, that the amendments, waivers and other modifications described in this clause (xii) shall not require the consent of any Lenders other than the Lenders holding the Majority in Interest with respect to any applicable Class, (xiii) modify or extend the maturity date of any Letter of Credit to a date that is later than the Maturity Date applicable to the Revolving Commitments, without the written consent of each Revolving Lender; provided, however, that the amendments, waivers and other modifications described in this clause (xiii) shall not require the consent of any Lenders other than each Revolving Lender, (xiv) amend, waive or modify any of the provisions of this Agreement

188 or any other Loan Document in a manner that subordinates any of the Obligations in right of payment, or the priority of the Liens securing any of the Obligations, to any other Indebtedness (other than any Indebtedness permitted to be senior to the Obligations in accordance with the terms of this Agreement as in effect on the Effective Date and any debtor-in-possession financings that does not roll-up or refinance any Indebtedness that is junior in priority from payments on account of the Collateral proceeds or in right of security or payment to any of the Revolving Facility Obligations) without the written consent of each Lender or (xv) amend, waive or modify any provision of Section 2.11(b), Section 4.02, Section 6.04(w) or Section 9.13(a), amend, waive or modify any provision of Section 2.20 with respect to any Incremental Revolving Commitments or Incremental Revolving Loans, amend or modify the definition of “Material IP”, amend, waive or modify the last two sentences of Section 5.10, or amend, waive or modify any material right of the Revolving Lenders under the Intercreditor Agreement or under any other Approved Intercreditor Agreement, in each case with respect to this clause (xv), without the written consent of the Required Revolving Lenders; provided, further, that (i) no such agreement shall amend, modify or otherwise affect the rights or duties of the Administrative Agent, the Collateral Agent, the Issuing Bank or the Swingline Lender hereunder without the prior written consent of the Administrative Agent, the Collateral Agent, the Issuing Bank or the Swingline Lender, as the case may be and (ii) Section 9.04(f) may not be amended, waived or otherwise modified without the consent of each Granting Lender all or any part of whose Loans are being funded by an SPC at the time of such amendment, waiver or other modification. (c) Notwithstanding the foregoing, this Agreement and any other Loan Document may be amended (or amended and restated) with the written consent of the Required Lenders, the Administrative Agent and the Borrower (x) to add one or more credit facilities (in addition to the Incremental Term Loans pursuant to an Incremental Amendment, Extended Loans pursuant to an Extension Amendment and any Credit Agreement Refinancing Indebtedness pursuant to a Refinancing Amendment, which, in each case, for the avoidance of doubt, shall not require the consent of the Required Lenders) to this Agreement and to permit extensions of credit from time to time outstanding thereunder and the accrued interest and fees in respect thereof to share ratably in the benefits of this Agreement and the other Loan Documents with the Revolving Loans, the Term Loans, the Incremental Loans, the Extended Loans and the accrued interest and fees in respect thereof and (y) to include appropriately the Lenders holding such credit facilities in any determination of the Required Lenders and Lenders, and for purposes of the relevant provisions of Section 2.18(b). (d) If, in connection with any proposed amendment, waiver or consent requiring the consent of “each Lender” or “each Lender directly affected thereby,” the consent of the Required Lenders or Lenders representing a Majority in Interest of any Class directly affected, as applicable, is obtained, but the consent of other necessary Lenders is not obtained (any such Lender whose consent is necessary but not obtained being referred to herein as a “Non-Consenting Lender”), then the Borrower may elect to replace a Non-Consenting Lender as a Lender party to this Agreement; provided that, concurrently with such replacement, (i) another bank or other entity (which is reasonably satisfactory to the Borrower and the Administrative Agent) shall agree, as of such date, to purchase for cash the Loans and other Obligations due to the Non-Consenting Lender pursuant to an Assignment and Assumption and to become a Lender for all purposes under this Agreement and to assume all obligations of the Non-Consenting Lender to be terminated as of such date and to comply with the requirements of clause (b) of Section 9.04, and (ii) the Borrower

189 shall pay to such Non-Consenting Lender in same day funds on the day of such replacement (1) all interest, fees and other amounts then accrued but unpaid to such Non-Consenting Lender by the Borrower hereunder to and including the date of termination, including without limitation payments due to such Non-Consenting Lender under Sections 2.15 and 2.17, (2) an amount, if any, equal to the payment which would have been due to such Lender on the day of such replacement under Section 2.16 had the Loans of such Non-Consenting Lender been prepaid on such date rather than sold to the replacement Lender and (3) any amounts owing to such Lender pursuant to Section 2.12(d). A Lender shall not be required to make any such assignment and delegation if, prior thereto, as a result of a waiver by such Lender or otherwise, the circumstances entitling the Borrower to require such assignment and delegation cease to apply. (e) Notwithstanding anything to the contrary herein, (i) if following the Effective Date, the Administrative Agent and any Loan Party shall have jointly identified an ambiguity, inconsistency, obvious error or any error or omission of a technical or immaterial nature, in each case, in any provision of the Loan Documents, then the Administrative Agent and the applicable Loan Parties shall be permitted to amend such provision and such amendment shall become effective without any further action or consent of any other party to any Loan Documents if the same is not objected to in writing by the Required Lenders within five (5) Business Days following receipt of notice thereof, (ii) guarantees, collateral security agreements, pledge agreements and related documents (if any) executed by the Loan Parties in connection with this Agreement may be in a form reasonably determined by the Administrative Agent and may be amended, supplemented and/or waived with the consent of the Administrative Agent at the request of Parent or any Borrower without the input or need to obtain the consent of any other Lenders if such amendment or waiver is delivered in order (x) to comply with local law or advice of local counsel, (y) to cure ambiguities, omissions or defects or (z) to cause such guarantees, collateral security agreements, pledge agreement or other documents to be consistent with this Agreement and the other Loan Documents and (iii) the Borrower and the Administrative Agent may amend any provision of the Loan Documents to effect any technical, administrative or operational changes reasonably necessary to permit Borrowings in any Agreed Currency other than Dollars and Canadian Dollars, and such amendment shall become effective without any further action or consent of any other party to any Loan Documents if the same is not objected to in writing by the Multicurrency Tranche Lenders holding a majority of the outstanding Multicurrency Tranche Commitments and Multicurrency Tranche Revolving Credit Exposure as of such date within five (5) Business Days following receipt of notice thereof. (f) Notwithstanding anything to the contrary herein, in connection with any determination as to whether the Required Revolving Lenders or the Required Lenders, as applicable, have (A) consented (or not consented) to any amendment or waiver of any provision of this Agreement or any other Loan Document or any departure by any Loan Party therefrom, otherwise acted on any matter related to any Loan Document, or (B) directed or required the Administrative Agent, the Collateral Agent or any Lender to undertake any action (or refrain from taking any action) with respect to or under any Loan Document, any Lender (other than (x) any Lender that is a regulated bank, (y) any Revolving Lender as of the Effective Date and (z) any Affiliate of the foregoing) or any of its Affiliates (other than Affiliates that (I) make independent investment decisions, (II) have customary information screens in place (that apply to the Borrower) and (III) have investment policies that are not directed by, and whose investment decisions are not influenced by, the holder or a common Affiliate acting in concert with the holder) that, as a result

190 of its interest in any total return swap, total rate of return swap, credit default swap or other derivative contract (other than any such total return swap, total rate of return swap, credit default swap or other derivative contract entered into pursuant to bona fide market making activities), has a net short position with respect to the Loans and/or Commitments (each, a “Net Short Lender”) shall have no right to vote any of its Loans and Commitments and shall be deemed to have voted its interest as a Lender without discretion in the same proportion as the allocation of voting with respect to such matter by Lenders who are not Net Short Lenders. For purposes of determining whether a Lender has a “net short position” on any date of determination: (i) derivative contracts with respect to the Loans and Commitments and such contracts that are the functional equivalent thereof shall be counted at the notional amount thereof in Dollars, (ii) notional amounts in other currencies shall be converted to the dollar equivalent thereof by such Lender in a commercially reasonable manner consistent with generally accepted financial practices and based on the prevailing conversion rate (determined on a mid-market basis) on the date of determination, (iii) derivative contracts in respect of an index that includes any of the Borrower or other Loan Parties or any instrument issued or guaranteed by any of the Borrower or other Loan Parties shall not be deemed to create a short position with respect to the Loans and/or Commitments, so long as (x) such index is not created, designed, administered or requested by such Lender and (y) the Borrower and other Loan Parties and any instrument issued or guaranteed by any of the Borrower or other Loan Parties, collectively, shall represent less than 5.0% of the components of such index, (iv) derivative transactions that are documented using either the 2014 ISDA Credit Derivatives Definitions or the 2003 ISDA Credit Derivatives Definitions (collectively, the “ISDA CDS Definitions”) shall be deemed to create a short position with respect to the Loans and/or Commitments if such Lender is a protection buyer or the equivalent thereof for such derivative transaction and (x) the Loans or the Commitments are a “Reference Obligation” under the terms of such derivative transaction (whether specified by name in the related documentation, included as a “Standard Reference Obligation” on the most recent list published by Markit, if “Standard Reference Obligation” is specified as applicable in the relevant documentation or in any other manner), (y) the Loans or the Commitments would be a “Deliverable Obligation” under the terms of such derivative transaction or (z) any of Parent, the Borrower or the other Loan Parties (or their respective successors) is designated as a “Reference Entity” under the terms of such derivative transactions, and (v) credit derivative transactions or other derivatives transactions not documented using the ISDA CDS Definitions shall be deemed to create a short position with respect to the Loans and/or Commitments if such transactions are functionally equivalent to a transaction that offers the Lender protection in respect of the Loans or the Commitments, or as to the credit quality of any of the Borrower or other Loan Parties other than, in each case, as part of an index so long as (x) such index is not created, designed, administered or requested by such Lender and (y) the Borrower and other Loan Parties and any instrument issued or guaranteed by any of the Borrower or other Loan Parties, collectively, shall represent less than 5.0% of the components of such index. In connection with any such determination, each Lender (other than any Lender that is a regulated bank and any Revolving Lender as of the Effective Date) shall promptly notify the Administrative Agent in writing that it is a Net Short Lender, or shall otherwise be deemed to have represented and warranted to the Borrower and the Administrative Agent that it is not a Net Short Lender. Section 9.03 Expenses; Indemnity; Damage Waiver. (a) Parent and the Borrower shall (and hereby jointly and severally agree to) pay (i) all reasonable and documented out-of-pocket expenses incurred by the Administrative Agent, the Lead Arrangers and their respective Affiliates, including the reasonable fees, charges and disbursements of counsel for the Administrative Agent,

191 in connection with the syndication and distribution (including, without limitation, via the internet or through a service such as Intralinks) of the credit facilities provided for herein, the preparation and administration of this Agreement and the other Loan Documents or any amendments, modifications or waivers of the provisions hereof or thereof (whether or not the transactions contemplated hereby or thereby shall be consummated), (ii) all reasonable and documented out- of-pocket expenses incurred by the Issuing Bank in connection with the issuance, amendment or extension of any Letter of Credit or any demand for payment thereunder and (iii) all documented out-of-pocket expenses incurred by the Administrative Agent, the Issuing Bank or any Lender, including the fees, charges and disbursements of any counsel (other than in-house counsel) for the Administrative Agent, the Issuing Bank or any Lender, in connection with the enforcement or protection of its rights in connection with this Agreement and any other Loan Document, including its rights under this Section 9.03, or in connection with the Loans made or Letters of Credit issued hereunder, including all such out-of-pocket expenses incurred during any workout, restructuring or negotiations in respect of such Loans or Letters of Credit; provided, however, that in no event shall Parent or the Borrower be required to reimburse the Lenders for more than one counsel to the Administrative Agent (and up to one local counsel in each applicable jurisdiction and regulatory counsel) and one counsel for all of the other Lenders (and up to one local counsel in each applicable jurisdiction and regulatory counsel), unless a Lender or its counsel determines that it would create actual or potential conflicts of interest to not have individual counsel, in which case each Lender may have its own counsel which shall be reimbursed in accordance with the foregoing. (b) Except in respect of Indemnified Taxes or Other Taxes otherwise covered by Section 2.17(c), Parent and the Borrower shall, and jointly and severally agree to, indemnify the Administrative Agent, the Lead Arrangers, the Issuing Bank and each Lender, and each Related Party of any of the foregoing Persons (each such Person being called an “Indemnitee”) against, and hold each Indemnitee harmless from, any and all losses, claims, damages, liabilities and related expenses, including the fees, charges and disbursements of any counsel for any Indemnitee (but excluding any Excluded Taxes), incurred by or asserted against any Indemnitee arising out of, in connection with, or as a result of (i) the execution or delivery of any Loan Document or any agreement or instrument contemplated thereby, (ii) the performance by the parties hereto of their respective obligations thereunder or the consummation of the Transactions or any other transactions contemplated hereby, (iii) any Loan or Letter of Credit or the use of the proceeds therefrom (including any refusal by the Issuing Bank to honor a demand for payment under a Letter of Credit if the documents presented in connection with such demand do not strictly comply with the terms of such Letter of Credit), (iv) any actual or alleged presence or release of Hazardous Materials on or from any property owned or operated by Parent or any of its Subsidiaries, or any Environmental Liability related in any way to Parent or any of its Subsidiaries, or (v) any actual or prospective claim, litigation, investigation or proceeding relating to any of the foregoing, whether based on contract, tort or any other theory and regardless of whether (x) any Indemnitee is a party thereto or (y) such matter is initiated by a third party or by Parent or any of its affiliates; provided that such indemnity shall not, as to any Indemnitee, be available to the extent that such losses, claims, damages, liabilities or related expenses are determined by a court of competent jurisdiction by final and nonappealable judgment to have resulted from (i) the gross negligence, bad faith or willful misconduct of such Indemnitee or any of its Related Indemnified Persons, (ii) a material breach in bad faith by such Indemnitee or any of its Related Indemnified Persons of the express contractual obligations under any Loan Document pursuant to a claim made by Parent or the Borrower or (iii) any disputes among the

192 Indemnitees or any of their Related Indemnified Persons (other than in their capacities as Lead Arrangers or Administrative Agent) and not arising from any act or omission by Parent or any of its Affiliates. (c) To the extent that Parent or any Borrower fails to pay any amount required to be paid by it to the Administrative Agent, the Issuing Bank or the Swingline Lender under paragraph (a) or (b) of this Section 9.03, each Lender severally agrees to pay to the Administrative Agent, as the case may be, and each Revolving Lender severally agrees to pay to the Issuing Bank or the Swingline Lender, as the case may be, such Lender’s Applicable Percentage (determined as of the time that the applicable unreimbursed expense or indemnity payment is sought) of such unpaid amount (it being understood that any Borrower’s failure to pay any such amount shall not relieve such Borrower of any default in the payment thereof); provided that the unreimbursed expense or indemnified loss, claim, damage, liability or related expense, as the case may be, was incurred by or asserted against the Administrative Agent, the Issuing Bank or the Swingline Lender in its capacity as such. (d) To the extent permitted by applicable law, (i) neither Parent nor any Borrower shall assert, and each hereby waives, any claim against any Indemnitee for any damages arising from the use by others of information or other materials obtained through telecommunications, electronic or other information transmission systems (including the Internet) other than damages that are determined by a court of competent jurisdiction by final and nonappealable judgment to have resulted from the gross negligence or willful misconduct of such Indemnitee or any of its Related Indemnified Persons, or (ii) no party hereto shall assert, and each party hereto hereby waives, any claim on any theory of liability, for special, indirect, consequential or punitive damages (as opposed to direct or actual damages) arising out of, in connection with, or as a result of, this Agreement, any other Loan Document or any agreement or instrument contemplated hereby or thereby, the Transactions, any Loan or Letter of Credit or the use of the proceeds thereof. (e) All amounts due under this Section 9.03 shall be payable not later than fifteen (15) days after written demand therefor. Section 9.04 Successors and Assigns. (a) The provisions of this Agreement shall be binding upon and inure to the benefit of the parties hereto and their respective successors and assigns permitted hereby (including any Affiliate of the Issuing Bank that issues any Letter of Credit), except that (i) Parent and the Borrower may not assign or otherwise transfer any of its rights or obligations hereunder without the prior written consent of each Lender, except in a transaction permitted by Section 6.03 (and any attempted assignment or transfer by Parent or a Borrower without such consent shall be null and void).Subject to the terms and conditions set forth in Sections 9.04(b) through (l) below, any Lender may assign to one or more assignees (other than any Person that is not an Eligible Transferee) all or a portion of its rights and obligations under this Agreement (including all or a portion of its Commitments and the Loans at the time owing to it). Nothing in this Agreement, expressed or implied, shall be construed to confer upon any Person (other than the parties hereto, their respective successors and assigns permitted hereby (including any Affiliate of the Issuing Bank that issues any Letter of Credit), Participants (to the extent provided in paragraph (c) of this Section 9.04) and, to the extent expressly contemplated hereby,

193 the Related Parties of each of the Administrative Agent, the Issuing Bank and the Lenders) any legal or equitable right, remedy or claim under or by reason of this Agreement. (b) (i) Subject to the conditions set forth in paragraph (b)(ii) below, any Lender may assign to one or more “accredited investors” (as defined in regulation D of the Securities Act) (other than (1) Parent and its Affiliates, except to the extent permitted in Sections 2.24, 9.04(j), 9.04(g) and 9.04(k), (2) any natural Person, (3) any Defaulting Lender, (4) a Person that would be a Foreign Lender but is not capable of making the representation contained in Section 2.17(l) on the date it becomes a Lender, (5) any Disqualified Lender or (6) any Net Short Lender) (each, an “Eligible Transferee”) all or a portion of its rights and obligations under this Agreement (including all or a portion of its Commitments and the Loans at the time owing to it) with the prior written consent of: (A) the applicable Borrower (such consent not to be unreasonably withheld or delayed); provided that such Borrower shall be deemed to have consented to any such assignment unless it shall object thereto by written notice to the Administrative Agent within ten (10) Business Days after having received written notice thereof; provided, further, that no consent of the applicable Borrower shall be required for (x) any assignment by any Agent or Lead Arranger (or any affiliate thereof) of Term Loans or related commitments pursuant to the primary syndication of such Term Loans and related commitments or (y) an assignment to a Lender, an Affiliate of a Lender, an Approved Fund or, if an Event of Default under clause (a), (b), (h), (i) or (j) of Section 7.01 has occurred and is continuing, any other assignee; (B) the Administrative Agent (such consent not to be unreasonably withheld or delayed); provided that no consent of the Administrative Agent shall be required for an assignment of all or any portion of a Term Loan to a Lender, an Affiliate of a Lender or an Approved Fund; and (C) the Issuing Bank and the Swingline Lender (such consent not to be unreasonably withheld or delayed); provided that no consent of the Issuing Bank or the Swingline Lender shall be required for an assignment of all or any portion of a Term Loan or any related commitment. (ii) Assignments shall be subject to the following additional conditions: (A) except in the case of an assignment to a Lender or an Affiliate of a Lender or an Approved Fund or an assignment of the entire remaining amount of the assigning Lender’s Commitment or Loans of any Class, the amount of the applicable Commitment or Loans of the assigning Lender subject to each such assignment (determined as of the date the Assignment and Assumption with respect to such assignment is delivered to the Administrative Agent) shall not be less than $5,000,000 (in the case of Revolving Commitments and Revolving Loans) or $1,000,000 (in the case of a Term Loan) unless each of the applicable Borrower and the Administrative Agent otherwise consent; provided that no such consent of

194 the applicable Borrower shall be required if an Event of Default has occurred and is continuing; (B) each partial assignment shall be made as an assignment of a proportionate part of all the assigning Lender’s rights and obligations under this Agreement; provided that this clause shall not be construed to prohibit the assignment of a proportionate part of all the assigning Lender’s rights and obligations in respect of one Class of Commitments or Loans; (C) the parties to each assignment shall execute and deliver to the Administrative Agent an Assignment and Assumption, together with a processing and recordation fee of $3,500, such fee to be paid by either the assigning Lender or the assignee Lender or shared between such Lenders; (D) the assignee, if it shall not be a Lender, shall deliver to the Administrative Agent an Administrative Questionnaire in which the assignee designates one or more credit contacts to whom all syndicate-level information (which may contain material non-public information about Parent and its affiliates and their Related Parties or their respective securities) will be made available and who may receive such information in accordance with the assignee’s compliance procedures and applicable laws, including Federal and state securities laws; (E) without the prior written consent of the Administrative Agent, no assignment shall be made to a prospective assignee that bears a relationship to the applicable Borrower described in Section 108(e)(4) of the Code; and (F) if, at the time of any assignment, the respective assignee would be entitled to greater increased cost payments pursuant to Section 2.15 than those that apply to the respective assignor, then the respective assignee shall not be entitled to charge the Borrower for any such increased costs which would otherwise by owed to it pursuant to Section 2.15, but in each case only to the extent in excess of those that would have applied to the respective assignor at the time of such assignment. For the purposes of this Section 9.04(b), the term “Approved Fund” has the following meaning: “Approved Fund” means any Person (other than a natural person) that is (or will be) engaged in making, purchasing, holding or investing in bank loans and similar extensions of credit in the ordinary course of its business and that is administered or managed by (a) a Lender, (b) an Affiliate of a Lender or (c) an entity or an Affiliate of an entity that administers or manages a Lender. (iii) Subject to acceptance and recording thereof pursuant to paragraph (b)(iv) of this Section 9.04 (and, in the case of an Affiliated Lender or a Person that, after giving effect to such assignment, would become an Affiliated Lender, to the requirements of clause (g) of this Section 9.04), from and after the effective date specified

195 in each Assignment and Assumption (or Affiliated Lender Assignment and Assumption) the assignee thereunder shall be a party hereto and, to the extent of the interest assigned by such Assignment and Assumption, have the rights and obligations of a Lender under this Agreement, and the assigning Lender thereunder shall, to the extent of the interest assigned by such Assignment and Assumption, be released from its obligations under this Agreement (and, in the case of an Assignment and Assumption covering all of the assigning Lender’s rights and obligations under this Agreement, such Lender shall cease to be a party hereto but shall continue to be entitled to the benefits of Sections 2.15, 2.16, 2.17 and 9.03). Any assignment or transfer by a Lender of rights or obligations under this Agreement that does not comply with this Section 9.04 shall be treated for purposes of this Agreement as a sale by such Lender of a participation in such rights and obligations in accordance with paragraph (c) of this Section 9.04. (iv) The Administrative Agent, acting for this purpose as a non- fiduciary agent of the Borrower, shall maintain at one of its offices in the United States a copy of each Assignment and Assumption delivered to it and a register for the recordation of the names and addresses of the Lenders, and the Commitment of, and principal amount of and interest on the Loans and LC Disbursements owing to, each Lender pursuant to the terms hereof from time to time (the “Register”). The entries in the Register shall be conclusive (absent manifest error), and the Borrower, the Administrative Agent, the Issuing Bank and the Lenders shall treat each Person whose name is recorded in the Register pursuant to the terms hereof as a Lender hereunder for all purposes, notwithstanding notice to the contrary. The Register shall be available for inspection by the Borrower, the Issuing Bank and any Lender, at any reasonable time and from time to time upon reasonable prior notice. (v) Upon its receipt of a duly completed Assignment and Assumption executed by an assigning Lender and an assignee, the assignee’s completed Administrative Questionnaire (unless the assignee shall already be a Lender hereunder), the processing and recordation fee referred to in paragraph (b) of this Section 9.04 and any written consent to such assignment required by paragraph (b) of this Section 9.04, the Administrative Agent shall accept such Assignment and Assumption and record the information contained therein in the Register; provided that if either the assigning Lender or the assignee shall have failed to make any payment required to be made by it pursuant to Section 2.05(c), 2.06(d) or (e), 2.07(b), 2.18(e) or 9.03(c), the Administrative Agent shall have no obligation to accept such Assignment and Assumption and record the information therein in the Register unless and until such payment shall have been made in full, together with all accrued interest thereon. No assignment shall be effective for purposes of this Agreement unless it has been recorded in the Register as provided in this paragraph. (c) (i) Any Lender may, without the consent of the applicable Borrower, the Administrative Agent, the Issuing Bank or the Swingline Lender, sell participations to one or more banks or other entities (excluding (x) Parent and its Affiliates (other than Debt Fund Affiliates) and (y) any Person that is not an Eligible Transferee) (a “Participant”) in all or a portion of such Lender’s rights and obligations under this Agreement (including all or a portion of its Commitment and the Loans owing to it); provided that (A) such Lender’s obligations under this

196 Agreement shall remain unchanged, (B) such Lender shall remain solely responsible to the other parties hereto for the performance of such obligations, and (C) Parent, the Borrower and the other Lenders shall continue to deal solely and directly with such Lender in connection with such Lender’s rights and obligations under this Agreement. Any agreement or instrument pursuant to which a Lender sells such a participation shall provide that such Lender shall retain the sole right to enforce this Agreement and to approve any amendment, modification or waiver of any provision of this Agreement; provided that such agreement or instrument may provide that such Lender will not, without the consent of the Participant, agree to any amendment, modification or waiver described in the first proviso to Section 9.02(b) that affects such Participant. Subject to paragraph (c)(ii) of this Section 9.04, the Borrower agrees that each Participant shall be entitled to the benefits of Sections 2.15, 2.16 and 2.17 to the same extent as if it were a Lender and had acquired its interest by assignment pursuant to paragraph (b) of this Section 9.04. To the extent permitted by law, each Participant also shall be entitled to the benefits of Section 9.08 as though it were a Lender; provided such Participant agrees to be subject to Section 2.18(d) as though it were a Lender. (ii) A Participant shall not be entitled to receive any greater payment under Section 2.15 or 2.17 than the applicable Lender would have been entitled to receive with respect to the participation sold to such Participant, unless the sale of the participation to such Participant is made with the applicable Borrower’s prior written consent. A Participant shall not be entitled to the benefits of Section 2.17 unless such Participant agrees, for the benefit of the applicable Borrower, to comply with Section 2.17(e), Section 2.17(f), Section 2.17(g), Section 2.17(h) and Section 2.17(k) as though it were a Lender (it being understood that the documentation required under Section 2.17(e), Section 2.17(f), Section 2.17(g), Section 2.17(h) and Section 2.17(k) shall be delivered to the participating Lender). In addition, a Participant that would be a Foreign Lender if it were a Lender shall not be entitled to the benefits of Section 2.17 unless the applicable Borrower is notified of the participation sold to such Participant. Each Lender that sells a participation shall, acting solely for this purpose as a non-fiduciary agent of the applicable Borrower, maintain a register on which it enters the name and address of each Participant and the principal amounts (and stated interest) of each Participant’s interest in the Loans or other obligations under the Loan Documents (the “Participant Register”); provided that no Lender shall have any obligation to disclose all or any portion of the Participant Register (including the identity of any Participant or any information relating to a Participant’s interest in any Commitments, Loans, Letters of Credit or its other obligations under any Loan Document) to any Person except to the extent that such disclosure is necessary to establish that such Commitment, Loan, Letter of Credit or other obligation is in registered form under Section 5f.103-1(c) of the United States Treasury Regulations or Proposed Section 1.163-5(b) of the United States Treasury Regulations (or, in each case, any amended or successor version). The entries in the Participant Register shall be conclusive absent manifest error, and such Lender shall treat each Person whose name is recorded in the Participant Register as the owner of such participation for all purposes of this Agreement notwithstanding any notice to the contrary. For the avoidance of doubt, the Administrative Agent (in its capacity as Administrative Agent) shall have no responsibility for maintaining a Participant Register. (d) Any Lender may at any time pledge or assign a security interest in all or any portion of its rights under this Agreement to secure obligations of such Lender, including

197 without limitation any pledge or assignment to secure obligations to a Federal Reserve Bank or any other central bank, and this Section 9.04 shall not apply to any such pledge or assignment of a security interest; provided that no such pledge or assignment of a security interest shall release a Lender from any of its obligations hereunder or substitute any such pledgee or assignee for such Lender as a party hereto. (e) In the case of any assignment, transfer or novation by a Lender to a new Lender, or any participation by such Lender in favor of a Participant, of all or any part of such Lender’s rights and obligations under this Agreement or any of the other Loan Documents, such Lender and the new Lender or Participant (as applicable) hereby agree that, for the purposes of Article 1278 and/or Article 1281 of the Luxembourg Civil Code (to the extent applicable), any assignment, amendment, transfer and/or novation of any kind permitted under, and made in accordance with the provisions of, this Agreement or any agreement referred to herein to which any Luxembourg Guarantor is a party (including any Collateral Document), any security created or guarantee given under or in connection with this Agreement or any other Loan Document shall be preserved and shall continue in full force and effect for the benefit of such new Lender or Participant (as applicable). (f) Notwithstanding anything to the contrary contained herein, any Lender (a “Granting Lender”) may grant to a special purpose vehicle identified as such in writing from time to time by the Granting Lender to the Administrative Agent and Parent (an “SPC”) the option to provide all or any part of any Loan that such Granting Lender would otherwise be obligated to make pursuant to this Agreement; provided that (i) nothing herein shall constitute a commitment by any SPC to fund any Loan, (ii) if an SPC elects not to exercise such option or otherwise fails to make all or any part of such Loan, the Granting Lender shall be obligated to make such Loan pursuant to the terms hereof and (iii) such SPC and the applicable Loan or any applicable part thereof shall be appropriately reflected in the Participant Register. Each party hereto hereby agrees that (i) neither the grant to any SPC nor the exercise by any SPC of such option shall increase the costs or expenses or otherwise increase or change the obligations of Parent or any Subsidiary under the Loan Documents (including its obligations under Sections 2.15 through 2.17), (ii) no SPC shall be liable for any indemnity or similar payment obligation under this Agreement for which a Lender would be liable (and such Lender shall remain liable for such indemnity or similar payment obligation on behalf of the SPC), and (iii) the Granting Lender shall for all purposes, including the approval of any amendment, waiver or other modification of any provision of any Loan Document, remain the Lender hereunder. The making of a Loan by an SPC hereunder shall utilize the Commitment of the Granting Lender to the same extent, and as if, such Loan were made by such Granting Lender. Notwithstanding anything to the contrary contained herein, any SPC may (i) with notice to, but without prior consent of any Borrower and the Administrative Agent and with the payment of a processing fee of $3,500 (which processing fee may be waived by the Administrative Agent in its sole discretion), assign all or any portion of its right to receive payment with respect to any Loan to the Granting Lender and (ii) disclose on a confidential basis any non-public information relating to its funding of Loans to any rating agency, commercial paper dealer or provider of any surety or Guarantee or credit or liquidity enhancement to such SPC. (g) Any Lender may, at any time, assign all or a portion of its rights and obligations with respect to Term Loans under this Agreement to a Person who is or will become,

198 after such assignment, an Affiliated Lender through (x) Dutch auctions or other offers to purchase or take by assignment open to all Lenders on a pro rata basis in accordance with procedures determined by such Affiliated Lender in its sole discretion or (y) open market purchase consisting solely of cash on a non-pro rata basis, in each case subject to the following limitations: (i) Affiliated Lenders will not (A) receive information provided solely to Lenders by the Administrative Agent or any Lender and will not be permitted to attend or participate in conference calls or meetings attended solely by the Lenders and the Administrative Agent, other than the right to receive notices of prepayments and other administrative notices in respect of its Loans or Commitments required to be delivered to Lenders pursuant to Article II or (B) make any challenge to the Administrative Agent’s or any other Lender’s attorney-client privilege on the basis of its status as a Lender; (ii) each Affiliated Lender that purchases any Loans this subsection (g) shall identify itself as an Affiliated Lender; (iii) each Lender (other than any other Affiliated Lender) that assigns any Loans to an Affiliated Lender pursuant to this subsection (g) shall deliver to the Administrative Agent and Parent a customary Big Boy Letter; (iv) the aggregate principal amount of Term Loans of any Class under this Agreement held by Affiliated Lenders at the time of any such purchase or assignment shall not exceed 25% of the aggregate principal amount of Term Loans of such Class outstanding at such time under this Agreement (such percentage, the “Affiliated Lender Cap”); provided that to the extent any assignment to an Affiliated Lender would result in the aggregate principal amount of all Term Loans of any Class held by Affiliated Lenders exceeding the Affiliated Lender Cap, the assignment of such excess amount will be void ab initio; (v) as a condition to each assignment pursuant to this subsection (g), the Administrative Agent and Parent shall have been provided a notice in connection with each assignment to an Affiliated Lender or a Person that upon effectiveness of such assignment would constitute an Affiliated Lender pursuant to which such Affiliated Lender (in its capacity as such) shall waive any right to bring any action in connection with such Loans against the Administrative Agent, in its capacity as such; and (vi) the assigning Lender and the Affiliated Lender purchasing such Lender’s Term Loans shall execute and deliver to the Administrative Agent an assignment agreement substantially in the form of Exhibit B-2 hereto (an “Affiliated Lender Assignment and Assumption”). Notwithstanding anything to the contrary contained herein, any Affiliated Lender that has purchased Term Loans pursuant to this subsection (g) may, in its sole discretion, contribute, directly or indirectly, the principal amount of such Term Loans or any portion thereof, plus all accrued and unpaid interest thereon, to a Borrower for the purpose of cancelling and extinguishing such Term Loans. Upon the date of such contribution, assignment or transfer, (x) the aggregate outstanding principal amount of Term Loans shall reflect such cancellation and

199 extinguishing of the Term Loans then held by such Borrower and (y) such Borrower shall promptly provide notice to the Administrative Agent of such contribution of such Term Loans, and the Administrative Agent, upon receipt of such notice, shall reflect the cancellation of the applicable Term Loans in the Register. Each Affiliated Lender agrees to notify the Administrative Agent and Parent promptly (and in any event within ten (10) Business Days) if it acquires any Person who is also a Lender, and each Lender agrees to notify the Administrative Agent and Parent promptly (and in any event within ten (10) Business Days) if it becomes an Affiliated Lender. The Administrative Agent may conclusively rely upon any notice delivered pursuant to the immediately preceding sentence or pursuant to clause (v) of this subsection (g) and shall not have any liability for any losses suffered by any Person as a result of any purported assignment to or from an Affiliated Lender. (h) Notwithstanding anything in Section 9.02 or the definition of “Required Lenders,” “Required Revolving Lenders” or “Majority in Interest” to the contrary, for purposes of determining whether the Required Lenders, Required Revolving Lenders and Majority in Interest in respect of a Class of Loans have (i) consented (or not consented) to any amendment, modification, waiver, consent or other action with respect to any of the terms of any Loan Document or any departure by any Loan Party therefrom, or subject to Section 9.04(i), any plan of reorganization pursuant to the U.S. Bankruptcy Code, (ii) otherwise acted on any matter related to any Loan Document, or (iii) directed or required the Administrative Agent or any Lender to undertake any action (or refrain from taking any action) with respect to or under any Loan Document, no Affiliated Lender shall have any right to consent (or not consent), otherwise act or direct or require the Administrative Agent or any Lender to take (or refrain from taking) any such action and, except with respect to any amendment, modification, waiver, consent or other action (x) in Section 9.02 requiring the consent of all Lenders, all Lenders directly and adversely affected or specifically such Lender, (y) that alters an Affiliated Lender’s pro rata share of any payments given to all Lenders, or (z) affects the Affiliated Lender (in its capacity as a Lender) in a manner that is disproportionate to the effect on any Lender in the same Class, the Loans held by an Affiliated Lender shall be disregarded in both the numerator and denominator in the calculation of any Lender vote (and shall be deemed to have been voted in the same percentage as all other applicable Lenders voted if necessary to give legal effect to this paragraph) (but, in any event, in connection with any amendment, modification, waiver, consent or other action, shall be entitled to any consent fee, calculated as if all of such Affiliated Lender’s Loans had voted in favor of any matter for which a consent fee or similar payment is offered). (i) Notwithstanding anything in this Agreement or the other Loan Documents to the contrary, each Affiliated Lender hereby agrees that, and each Affiliated Lender Assignment and Assumption shall provide a confirmation that, if a proceeding under any Debtor Relief Law shall be commenced by or against Parent or any other Loan Party at a time when such Lender is an Affiliated Lender, such Affiliated Lender irrevocably authorizes and empowers the Administrative Agent to vote on behalf of such Affiliated Lender with respect to the Term Loans held by such Affiliated Lender in any manner in the Administrative Agent’s sole discretion, unless the Administrative Agent instructs such Affiliated Lender to vote, in which case such Affiliated Lender shall vote with respect to the Term Loans held by it as the Administrative Agent directs; provided that such Affiliated Lender shall be entitled to vote in accordance with its sole discretion

200 (and not in accordance with the direction of the Administrative Agent) in connection with any plan of reorganization to the extent any such plan of reorganization proposes to treat any Obligations held by such Affiliated Lender in a disproportionately adverse manner than the proposed treatment of similar Obligations held by Term Lenders that are not Affiliated Lenders. (j) Although any Debt Fund Affiliate(s) shall be Eligible Transferees and shall not be subject to the provisions of Section 9.04(g), (h) and (i), any Lender may, at any time, assign all or a portion of its rights and obligations with respect to Term Loans under this Agreement to a Person who is or will become, after such assignment, a Debt Fund Affiliate. Notwithstanding anything in Section 9.02 or the definition of “Required Lenders” or “Required Revolving Lenders” to the contrary, for purposes of determining whether the Required Lenders or the Required Revolving Lenders, as applicable, have (i) consented (or not consented) to any amendment, modification, waiver, consent or other action with respect to any of the terms of any Loan Document or any departure by any Loan Party therefrom, (ii) otherwise acted on any matter related to any Loan Document or (iii) directed or required the Administrative Agent, the Collateral Agent or any Lender to undertake any action (or refrain from taking any action) with respect to or under any Loan Document, all Term Loans, Revolving Commitments and Revolving Loans held by Debt Fund Affiliates, as applicable, in the aggregate, may not account for more than 49.9% of the Term Loans, Revolving Commitments and Revolving Loans, as applicable, of consenting Lenders included in determining whether the Required Lenders or Required Revolving Lenders have consented to any action pursuant to Section 9.02. (k) Any Lender may, so long as no Event of Default has occurred and is continuing, at any time, assign all or a portion of its rights and obligations with respect to Term Loans under this Agreement to Parent or any Subsidiary through (x) Dutch auctions or other offers to purchase open to all Lenders on a pro rata basis in accordance with Auction Procedures or (y) open market purchases consisting solely of cash on a non-pro rata basis; provided that: (i) (a) the principal amount of such Term Loans, along with all accrued and unpaid interest thereon, so contributed, assigned or transferred to Parent or any Subsidiary shall be deemed automatically cancelled and extinguished on the date of such contribution, assignment or transfer, (b) the aggregate outstanding principal amount of Term Loans of the remaining Lenders shall reflect such cancellation and extinguishing of such Term Loans and (c) Parent or a Borrower shall promptly provide notice to the Administrative Agent of such contribution, assignment or transfer of such Term Loans, and the Administrative Agent, upon receipt of such notice, shall reflect the cancellation of the applicable Term Loans in the Register; (ii) each Person that purchases any Loans pursuant to this subsection (k) shall identify itself as Parent or a Subsidiary of Parent; (iii) each Lender (other than an Affiliated Lender) that assigns any Loans to Parent, a Borrower or any Subsidiary of Parent pursuant to this subsection (k) shall deliver to the Administrative Agent and Parent a customary Big Boy Letter; and (l) purchases of Term Loans pursuant to this subsection (l) may not be funded with the proceeds of Revolving Loans.

201 Section 9.05 Survival. All covenants, agreements, representations and warranties made by the Loan Parties in the Loan Documents and in the certificates or other instruments delivered in connection with or pursuant to this Agreement or any other Loan Document shall be considered to have been relied upon by the other parties hereto and shall survive the execution and delivery of the Loan Documents and the making of any Loans and issuance of any Letters of Credit, regardless of any investigation made by any such other party or on its behalf and notwithstanding that the Administrative Agent, the Issuing Bank or any Lender may have had notice or knowledge of any Default or incorrect representation or warranty at the time any credit is extended hereunder, and shall continue in full force and effect as long as the principal of or any accrued interest on any Loan or any fee or any other amount payable under this Agreement or any other Loan Document is outstanding and unpaid (except for Unliquidated Obligations) or any Letter of Credit is outstanding (unless such Letter of Credit has been Cash Collateralized) and so long as the Commitments have not expired or terminated. The provisions of Sections 2.15, 2.16, 2.17 and 9.03 and Article VIII shall survive and remain in full force and effect regardless of the consummation of the transactions contemplated hereby, the repayment of the Loans, the expiration or termination of the Letters of Credit and the Commitments or the termination of this Agreement or any other Loan Document or any provision hereof or thereof. Section 9.06 Integration; Counterparts; Electronic Signature. This Agreement may be executed in counterparts (and by different parties hereto in different counterparts), each of which shall constitute an original, but all of which when taken together shall constitute a single contract. This Agreement, the other Loan Documents and any separate letter agreements with respect to fees payable to the Administrative Agent constitute the entire contract among the parties relating to the subject matter hereof and supersede any and all previous agreements and understandings, oral or written, relating to the subject matter hereof. Delivery of an executed counterpart of a signature page of this Agreement by facsimile or by email as a “.pdf” or “.tif” attachment shall be effective as delivery of a manually executed counterpart of this Agreement. Delivery of an executed counterpart of a signature page of this Agreement, and/or any document, amendment, approval, consent, information, notice, certificate, request, statement, disclosure or authorization related to this Agreement and/or the transactions contemplated hereby and/or thereby (each an “Ancillary Document”) that is an electronic sound, symbol, or process attached to, or associated with, a contract or other record and adopted by a Person with the intent to sign, authenticate or accept such contract or record (an “Electronic Signature”) transmitted by telecopy, emailed pdf or any other electronic means that reproduces an image of an actual executed signature page shall be effective as delivery of a manually executed counterpart of this Agreement or such Ancillary Document, as applicable. The words “execution,” “signed,” “signature,” “delivery,” and words of like import in or relating to this Agreement and/or any Ancillary Document shall be deemed to include Electronic Signatures, deliveries or the keeping of records in any electronic form (including deliveries by telecopy, emailed pdf or any other electronic means that reproduces an image of an actual executed signature page), each of which shall be of the same legal effect, validity or enforceability as a manually executed signature, physical delivery thereof or the use of a paper-based recordkeeping system, as the case may be. Nevertheless, upon the request of the Administrative Agent or any Lender, any Electronic Signature shall be promptly followed by a manually executed counterpart. Section 9.07 Severability. Any provision of any Loan Document held to be invalid, illegal or unenforceable in any jurisdiction shall, as to such jurisdiction, be ineffective to the extent of such invalidity, illegality or unenforceability without affecting the validity, legality and

202 enforceability of the remaining provisions thereof; and the invalidity of a particular provision in a particular jurisdiction shall not invalidate such provision in any other jurisdiction. Section 9.08 Right of Setoff. If an Event of Default shall have occurred and be continuing, each Lender and each of its Affiliates is hereby authorized at any time and from time to time, to the fullest extent permitted by law, to set off and apply any and all deposits (general or special, time or demand, provisional or final and in whatever currency denominated) at any time held and other obligations at any time owing by such Lender or Affiliate to or for the credit or the account of Parent, any Borrower or any Subsidiary Guarantor against any of and all of the Secured Obligations held by such Lender, irrespective of whether or not such Lender shall have made any demand under the Loan Documents and although such obligations may be unmatured; provided that any recovery by any Lender or any Affiliate pursuant to its setoff rights under this Section 9.08 is subject to the provisions of Section 2.18(d). The rights of each Lender under this Section 9.08 are in addition to other rights and remedies (including other rights of setoff) which such Lender may have. Section 9.09 Governing Law; Jurisdiction; Consent to Service of Process; Foreign Process Agent. (a) This Agreement shall be construed in accordance with and governed by the law of the State of New York. (b) Parent and the Borrower hereby irrevocably and unconditionally submit, for themselves and their property, to the exclusive jurisdiction of the Supreme Court of the State of New York sitting in New York County and of the United States District Court of the Southern District of New York sitting in New York County, and any appellate court from any thereof, in any action or proceeding arising out of or relating to any Loan Document (other than any Collateral Documents which specify a different jurisdiction), or for recognition or enforcement of any judgment, and each of the parties hereto hereby irrevocably and unconditionally agrees that all claims in respect of any such action or proceeding may be heard and determined in such New York State or, to the extent permitted by law, in such Federal court. Each of the parties hereto agrees that a final judgment in any such action or proceeding shall be conclusive and may be enforced in other jurisdictions by suit on the judgment or in any other manner provided by law. Nothing in this Agreement or any other Loan Document shall affect any right that the Administrative Agent, the Issuing Bank or any Lender may otherwise have to bring any action or proceeding relating to this Agreement or any other Loan Document against any Loan Party or its properties in the courts of any jurisdiction. (c) Parent and the Borrower hereby irrevocably and unconditionally waive, to the fullest extent they may legally and effectively do so, any objection which they may now or hereafter have to the laying of venue of any suit, action or proceeding arising out of or relating to this Agreement or any other Loan Document in any court referred to in paragraph (b) of this Section 9.09. Each of the parties hereto hereby irrevocably waives, to the fullest extent permitted by law, the defense of an inconvenient forum to the maintenance of such action or proceeding in any such court. (d) Each party to this Agreement irrevocably consents to service of process in the manner provided for notices in Section 9.01. Nothing in this Agreement or any

203 other Loan Document will affect the right of any party to this Agreement to serve process in any other manner permitted by law. Section 9.10 WAIVER OF JURY TRIAL. EACH PARTY HERETO HEREBY WAIVES, TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY RIGHT IT MAY HAVE TO A TRIAL BY JURY IN ANY LEGAL PROCEEDING DIRECTLY OR INDIRECTLY ARISING OUT OF OR RELATING TO THIS AGREEMENT, ANY OTHER LOAN DOCUMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY OR THEREBY (WHETHER BASED ON CONTRACT, TORT OR ANY OTHER THEORY). EACH PARTY HERETO (A) CERTIFIES THAT NO REPRESENTATIVE, AGENT OR ATTORNEY OF ANY OTHER PARTY HAS REPRESENTED, EXPRESSLY OR OTHERWISE, THAT SUCH OTHER PARTY WOULD NOT, IN THE EVENT OF LITIGATION, SEEK TO ENFORCE THE FOREGOING WAIVER AND (B) ACKNOWLEDGES THAT IT AND THE OTHER PARTIES HERETO HAVE BEEN INDUCED TO ENTER INTO THIS AGREEMENT BY, AMONG OTHER THINGS, THE MUTUAL WAIVERS AND CERTIFICATIONS IN THIS SECTION 9.10. Section 9.11 Headings. Article and Section headings and the Table of Contents used herein are for convenience of reference only, are not part of this Agreement and shall not affect the construction of, or be taken into consideration in interpreting, this Agreement. Section 9.12 Confidentiality. Each of the Administrative Agent, the Collateral Agent, the Swingline Lender, the Issuing Bank and the Lenders agrees to maintain the confidentiality of the Information (as defined below), except that Information may be disclosed (a) to its Affiliates and its and their respective directors, officers, employees and agents, including accountants, legal counsel and other advisors (it being understood that the Persons to whom such disclosure is made will be informed of the confidential nature of such Information and instructed to keep such Information confidential), (b) to the extent required or requested by any regulatory authority purporting to have jurisdiction over such Person or its Related Parties (including any self- regulatory authority, such as the National Association of Insurance Commissioners), (c) to the extent required by applicable laws or regulations or by any subpoena or similar legal process, (d) to any other party to this Agreement, (e) in connection with the exercise of any remedies under this Agreement or any other Loan Document or any suit, action or proceeding relating to this Agreement or any other Loan Document or the enforcement of rights hereunder or thereunder, (f) subject to an agreement containing provisions substantially the same as those of this Section 9.12, to (i) any assignee of or Participant in, or any prospective assignee of or Participant in, any of its rights or obligations under this Agreement or (ii) any actual or prospective counterparty (or its Related Parties) to any swap, derivative or other transaction relating to Parent or its Restricted Subsidiaries and their obligations, (g) on a confidential basis to (i) any rating agency in connection with rating Parent or its Subsidiaries or the facilities evidenced by this Agreement or (ii) the CUSIP Service Bureau or any similar agency in connection with the issuance and monitoring of CUSIP numbers with respect to the facilities evidenced by this Agreement, (h) with the prior written consent of Parent or (i) to the extent such Information (i) becomes publicly available other than as a result of a breach of this Section 9.12 by the disclosing party or its Affiliates or (ii) becomes available to the Administrative Agent, the Issuing Bank or any Lender on a nonconfidential basis from a source other than Parent or the Borrower. In addition, the Administrative Agent, the

204 Swingline Lender, the Issuing Bank and each of the Lenders may disclose the existence of this Agreement and the information about this Agreement to market data collectors, similar service providers to the lending industry, and service providers to the Administrative Agent and the Lenders in connection with the administration and management of this Agreement and the other Loan Documents. For the purposes of this Section 9.12, “Information” means all information received from Parent, the Borrower or their Affiliates relating to Parent, the Borrower or their respective Affiliates and businesses, other than any such information that is available to the Administrative Agent, the Issuing Bank or any Lender on a nonconfidential basis prior to disclosure by Parent or the Borrower. Any Person required to maintain the confidentiality of Information as provided in this Section 9.12 shall be considered to have complied with its obligation to do so if such Person has exercised the same degree of care to maintain the confidentiality of such Information as such Person would accord to its own confidential information. For the avoidance of doubt, nothing herein prohibits any individual from communicating or disclosing information regarding suspected violations of laws, rules, or regulations to a governmental, regulatory or self-regulatory authority without any notification to any person. Section 9.13 Release of Liens and Guarantees. (a) A Guarantor (other than Parent and any Borrower) shall automatically be released from its obligations under the Loan Documents upon (i) the consummation of any transaction permitted by this Agreement as a result of which such Guarantor ceases to be a Restricted Subsidiary; provided that, if so required by this Agreement, the Required Lenders shall have consented to such transaction and the terms of such consent shall not have provided otherwise or (ii) such Guarantor becoming an Excluded Subsidiary; provided that Parent has elected for such Excluded Subsidiary to be released from its Guaranty; provided, further to the extent any such Guarantor becomes an Excluded Subsidiary pursuant to clause (a) of the definition thereof, the transaction resulting in such Guarantor becoming an Excluded Subsidiary shall be (1) with a Person that is not an Affiliate of the Borrower, (2) for a bona fide business purpose in the good faith determination of the Borrower Representative and (3) for fair market value. Upon (a) the occurrence of the Termination Date (as defined in the US Security Agreement), (b) any Disposition (other than any lease or license) by any Loan Party (other than to Parent or any Restricted Subsidiary) of any Collateral (i) in a transaction permitted under this Agreement or (ii) in connection with any exercise of remedies of the Administrative Agent and the Lenders pursuant to Article VII or by the Collateral Agent pursuant to the US Security Agreement, the Intercreditor Agreement or any Approved Intercreditor Agreement, (c) any Disposition by any Loan Party to a Receivables Entity of any Permitted Receivables Facility Assets in connection with a Permitted Receivables Facility, (d) any property of a Loan Party becoming an Excluded Asset or (e) the effectiveness of any written consent to the release of the security interest created under any Collateral Document in any Collateral pursuant to Section 9.02, the security interests in such Collateral shall be automatically released. Any termination or release pursuant to this Section 9.13 shall not in any manner discharge, affect or impair the Secured Obligations or any Liens upon (or obligations of Parent or any Subsidiary in respect of) all interests retained by Parent or any Subsidiary, including (without limitation) the proceeds of the sale, all of which shall continue to constitute part of the Collateral. Any execution and delivery of documents pursuant to this Section 9.13 shall be without recourse to or warranty by the Collateral Agent.

205 (b) The Collateral Agent is irrevocably authorized to subordinate any Lien on any property granted to or held by the Collateral Agent under any Loan Document to the holder of any Lien on such property that is permitted by clauses (b), (d) or (h) of the definition of “Permitted Encumbrances” or Section 6.02(c), (d), (e), (g), (h), (k), (l), (m), (o), (p), (r), (y) (to the extent that the relevant Lien is of the type to which the Lien of the Collateral Agent may otherwise be required to be subordinated under this clause (b) pursuant to any of the other exceptions to Section 6.02 that are expressly included in this clause (b)), (bb) or (cc). Section 9.14 USA Patriot Act. Each Lender that is subject to the requirements of the USA Patriot Act (Title III of Pub. L. 107-56 (signed into law October 26, 2001)) (the “USA Patriot Act”) hereby notifies each Loan Party that pursuant to the requirements of the USA Patriot Act, it is required to obtain, verify and record information that identifies such Loan Party, which information includes the name and address of such Loan Party and other information that will allow such Lender to identify such Loan Party in accordance with the USA Patriot Act and the Beneficial Ownership Regulation. Section 9.15 Appointment for Perfection. Each Lender hereby appoints each other Lender as its agent for the purpose of perfecting Liens, for the benefit of the Secured Parties, in assets which, in accordance with Article 9 of the UCC or any other applicable law can be perfected by possession. Should any Lender (other than the Administrative Agent) obtain possession of any such Collateral, such Lender shall notify the Administrative Agent and the Collateral Agent thereof, and, promptly upon the Administrative Agent’s request therefor shall deliver such Collateral to the Collateral Agent or otherwise deal with such Collateral in accordance with the Administrative Agent’s instructions. Section 9.16 No Fiduciary Relationship. Parent, on behalf of itself and its Subsidiaries, agrees that, in connection with all aspects of the transactions contemplated hereby (including in connection with any amendment, waiver or other modification hereof or of any other Loan Document) and any communications in connection therewith: (i) (A) the arranging and other services regarding this Agreement provided by the Lenders are arm’s-length commercial transactions between Parent and its Affiliates, on the one hand, and the Lenders and their Affiliates, on the other hand, (B) Parent has consulted its own legal, accounting, regulatory and tax advisors to the extent it has deemed appropriate, and (C) Parent is capable of evaluating, and understands and accepts, the terms, risks and conditions of the transactions contemplated hereby and by the other Loan Documents; (ii) (A) each of the Lenders and their Affiliates is and has been acting solely as a principal and, except as expressly agreed in writing by the relevant parties, has not been, is not, and will not be acting as an advisor, agent or fiduciary for Parent, the Borrower or any of their respective Affiliates, or any other Person and (B) no Lender or any of its Affiliates has any obligation to Parent, the Borrower or any of their respective Affiliates with respect to the transactions contemplated hereby except, in the case of a Lender, those obligations expressly set forth herein and in the other Loan Documents; and (iii) each of the Lenders and their respective Affiliates may be engaged in a broad range of transactions that involve interests that differ from those of Parent and its Affiliates, and no Lender or any of its Affiliates has any obligation to disclose any of such interests to Parent, the Borrower or their respective Affiliates. To the fullest extent permitted by law, Parent and the Borrower hereby agree not to assert any claims that they may have against each of the Lenders and their Affiliates with respect to any breach or alleged

206 breach of agency or fiduciary duty in connection with any aspect of any transaction contemplated hereby. Section 9.17 Interest Rate Limitation. Notwithstanding anything to the contrary contained in any Loan Document, the interest paid or agreed to be paid under the Loan Documents shall not exceed the maximum rate of non-usurious interest permitted by applicable law (the “Maximum Rate”). If the Administrative Agent or any Lender shall receive interest in an amount that exceeds the Maximum Rate, the excess interest shall be applied to the principal of the Loans or, if it exceeds such unpaid principal, refunded to the applicable Borrower. In determining whether the interest contracted for, charged, or received by the Administrative Agent or a Lender exceeds the Maximum Rate, such Person may, to the extent permitted by applicable law, (a) characterize any payment that is not principal as an expense, fee, or premium rather than interest, (b) exclude voluntary prepayments and the effects thereof, and (c) amortize, prorate, allocate, and spread in equal or unequal parts the total amount of interest throughout the contemplated term of the Obligations hereunder. Section 9.18 Additional Borrowers. (a) Parent may designate any wholly-owned Restricted Subsidiary as a Borrower hereunder with respect to the Revolving Facility and/or any Incremental Revolving Commitments (and Incremental Revolving Loans) or any Incremental Term Loan Commitments or Incremental Term Loans (other than Incremental Term Loans that are not Other Term Loans); provided, however, that such wholly-owned Restricted Subsidiary shall be organized under the laws of (i) the same jurisdiction under which any other Borrower is organized or (ii) otherwise, a jurisdiction that is reasonably acceptable to the (x) Administrative Agent and (y)(1) in the case of an Additional Borrower with respect to the Revolving Facility, each of the Lenders under the Revolving Facility and (2) in the case of an Additional Borrower with respect to any Incremental Term Loans that are Other Term Loans, the Incremental Term Lenders with respect to such Incremental Term Loans. Such wholly-owned Restricted Subsidiary shall become an Additional Borrower and a party to this Agreement by delivering to the Administrative Agent an Additional Borrower Joinder, and all references to the “Borrower” shall also include such Additional Borrower, as applicable, upon (a) the receipt by the Administrative Agent of (i) copies, certified by the secretary or assistant secretary of such Additional Borrower, of resolutions of the board of directors or similar governing body of such Additional Borrower approving this Agreement and any other Loan Documents to which such Additional Borrower is becoming a party and performing the obligations thereunder and such other documents and certificates as the Administrative Agent or its counsel may reasonably request relating to the organization and existence of such Additional Borrower; (ii) an incumbency certificate, executed by the secretary or assistant secretary of such Additional Borrower, which shall identify by name and title and bear the signature of the officers of such Additional Borrower authorized to request Borrowings hereunder and sign this Agreement and the other Loan Documents to which such Additional Borrower is becoming a party, upon which certificate the Administrative Agent and the Lenders shall be entitled to rely until informed of any change in writing by Parent or such Additional Borrower, as applicable; (iii) opinions of counsel to such Additional Borrower, in form and substance reasonably satisfactory to the Administrative Agent and its counsel, with respect to the laws of its jurisdiction of organization and such other customary matters as are reasonably requested by counsel to the Administrative Agent and addressed to the Administrative Agent and the Lenders; (iv) at least three (3) Business

207 Days prior to such designation, any other instruments and documents reasonably requested by the Administrative Agent and each Lender under applicable “know-your-customer” or similar rules and regulations, including the USA Patriot Act and the Beneficial Ownership Regulation; and (v) a certificate from Parent and such Additional Borrower certifying that as of the date of such joinder, the conditions set forth in Section 4.02(a) and (b) shall be met as if a Credit Event were to occur on such date and (b) the Lenders being provided with ten (10) Business Days’ prior notice (or such shorter period of time as the Administrative Agent shall reasonably agree) of any Additional Borrower being proposed to be added pursuant to this Section 9.18(a). This Agreement may be amended as necessary or appropriate, in the reasonable opinion of the Administrative Agent and Parent to effect the provisions of or be consistent with this Section 9.18(a). Notwithstanding any other provision of this Agreement to the contrary, any such deemed amendment may be memorialized in writing by the Administrative Agent with Parent’s consent, but without the consent of any other Lenders and furnished to the other parties hereto. (b) Notwithstanding anything to the contrary contained in this Agreement, the parties hereto agree that any US Borrower shall only be jointly and severally liable with respect to the US Borrowings and shall not be jointly and severally liable with respect to any Loans and Obligations of any Borrower that is not a US Borrower. (c) Notwithstanding anything to the contrary contained in this Agreement (but subject to subsection (b) of this Section 9.18), the parties hereto agree that the Borrower Representative shall be a co-borrower with respect to all Loans and other Obligations of any Additional Borrowers hereunder, and each reference herein to the “Additional Borrower(s)” or the “Borrower(s)” with respect to any Loans (other than Revolving Loans and related extensions of credit incurred directly by any Additional Borrower) or Obligations of any Additional Borrower hereunder shall be deemed to be a reference to any Additional Borrower and the Borrower Representative, jointly and severally. Subject to subsection (b) of this Section 9.18, each Additional Borrower and the Borrower Representative shall be jointly and severally liable for all such Loans and other Obligations, regardless of which Borrower actually receives the benefit thereof or the manner in which they account for such Loans and Obligations on their books and records. Upon the commencement and during the continuation of any Event of Default, the Administrative Agent and the applicable Lenders may (in accordance with the terms of this Agreement and the other Loan Documents) proceed directly and at once, without notice, against any Additional Borrower or the Borrower Representative to collect and recover the full amount, or any portion of, such Obligations, without first proceeding against the other Borrower(s) or any other Person, or any security or collateral for such Obligations, subject, however, to subsection (b) of this Section 9.18. Each Additional Borrower and the Borrower Representative consents and agrees that neither the Administrative Agent nor the Lenders shall be under any obligation to marshal any assets in favor of any Borrower or against or in payment of any or all of such Obligations. Section 9.19 Acknowledgement and Consent to Bail-In of Affected Financial Institution. Notwithstanding anything to the contrary in any Loan Document or in any other agreement, arrangement or understanding among any such parties, each party hereto acknowledges that any liability of any Affected Financial Institution arising under any Loan Document, to the extent such liability is unsecured, may be subject to the write-down and conversion powers of the

208 applicable Resolution Authority and agrees and consents to, and acknowledges and agrees to be bound by: (a) the application of any Write-Down and Conversion Powers by the applicable Resolution Authority to any such liabilities arising hereunder which may be payable to it by any party hereto that is an Affected Financial Institution; and (b) the effects of any Bail-In Action on any such liability, including, if applicable: (i) a reduction in full or in part or cancellation of any such liability; (ii) a conversion of all, or a portion of, such liability into shares or other instruments of ownership in such Affected Financial Institution, its parent undertaking, or a bridge institution that may be issued to it or otherwise conferred on it, and that such shares or other instruments of ownership will be accepted by it in lieu of any rights with respect to any such liability under this Agreement or any other Loan Document; or (iii) the variation of the terms of such liability in connection with the exercise of the write-down and conversion powers of the applicable Resolution Authority. Section 9.20 Intercreditor Agreement. Notwithstanding anything to the contrary contained herein, the Administrative Agent, the Collateral Agent, the Issuing Bank and each Lender hereby acknowledges that the Liens and security interests securing the Secured Obligations, the exercise of any right or remedy by the Collateral Agent under the Loan Documents or with respect thereto, and certain rights of the parties thereto are subject to the provisions of the Intercreditor Agreement and any Approved Intercreditor Agreement that has been entered into by the Administrative Agent and the Collateral Agent pursuant to the terms hereof. In the event of any conflict between the terms of the Intercreditor Agreement or any such Approved Intercreditor Agreement and the terms of this Agreement or any other Loan Document with respect to the priority of any Liens granted to the Collateral Agent or the exercise of any rights and remedies of the Collateral Agent, the terms of the Intercreditor Agreement and such applicable Approved Intercreditor Agreements shall govern and control. Section 9.21 Certain ERISA Matters. (a) Each Lender (x) represents and warrants, as of the date such person became a Lender party hereto, to, and (y) covenants, from the date such person became a Lender party hereto to the date such person ceases being a Lender party hereto, for the benefit of the Administrative Agent, the Collateral Agent and their respective Affiliates, and not, for the avoidance of doubt, to or for the benefit of any Loan Party, that at least one of the following is and will be true:

209 (i) such Lender is not using “plan assets” (within the meaning of Section 3(42) of ERISA or otherwise) of one or more Benefit Plans in connection with the Loans, the Letters of Credit, the Commitments or this Agreement, (ii) the transaction exemption set forth in one or more PTEs, such as PTE 84-14 (a class exemption for certain transactions determined by independent qualified professional asset managers), PTE 95-60 (a class exemption for certain transactions involving insurance company general accounts), PTE 90-1 (a class exemption for certain transactions involving insurance company pooled separate accounts), PTE 91- 38 (a class exemption for certain transactions involving bank collective investment funds) or PTE 96-23 (a class exemption for certain transactions determined by in-house asset managers), is applicable with respect to such Lender’s entrance into, participation in, administration of and performance of the Loans, the Letters of Credit, the Commitments, this Agreement and the Loan Documents, (iii) (A) such Lender is an investment fund managed by a “Qualified Professional Asset Manager” (within the meaning of Part VI of PTE 84-14), (B) such Qualified Professional Asset Manager made the investment decision on behalf of such Lender to enter into, participate in, administer and perform the Loans, the Letters of Credit, the Commitments and this Agreement, (C) the entrance into, participation in, administration of and performance of the Loans, the Letters of Credit, the Commitments and this Agreement satisfies the requirements of sub-sections (b) through (g) of Part I of PTE 84-14 and (D) to the best knowledge of such Lender, the requirements of subsection (a) of Part I of PTE 84-14 are satisfied with respect to such Lender’s entrance into, participation in, administration of and performance of the Loans, the Letters of Credit, the Commitments, this Agreement and the Loan Documents, or (iv) such other representation, warranty and covenant as may be agreed in writing between the Administrative Agent, in its sole discretion, and such Lender. (b) In addition, unless either (1) sub-clause (i) in the immediately preceding clause (a) is true with respect to a Lender or (2) a Lender has provided another representation, warranty and covenant in accordance with sub-clause (iv) in the immediately preceding clause (a), such Lender further (x) represents and warrants, as of the date such person became a Lender party hereto, to, and (y) covenants, from the date such person became a Lender party hereto to the date such person ceases being a Lender, for the benefit of the Administrative Agent, each Arranger and their respective Affiliates, and not, for the avoidance of doubt, to or for the benefit of the Loan Parties, that none of the Administrative Agent, any Arranger or any of their respective Affiliates is a fiduciary with respect to the assets of such Lender involved in such Lender’s entrance into, participation in, administration of and performance of the Loans, the Letters of Credit, the Commitments, this Agreement and the Loan Documents (including in connection with the reservation of exercise or any rights by the Administrative Agent under this Agreement, any Loan Document or any documents related hereto or thereto). Section 9.22 Acknowledgment Regarding Any Supported QFCs. To the extent that the Loan Documents provide support, through a guarantee or otherwise, for Swap Agreements or any other agreement or instrument that is a QFC (such support, “QFC Credit Support” and, each such

210 QFC, a “Supported QFC”), the parties acknowledge and agree as follows with respect to the resolution power of the Federal Deposit Insurance Corporation under the Federal Deposit Insurance Act and Title II of the Dodd-Frank Wall Street Reform and Consumer Protection Act (together with the regulations promulgated thereunder, the “U.S. Special Resolution Regimes”) in respect of such Supported QFC and QFC Credit Support (with the provisions below applicable notwithstanding that the Loan Documents and any Supported QFC may in fact be stated to be governed by the laws of the State of New York and/or of the United States or any other state of the United States): (a) In the event a Covered Entity that is party to a Supported QFC (each, a “Covered Party”) becomes subject to a proceeding under a U.S. Special Resolution Regime, the transfer of such Supported QFC and the benefit of such QFC Credit Support (and any interest and obligation in or under such Supported QFC and such QFC Credit Support, and any rights in property securing such Supported QFC or such QFC Credit Support) from such Covered Party will be effective to the same extent as the transfer would be effective under the U.S. Special Resolution Regime if the Supported QFC and such QFC Credit Support (and any such interest, obligation and rights in property) were governed by the laws of the United States or a state of the United States. In the event a Covered Party or a BHC Act Affiliate of a Covered Party becomes subject to a proceeding under a U.S. Special Resolution Regime, Default Rights under the Loan Documents that might otherwise apply to such Supported QFC or any QFC Credit Support that may be exercised against such Covered Party are permitted to be exercised to no greater extent than such Default Rights could be exercised under the U.S. Special Resolution Regime if the Supported QFC and the Loan Documents were governed by the laws of the United States or a state of the United States. Without limitation of the foregoing, it is understood and agreed that rights and remedies of the parties with respect to a Defaulting Lender shall in no event affect the rights of any Covered Party with respect to a Supported QFC or any QFC Credit Support; and (b) As used in this Section 9.22, the following terms have the following meanings: “BHC Act Affiliate” of a party means an “affiliate” (as such term is defined under, and interpreted in accordance with, 12 U.S.C. 1841(k)) of such party. “Covered Entity” means any of the following: (i) a “covered entity” as that term is defined in, and interpreted in accordance with, 12 C.F.R. § 252.82(b); (ii) a “covered bank” as that term is defined in, and interpreted in accordance with, 12 C.F.R. § 47.3(b); or (iii) a “covered FSI” as that term is defined in, and interpreted in accordance with, 12 C.F.R. § 382.2(b). “Default Right” has the meaning assigned to that term in, and shall be interpreted in accordance with, 12 C.F.R. §§ 252.81, 47.2 or 382.1, as applicable.

211 “QFC” has the meaning assigned to the term “qualified financial contract” in, and shall be interpreted in accordance with, 12 U.S.C. 5390(c)(8)(D). ARTICLE X PARENT GUARANTY Section 10.01 Guaranty. Parent hereby guarantees to each Secured Party as hereinafter provided, as primary obligor and not as surety, the payment of the Secured Obligations in full in cash when due (whether at stated maturity, as a mandatory prepayment, by acceleration, as a mandatory cash collateralization or otherwise) strictly in accordance with the terms thereof. Parent hereby further agrees that if any of the Secured Obligations are not paid in full in cash when due (whether at stated maturity, as a mandatory prepayment, by acceleration, as a mandatory cash collateralization or otherwise), Parent will promptly pay the same, without any demand or notice whatsoever, and that in the case of any extension of time of payment or renewal of any of the Secured Obligations, the same will be promptly paid in full in cash when due (whether at extended maturity, as a mandatory prepayment, by acceleration, as a mandatory cash collateralization or otherwise) in accordance with the terms of such extension or renewal. Section 10.02 Obligations Unconditional. (a) The obligations of Parent under Section 10.01 are absolute and unconditional, irrespective of the value, genuineness, validity, regularity or enforceability of any of the Loan Documents or other documents relating to the Secured Obligations, or any substitution, release, impairment or exchange of any other guarantee of or security for any of the Secured Obligations, and, to the fullest extent permitted by applicable law, irrespective of any other circumstance whatsoever which might otherwise constitute a legal or equitable discharge or defense of a surety or guarantor (other than payment in full in cash of the Secured Obligations, other than contingent indemnification, tax gross up, expense reimbursement or yield protection obligations, in each case, for which no claim has been made), it being the intent of this Section 10.02 that the obligations of Parent hereunder shall be absolute and unconditional under any and all circumstances. Parent agrees that it shall have no right of subrogation, indemnity, reimbursement or contribution against a Borrower or any other Guarantor for amounts paid under this Article X until such time as the Secured Obligations have been paid in full in cash and the Commitments have expired or terminated. (b) Without limiting the generality of the foregoing, it is agreed that, to the fullest extent permitted by law, the occurrence of any one or more of the following shall not alter or impair the liability of Parent hereunder, which shall remain absolute and unconditional as described above: (i) at any time or from time to time, without notice to Parent the time for any performance of or compliance with any of the Secured Obligations shall be extended, or such performance or compliance shall be waived; (ii) any of the acts mentioned in any of the provisions of any of the Loan Documents or other documents relating to the Secured Obligations shall be done or omitted;

212 (iii) the maturity of any of the Secured Obligations shall be accelerated, or any of the Secured Obligations shall be modified, supplemented or amended in any respect, or any right under any of the Loan Documents or other documents relating to the Secured Obligations shall be waived or any other guarantee of any of the Secured Obligations or any security therefor shall be released, impaired or exchanged in whole or in part or otherwise dealt with; (iv) any Lien granted to, or in favor of, the Collateral Agent or any other holder of the Secured Obligations as security for any of the Secured Obligations shall fail to attach or be perfected; or (v) any of the Secured Obligations shall be determined to be void or voidable (including, without limitation, for the benefit of any creditor of Parent) or shall be subordinated to the claims of any Person (including, without limitation, any creditor of Parent). (c) With respect to its obligations hereunder, Parent hereby expressly waives diligence, presentment, demand of payment, protest and all notices whatsoever, and any requirement that the Administrative Agent, the Collateral Agent or any other holder of the Secured Obligations exhaust any right, power or remedy or proceed against any Person under any of the Loan Documents or other documents relating to the Secured Obligations, or against any other Person under any other guarantee of, or security for, any of the Secured Obligations. Section 10.03 Reinstatement. The obligations of Parent under this Article X shall be automatically reinstated if and to the extent that for any reason any payment by or on behalf of any Person in respect of the Secured Obligations is rescinded or must be otherwise restored by any holder of any of the Secured Obligations, whether as a result of any proceedings under any Debtor Relief Law, and Parent agrees that it will indemnify the Administrative Agent and each holder of the Secured Obligations on demand for all reasonable costs and expenses (including, without limitation, the fees, charges and disbursements of counsel) incurred by the Administrative Agent or such holder of the Secured Obligations in connection with such rescission or restoration, including any such costs and expenses incurred in defending against any claim alleging that such payment constituted a preference, fraudulent transfer or similar payment under any proceedings under any debtor relief law. Section 10.04 Certain Additional Waivers. Parent further agrees that it shall have no right of recourse to security for the Secured Obligations, except through the exercise of rights of subrogation pursuant to Section 10.02 and through the exercise of rights of contribution pursuant to Section 10.06. Section 10.05 Remedies. Parent agrees that, to the fullest extent permitted by law, as between Parent, on the one hand, and the Administrative Agent, the Collateral Agent and the other holders of the Secured Obligations, on the other hand, the Secured Obligations may be declared to be forthwith due and payable as provided in Section 7.01 (and shall be deemed to have become automatically due and payable in the circumstances provided in said Article VII) for purposes of Section 10.01 notwithstanding any stay, injunction or other prohibition preventing such declaration (or preventing the Secured Obligations from becoming automatically due and payable)

213 as against any other Person and that, in the event of such declaration (or the Secured Obligations being deemed to have become automatically due and payable), the Secured Obligations (whether or not due and payable by any other Person) shall forthwith become due and payable by Parent for purposes of Section 10.01. Parent acknowledges and agrees that its obligations hereunder are secured in accordance with the terms of the Collateral Documents and that the holders of the Secured Obligations may exercise their remedies thereunder in accordance with the terms thereof. Section 10.06 Rights of Contribution. Parent agrees that, in connection with payments made hereunder, Parent shall have contribution rights against the other Guarantors as permitted under applicable law. Such contribution rights shall be subordinate and subject in right of payment to the obligations of such Guarantors under the Loan Documents and no Guarantor shall exercise such rights of contribution until all Secured Obligations have been paid in full in cash and the Commitments have terminated. Section 10.07 Guarantee of Payment; Continuing Guarantee. The guarantee given by Parent, in this Article X is a guaranty of payment and not of collection, is a continuing guarantee, and shall apply to all Secured Obligations whenever arising. [Remainder ofSignature Pages Intentionally Left BlankOmitted]

[Signature Page to Credit Agreement] IN WITNESS WHEREOF, the parties hereto have caused this Agreement to be duly executed and delivered by their duly authorized Responsible Officers as of the date first set forth above. ENDO FINANCE HOLDINGS, INC., as the Borrower Representative By: Name: Title: ENDO, INC., as Parent By: Name: Title:

[Signature Page to Credit Agreement] GOLDMAN SACHS BANK USA, as Administrative Agent, Collateral Agent, Issuing Bank and Swingline Lender By: Name: Title:

[Signature Page to Credit Agreement] [LENDER], as a Lender By: Name: Title:
| Exhibit 10.51 | ||
Execution Version | ||
First Lien Intercreditor Agreement Joinder
SUPPLEMENT NO. 4 dated as of December 17, 2025 (this “Supplement”), to the FIRST LIEN INTERCREDITOR AGREEMENT dated as of April 23, 2024 (the “First Lien Intercreditor Agreement”), among Endo LP (f/k/a Endo, Inc.) (“Holdings”), Endo Finance Holdings LP (f/k/a Endo Finance Holdings, Inc.) (“Borrower”), the other Grantors party thereto, Goldman Sachs Bank USA, as collateral agent for the Credit Agreement Secured Parties (in such capacity and together with its successors in such capacity, the “Bank Collateral Agent”), and Computershare Trust Company, National Association, as collateral agent for the Indenture Secured Parties (in such capacity and together with its successors in such capacity, the “Notes Collateral Agent”), and each Additional Agent from time to time party thereto.
A Capitalized terms used herein and not otherwise defined herein shall have the meanings assigned to such terms in the First Lien Intercreditor Agreement.
B. The Grantors have entered into the First Lien Intercreditor Agreement. Pursuant to certain Secured Debt Documents, certain newly acquired or organized Subsidiaries of Holdings are required to enter into the First Lien Intercreditor Agreement. Section 5.16 of the First Lien Intercreditor Agreement provides that such Subsidiaries may become party to the First Lien Intercreditor Agreement by execution and delivery of an instrument in the form of this Supplement. The undersigned Subsidiaries listed on Schedules I and II attached hereto (the “New Grantors” and each a “New Guarantor”) are each executing this Supplement in accordance with the requirements of the Credit Agreement, the Indenture and the Additional First Lien Documents.
Accordingly, the Controlling Collateral Agent and each New Grantor agree as follows:
SECTION 1. In accordance with Section 5.16 of the First Lien Intercreditor Agreement, each New Grantor by its signature below becomes a Grantor under the First Lien Intercreditor Agreement with the same force and effect as if originally named therein as a Grantor, and each New Grantor hereby agrees to all the terms and provisions of the First Lien Intercreditor Agreement applicable to it as a Grantor thereunder. Each reference to a “Grantor” in the First Lien Intercreditor Agreement shall be deemed to include the New Grantors. The First Lien Intercreditor Agreement is hereby incorporated herein by reference.
SECTION 2. Each New Grantor represents and warrants to the Controlling Collateral Agent and the other Secured Parties that this Supplement has been duly authorized, executed and delivered by it and constitutes its legal, valid and binding obligation, enforceable against it in accordance with its terms.
SECTION 3. This Supplement may be executed in counterparts, each of which shall constitute an original, but all of which when taken together shall constitute a single contract. This Supplement shall become effective when the Controlling Collateral Agent shall have received a counterpart of this Supplement that bears the signature of each New Grantor. Delivery of an executed signature page to this Supplement by facsimile transmission shall be as
effective as delivery of a manually signed counterpart of this Supplement. Delivery of an executed signature page to this Joinder by facsimile transmission shall be effective as delivery of a manually signed counterpart of this Joinder. Delivery of an executed counterpart of a signature page of this Joinder that is an electronic sound, symbol, or process attached to, or associated with, a contract or other record and adopted by a person with the intent to sign, authenticate or accept such contract or record (an “Electronic Signature”) transmitted by telecopy, emailed pdf or any other electronic means that reproduces an image of an actual executed signature page shall be effective as delivery of a manually executed counterpart of this Joinder. The words “execution,” “signed,” “signature,” “delivery,” and words of like import in or relating to this Joinder shall be deemed to include Electronic Signatures, deliveries or the keeping of records in any electronic form (including deliveries by telecopy, emailed pdf or any other electronic means that reproduces an image of an actual executed signature page), each of which shall be of the same legal effect, validity or enforceability as a manually executed signature, physical delivery thereof or the use of a paper-based recordkeeping system, as the case may be.
SECTION 4. Except as expressly supplemented hereby, the First Lien Intercreditor Agreement shall remain in full force and effect.
SECTION 5. THIS SUPPLEMENT SHALL BE GOVERNED BY, AND CONSTRUED IN ACCORDANCE WITH, THE LAWS OF THE STATE OF NEW YORK.
SECTION 6. In case any one or more of the provisions contained in this Supplement should be held invalid, illegal or unenforceable in any respect, no party hereto shall be required to comply with such provision for so long as such provision is held to be invalid, illegal or unenforceable, but the validity, legality and enforceability of the remaining provisions contained herein and in the First Lien Intercreditor Agreement shall not in any way be affected or impaired. The parties hereto shall endeavor in good-faith negotiations to replace the invalid, illegal or unenforceable provisions with valid provisions the economic effect of which comes as close as possible to that of the invalid, illegal or unenforceable provisions.
SECTION 7. All communications and notices hereunder shall be in writing and given as provided in Section 5.01 of the First Lien Intercreditor Agreement. All communications and notices hereunder to the New Grantor shall be given to it in care of the Company as specified in the First Lien Intercreditor Agreement.
SECTION 8. The Borrower agrees to reimburse the Controlling Collateral Agent for its reasonable and documented out of pocket expenses in connection with this Supplement, including the reasonable and documented fees, other charges and disbursements of counsel for the Controlling Collateral Agent; provided that in no event shall the Borrower be required to reimburse the Controlling Collateral Agent for more than one counsel (and up to one local counsel in each applicable jurisdiction and regulatory counsel).
[Signature pages follow]
IN WITNESS WHEREOF, the New Grantors, and the Controlling Collateral Agent have duly executed this Supplement No. 4 to the First Lien Intercreditor Agreement as of the day and year first above written.
Keenova International Holdings LLC
By: /s/ Matthew Peters___________________
Name: Matthew Peters
Title: Vice President of Tax and Treasurer
Name: Matthew Peters
Title: Vice President of Tax and Treasurer
Sonorant Therapeutics Limited
By: /s/ Alasdair Fenlon___________________
Name: Alasdair Fenlon
Title: Director
Name: Alasdair Fenlon
Title: Director
[Signature Page to First Lien Intercreditor Agreement Joinder Supplement No. 4]
Acknowledged by:
GOLDMAN SACHS BANK USA, as Controlling Collateral Agent, | ||||||||
| By: | /s/ Luke Qiu | |||||||
| Name: Luke Qiu | ||||||||
| Title: Authorized Signatory | ||||||||
[Signature Page to First Lien Intercreditor Agreement Joinder Supplement No. 4]
Schedule I
Domestic Joinder Parties
Company | Jurisdiction of Organization | Type of Organization | ||||||
Keenova International Holdings LLC | Delaware | Limited Liability Company | ||||||
Schedule II
Foreign Joinder Parties
Company | Jurisdiction of Organization | Type of Organization | ||||||
Sonorant Therapeutics Limited | Ireland | Private Limited Company | ||||||
Exhibit 19.1


Effective Date: February 20, 2026
Table of Contents
2
Section 1: Statement of Policy
U.S. securities laws impose strict requirements regarding how we use and disclose material information of or concerning Keenova Therapeutics plc (“Keenova” the “Company”). The Securities and Exchange Commission, U.S. Attorneys and enforcement agencies of other jurisdictions vigorously pursue and punish violations of these insider trading laws. In the course of your job at the Company, you may be privy to nonpublic information about the Company or its subsidiaries, customers, suppliers, acquisition targets or other entities with which the Company does business. For that reason, the Company has adopted this Global Insider Trading Policy, which prohibits trading the Company’s securities, or the securities of any such other publicly traded company, based on material, non-public information or providing such material, non-public information to other persons who may trade on the basis of that information (commonly known as “tipping”).
Section 2: Purpose of this Policy
The purpose of this policy is to ensure that all Covered Persons (as defined below) comply with all applicable laws and regulations concerning securities trading, commonly known as “insider trading.” Insider trading and tipping, are criminal offenses subject to severe criminal and civil consequences as well as possible discipline or dismissal under this policy.
Section 3: Policy Owner and Statement of Responsibility
This policy is owned by the Keenova Legal Department and applies to all Covered Persons. All employees of the Company are responsible for implementation of this policy. The office of the Chief Legal Officer (“CLO”)/General Counsel (“GC”) is specifically responsible for interpreting this policy and implementing the Trading Window provisions of this policy.
Section 4: Procedures for all Covered Persons
This Section 4 shall apply to all of the following (collectively, the “Covered Persons”):
•All members of the Board of Directors of Keenova (“Directors”);
•All employees of Keenova and any of its subsidiaries (“Employees”);
•Any other persons that the Company determines should be Covered Persons for purposes of this policy, such as contractors or consultants, who have access to material nonpublic information (the “Designated Persons”);
•All family members of Directors, Employees and Designated Persons who share the same address as, or are financially dependent on, the Director, Employee or Designated Person and any other person who shares the same address as the Director, Employee or Designated Person (other than (x) an employee or tenant of the Director, Employee or Designated Person or (y) another unrelated person whom the CLO/GC determines should not be covered by this policy). For the purposes of this policy, family members include a spouse, domestic partner, romantic partner, child, child away at college, stepchildren, grandchildren, parents, stepparents, grandparents, siblings, and in-laws, and any family members whose transactions are directed by or under the influence or control of the Director, Employee or Designated Person, such as parents or children who consult with the Director, Employee, or Designated Person before they trade in Keenova securities (“Family Members”);
•All corporations, partnerships, trusts or other entities controlled by any of the above persons, unless the entity has implemented policies or procedures designed to ensure that such person cannot influence securities transactions by the entity (“Controlled Entities”).
Keenova requires all Covered Persons to comply with applicable securities laws. Covered Persons must never:
1.Buy, sell or engage in other transactions in, or recommend the purchase or sale of, Keenova securities while aware of material non-public information about the Company or its subsidiaries.
2.Buy, sell, or engage in other transactions in, or recommend the purchase or sale of, securities of other companies while aware of material non-public information about those companies of which the Covered Person becomes aware in the course of working for the Company, until the information becomes public or is no longer material.
3
3.Disclose material non-public information to persons within Keenova whose jobs do not require them to have that information, or any unauthorized persons outside Keenova, including family, friends, business associates, investors and expert consulting firms – commonly known as “tipping”. Covered Persons are prohibited from “tipping” other persons about material non-public information or otherwise making unauthorized disclosures or use of such information, regardless of whether the person profits or intends to profit by such tipping, disclosure or use. You must take steps to prevent the inadvertent disclosure of material non-public information to persons within Keenova whose jobs do not require them to have that information, or unauthorized persons outside Keenova. If you believe that the disclosure of material non-public information is necessary or appropriate for business reasons, you must consult with Company counsel to ensure that they concur that such disclosure is necessary, and to ensure that any such disclosure will comply with all applicable laws.
4.Assist anyone engaged in the above activities.
Securities transactions executed pursuant to “limit orders,” “good until cancelled orders” or similar market orders are subject to this policy, regardless of when the order was placed. If a Covered Person comes into possession of material non-public information while such a “limit order,” “good until cancelled order” or similar market order is in place, the Covered Person has an affirmative obligation to cancel such order as soon as reasonably practicable. Keenova discourages placing standing or limit orders on the Company’s securities. If a Covered Person determines that they must use a standing order or limit order, the order should be limited to a short duration and should otherwise comply with the restrictions and procedures outlined below in Section 5.
Non-public information is sometimes referred to as confidential information and means information that is not known to the public-at-large. For purposes of this policy, all information about the Company is considered non-public until two full trading days after it has been widely disseminated through a press release, news wire or a public disclosure document filed with the U.S. Securities and Exchange Commission. By contrast, information would likely not be considered widely disseminated if it is available only to the Employees, or if it is only available to a select group of persons. In addition, please be aware that disclosure on the Company’s website or social media channel, by itself, may not necessarily be considered wide dissemination.
Information is considered material if there is a substantial likelihood that a reasonable investor would consider it important in deciding whether to buy, hold, or sell securities. If a Covered Person is motivated to buy, hold or sell a stock because of information they possess, the information likely will be considered material. Any information that reasonably could be expected to affect the price of securities of the Company to which the information pertains will often be viewed as material. It is important also to remember that materiality will be judged in hindsight, typically after the price of securities has moved following the release of information. Material information may be positive or negative.
Examples of material information include:
•expected earnings or revenues for a particular period, as well as Company projections as to future earnings or revenues, or other financial guidance;
•changes to previously announced financial guidance, or the decision to suspend guidance;
•impending bankruptcy or the existence of severe liquidity problems;
•significant new products or product development milestones (such as major clinical trial results or FDA approvals or other actions);
•a proposed major acquisition, disposition or joint venture;
•substantial purchases, sales or write-offs of assets;
•a planned offering of additional new classes of securities or changes in dividend policy or declaration of a stock split;
•pending or threatened significant litigation or investigation, or the resolution of such litigation or investigation;
•significant related party transactions;
•bank borrowings or other financing transactions out of the ordinary course;
•a change in senior management;
•a change in auditors or notification that the auditor’s reports may no longer be relied upon;
•a gain or loss of a major customer or substantial contract award or termination;
•a significant or potentially significant cybersecurity incident; or
•the imposition of a restriction on trading in Company securities or the extension or termination of such restriction.
4
This is only an illustrative list of examples of information that could be material and is not intended to be comprehensive.
While in possession of material non-public information, Covered Persons are prohibited from trading in any Keenova securities as to which such Covered Person has a “beneficial” or financial interest, or over which such Covered Person exercises investment control.
Event-Specific Trading Restrictions
From time to time, the Company may decide to impose a special trading blackout on those who are aware of particular information that the Company determines may be considered material non-public information. This kind of trading blackout may be imposed in connection with a potential acquisition or divestiture or other material development. Persons subject to the blackout may not enter into transactions in Company securities (other than transactions made pursuant to an approved 10b5-1(c) plan, if any) until notified that the blackout has ended.
The CLO/GC, in consultation with senior management, will determine whether an event-specific trading blackout should be imposed. If a person is covered by the event-specific trading blackout, he or she will be notified by the CLO/GC’s office. The existence of an event-specific trading blackout will not be generally announced, and persons made aware of it should not disclose its existence to anyone else. Even if a Covered Person has not been designated by the CLO/CG’s office as a person who should not trade due to an event-specific trading blackout, that Covered Person is still not permitted to trade while aware of material non-public information.
Specific Prohibition Regarding Hedging Transactions
Covered Persons are prohibited from engaging at any time in short sales of Company securities (i.e., the sale of a security that the seller does not own), including short sales “against the box,” or any transactions in puts, calls, equity swaps, prepaid variable forward contracts, cashless collars, options, call options, or similar rights and obligations or any other hedging activity involving Keenova securities, other than the exercise of a Company-issued stock option. In addition, Section 16(c) of the Securities Exchange Act of 1934, as amended ("Exchange Act”) prohibits executive officers and directors from engaging in short sales.
Specific Prohibition Regarding Margin Accounts and Pledges
No Covered Person may purchase Company securities on margin, borrow against Company securities held in a margin account, or pledge Company securities as collateral for a loan. However, an exception may be granted where a person wishes to pledge Company securities as collateral for a loan and clearly demonstrates the financial capacity to repay the loan without resort to the pledged securities. Any person who wishes to pledge Company securities as collateral for a loan must submit a request for approval to the CLO/GC at least 10 business days prior to entering into the transaction.
Transactions Under Company Plans
This policy does not apply in the case of the following transactions, except as specifically noted:
•Company-issued Stock Options. This policy does not apply to the grant, vesting or exercise of a Company-issued stock option, or to the exercise of a tax withholding event pursuant to which a person elected to have the Company withhold shares subject to an option to satisfy tax withholding requirements. This policy does apply, however, to any sale of shares by a Covered Person as part of a broker-assisted cashless exercise of an option, or any other sale of shares received upon exercise of an option.
•Restricted Unit or Performance Unit Awards. This policy does not apply to the grant or vesting of restricted units or performance units, or the withholding of shares to satisfy tax withholding requirements upon vesting. This policy does apply, however, to any sale of shares to third parties (i.e., parties other than the Company) received upon vesting of restricted units or performance units.
•Other Similar Transactions. Any other purchases of Company securities from the Company (such as pursuant to a “cash bonus for stock exchange” program) or sales of Company securities to the Company are not subject to this policy.
Gifts
Bona fide gifts of Company securities to persons covered by this policy (e.g., family members and family trusts) are not transactions prohibited by this policy. Such recipient's sale of the gifted securities would be subject to this policy, however. Gifts to persons or entities not covered by this policy, including charitable gifts, are not permitted while the Covered Person is aware of material non-public information or while the Covered Person is subject to trading restrictions under this policy. Members of the Window Group (as defined below) must pre-clear any gift transactions with the CLO/GC’s office.
5
External Inquiries
In the event that a Covered Person is contacted by any governmental entity or any other external party regarding any transactions in Company securities undertaken by such Covered Person, then the Covered Person must promptly notify the CLO/GC of such contact, including all material details. Notwithstanding the foregoing, a Covered Person is not required to notify the CLO/GC if they reported any alleged violations of this policy or violations of law to any governmental entity or the details of what they reported to a governmental entity.
Section 5: Special Procedures for the Window Group
The Company imposes certain special restrictions on specified senior officers, management and directors in trading Keenova securities. These restrictions govern even though the transactions may be permissible under law, and apply to the following persons, hereafter defined as the “Window Group”:
•All Directors;
•All executive officers of the Company (“Executives”);
•All Presidents of Global Business Units (“Presidents”);
•Any other employees designated by members of the Window Group (“Designees”);
•All Family Members of Directors, Executives, Presidents or Designees; and
•All Controlled Entities of any of the above persons.
The CLO/GC shall have the discretion to determine if any employee shall be designated as a member of the Window Group and thus subject to the Special Procedures set forth in this Section 5.
Other than transactions made subject to an approved 10b5-1(c) plan, members of the Window Group may only enter into transactions in Keenova securities during an open trading window that commences two full trading days after the public release of the Company’s quarterly or annual financial results and ends at 11:59 p.m. U.S. Eastern Time on the 17th day of the third month of the fiscal quarter in which the release occurs.
The Company will conduct an evaluation each quarter to determine whether the scheduled trading window should be cancelled. The Company may close an open trading window early at any time, as deemed appropriate by the CLO/GC in consultation with senior management. Even when the trading window is open, members of the Window Group cannot trade if they in fact possess material non-public information (unless the trade is pursuant to a previously approved 10b5-1(c) plan, as described below).
The CLO/GC or his or her designee may implement procedures to inform the Window Group each quarter of the opening and closing of the trading window.
Pre-Approval for Directors, Section 16 Officers and Certain Finance Employees
Even within the trading window, all Directors, Section 16 Officers (as defined below), the Corporate Controller, Head of Investor Relations, Corporate Treasurer and Vice President of Financial Planning and Analysis must pre-clear all trades with the CLO/GC’s office. A request for pre-clearance should be submitted to the CLO/GC’s office at least two business days before the proposed transaction. The CLO/GC and any of his or her designees are under no obligation to approve a transaction submitted for pre-clearance and may determine not to permit the transaction. Furthermore, pre-clearance of a trade does not, in any circumstance, relieve anyone of their legal obligation to refrain from trading while in possession of material non-public information and therefore is not a defense to a claim of insider trading and does not excuse anyone from otherwise complying with insider trading laws or this policy. Any transaction pre-cleared must be executed within five business days after receiving pre-clearance approval. If the transaction does not take place during that time, the individual must re-request pre-clearance through the same process. If a request for pre-clearance is denied, then the member of the Window Group must refrain from engaging in any transaction in Company securities, and should not inform any other person of the restriction.
Transactions under Rule 10b5-1(c)
Rule 10b5-1(c) under the Exchange Act provides a defense from insider trading liability if certain conditions are met. If a member of the Window Group wishes to trade pursuant to a 10b5-1(c) plan, he or she must obtain the approval of the CLO/GC prior to entering into the 10b5-1(c) plan. The CLO/GC or his or her designee, must review the 10b5-1(c) plan for compliance with applicable law, including Rule 10b5-1(c), this policy and the Company’s securities ownership and retention guidelines (if any). In addition, a member of the Window Group may only enter into, modify or terminate the 10b5-1(c) plan during an open trading window and while not in possession of any material non-public information, and once the 10b5-1(c) plan is adopted, may not exercise any influence over the amount of securities to be traded, the price at
6
which they are to be traded, or the trade date. The plan must (a) specify the amount, pricing and timing of transactions in advance, (b) include a written formula, algorithm or computer program for determining the amount, pricing and timing of transactions, or (c) delegate discretion on these matters to an independent third party such that the member of the Window Group is not permitted to exercise any subsequent influence over such matters, and otherwise meet the requirements of Rule 10b5-1(c). Any member of the Window Group who enters into a 10b5-1(c) plan pursuant to the requirements set forth above must also obtain approval of the CLO/GC prior to modifying or terminating a 10b5-1(c) plan. A request for approval of the entry into or modification or termination of a 10b5-1(c) plan should be submitted to the CLO/GC’s office at least ten business days in advance of the proposed date of such action.
Director and Section 16 Officer Reporting Requirements
Certain members of the Window Group are subject to Section 16 of the Exchange Act (“Directors and Section 16 Officers”) and Rule 144 of the Securities Act of 1933, as amended, applicable to affiliates of the Company. Directors and Section 16 Officers are required to notify the CLO/GC of the occurrence of any purchase, sale or other acquisition or disposition of Company securities as soon as possible following the transaction, but in any event within one business day after executing the transaction. Such notification may be oral or in writing (including by e-mail) and should include the identity of the Director or Section 16 Officer, the type of transaction, the date of the transaction, the number of shares involved and the purchase or sale price (if any).
Section 6: Post-Termination Transactions
The prohibitions in this policy apply to transactions in Company securities even after termination of service to or employment with Keenova. If a Covered Person is in possession of material non-public information when his or her service or employment terminates, that individual may not trade in Company securities until that information becomes public or is no longer material.
Section 7: Exceptions
There are no exceptions to this policy. Transactions that may be necessary or justifiable for independent reasons (such as the need to raise money for an emergency expenditure), or small transactions, are not exempted from this policy. The securities laws do not recognize any mitigating circumstances, and, in any event, even the appearance of an improper transaction must be avoided to preserve Keenova’s reputation for adhering to the highest standards of conduct. Each individual is responsible for making sure that they comply with this policy, and that any Family Member or Controlled Entity whose transactions are subject to this policy also comply with this policy.
Section 8: Consequences of Violating the Policy
In addition to the serious sanctions, including significant fines and imprisonment, imposed by law or regulation, violations of this policy are grounds for disciplinary action, up to and including immediate termination of employment for cause.
Section 9: Who to Contact
For any questions related to this policy or its application to you, please contact either the CLO/GC or the Office of Corporate Secretary.
Section 10: Certification
All persons subject to this policy must certify their understanding of, and intent to comply with, this policy in the form attached hereto as Exhibit A. The Company may require you to sign this Certification on an annual basis, including in electronic format. Please note that you are bound by the policy whether or not you sign the Certification.
Section 11: Revision History
7
| Revision Number | Effective Date | Author(s) | ||||||
| Adopted as part of the Company’s separation from Covidien | July 1, 2013 | Miriam Singer, Corporate Secretary | ||||||
| Amended and Restated | September 1, 2016 | Kenneth L. Wagner, Corporate Secretary | ||||||
| Amended and Restated | September 6, 2019 | Stephanie D. Miller, Corporate Secretary | ||||||
| Amended and Restated | December 17, 2020 | Stephanie D. Miller, Corporate Secretary | ||||||
| Amended and Restated | June 1, 2023 | Mark Tyndall, EVP, Chief Legal Officer & Corporate Secretary | ||||||
| Amended and Restated | January 7, 2025 | Mark Tyndall, EVP, Chief Legal Officer & Corporate Secretary | ||||||
| Amended and Restated | February 20, 2026 | Mark Tyndall, EVP, Chief Legal Officer & Corporate Secretary | ||||||
8
EXHIBIT A
CERTIFICATION
I hereby certify that:
•I have read and understand the Global Insider Trading Policy to which this Certification is attached.
•Since the effective date of the Global Insider Trading Policy, or such shorter period of time that I have been a director, officer, employee or independent contractor of the Company, I have complied in all respects with the Global Insider Trading Policy.
•I will continue to comply with the Global Insider Trading Policy for as long as I am a director, officer, employee or independent contractor of the Company.
| Signature | Date | |||||||
| Printed Name (Please print legibly) | ||||||||
| Title | ||||||||
1
Exhibit 21.1
SUBSIDIARIES OF KEENOVA THERAPEUTICS PLC
The following is a list of subsidiaries of Keenova Therapeutics plc as of December 31, 2025.
| Name of Subsidiary | Jurisdiction of Incorporation/Organization | |||||||
| BP USA Holdings, LLC | United States | |||||||
| Cache Holdings Limited | Bermuda | |||||||
| Carnforth Limited | Bermuda | |||||||
| CPEC, LLC | United States | |||||||
| EFHI GP Limited | Ireland | |||||||
| ELP 2025 GP Limited | Ireland | |||||||
| Endo Biologics Limited | Ireland | |||||||
| Endo Enterprise, Inc. | United States | |||||||
| Endo Finance Holdings LP | United States | |||||||
| Endo LP | United States | |||||||
| Endo Operations Limited | Ireland | |||||||
| Endo USA, Inc. | United States | |||||||
| Ikaria Australia Pty Ltd | Australia | |||||||
| Ikaria Canada Inc. | Canada | |||||||
| Infacare Pharmaceutical Corporation | United States | |||||||
| INO Therapeutics LLC | United States | |||||||
| Keenova International Holdings LLC | United States | |||||||
| Keenova Therapeutics Group Limited | Ireland | |||||||
| Keenova Therapeutics Public Limited Company | Ireland | |||||||
| KT Finance Inc. | United States | |||||||
| Ludlow LLC | United States | |||||||
| MAK LLC | United States | |||||||
| Mallinckrodt ARD Holdings Inc. | United States | |||||||
| Mallinckrodt ARD Holdings Limited | United Kingdom | |||||||
| Mallinckrodt ARD LLC | United States | |||||||
| Mallinckrodt Brand Pharmaceuticals LLC | United States | |||||||
| Mallinckrodt Canada Cooperatie U.A. | Netherlands | |||||||
| Mallinckrodt CB LLC | United States | |||||||
| Mallinckrodt Critical Care Finance LLC | United States | |||||||
| Mallinckrodt Equinox Finance LLC | United States | |||||||
| Mallinckrodt Equinox Limited | United Kingdom | |||||||
| Mallinckrodt Finance Management Ireland Limited | Ireland | |||||||
| Mallinckrodt Group S.à r.l. | Luxembourg | |||||||
| Mallinckrodt Group S.à r.l., Luxembourg (LU) Schaffhausen Branch | Switzerland | |||||||
| Mallinckrodt Holdings GmbH | Switzerland | |||||||
| Mallinckrodt Hospital Products Inc. | United States | |||||||
| Mallinckrodt International Finance SA | Luxembourg | |||||||
| Mallinckrodt International Holdings S.à r.l. | Luxembourg | |||||||
| Mallinckrodt Lux IP S.à r.l. | Luxembourg | |||||||
| Mallinckrodt Manufacturing LLC | United States | |||||||
| Mallinckrodt Medical Holdings (UK) Limited | United Kingdom | |||||||
| Mallinckrodt Medical Holdings (UK) Limited, Zweigniederlassung Deutschland (the German Branch) | Germany | |||||||
| Mallinckrodt Petten Holdings B.V. | Netherlands | |||||||
| Mallinckrodt Pharma IP Trading Limited | Ireland | |||||||
| Mallinckrodt Pharma K.K. | Japan | |||||||
| Mallinckrodt Pharmaceuticals Ireland Limited | Ireland | |||||||
| Mallinckrodt Pharmaceuticals Limited | United Kingdom | |||||||
| Mallinckrodt Quincy S.à r.l. | Luxembourg | |||||||
| Mallinckrodt SAG Holdings GmbH | Switzerland | |||||||
| Mallinckrodt UK Finance LLP | United Kingdom | |||||||
| Mallinckrodt UK Ltd | United Kingdom | |||||||
| Mallinckrodt US Pool LLC | United States | |||||||
| Mallinckrodt Windsor S.à r.l. | Luxembourg | |||||||
| MCCH LLC | United States | |||||||
| MHP Finance LLC | United States | |||||||
| MNK 2011 LLC | United States | |||||||
| Montjeu Limited | Ireland | |||||||
| OCERA Therapeutics LLC | United States | |||||||
| Operand Pharmaceuticals Holdco I Unlimited Company | Ireland | |||||||
| Paladin Pharma Inc. | Canada | |||||||
| Petten Holdings Inc. | United States | |||||||
| Profibrix B.V. | Netherlands | |||||||
| Questcor International Limited | Ireland | |||||||
| Salvare Merger Sub LLC | United States | |||||||
| Sonorant Therapeutics Limited | Ireland | |||||||
| ST Operations LLC | United States | |||||||
| ST Shared Services LLC | United States | |||||||
| ST US AR Finance LLC | United States | |||||||
| ST US Holdings LLC | United States | |||||||
| ST US Pool LLC | United States | |||||||
| Stratatech Corporation | United States | |||||||
| Sucampo Finance Inc. | United States | |||||||
| Sucampo GmbH | Switzerland | |||||||
| Sucampo Holdings Inc. | United States | |||||||
| Sucampo International Holdings Limited | United Kingdom | |||||||
| Sucampo Pharma Americas LLC | United States | |||||||
| Sucampo Pharma, LLC | Japan | |||||||
| Sucampo Pharmaceuticals LLC | United States | |||||||
| Vtesse LLC | United States | |||||||
Exhibit 23.1
CONSENT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
We hereby consent to the incorporation by reference in the Registration Statement on Form S-8 (Nos. 333-289602 and 333-286694) of Keenova Therapeutics plc of our report dated April 15, 2026 relating to the financial statements, which appears in this Form 10-K.
/s/ PricewaterhouseCoopers LLP
Florham Park, New Jersey
April 15, 2026
Florham Park, New Jersey
April 15, 2026
Exhibit 23.2
CONSENT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
We consent to the incorporation by reference in Registration Statement Nos. 333-289602 and 333-286694 on Form S-8 of our report dated March 26, 2024 (March 12, 2025 as to the segment disclosures in Note 22 and April 15, 2026 as to the divestitures disclosures in Note 6), relating to the financial statements of Keenova Therapeutics plc (formerly known as Mallinckrodt plc) appearing in this Annual Report on Form 10-K for the year ended December 31, 2025.
/s/ Deloitte & Touche LLP
St. Louis, Missouri
April 15, 2026
Exhibit 31.1
CERTIFICATION OF CHIEF EXECUTIVE OFFICER
PURSUANT TO RULE 13A-14(A) OF THE SECURITIES EXCHANGE ACT OF 1934
I, Sigurdur Olafsson, certify that:
1.I have reviewed this annual report on Form 10-K of Keenova Therapeutics plc;
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant's other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
a.Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
b.Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c.Evaluated the effectiveness of the registrant's disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d.Disclosed in this report any change in the registrant's internal control over financial reporting that occurred during the registrant's most recent fiscal quarter (the registrant's fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant's internal control over financial reporting; and
5.The registrant's other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant's auditors and the audit committee of the registrant's board of directors (or persons performing the equivalent functions):
a.All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant's ability to record, process, summarize and report financial information; and
b.Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal control over financial reporting.
| Date: April 15, 2026 | By: | /s/ Sigurdur Olafsson | |||||||||
| Sigurdur Olafsson | |||||||||||
| President, Chief Executive Officer and Director | |||||||||||
| (principal executive officer) | |||||||||||
Exhibit 31.2
CERTIFICATION OF CHIEF FINANCIAL OFFICER
PURSUANT TO RULE 13A-14(A) OF THE SECURITIES EXCHANGE ACT OF 1934
I, Christiana Stamoulis, certify that:
1.I have reviewed this annual report on Form 10-K of Keenova Therapeutics plc;
2.Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3.Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the registrant as of, and for, the periods presented in this report;
4.The registrant's other certifying officer and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the registrant and have:
a.Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the registrant, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
b.Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
c.Evaluated the effectiveness of the registrant's disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation; and
d.Disclosed in this report any change in the registrant's internal control over financial reporting that occurred during the registrant's most recent fiscal quarter (the registrant's fourth fiscal quarter in the case of an annual report) that has materially affected, or is reasonably likely to materially affect, the registrant's internal control over financial reporting; and
5.The registrant's other certifying officer and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the registrant's auditors and the audit committee of the registrant's board of directors (or persons performing the equivalent functions):
a.All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the registrant's ability to record, process, summarize and report financial information; and
b.Any fraud, whether or not material, that involves management or other employees who have a significant role in the registrant's internal control over financial reporting.
| Date: April 15, 2026 | By: | /s/ Christiana Stamoulis | |||||||||
| Christiana Stamoulis | |||||||||||
| President and Chief Financial Officer, | |||||||||||
| (principal financial officer) | |||||||||||
Exhibit 32.1
CERTIFICATION PURSUANT TO
18 U.S.C. SECTION 1350,
AS ADOPTED PURSUANT TO
SECTION 906 OF THE SARBANES-OXLEY ACT OF 2002
The undersigned officers of Keenova Therapeutics plc (“the Company”) hereby certify to their knowledge that the Company's annual report on Form 10-K for the annual period ended December 31, 2025 (“the Report”), as filed with the Securities and Exchange Commission on the date hereof, fully complies with the requirements of Section 13(a) or 15(d), as applicable, of the Securities Exchange Act of 1934, as amended, and that the information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
| By: | /s/ Sigurdur Olafsson | ||||
| Sigurdur Olafsson | |||||
| President and Chief Executive Officer and Director (principal executive officer) | |||||
April 15, 2026
| By: | /s/ Christiana Stamoulis | ||||
| Christiana Stamoulis | |||||
| President and Chief Financial Officer (principal financial officer) | |||||
April 15, 2026
